Abstract
The purpose of this study was to determine whether ischemic postconditioning (IPC) could improve peripheral endothelial function in patients with acute ST-segment elevation myocardial infarction (STEMI) undergoing primary percutaneous coronary intervention (PCI). Of 102 patients randomly assigned to an IPC or standard protocol to study infarct size utilizing cardiovascular magnetic resonance imaging, 84 patients had peripheral endothelial function assessed with brachial ultrasound measures and peripheral arterial tonometry (PAT) during reactive hyperemia 3 days after PCI. Overall IPC was not associated with a smaller infarct size compared to controls, though there was a trend towards greater myocardial salvage with IPC. Patients randomized to IPC (n=43; age 56 ± 11 years; 85% male) and standard protocol (n=41; age 56 ± 10 years; 88% male) underwent endothelial function assessment. Flow mediated vasodilatation was not significantly greater in the IPC group than in the standard group (7.4 ± 4.9% versus 6.6 ± 4.0% respectively, p=0.40) nor was peak hyperemic velocity-time integral (78 ± 26 cm versus 71 ± 30 cm respectively, p=0.28). Similarly, the PAT hyperemic ratio was not significantly greater in the IPC group than in the standard group (2.0 ± 0.9 versus 1.8 ± 0.6 respectively, p=0.14). In conclusion, IPC did not improve early peripheral endothelial function in patients with STEMI undergoing primary PCI.
Keywords
Introduction
The endothelium plays a vital role in the regulation of vascular tone and protection from atherosclerosis. 1 The acute increases in oxidative stress and pro-inflammatory markers following myocardial infarction have been shown to have detrimental effects on peripheral endothelial function.2–4 Coronary endothelial dysfunction is related to peripheral vasodilatory dysfunction, allowing peripheral endothelial function to be a surrogate measure of coronary endothelial function.5,6
Ischemic postconditioning (IPC), a sequence of brief repeated episodes of ischemia-reperfusion applied before allowing permanent reperfusion, has demonstrated marked cardioprotective effects in animal models of myocardial ischemia. 7 It is recognized that IPC improves the coronary endothelial dysfunction that accompanies reperfusion injury in animals.8,9 However, there are only a limited number of studies that have evaluated the effect of IPC on measures of vascular function in humans.
We have previously published data on the influence of IPC on myocardial salvage and infarct size in patients undergoing primary percutaneous coronary intervention (PCI) for STEMI 10 and hypothesized that those patients undergoing IPC would have improved endothelial function assessed utilizing the well established non-invasive tools of brachial artery ultrasound 11 and finger arterial pulse wave amplitude. 12
Methods
This study was performed according to the Declaration of Helsinki and Canadian Guidelines of Good Clinical Practice. The Conjoint Ethics Committee of the University of Calgary approved the protocol (E20039). The study was registered with the Clinical Trials Protocol registration system (clinicaltrials.gov – NCT00334373). All patients gave written informed consent before enrolment.
Study design and post conditioning protocol
Patients ≥ 18 years of age presenting within 6 hours of the onset of chest pain with ST-segment elevation undergoing primary PCI were considered for inclusion. Details of the inclusion and exclusion criteria have been published previously. 10 This was conducted as a single centre, prospective, randomized trial with blinded evaluation of all endpoints. All patients received standard doses of antiplatelet and anticoagulation agents. After establishing reflow with guidewire passage or balloon inflation patients were randomized to standard PCI alone or IPC protocol utilizing a computerized 1:1 randomization 4 block sequence placed in a numbered sealed envelope. IPC was performed by 4 cycles of 30-second low pressure balloon inflations (4-6 atm) within the previously occluded segment each separated by 30 seconds of reflow. PCI was then performed according to the preferences of experienced operators.
