Abstract
Post-thrombotic syndrome (PTS) is a common complication of deep venous thrombosis (DVT) of the iliofemoral venous system leading to significant morbidity and high health care costs. It has been recently shown that percutaneous endovenous intervention (PEVI) can effectively reduce the incidence of PTS. The role of new oral anticoagulants (NOACs) in combination with PEVI in the reduction of PTS has not been previously studied. This report sought to evaluate the role of PEVI plus NOACs in the reduction of PTS in acute symptomatic femoropopliteal and iliac DVT. We studied 127 patients with acute lower extremity DVT who had undergone PEVI plus administration of NOACs. All had received a minimum of 3 months of anticoagulation with a NOAC following PEVI. The mean follow-up was 22±5 months. The patients were evaluated for development of PTS, bleeding, recurrent venous thromboembolism (VTE), duration of hospitalization and mortality. There was no in-hospital bleeding. The mean duration of hospitalization was 46±9 hours. DVT occurred in two patients who had been later switched to warfarin. There were four non-VTE-related deaths. PTS developed in five patients (3%), two of whom were those who had been switched to warfarin. Their mean Villalta score was 6.2±0.9. We conclude that the combination of PEVI plus NOAC and low dose aspirin is associated with a very low rate of PTS with the severity being only mild. This approach leads to very low rates of bleeding and recurrent VTE and promotes early discharge.
Introduction
Post-thrombotic syndrome (PTS) develops in approximately 25–60% of patients with acute lower extremity deep venous thrombosis (DVT) depending on severity, chronicity, anatomic level of involvement and efficacy of anticoagulation.1,2 The frequency increases with occlusive iliac venous thrombosis. PTS results in significant morbidity and a staggering toll on health care resources. 3 There is a growing body of evidence demonstrating that thromboreductive strategies including percutaneous endovenous intervention (PEVI) reduce this incidence.4 –6 The new oral anticoagulants (NOACs) that have become available over the last few years have shown promising results in the treatment of DVT. 7 Since they can directly and indirectly target clot-bound thrombin, they may be superior to vitamin K antagonists (VKAs). All related studies evaluating these drugs thus far have excluded patients undergoing catheter-directed thrombolysis (CDT). It is plausible to postulate that the synergistic effect of rapid thrombus reduction by CDT and subsequent treatment with NOACs can further reduce the PTS rate. The role of PEVI, which usually involves some form of CDT plus NOAC in the reduction of PTS, has not been previously investigated. We thus embarked to evaluate this role in patients presenting with acute DVT of the lower extremities.
Materials and methods
From September 2011 through June 2013, 127 patients with acute lower extremity DVT involving femoropopliteal and iliac veins underwent PEVI plus administration of NOACs by our group. These patients had significant symptoms warranting admission to the hospital. They received the treatment that we offer in our usual practice. This cohort was retrospectively identified and followed in a prospective fashion for development of outcomes. The study was approved by the institutional review board of A.T. Still University. Informed written consent was obtained from all patients for the procedure and observational follow up. The patients’ clinical characteristics are shown in Table 1.
Clinical characteristics of patients.
