Abstract
Treatment options for established post-thrombotic syndrome (PTS) are limited. Complex lymphedema therapy (CLT), a non-invasive treatment that improves lymphatic flow, may have the potential to improve PTS. We conducted a single-center, investigator-blind, randomized controlled trial of 31 patients with a clinically established diagnosis of PTS and compared the efficacy of graduated compression stockings alone (30–40 mmHg) with CLT, a treatment that includes compression stockings, exercise, patient education, skin care and lymphatic drainage. Primary outcomes were the 1- and 3-month changes in PTS severity by the Villalta score and disease-specific quality of life using the VEINES-QOL (Venous Insufficiency Epidemiological and Economic Study Quality of Life) questionnaire. Analysis was by intent-to-treat. We found from a baseline average score of 9.9 points, CLT reduced mean PTS severity scores by −2.4 points (p=0.02) at the 1-month and −2.3 points (p=0.05) at the 3-month follow-up. Score reductions with stockings only were similar at −2.1 (p=0.03) and −3.3 points (p=0.03) at 1 and 3 months. The differences in score between treatments were not significant. Neither treatment significantly changed the VEINES-QOL score except in patients with severe disease. Patients with moderate to severe PTS derived the greatest benefit from either therapy and the two therapies differentially impacted PTS signs and symptoms. We found a short course of lymphedema therapy and compression stockings offer similar benefit in patients with PTS; however, larger studies are needed to further explore the potential use of CLT in PTS, particularly in patients with more severe disease.
Introduction
Post-thrombotic syndrome (PTS) occurs in 20–50% of patients who experience a deep venous thrombosis (DVT).1,2 The pathophysiology of PTS is not well understood but involves venous hypertension, capillary leak and tissue hypoxia, ultimately leading to venous insufficiency following a DVT.3,4 Pain is a prominent symptom of PTS, while other signs and symptoms include itching, heaviness, edema, skin discoloration and leg ulcers. The development of PTS is associated with a diminished quality of life.5,6 Importantly, established PTS has no proven effective therapy that stops long-term progression of the disease or improves symptoms.
New treatment approaches are needed in the care of patients with PTS. The current standard of care in patients with DVT includes efforts at prevention of PTS through early initiation of anticoagulation and the use of elastic compression stockings to reduce the risk of developing PTS, although findings on stocking use are mixed.7,8 The use of mechanical intermittent compression devices is efficacious in the treatment of severe PTS symptoms; however, these treatments can be cumbersome.9,10 Compression stocking use remains the standard of care in treating established PTS but is not well supported in the literature.11,12 Pharmacologic treatment of established PTS has also not proven consistently beneficial.13–16
Complex lymphedema therapy (CLT), a treatment originally developed to treat congenital defects of the lymphatic system and secondary lymphedema due to disruption of lymphatic drainage (such as by axillary lymph node dissection), is an effective non-invasive treatment designed to enhance lymph flow through intact cutaneous lymphatics, thereby reducing swelling and restoring function in the affected extremity.17–19 We are aware of no formal evaluations of CLT efficacy for the treatment of PTS. However, given that venous hypertension that develops in PTS may lead to lymphatic overload and secondary lymphedema, we hypothesized that therapy which decreases lymphedema might improve PTS symptoms. Based on the lack of effective treatments for patients with a diagnosis of established PTS, we completed a clinical trial to compare the effect of CLT with compression stocking use on disease severity and quality of life.
Clinical study methods
Patients
Patients with a prior history of ipsilateral DVT and a clinical diagnosis of PTS of the lower extremity were eligible for trial participation. Other eligibility criteria included being age 18 years or older and having a median life expectancy greater than 2 years. Patients were excluded if they had active congestive heart failure, symptomatic lung disease, active bacterial infections, active angina pectoris, cardiac arrhythmia, DVT within the last 180 days, were pregnant or within 1 month post-partum, or were unable to participate in therapy due to monetary, physical, psychiatric or transportation limitations.
The study was approved by the institutional review board of University of Vermont and written informed consent meeting federal, state and institutional guidelines was obtained from all patients prior to enrollment. The trial was registered at ClinicalTrials.gov (NCT00633971).