Brachial artery ultrasound measurements
Endothelial function was assessed 3 days following primary PCI after all vasoactive medicines were stopped for 24 hours. Subjects underwent assessment of brachial artery flow-mediated vasodilation as previously published by our group.13–15 Studies were performed in a quiet clinical laboratory with the temperature maintained at 21-23 °C. Subjects rested for at least 10 minutes before the first scan and remained supine throughout the study. Baseline longitudinal 2-dimensional images of the right brachial artery were acquired with a 7.5 MHz linear phase-arrayed ultrasound transducer attached to a Hewlett-Packard 5500 ultrasound machine. Pulse-wave Doppler was used to record brachial artery velocity. A blood pressure cuff was placed on the right lower arm with a 5-minute occlusion time. On cuff release, hyperemic velocity was recorded for 30 seconds, and 2-dimensional imaging was resumed for up to 2 minutes after cuff release.
Analysis of the brachial artery images was performed in our core laboratory using automated edge detection software (DEA, Montreal PQ). During the procedure, 2 images were digitized during baseline from 5 seconds to 60 seconds. During reactive hyperemia, 2 arterial diameters were sampled starting 60 seconds after cuff release, and repeated every 10 seconds, ending at the 90 second mark, for a total of 8 images. Any poor quality images were discarded. The maximum dilation occurring between 60 and 90 seconds was determined.
Flow-mediated vasodilatation (FMD) was calculated from the diameters as (reactive hyperemia – baseline)/baseline × 100%. The intra- and inter-observer variability in our laboratory is 1% for the measurement of baseline brachial artery diameter. Brachial artery flow was calculated as the product of velocity and cross-sectional arterial area. Hyperemic velocity was determined as the peak velocity-time integral (VTI) of the first complete velocity envelope obtained after cuff release. Higher values represent better microvascular dilation. The co-efficient of variation for intra-observer variability for the measurement of VTI is 2%. Flow velocity (V) was converted to local shear stress (SS) using the following equation: SS = 8 × μ × V/D, where D is brachial artery diameter and μ was viscosity of blood, which was assumed to be 0.035 dyne.s.cm−2. The hyperemic ratio was determined for volumetric flow, VTI and SS compared to the respective baseline values.
Pulse wave amplitude measurements
Finger pulse wave amplitude (PWA) was recorded with peripheral arterial tonometry (PAT) using a plethysmographic device (Itamar-Medical Inc., Caesaria, Israel). 12 PAT testing was performed simultaneously with brachial ultrasound measures. One PAT finger probe was placed on the right index finger that would undergo hyperemia testing. Another probe was placed on the contralateral index finger. The PAT hyperemia ratio was defined as the ratio of the average PWA during the 1-minute period beginning 1-minute after reactive hyperemia compared with the average PWA during a 3.5-minute pre-occlusion baseline period. The PWA signal was fed to a personal computer, band-filtered (0.3 to 30 Hz), amplified, displayed, and analyzed with automated software that generates a PAT ratio and normalizes to the control arm to compensate for potential systemic changes.
Statistical analysis
Continuous variables were reported as mean ± SD. Categorical data were reported as frequencies and percentages. Differences between groups were assessed by the chi-square test for categorical variables and by the non-paired Student’s t test for continuous data with normal distribution. Otherwise, the non-parametric Wilcoxon rank-sum test was used. All analysis was conducted using SPSS for Macintosh, version 19.0 (SPSS, Inc., Chicago, Illinois). A two-tailed p value of less than 0.05 was considered statistically significant.
Results
Study population
Of the 102 patients randomized in our original myocardial IPC study, 84 patients consented to participate in the endothelial function sub-study with 43 patients in the IPC group (age 56 ± 11 years, 85% male) and 41 patients in the standard group (age 56 ± 10 years, 88% male). The two groups were well balanced with regard to baseline demographic and angiographic characteristics (Table 1). A high proportion of patients had dyslipidemia, systemic hypertension, and were active smokers, but were balanced across both groups (Table 1). There was very high utilization of standard medical therapy prescribed post myocardial infarction (Table 2).
Baseline characteristics.