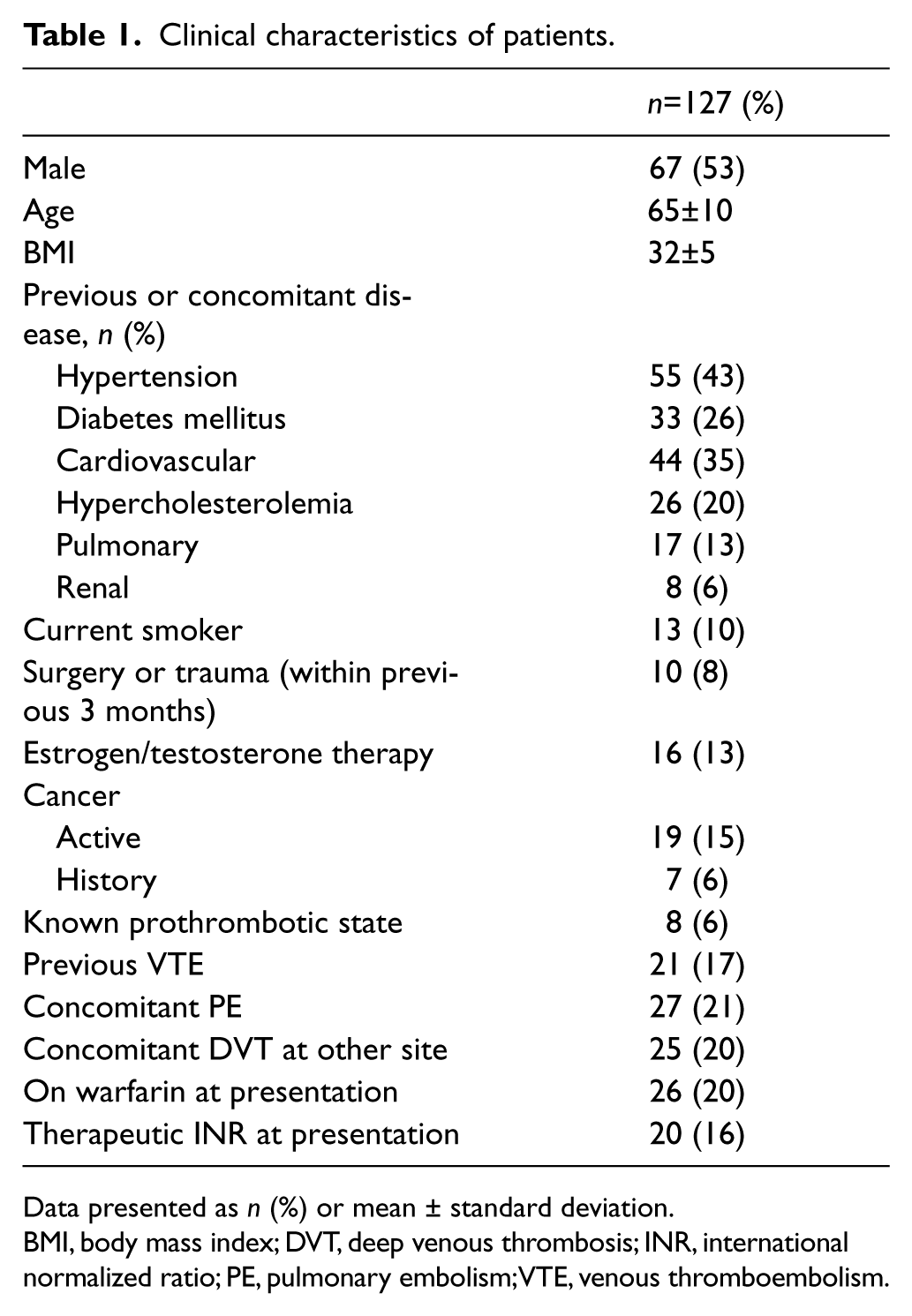
Data presented as n (%) or mean ± standard deviation.
BMI, body mass index; DVT, deep venous thrombosis; INR, international normalized ratio; PE, pulmonary embolism; VTE, venous thromboembolism.
The patients underwent PEVI from 2 to 32 hours after admission, with the onset of symptoms from 0 to 95 days of admission. Parenteral anticoagulation continued before and throughout PEVI and stopped at the end of the procedure. Two hours thereafter, oral anticoagulation was started with NOACs, which was continued for a minimum of 3 months. No warfarin or other VKAs were used for the initial 3 months. All patients received a minimum of 6 months of oral anticoagulation. There were 25 patients who after 3 months were subsequently bridged to warfarin due to insurance reasons. Initial parenteral anticoagulation during hospitalization was with enoxaparin in 27 patients (21%) and unfractionated heparin (UFH) in the remainder. Enoxaparin was given at 1 mg/kg subcutaneously twice daily. The dose of UFH was reduced after the initial bolus of 70 U/kg to 8–10 U/kg/hour during PEVI including the CDT period. The activated prothrombin time (PTT) was maintained at 60–100 seconds. No bolus of UFH was given during CDT. If the PTT was less than 60 seconds, only the maintenance dose was increased. Following termination of PEVI, no further parenteral anticoagulation was used. The duration of parenteral anticoagulation was thus not more than 24 hours.