Study design
This was a single-center, investigator-blind, randomized controlled trial. Participant demographics were recorded at study entry. Data regarding prior standard laboratory testing for hypercoagulable disorders were obtained from medical records. The primary outcomes were the 1- and 3-month changes in PTS severity as measured by the Villalta score. A second outcome was the change in disease-specific quality of life using the VEINES-QOL (Venous Insufficiency Epidemiological and Economic Study Quality of Life) questionnaire.
Patients were randomized to receive CLT or compression stockings in a 1:1 fashion using blocked randomization stratified by current anticoagulation use (yes or no) and time since initial thrombosis (6 months to 2 years versus greater than 2 years). Randomization occurred through use of sealed envelopes with randomization allocation concealed until the intervention was assigned. The allocation sequence was generated by the statistician assigned to the study. Clinicians enrolled participants at the time of their clinic visit. Allocation to treatment assignment was done by a study research nurse following opening of the sealed envelope. Investigator but not patient blinding was maintained throughout the study. Study compliance was obtained by verbal report and patient interview at 1- and 3-month visits. All adverse events reported by the patient during the study were recorded.
Treatments
Patients randomized to compression stocking use were prescribed a 30–40 mmHg elastic graduated compression stocking and instructed to wear the garment during waking hours throughout the duration of the study (3 months). Patients randomized to CLT underwent a standard four-component program including skin care, compression stocking and device use, manual lymphatic drainage and exercises. Therapists who performed CLT were licensed and trained as lymphedema therapists, and referrals were made based on the proximity of the therapist to the patient’s home or work. CLT was done in the outpatient setting and treatment consisted of two phases. Phase 1 incorporated visits to the lymphedema therapist where manual drainage, compressive therapy with bandaging and compression garments, lymphedema exercise and patient education were performed. Phase 2 involved maintenance in which patients performed their own exercises and self-administered compressive therapy as instructed. 20 The duration of Phase 1 of therapy (e.g. number and frequency of visits) was left to the discretion of the certified lymphedema therapist. For patients randomized to CLT, compression stocking therapy was encouraged throughout Phase 2.
For all enrolled patients, compression stockings and specialized garments not covered by insurance were provided by the study. Patients electing not to enroll in the study were also offered financial assistance for garments. Pre-randomization therapy with compression stockings was not an exclusion criterion. Patients using compression stocking therapy at the time of study enrollment were offered a prescription for 30–40 mmHg knee-high stockings (if they were currently wearing less compressive garments) and standard education on stocking use and care (if they were currently using 30–40 mmHg stockings, knee or thigh high). Patients not wearing garments at study entry were given a prescription for 30–40 mmHg knee-high stockings and standard education on stocking use and care.
PTS severity and quality of life assessments
All patients underwent an initial assessment of PTS severity and quality of life prior to randomization. Subsequent assessment was done 30 and 90 days following the initiation of therapy.
PTS severity was measured by the Villalta score. 21 This validated scale contains five patient-rated symptoms (heaviness, pruritus, pain, cramps and paresthesia) and six clinically rated signs of disease (edema, hyperpigmentations, venous ectasia, skin induration, skin redness and pain during calf compression).22,23 For clinician-rated signs, investigators were blind to the patient treatment arm but did know the study visit number (baseline, 1 month or 3 months). Investigators did not have access to prior scores at subsequent study visits. Villalta scores of 5–9 indicate mild PTS, 10–14 moderate PTS and a score greater than or equal to 15 or a venous ulcer is severe PTS.
The VEINES-QOL/symptom questionnaire, a validated self-administered questionnaire, was used to assess quality of life.24,25 This questionnaire measures several aspects related to quality of life, including multiple leg symptoms, the psychological impact of venous disease and limitations of daily activity. The questionnaire rates quality of life over the last 4 weeks and current symptoms. Lower scores indicate a poorer quality of life.
Statistical analysis
Patients randomized to CLT or compression stocking therapy were compared on baseline characteristics by two-sample t-tests for continuous variables and Fisher’s exact test for categorical variables. Changes in PTS severity and VEINES-QOL from baseline to 1 month and 3 months were tested within each group using paired t-tests. Two-sample t-tests were used to compare the changes in each measure between the two groups. Analyses were conducted based on intention-to-treat, with last observation carried forward for missing data. Thus, patients who did not complete the study were assumed to have not improved or worsened after dropping out.
