Abstract
Fibromuscular dysplasia (FMD) is a non-atherosclerotic vascular disease commonly affecting the renal and internal carotid arteries (ICAs). A previously unrecognized finding is a redundancy of the mid-distal ICA in FMD patients causing an ‘S’-shaped curve. Carotid artery duplex ultrasounds were reviewed in 116 FMD patients to determine S-curve prevalence. FMD patients with an S curve were matched to four control patients divided equally into two groups: (1) age and sex-matched and (2) age ≥70 and sex-matched. S curves were present in 37 (32%) FMD patients. Of these, nine (24%) had angiographic evidence of FMD in their ICA only, 13 (35%) had renal artery FMD only, and 15 (41%) had both ICA and renal FMD. Two patients in the age and sex-matched group had S curves (odds ratio 16.86, 95% CI 3.92–72.48;
Introduction
Fibromuscular dysplasia (FMD) is a non-inflammatory, non-atherosclerotic vascular disease. While most commonly affecting the renal and internal carotid arteries (ICAs), FMD may lead to stenosis, occlusion, aneurysm and/or dissection of any involved artery.1–3 Affecting women approximately 90% of the time, the cause of this disease remains unknown. 4 Although classified as a ‘rare’ disease, evidence suggests that FMD is more common than previously thought.2,5 Summarizing the results of four studies, it has been reported that 139 (4.4%) of 3181 angiograms in asymptomatic potential kidney donors had angiographic evidence for FMD.5–9
Historical data suggest that carotid artery FMD was found in 25–30% of patients with FMD.1,10–12 However, recent data from the first 447 patients enrolled in the United States Registry for Fibromuscular Dysplasia found carotid FMD in 74% of patients in whom neuroimaging was obtained. 4 The underestimated disease prevalence of FMD in the carotid and vertebral arteries is probably related to the fact that patients who present with hypertension and renal artery FMD are often not screened for the presence of asymptomatic carotid and vertebral FMD. The typical morphological appearance of carotid FMD is shown in Figure 1. Whereas atherosclerosis typically occurs at the origin or proximal portion of an artery, FMD most commonly occurs in the mid to distal portion of the vessel, 2–3 cm (or more) distal to the carotid bifurcation.1,2