The diagnosis of DVT was made by venous duplex sonography. All procedures were performed via the popliteal vein with the patient in the prone position. Access to the popliteal vein was obtained using a micropuncture needle with ultrasound guidance, with subsequent placement of a 6–8 French sheath. Patients having occlusive DVT involving two or more contiguous venous segments (femoropopliteal and iliac veins) underwent CDT for 24 hours and were re-evaluated the next day. No thrombectomy catheters were used in these patients and only an infusion catheter placed. CDT was completed within 24 hours and not extended beyond this period. The dose of tPA was 1 mg/hour through the infusion catheter. Patients having DVT in only one venous segment underwent same-day completion of treatment with a thrombectomy device. Thrombectomy was performed using a Trellis device (Bacchus Vascular, Santa Clara, CA, USA), Cleaner Catheter (Argon Medical Devices Inc., Athens, TX, USA), or placement of an infusion catheter with spraying of the thrombus by forceful hand injection and subsequent manual aspiration with an 8 French guide catheter. The mean dose of tPA used with thrombectomy catheters was 18 ± 3 mg. Fixed lesions deemed to be flow-limiting >50% stenosis on venography or intravascular ultrasound underwent stenting. Following the termination of PEVI, the sheath was removed and hemostasis achieved by manual compression for 5–10 minutes. Thigh-high compression stockings at 30–40 mmHg were applied and all patients were encouraged to ambulate in 1 hour. Inferior vena cava filters were placed in only 11 patients who were deemed to be at high risk for iatrogenic pulmonary embolism (PE) based on predictors described previously. 8
Two hours after PEVI, NOACs were given to the patients. Dabigatran was given at 75–150 mg orally twice daily to 35 patients; rivaroxaban at 20 mg orally daily to 76 patients; and apixaban at 5 mg orally twice daily in the remainder. New administration of aspirin at 81 mg daily was given to 75 patients for a minimum of 1 month. This included patients who had received a stent or were deemed to be of low bleeding risk. An additional 26 patients were already on aspirin for other reasons in whom aspirin was also continued. There were eight other patients on clopidogrel and prasugrel for coronary stents. No changes in the patients’ antiplatelet therapy was made, with the exception of reducing the dose of aspirin to 81 mg for those who were on higher doses. All patients were given graded compression stockings at 30–40 mmHg and instructed to wear them for 2 years.
During follow-up, patients underwent a thorough clinical assessment and a venous duplex by which the treated veins were evaluated for reflux and recurrent DVT. Reflux was defined as greater than 1 second of flow reversal on the spectral Doppler with standing or manual compression.
PTS was evaluated by two methods: our previous definition (Table 2) and the Villalta score.5,9 Two separate experienced investigators independently evaluated PTS with one using the Villalta score and the other using our definition. The assessors were physicians certified by the American Board of Internal Medicine in cardiovascular diseases or internal medicine with an interest in vascular medicine who were well trained in the assessment of PTS. The assessors were not personally involved in the interventional procedures and subsequent treatment. The utilized interventional approaches and site of DVT are shown in Table 3.
Definition of PTS a .

Definition of PTS is the presence of at least two new symptoms (leg burning, pain, aches, discomfort, restlessness, tingling), plus the
More than 1000 ms of reflux on venous spectral Doppler in a segment of the deep venous system without previous reflux or an increase of more than 500 ms in the extent of reflux if previously present.
Finding must be present for diagnosis; – finding must be absent for diagnosis; ± presence or absence of finding would not affect diagnosis.
PTS, post-thrombotic syndrome.
Interventional approaches used and site of DVT.