Two sensitivity analyses were performed to assess the potential impact of missing data on the results. First, all analyses were repeated using only participants who completed all visits. Second, the outcomes were analyzed using mixed (hierarchical) modeling, with time as a within-subject factor and treatment group as a between-subject factor. This approach uses all available data for each subject.
Results
Patient characteristics
Trial participants were recruited from the thrombosis clinic from April 2008 to October 2010. The characteristics of trial participants are given in Table 1. No significant differences were seen by treatment assignment. Fifty-five percent of participants were previously diagnosed with a hypercoagulable state including heterozygous factor V Leiden (R506Q), heterozygous prothrombin G20210A, elevated factor VIII, protein C deficiency, protein S deficiency, antithrombin deficiency and antiphospholipid antibody syndrome. The majority of patients had a single disorder, with one patient having three disorders and two patients having two disorders. Nearly all patients (94%) were overweight or obese (BMI >24.9).
Characteristics of study participants.
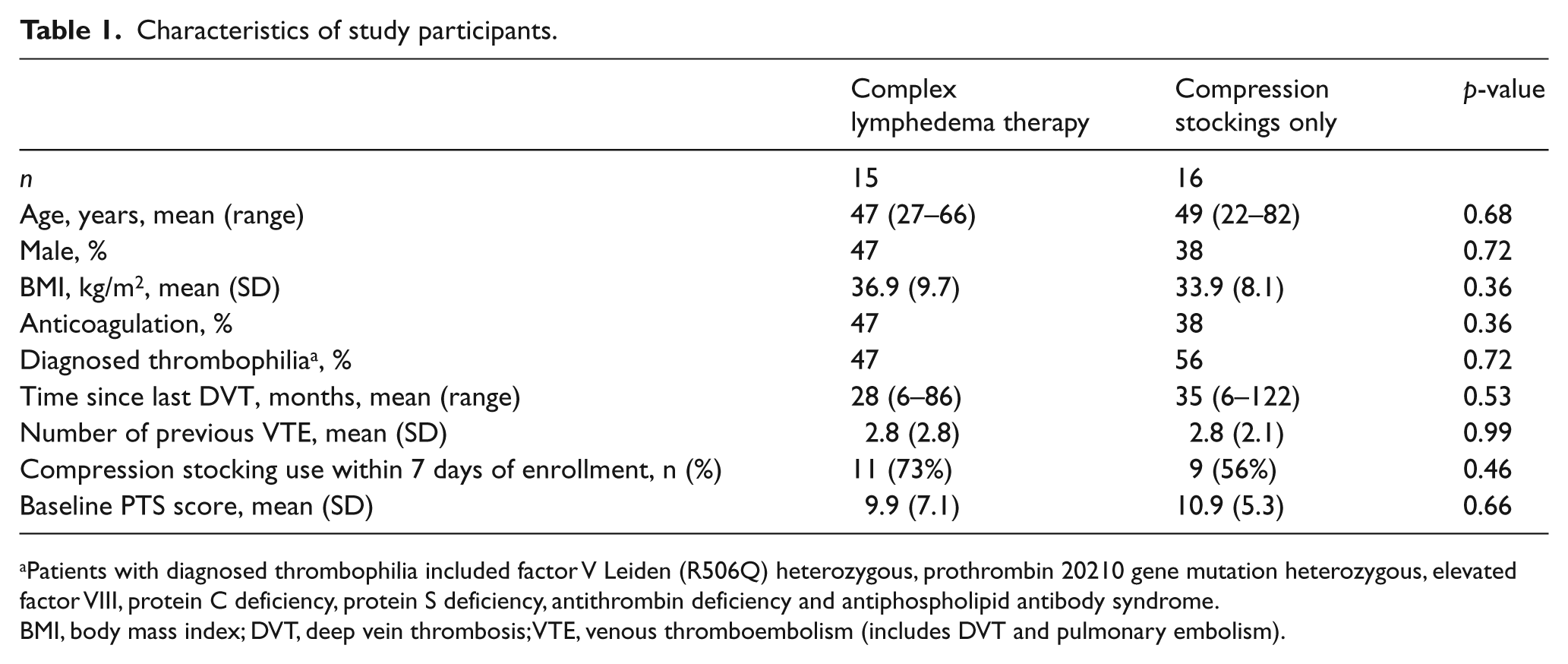
Patients with diagnosed thrombophilia included factor V Leiden (R506Q) heterozygous, prothrombin 20210 gene mutation heterozygous, elevated factor VIII, protein C deficiency, protein S deficiency, antithrombin deficiency and antiphospholipid antibody syndrome.