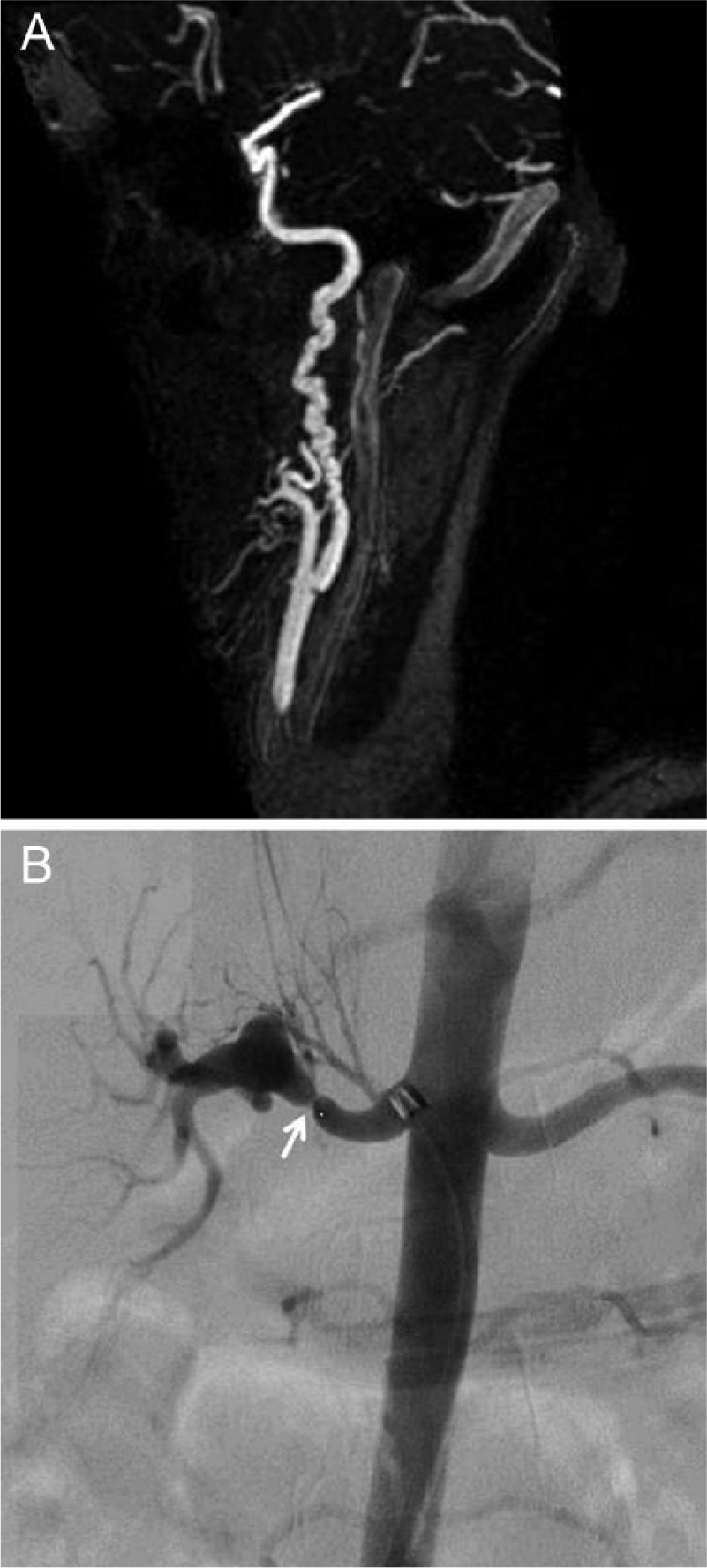
(A) Magnetic resonance angiogram (MRA) of the left internal carotid artery showing typical appearance of medial fibroplasia (multifocal FMD). Note multiple alternating areas of constriction and dilatation (string of beads) in the mid and distal part of the internal carotid artery. (B) Catheter-based angiogram demonstrating a focal severe stenosis (arrow) in the mid renal artery consistent with intimal fibroplasia (focal FMD).
Common clinical presentations of patients with carotid or vertebral artery FMD include hypertension, headache, pulsatile tinnitus, dizziness, cervical bruit, neck pain, transient ischemic attack (TIA) and stroke.4,10,11 Less common manifestations include non-pulsatile tinnitus, amaurosis fugax, and Horner’s syndrome.4,10–12 However, many patients have no symptoms and are discovered incidentally when imaging is performed for another reason.2,4 While high rates of stenosis, dissection, and aneurysm in FMD have been observed, 4 concrete data linking ICA tortuosity to FMD have been limited.13–15
Although catheter-based angiography is the gold standard for the diagnosis of FMD, non-invasive imaging such as computed tomographic angiography (CTA) and magnetic resonance angiography (MRA) have been used for diagnostic purposes. 3 Carotid duplex ultrasound has not been directly compared to the gold standard, but velocity shifts, turbulence, and tortuosity in the mid to distal artery can be well visualized on color duplex ultrasound (Figure 2). Patients with FMD may have extremely tortuous or redundant arteries. The elongation and tortuosity of the mid to distal ICA may be easily visualized on carotid duplex ultrasound and has a distinct morphological appearance forming the shape of an ‘S’ (S curve) (Figure 3). We hypothesized that the S curve is present more frequently in FMD patients than case-matched controls and, in fact, may be another anatomic manifestation of FMD.