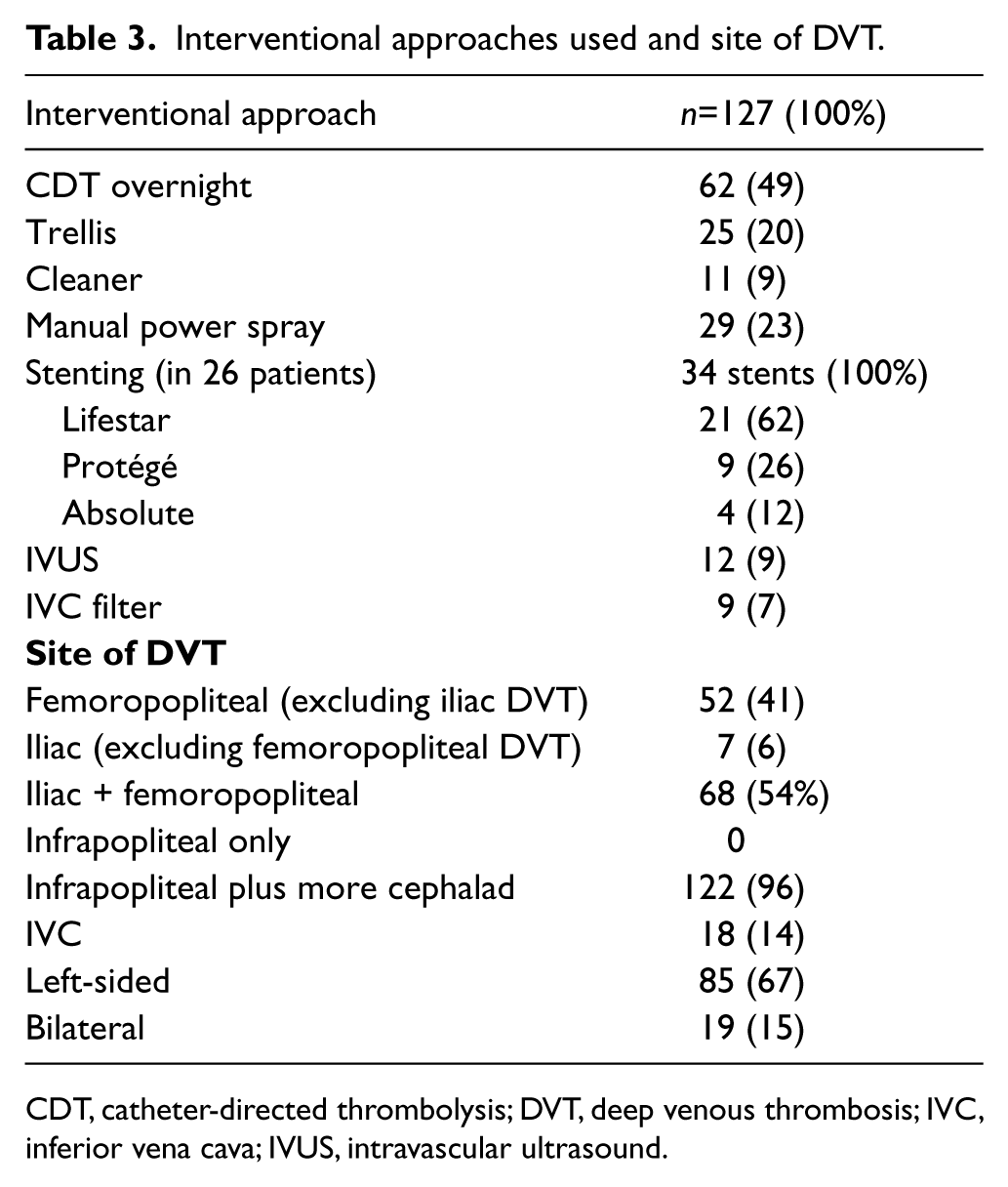
CDT, catheter-directed thrombolysis; DVT, deep venous thrombosis; IVC, inferior vena cava; IVUS, intravascular ultrasound.
Follow-up
The patients were seen at 1 week, 1 month, and then at 6-month intervals after discharge.
At each visit, they were evaluated for signs and symptoms of recurrent VTE, PTS and bleeding. All patients underwent a venous duplex scan of the treated venous segments at 1 month and every 6 months post procedure and at any time if suspicion of DVT was raised. If PE was suspected, objective testing was performed by pulmonary CT angiography or radioisotope ventilation perfusion (V/Q) scanning. Patients who had received a stent had annual radiography to monitor for stent fracture or kinks. Seven patients were lost to follow-up. The data are expressed as mean ± SD.