BMI, body mass index; DVT, deep vein thrombosis; VTE, venous thromboembolism (includes DVT and pulmonary embolism).
Impact of therapy on PTS severity
At 1 month, patients randomized to CLT reported wearing compression stockings an average of 5.7 days per week, while the compression stockings only group reported wearing stockings an average of 5.9 days per week. Similar high rates of stocking use were reported at the 3-month visit (6.7 and 5.9 days, respectively). For patients randomized to CLT, the average number of visits to the lymphedema therapist was 12 over the course of the 3-month period. At the 1-month visit, CLT participants reported performing lymphedema therapy at home an average of 3.5 days per week; this number decreased to 2.4 days per week at the 3-month visit.
At baseline, participants had moderate PTS with mean Villalta scores of 9.9 in the CLT group (n=15 patients) and 10.9 in the compression stockings only group (n=16 patients). As seen in Table 2, PTS severity decreased at 1 month of therapy for patients randomized to both CLT and compression stockings only (n=10 patients in each group). This improvement in PTS severity was maintained at 3 months of therapy, with the average score indicating mild PTS in both patient groups (n=7 patients in the CLT group and n=9 patients in the compression stockings arm). There was no difference between the study arms at 1 and 3 months. Results from sensitivity analyses were the same as for the intent-to-treat findings.
The impact of complex lymphedema therapy and compression stockings only on PTS severity and quality of life in patients with post-thrombotic syndrome.
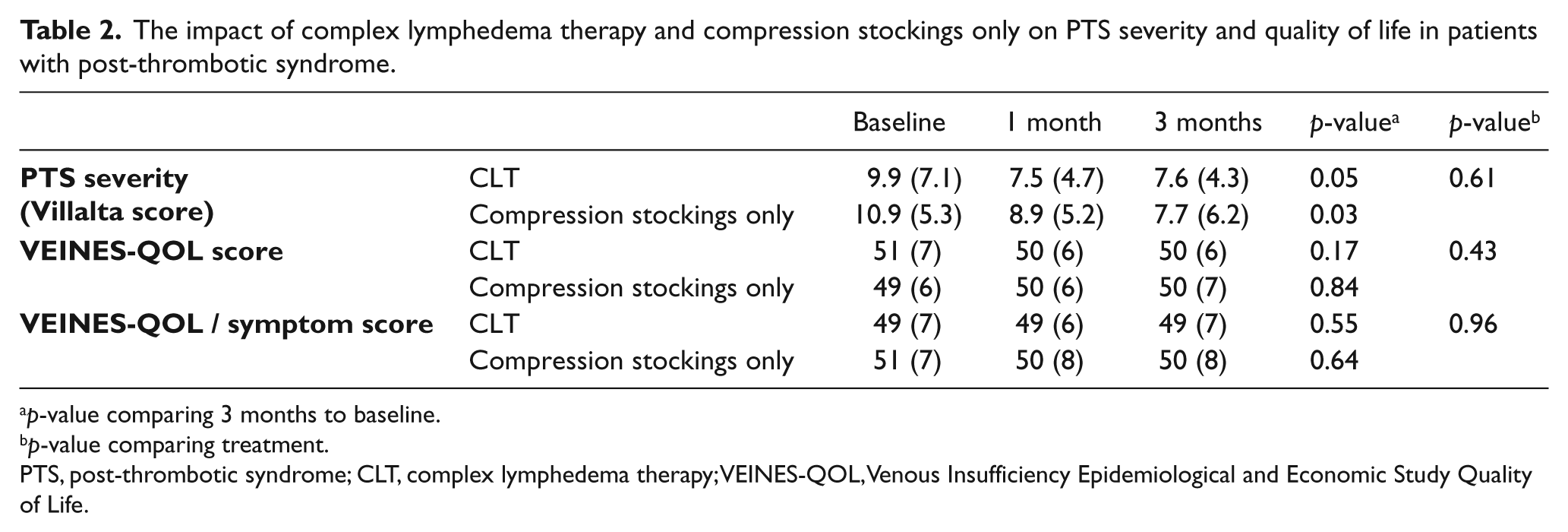
p-value comparing 3 months to baseline.