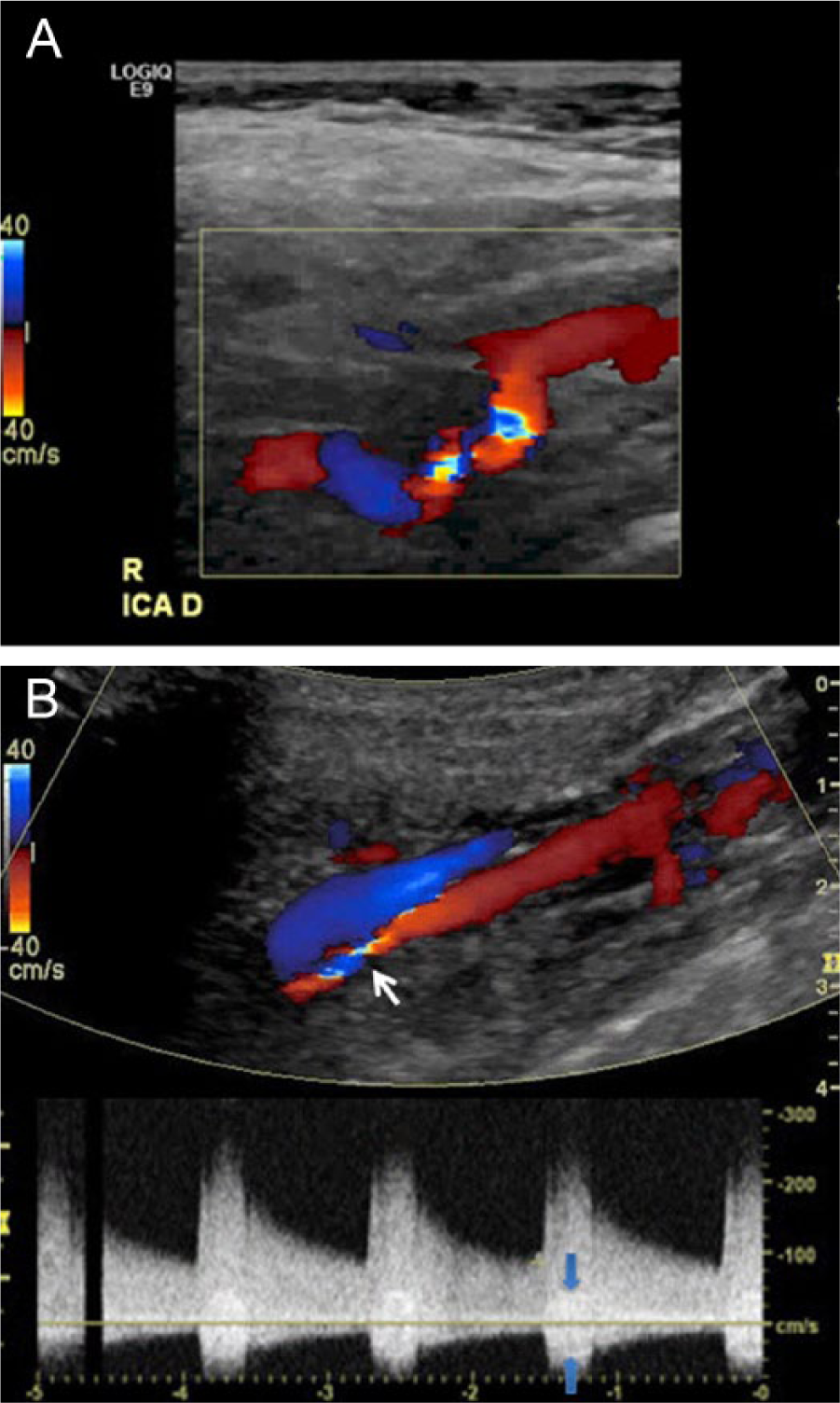
The appearance of fibromuscular dysplasia of the carotid artery duplex ultrasound. (A) There is turbulence and tortuosity in the distal internal artery. (B) There is turbulence (white arrow) in the distal internal carotid artery. The spectral waveform shows markedly increased velocity of blood flow (peak systolic velocity 268 cm/s) and the presence of a spectral bruit (blue arrows), indicating the stenosis is severe.