Results
The mean follow-up was 22±5 months. Using our definition, PTS developed in five patients (3%), two of whom were those who had been switched to warfarin. It developed at 9, 12, 14, 20 and 26 months after PEVI. The patients’ mean Villalta score was 6.2±0.9. Using the Villalta score as the initial diagnostic tool, PTS was found in seven patients (5.5%) with a mean score of 6.1±0.8. This included all five patients diagnosed using our criteria plus two additional patients. The degree of PTS was mild in all irrespective of the methodology used. There was no in-hospital bleeding. At follow-up, 99 patients were still on anticoagulation. There were four deaths related to cancer and congestive heart failure. One patient developed bleeding in the iliopsoas muscle at the time of transition to warfarin and while being on low-molecular-weight heparin. Another developed minor bleeding from hemorrhoids, again during the transition period. Four patients on dabigatran were later switched to rivaroxaban due to dyspepsia and abdominal pain. Venographic evidence of previous venous injury was found in 57 patients (45%), many of whom had no history of such event. This included obliteration of the truncal vein with presence of collaterals, venous stenosis, multiple tandem lesions, small diameter vessels, and venous fibrosis and sclerosis as previously defined. 4 There was no PE. Recurrent DVT was found in two patients who had been switched to warfarin. The mean duration of hospitalization was 46±9 hours. In 26 patients, 34 self-expandable stents were placed, six of which were in the femoropopliteal vein and the remainder in the iliac vein. Venous reflux in the treated venous segment was found in 29 patients (23%). Compliance with compression stockings was low. At 6 months only 50 patients were wearing compression stockings. Only 12 of them were at the recommended 30–40 mmHg pressure; five were at 15–20 and the remainder at 20–30 mmHg pressure. At 24 months, 28 patients were still wearing compression stockings.
Discussion
The results indicate that in patients undergoing PEVI for acute DVT, addition of NOACs is highly safe and effective and associated with a very low PTS rate of 3% or 5.5% based on the definition used. This rate is lower than the 6.8% observed at 30 months in our previous experience using the same definition of PTS wherein extended treatment was with warfarin. 5 It is substantially less than in the CaVenT study where the incidence of PTS was 40%. 6 NOACs target factor X and II and can dissolve clot-bound thrombin, a characteristic not seen with VKAs. 7 Early thromboreduction resulting from PEVI reduces the likelihood for valvular damage and associated reflux. Indeed, the rate of reflux was low at 23%. In a recent meta-analysis of three select papers, a trend toward reduction of venous reflux was noted with a relative risk (RR) of 0.39, although this value did not reach statistical significance (RR, 0.39; 95% CI, 0.16–1.00). 4
The duration of hospitalization was short (less than 2 days). This is in part due to the restriction of CDT to less than 24 hours, limitation of parenteral anticoagulation up to the end of sheath removal and early transition to NOACs. Patients can undergo PEVI while fully anticoagulated. 5 There is no need to withhold anticoagulation as is usually required when intervening in the arterial system.
In the current era, bleeding complications from PEVI should be very low. In this study and with adherence to the anticoagulation regimen no in-hospital bleeding was observed. In the TORPEDO trial, the bleeding rate was 2/91 (2.2%) and all were minor. 5 In the CaVenT study, total bleeding was unacceptably high (at 20%). 6 In this trial, the anticoagulation protocol was substantially different than our practice. The mean duration of CDT was 2.3±1.2 days and in some cases up to 6 days. Parenteral anticoagulation was given for 5 days prior to PEVI; in other words some patients were receiving over 11 days of parenteral anticoagulation and CDT combined, a hospitalization duration which is not acceptable in the current health economy. Furthermore, the anticoagulation was stopped several hours before PEVI, only to be started with bolus administration of 5000 units of UFH and a maintenance dose of 15 U/kg/hour. One hour after sheaths were pulled, full-dose low-molecular-weight heparin was given to a patient who had just received full-dose anticoagulation with UFH. 6
In a retrospective review of six case series using thrombectomy devices, the frequency of bleeding requiring transfusion was 7.5%. 10
The significant variation in the bleeding rate therefore stems from the differences in the duration and dose of anticoagulation and thrombolysis regimens.