p-value comparing treatment.
PTS, post-thrombotic syndrome; CLT, complex lymphedema therapy; VEINES-QOL, Venous Insufficiency Epidemiological and Economic Study Quality of Life.
The effect of the two treatments on individual signs and symptoms captured by the Villalta score are shown in Table 3. Decreases in leg cramps and leg heaviness were seen in the compression stockings only group. In patients receiving CLT, decreases in leg pain and the appearance of venous ectasia were observed. However, it is important to note that given the small sample size and number of items in the Villalta score, these observations must be interpreted with caution and require follow-up in larger clinical studies.
The effect of treatment on individual components of the Villalta score.
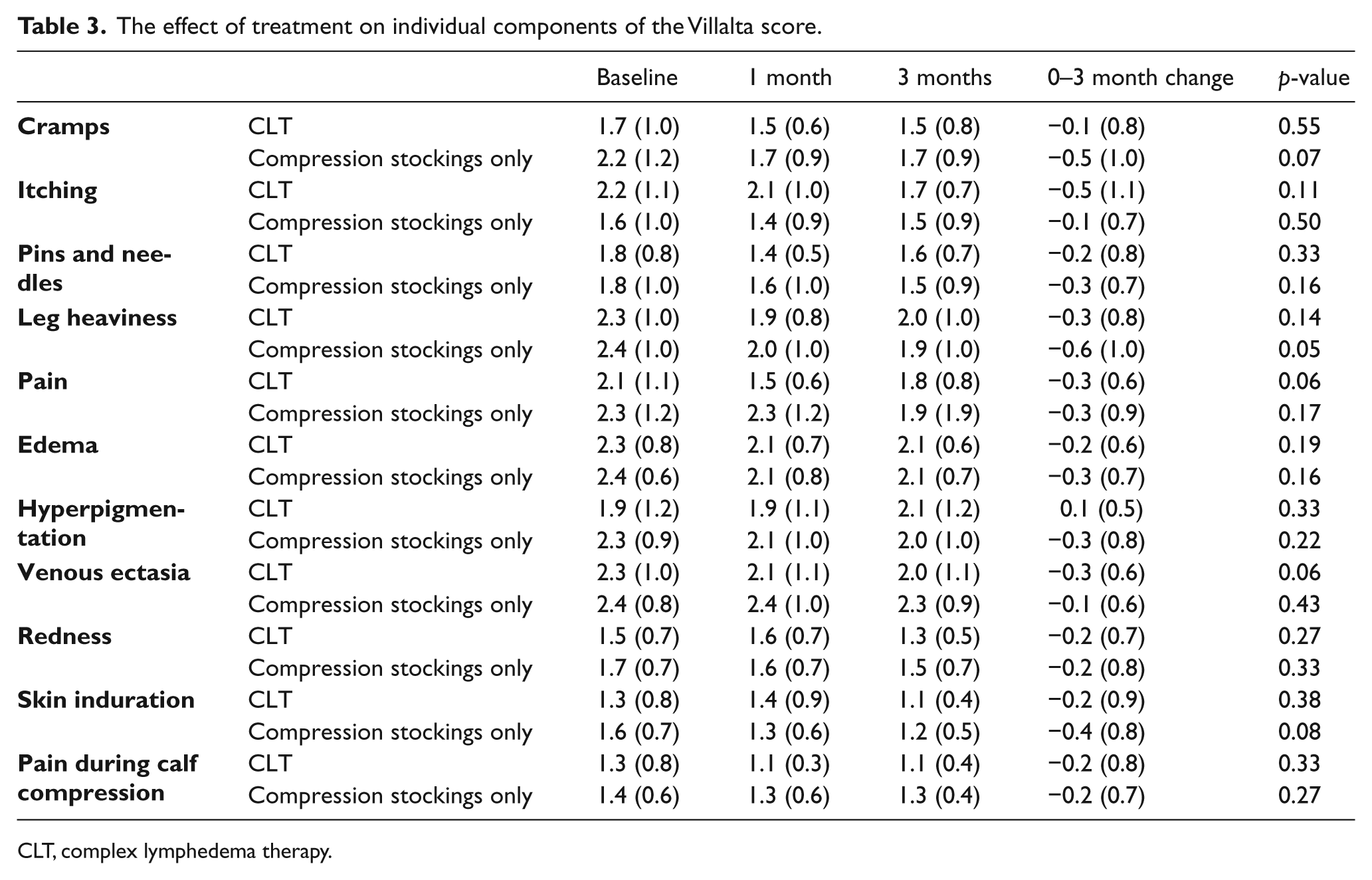
CLT, complex lymphedema therapy.