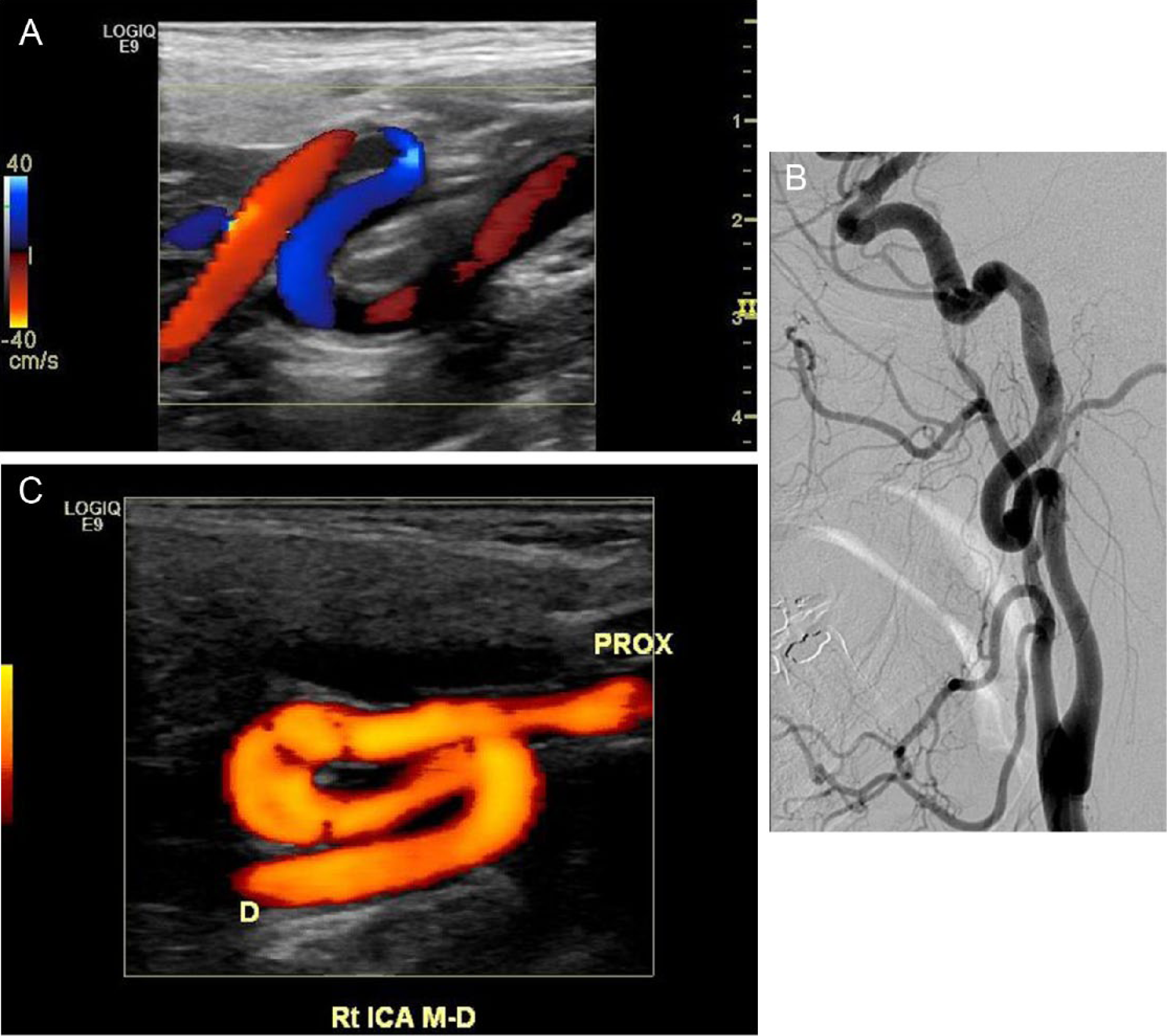
Examples of S-curve morphologies from three different patients. (A) Carotid duplex ultrasound demonstrating the color Doppler appearance of the S curve in the right internal carotid artery. The head is to the left and the feet are to the right. (B) Catheter angiogram of a patient with FMD and the S curve. (C) Carotid duplex ultrasound – color power angiogram of an S curve in the right ICA. The head is to the left and the feet are to the right.