In this study, all available NOACs were used based on their availability and operator’s discretion. The United States Food and Drug Administration (FDA) approved dabigatran, rivaroxaban and apixaban for the treatment of non-valvular atrial fibrillation in October 2010, November 2011 and December 2012, respectively. Rivaroxaban, dabigatran and apixaban were approved for treatment of VTE in November 2012, April 2014 and August 2014, respectively. Patients with renal insufficiency, bleeding tendencies and recent surgeries received apixaban. If none of the clinical conditions described above was present rivaroxaban was given. Earlier in the study dabigatran was the only available NOAC. It was given at 150 mg orally twice daily to most patients. If the patient was frail, had up to moderate renal insufficiency, weighed less than 50 kg or was over 80 years of age, the lower dose of dabigatran was used. In this study, the dose of NOAC used for treatment of DVT was less than the recommended initial dose. This was due to the fact that all reported studies had uniformly excluded patients who had received thrombolysis. We postulated that a modified dose of NOAC would be appropriate for our patients as all of them had received some form of thrombolysis. Furthermore, we did not adhere to the conventional 5 days of parenteral anticoagulation recommended by the American College of Chest Physicians. 2 Specifically, once PEVI was finished, no further parenteral anticoagulation was given. The rationale behind this approach was our previous observation that with substantial reduction in the thrombotic mass by PEVI, the requirement for parenteral anticoagulant therapy is reduced. 5 The anticoagulation regimen described resulted in no in-hospital bleeding and proved to be highly effective.
Role of aspirin
One potential reason for the low PTS rates in this study was the concomitant use of aspirin. The majority of patients received at least 81 mg of aspirin. The WARFASA and combination of WARFASA and ASPIRE trials demonstrated that use of aspirin for extended therapy for DVT leads to a 32–40% reduction in the development of VTE as compared to patients on no therapy.11,12 Prior to the publication of the above trials we had shown a 37% risk reduction in the development of PTS in the control arm of the TORPEDO trial. 5 In that arm, patients who were on aspirin had a statistically lower PTS rate than those not receiving aspirin. We now add 81 mg of aspirin to our treatment regimen for patients presenting for VTE unless there is a clear contraindication to its use.
Definition of PTS
Our definition of PTS was based on simplicity and objectivity. The scoring system developed by Villalta and colleagues has been shown to be valid when measured against quality of life and anatomic and physiologic markers of PTS with a high inter-observer agreement.9,13
The presence of five leg symptoms (pain, cramps, paresthesia, pruritus, and heaviness) and six signs (skin hyperpigmentation, induration, edema, new venous ectasia, redness, and pain during calf compression) is given a score of 0–3 using the contralateral unaffected leg as the basis for all evaluations.9,13 This means that 11 items have to be assessed, each with four possible scores; that is, 44 potential choices. Notwithstanding its previous validation, the Villalta score still incorporates substantial amounts of subjective data. Our definition of PTS encompassed the major features of the above scoring system, yet it was simpler to assess and included objective evidence of venous reflux. However, we ‘underdiagnosed’ two cases of mild PTS when compared against the Villalta score. PTS was of mild grade in all five (or seven) patients. By definition, all potential moderate and severe PTS cases using our definition would be inherently present in their corresponding severity category using the Villalta score. Thus, our PTS definition provides a simple and accurate tool to define PTS.
Limitations of the study
This study was an open-label single-arm study from our center which prospectively followed patients for outcomes after being treated by our group. The identification of the cohort was in a retrospective fashion. Consequently it did not have the rigor of a prospective randomized trial. Specifically there was no control group and no independent adjudication committee. It included all patients whose presentation was deemed to be that of an acute DVT. All available NOACs were included and their use was governed by their availability and the discretion of the operator. Nonetheless this report was a reflection of treatment in the ‘real world’ and for all comers. No patient was excluded for previous VTE, extent of involvement or chronicity of disease. All patients, however, had significant symptoms to warrant admission. Although there was variation in the extent of DVT and use of NOACs, little heterogeneity in the interventional treatment was present as it was based on the current practice approach that we offer to our patients. Owing to the existing limitations, comparisons between the outcomes of this report versus other studies should be made with caution.
Conclusion
We conclude that the combination of PEVI plus NOACs and aspirin is associated with very low rates of PTS when compared to historic controls. The resulting PTS severity is only mild and higher severity levels are eliminated. This approach leads to very low rates of bleeding and recurrent VTE and promotes early discharge.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