A stratified analyses was performed to determine if baseline PTS severity and use of compression stockings at study entry impacted observed responses to either therapy. As seen in Figure 1, patients (n=3 with data available at follow-up for analysis) with a higher initial PTS severity score (>10) who received CLT experienced a greater improvement in PTS severity as compared to those with less severe disease (n=7) (–9.3 vs −0.5 points, p=0.02). This same trend was seen in patients receiving compression stockings only (−7.8 vs −1.7, p=0.08). An analysis based on compression stocking use at study entry was also performed as it was anticipated that ongoing therapy prior to enrollment may impact our observed results. In patients not using compression stockings in the 7 days prior to study enrollment (n=9 patients with 3-month data available for analysis), treatment arms derived a greater benefit from either intervention (CLT or compression stockings only), with a decrease in the PTS severity score of −8.8 points for non-users and −1.5 for users (p=0.07).
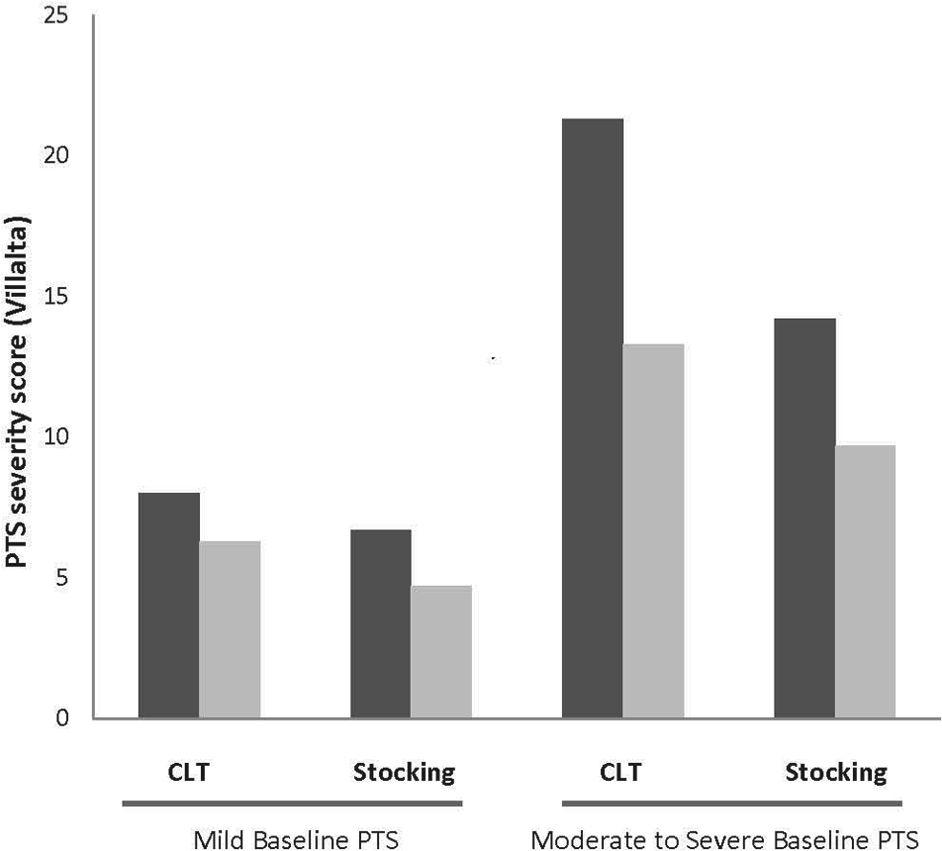
The impact of complex lymphedema therapy (CLT) and compression stockings only treatment on post-thrombotic syndrome (PTS) based on baseline PTS severity. Severity refers to baseline PTS severity at study entry. Data plotted are baseline Villalta score (dark bars) and 3-month Villalta score (light bars) for each severity group based on therapy received (CLT or compression stockings only). Mild baseline PTS = Villalta score <10, moderate and severe PTS = Villalta score ≥10.