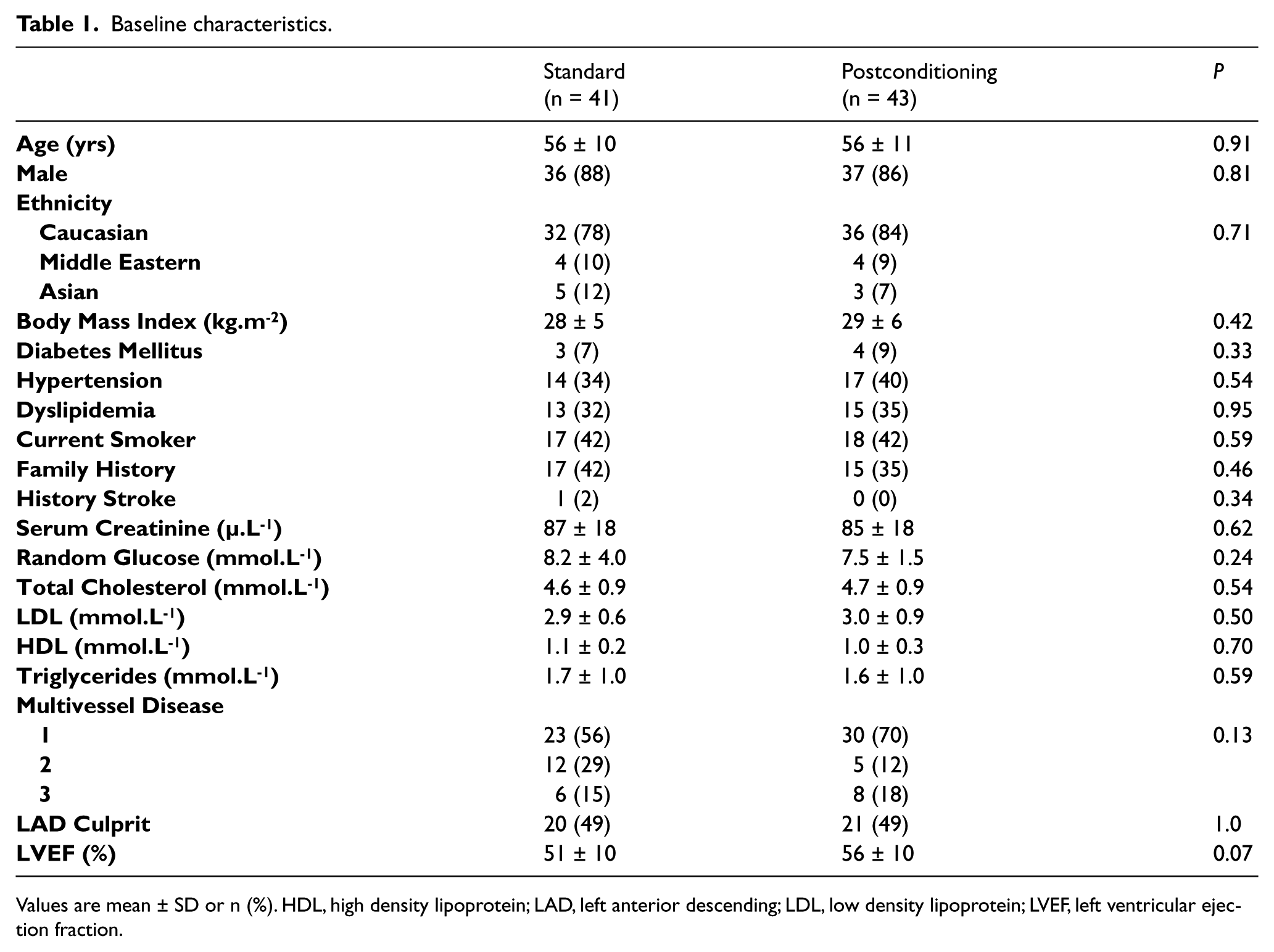
Values are mean ± SD or n (%). HDL, high density lipoprotein; LAD, left anterior descending; LDL, low density lipoprotein; LVEF, left ventricular ejection fraction.
Medical therapy utilized.