Methods
Study design
The study utilized a case-matched, cross-sectional design. Carotid artery duplex ultrasounds performed at a single center between 1 January 2008 and 1 November 2011 were reviewed in 116 patients diagnosed with FMD. If more than one ultrasound existed in the system, the most recent ultrasound was reviewed. The ultrasounds were evaluated for the presence of an S curve as determined by two independent reviewers (SSS, JFL). All ultrasounds with an S curve were reviewed by the principal investigator (JWO). The S curve was defined as a distinct morphological entity of the mid to distal portion of the ICA formed by an elongation causing two markedly tortuous turns in the shape of the letter ‘S’. If there was only one turn, or if there was less severe tortuosity (Figure 2A), the patient was not placed in the S-curve group. Patients determined to have an S curve were matched to carotid artery ultrasounds of non-FMD control patients in a 2:1 fashion into two groups: (1) age and sex-matched and (2) age ≥70 and sex-matched (resulting in four controls per case). Since arterial redundancies increase with age, the ≥70-year-old group was included to explore the comparison between age-related redundancies and FMD. This study was approved by the institutional review board of the Mount Sinai Medical Center.
Control patients were selected from 5676 patients who had a carotid duplex ultrasound from 1 January 2008 to 1 November 2011 in the Vascular Diagnostic Laboratory at the Mount Sinai Medical Center. In control group 1, age was matched to each case within 2 calendar years. In control group 2, an age ≥70 was included. Both control groups were sex-matched. Ultrasounds were excluded from the control group if the patients had known fibromuscular dysplasia or another arteriopathy in the vessel such as Loeys-Deitz syndrome, arterial tortuosity syndrome, Moyamoya disease, carotid dissection, carotid aneurysm and Takayasu’s arteritis. In searching the database for control subjects, the first ultrasound that met the age and sex specifications that occurred prior to the date of each index case ultrasound and the first ultrasound that occurred after the date of each index case ultrasound was used as long as they did not meet any of the exclusion criteria.
All FMD patients were clinically evaluated by the principal investigator (JWO), prospectively followed, and entered into the United States Registry for Fibromuscular Dysplasia. 4 S-curve morphology was not part of the initial registry database. The patient was considered to have FMD if they met one of the following two criteria on a CTA, MRA, or catheter-based angiogram: the appearance of a string of beads (Figure 1A) in the mid or distal portion of the renal or carotid (multifocal FMD, previously called medial fibroplasia); or the appearance of a focal severe stenosis of the renal or carotid in the absence of atherosclerosis (focal FMD, previously called intimal fibroplasia). 16
Other arteriopathies were excluded based on clinical evaluation, imaging, patients with aneurysms or dissections, and genetic testing for Loeys-Deitz syndrome or Ehlers-Danlos syndrome type IV. In our FMD clinic, patients routinely undergo a carotid artery and renal artery duplex ultrasound during their first office visit leading to the identification of FMD (often asymptomatic) in more than one arterial bed in the majority of patients. 4
Carotid duplex ultrasound
All carotid artery duplex ultrasound examinations were performed on a GE LOGIQ E9 (Milwaukee, WI, USA) ultrasound machine. Images were saved as dynamic clips and stored on the syngo Dynamics Cardiovascular Imaging and Information System (Siemens Medical Solutions, Muenchen, Germany). The Mount Sinai Vascular Diagnostic Laboratory performs approximately 8000 studies per year and is accredited by the Intersocietal Accreditation Commission (http://intersocietal.org/vascular/). All vascular technologists and the physicians interpreting the vascular ultrasounds are registered vascular technologists (RVT) or registered physicians in vascular interpretation (RPVI). The bilateral cervical ICAs and bilateral vertebral arteries were imaged from multiple approaches in the sagittal and transverse planes throughout the cervical segments using a 9L-D linear transducer (9–5 MHz). A C1-5-D convex transducer (1–5 MHz) was utilized as needed for imaging distally under the mandible. Vessels were evaluated for the presence of plaque, stenosis, tortuosity, turbulence, aneurysm and dissection. Color and pulsed-wave Doppler interrogation was performed with appropriate beam-to-vessel alignment and an angle-correction technique of ≤60 degrees for acquisition of velocity flow values. Multiple samples of peak systolic and end-diastolic velocities within the internal carotid and vertebral arteries were obtained within each of the proximal, mid and distal segments bilaterally.