Impact of therapy on patient quality of life
As seen in Table 2, there was no impact of either treatment on quality of life over the 3-month study period. This was true both for the VEINES-QOL summary score and for the VEINES-QOL/symptom score. A stratified analysis by baseline use of compression stockings in patients receiving either therapy revealed that participants who did not wear stockings prior to study enrollment (n=9) had an increase in their overall quality of life (summary QOL +1.9 points) as compared to participants who wore stockings prior to enrollment (summary QOL −2 points), p=0.02. Similar results were found for the symptom QOL: +1.0 points (prior stocking users) versus −2.3 points (non-users), p=0.06. Further, those with Villalta PTS scores >10 were associated with a greater change (improvement) in overall QOL (Spearman correlation coefficient = +0.39, p=0.10) and QOL/symptom score (Spearman correlation coefficient = +0.42, p=0.07) following either therapy, although these correlations did not reach statistical significance.
Adverse events and study discontinuation
Adverse events that required treatment discontinuation included new DVT in two participants in the compression stockings only arm (13%) and one in the CLT arm (7%), superficial thrombophlebitis in one patient in the compression stockings arm and stocking allergy in one patient in the compression stockings only group. Four patients (12%) randomized to CLT identified travel and time constraints as barriers to study continuation and discontinued study participation. Two patients (6%) randomized to compression stockings only withdrew without explanation and were lost to follow-up. No protocol deviations were reported. No cardiac or pulmonary complications were observed in either group.
Discussion
This trial demonstrated that both CLT and compression stocking use had similar efficacy in the treatment of established PTS. On average, PTS scores improved from moderate to mild over 3 months of either therapy. Patients with more severe disease and those not actively wearing compression stockings prior to study enrollment had more benefit from therapy with both improved disease severity and a trend toward improvement in quality of life.
The clinical care of patients with established PTS is challenging given the lack of available literature to support an efficacious approach to this chronic disease. Options available to patients are few in number and management is usually based on standard approaches to lower extremity wound care such as limb elevation, stockings, wraps or medicated bandages.26,27 Compression stockings are used empirically but there are conflicting results regarding efficacy.11,28–31 Recently, exercise training has shown reasonable clinical benefit but a prolonged exercise commitment from participants is required. 32 Therefore, there is a clinical need to establish additional effective therapy options in patients with established PTS of varying severity.
We are not aware of other studies that have evaluated the efficacy of CLT in the treatment of established PTS. The use of CLT was previously established in the treatment of patients with malignancy and congenital lymphedema syndromes.17–19 CLT is well tolerated and has been standardized in the United States using a credentialing program. Additionally, most of the time, CLT can be administered by the patient or family at home following training by a specialist. These aspects of CLT make it particularly attractive as a potential therapeutic option for patients with PTS. We believe this study is the first to evaluate the efficacy of CLT in the treatment of established PTS.
We observed a similar benefit from both compression stockings only and CLT on established PTS. The results observed for stockings only (and CLT) were similar in magnitude to those seen by Kahn et al. for a supervised exercise program, where the average improvement in the Villalta score was −3.6 points. 32 Our results in the stockings only arm of our trial, however, are in contrast to prior work that also assessed the impact of compression stockings (30–40 mmHg) on PTS. 28 In this study, no benefit from stocking use was observed; however, the endpoints for this study differed significantly from ours and largely focused on patient-reported symptom deterioration.