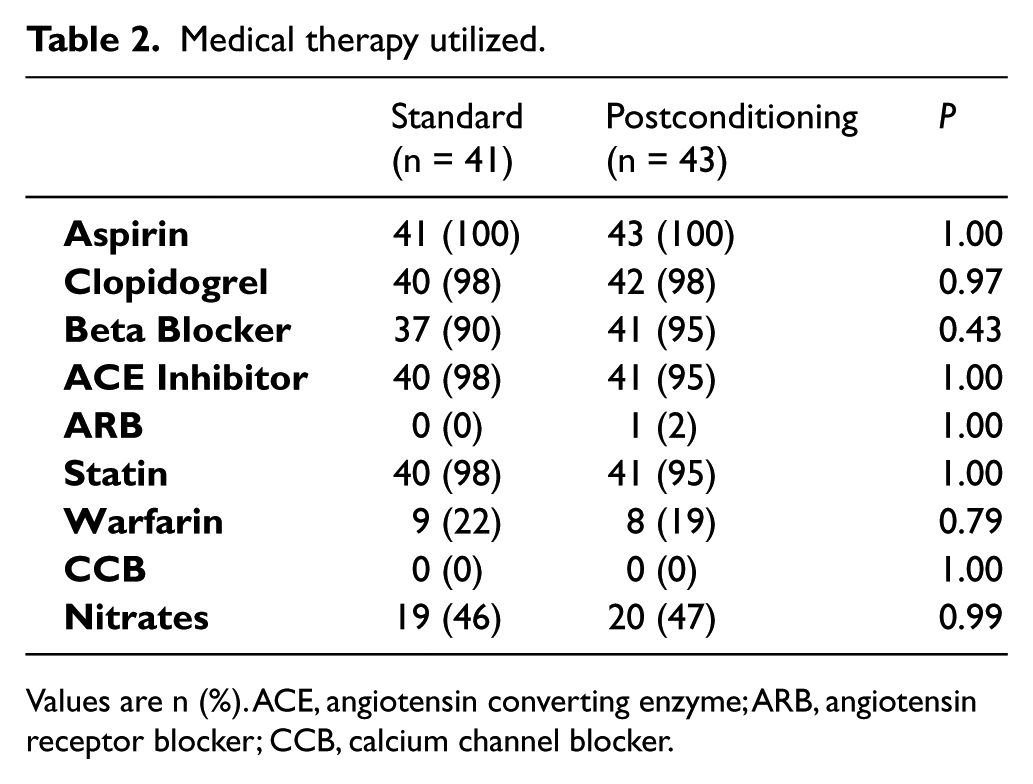
Values are n (%). ACE, angiotensin converting enzyme; ARB, angiotensin receptor blocker; CCB, calcium channel blocker.
Endothelial function assessments
Endothelial function assessment was performed 3.5 ± 1.3 days after PCI in the IPC group and 3.3 ± 1.1 days after PCI in the standard group (p = 0.40). The baseline and hyperemic brachial ultrasound measures are shown in Table 3. Resting brachial artery diameter was marginally but significantly smaller in the IPC group than the standard group (4.4 ± 0.8 mm versus 4.8 ± 0.6 mm respectively, p=0.02) and there was a trend towards lower resting brachial flow in the IPC group (145 ± 53 ml.min−1 versus 183 ± 113 ml.min−1 respectively, p=0.06).
Peripheral endothelial function parameters post PCI.