Statistical analysis
Prior to the study onset, power was calculated by estimating a prevalence of S curves to be 25% among FMD patients and 5% among control patients. This estimate was based on historical data estimating carotid FMD prevalence,10–12 as this study was conceived prior to the more recently released registry data indicating that FMD occurs in the carotid artery approximately 70% of the time. 4 With the aforementioned assumptions, there was an 80% power to detect the 20% difference if 46 FMD patients and 92 controls were evaluated. With carotid FMD being more frequent than previously thought, our initial sample size estimate was larger than required to meet the 80% power threshold.
Baseline differences between groups were analyzed using the chi-squared test for dichotomous variables and
Results
A total of 116 patients were diagnosed with FMD during this time period. In this cohort, 108 of 116 (93%) patients were women and the average age was 51.0 years (Table 1). There was no difference in age between those FMD patients with S curves and those without S curves. There were proportionally more men in the S-curve versus non-S-curve groups (13.5% vs 3.8%), but with small absolute numbers in men, this difference did not reach statistical significance (
Baseline characteristics.
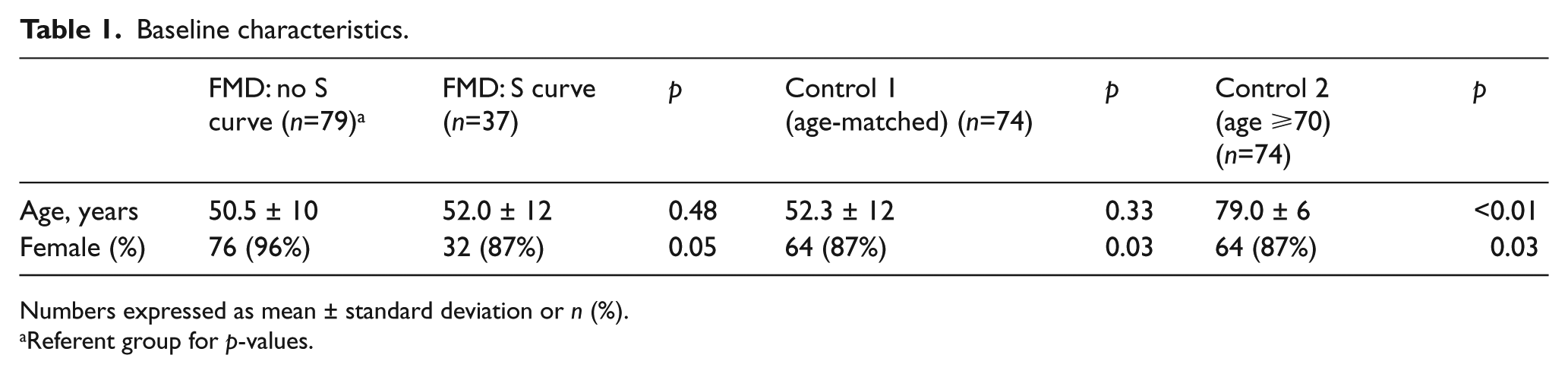
Numbers expressed as mean ± standard deviation or
Referent group for
The prevalence of S curves in FMD patients versus control groups
An S curve was present in 37 (31.8%) patients with FMD. Figure 3 is an example of carotid artery duplex ultrasounds and a catheter-based angiogram showing the S curve. Two patients (2.7%) in control group 1 (age and sex-matched,
The prevalence of S curves based on location of FMD
Of the entire group of FMD patients, 28 (24%) had FMD in the carotid artery only, 40 (34%) had renal artery FMD and 48 (42%) had both carotid artery and renal artery FMD. There was no difference in the prevalence of S curves based on the arterial bed affected by FMD (Table 2).