In the current trial, patients with the most severe disease appeared to benefit most from either intervention, suggesting this patient group might be considered ideal candidates for treatment as well as further study. Our findings are consistent with a prior trial evaluating twice daily lower extremity pump therapy in patients with severe PTS. 9 Similarly, in the VENOPTS trial evaluating a novel venous-return assist device in patients with severe PTS, a large improvement in PTS severity (based on the Villalta score) was observed. 10 Taken together, these results suggest that improving venous return as well as lymphatic return may provide clinical benefit for patients with severe disease. Because CLT includes compression stocking use, one conclusion that might be drawn from our study is that the majority of benefit seen with CLT is due to compression stocking use. While additional studies are needed to ascertain definitive answers to these questions, the findings of differences in improvement of individual symptoms suggests patients with different symptoms may benefit from different treatment strategies.
Limitations of our study include its small sample size (31 patients), single-center design and that one-third of patients were not available for follow-up analysis due to new venous thrombosis or lack of follow-up as scheduled. Notably, four patients (12%) randomized to CLT stated that time and travel constraints contributed to study discontinuation, emphasizing the additional commitment required for this therapeutic intervention. These factors significantly limit the generalizability of our findings to large populations at this time. Our study was similar in size to other published trials in PTS, and despite the small sample size we were able to detect changes in PTS scores with either treatment. In planning the study, we used estimates from prior studies to determine the sample size needed for 80% power to detect a 20% difference between treatments in PTS severity score (with two-sided alpha=0.05). However, our observed baseline PTS scores were lower than anticipated, the standard deviation of the score was higher than in prior studies, and improvement in the compression therapy group was larger than expected, decreasing our ability to detect a difference between the groups.
Additional limitations of our study include the relatively large number of patients enrolled with mild PTS, as it is likely that these patients do not require more aggressive treatment with CLT. Because compression stockings were recommended in patients for 2 years following a DVT at the time of study initiation, a large percentage of our patients were also wearing compression garments prior to randomization, which likely impacted the overall effect seen for either intervention (as illuminated by the stratified analysis). Also, our results reflect only a short follow-up of 3 months following therapy initiation. As PTS is a chronic disease, this follow-up period may be too short to see clinically significant results and does not address potential issues regarding compliance over longer time periods in this patient population. Additional patient characteristics of note in our study include that almost all patients were overweight and many had experienced several venous thrombosis episodes, which introduces a potential selection bias that must be taken into account and may limit the generalizability of our findings. Finally, a clinical diagnosis of PTS made by an experienced thrombosis physician was required for enrollment in the study; however, a formal Villalta score >4 was not used as an inclusion criteria.
Strengths of the study include the blinded ascertainment of PTS using a validated severity scale, and assessment of quality of life, which can be impaired in this condition. 33 The average VEINES-QOL score in both treatment groups was similar to estimates previously reported for patients with PTS and suggests our results may be generalizable. 33 Further, our findings on study retention and effect size inform the feasibility of larger trials for patients with established PTS.
In conclusion, CLT and compression stocking therapy alone appear to be safe, well-tolerated and effective treatments in patients with established PTS. Additional trials assessing CLT are essential to potentially expand treatment options for patients with PTS.
Footnotes
Acknowledgements
The authors wish to thank Dr Susan Kahn for providing the assessment tools used in the study and for her input regarding study design and analysis. We also thank Ms Karen Libby for her excellent work as study coordinator, Dr Patricia O’Brien for her expertise in lymphedema care, and the study participants. CEH conceived of and designed the research study, performed and supervised research, participated in data analysis and wrote the manuscript; NMB aided in study recruitment, data analysis and manuscript preparation; PL aided in study recruitment and manuscript preparation; PWC performed the data analysis and aided in manuscript preparation; and MC participated in study design, recruitment, data analysis and manuscript preparation
Declaration of conflicting interest
The authors have no conflict of interest to declare.
Funding
This work was supported by a grant from The Victoria Buffum Fund at the University of Vermont / Fletcher Allen Health Care and R01 HL083926 from the National Institute of Health.