Prevalence of S curve by location of FMD in FMD patients.

Discussion
This case–control study of 116 FMD patients demonstrated a strong association between the S curve and FMD as compared to age and sex-matched non-FMD patients. When comparing FMD patients to patients over the age of 70, the association of an S curve with FMD was still present, albeit a bit weaker. This is the first study to demonstrate these associations in FMD patients.
Redundancy of the ICAs may be a congenital or acquired phenomenon. Resulting from an elongation of the vessels in a fixed space, this redundancy gives the ICA a tortuous appearance on imaging tests. Embryologically, the proximal ICA originates from the third aortic arch, whereas the mid-distal ICA originates from the dorsal aorta.17–19 This causes a normal bend in the embryonic carotid arteries. As the fetus develops and the heart recedes into the thorax, stretch of the ICA eliminates any remaining bends or tortuosity. Failure of this developmental process is one causative explanation for ICA tortuosity as supported by the initial studies by Weibel and Fields that found instances of ICA tortuosity in patients as young as 6 weeks of age. 19 Based on carotid arteriograms, Weibel and Fields classified arterial tortuosity into three categories: (1) tortuosity, which describes vessel elongation or undulation; (2) coiling, which describes vessel elongation or redundancy; and (3) kinking, which describes vessel angulation associated with stenosis. 19 Various iterations of this classification scheme have been used since then, usually attempting to classify based on degree of angulation.20–23 The true prevalence of the S curve in the general population remains unknown.
ICA redundancy or tortuosity occurs with greater frequency in elderly individuals and increases with age.18,19,24 Strain due to prolonged arterial hypertension, and vessel wall alterations within the tunica media, have been two postulated mechanisms of acquired arterial tortuosity.25–27 Animal and human studies have revealed that failure of components within the tunica media plays a role in the tortuosity seen with age and hypertension, aneurysms, and ectasias (arteriomegaly).26,28 Several case reports have also identified disrupted and dysplastic elastic tissue localized to the area of elongation in histological/pathological specimens.29,30 These pathologic disturbances in the elastic tissue may explain the previously identified clinical disease states that have been linked to arterial tortuosity or redundancy, including Loeys-Deitz syndrome, arterial tortuosity syndrome, Ehlers-Danlos syndrome type IV, and Marfan’s syndrome.31–33
Along with the association between ICA redundancy and connective tissue disease, there has long been postulated to be overlap between some of these disease states and FMD.2,34,35 This potential link between connective tissue disturbances in arterial tortuosity and FMD was explored in a histopathological study of 139 symptomatic patients with carotid elongations that underwent surgical correction. 15 They found histologic abnormalities in all patients, classifying these pathologic disturbances into: (1) non-specific degeneration in the tunica media (43.9%); (2) tunica media hyperplasia with increased extracellular mucoid Alcian-Pas-positive matrix surrounding variously oriented smooth muscle cells (38.9%); and (3) typical patterns of fibromuscular dysplasia (17.2%). Combining the latter two categories into ‘atypical’ and ‘typical’ forms, the authors found evidence of fibromuscular dysplasia involving the tunica media in over 50% of patients that underwent surgery for carotid artery elongation. 15
Clinically, ICA redundancy has been associated with an increased incidence of cerebrovascular symptoms,15,36 particularly spontaneous carotid artery dissection. 37 In a small study of 13 dissection patients identified over 6 years, Barbour et al. found that imaging findings of carotid redundancy were associated with spontaneous carotid artery dissection in 8 of 13 (62%) patients, and 13 of 20 vessels (65%) with dissection, but the same findings existed in only 20 of 108 (19%) age-matched stroke patients without dissection. Similarly, fibromuscular dysplasia has been implicated to be a predisposing factor for spontaneous carotid artery dissection.1–3,38,39 This was recently confirmed in the first publication of the United States Registry for Fibromuscular Dysplasia, which found a prevalence of nearly 20% for arterial dissection in FMD patients. 4 Although a recent case–control study did not support the association between ICA redundancy and dissection, 40 the finding that both FMD and ICA redundancy have a positive association with spontaneous carotid artery dissection creates the plausible hypothesis that ICA tortuosity is a manifestation of FMD and therefore accounts for the association of FMD with carotid artery dissection.
The associations between FMD and the S curve, FMD and carotid artery dissection, and carotid artery redundancy and dissection create a possible theory for carotid artery dissection and the S curve both being related to abnormalities in the blood vessel wall that may occur in FMD. Aneurysms and/or dissections occur in approximately 20% of individuals with FMD. 4 The S curve occurs in approximately 32% of FMD patients. The presence of aneurysm, dissection, or S curve may merely represent different phenotypic expressions of FMD. However, it should be emphasized that while the S curve occurs with increased frequency in FMD patients, it is not specific for FMD. Given the overlap between these clinical syndromes, FMD, and multiple arteriopathies, further research is warranted to identify and clarify these complex interrelationships. Reports of general arterial tortuosity increasing with age or varying with sex are conflicted.18–21,24 However, in this population the S curve occurred more frequently in older patients, similar to those reports that indicated increasing arterial tortuosity with age.19,24
It is interesting to note that the presence of an S curve in FMD patients was not dependent on the arterial bed affected by FMD. Furthermore, S curves were found just as frequently in patients with typical FMD manifestations in the renal artery or the carotid artery. This confirms prior reports finding histopathological evidence of FMD in patients with arterial tortuosity.15,29 Similarly, a series of surgically corrected carotid arteries in FMD patients found a high prevalence of tortuosity. 41 This finding provides preliminary evidence that the S curve may be a marker of underlying FMD and should be investigated when encountered in routine clinical practice.
Limitations of the current study
This was a single-site study, but our database of FMD patients is relatively large. Owing to the technical limitations of our electronic storage of images and electronic medical records, carotid artery ultrasound reviewers were not blinded to FMD status. While genetic testing for Loeys-Deitz syndrome or Ehlers-Danlos syndrome type IV was performed in those FMD patients with aneurysms or dissections, there is no available data on the exact percentages of those tested. It should also be noted that the control patients had a carotid artery ultrasound performed for varying clinical indications, which may have led to overestimation of the prevalence of the S curve in these groups. The most common reasons for testing in the age and sex-matched control group were one of the following: carotid bruit, syncope or TIA. In the age ≥70 control group, the most frequent reasons were known carotid disease, bruit, TIA/stroke or prior to coronary artery bypass graft (CABG). We do not know with certainty that the two age and sex-matched control patients with the S curve do not in fact have asymptomatic FMD. The prevalence of S curves in a de novo healthy population is unknown. The clinical relevance of the S curve is not completely understood; however, some reports have suggested an increased cerebrovascular event rate, particularly carotid dissection.15,36,42,43 While our cross-sectional study is not positioned to answer this question, our analysis forms the basis for a planned prospective study that evaluates the impact of the S curve on relevant clinical endpoints.
Conclusion
The S curve is a distinct morphological pattern visualized on duplex ultrasonography in the mid-distal ICA. There was a significantly higher prevalence of the S curve in FMD patients as compared to both age-matched and age ≥70-matched control groups. While the S curve may not be specific to FMD, its presence in individuals <70 years old should alert the clinician to the possibility that FMD is present.
Footnotes
Declaration of conflicting interest
Dr Olin is chair of the medical advisory board (volunteer position) of the Fibromuscular Dysplasia Society of America. He receives no compensation for this position.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