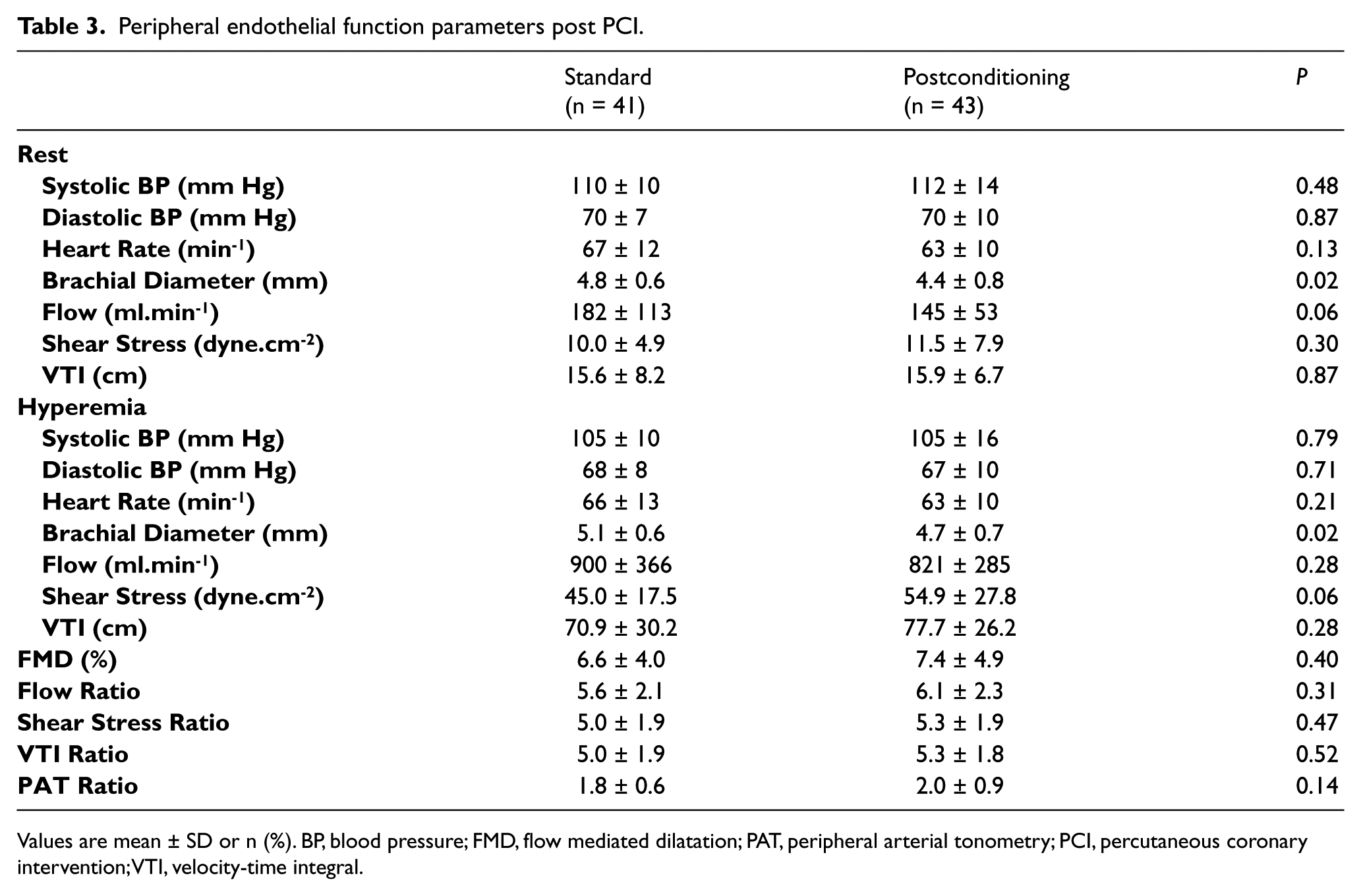
Values are mean ± SD or n (%). BP, blood pressure; FMD, flow mediated dilatation; PAT, peripheral arterial tonometry; PCI, percutaneous coronary intervention; VTI, velocity-time integral.
The mean flow-mediated dilatation (FMD) in the IPC group was 7.4 ± 4.9 % as compared with 6.6 ± 4.0 % in the standard group, for a difference of 0.8% (95% confidence interval [CI], -1.1 to 2.8%, p = 0.40) (Figure 1). There was no difference in the absolute brachial artery diameter change with hyperemia between the IPC group (0.30 ± 0.18 mm) and standard groups (0.29 ± 0.18 mm), for a mean difference of 0.01 mm (95% CI, -0.07 to 0.08 mm, p=0.88).
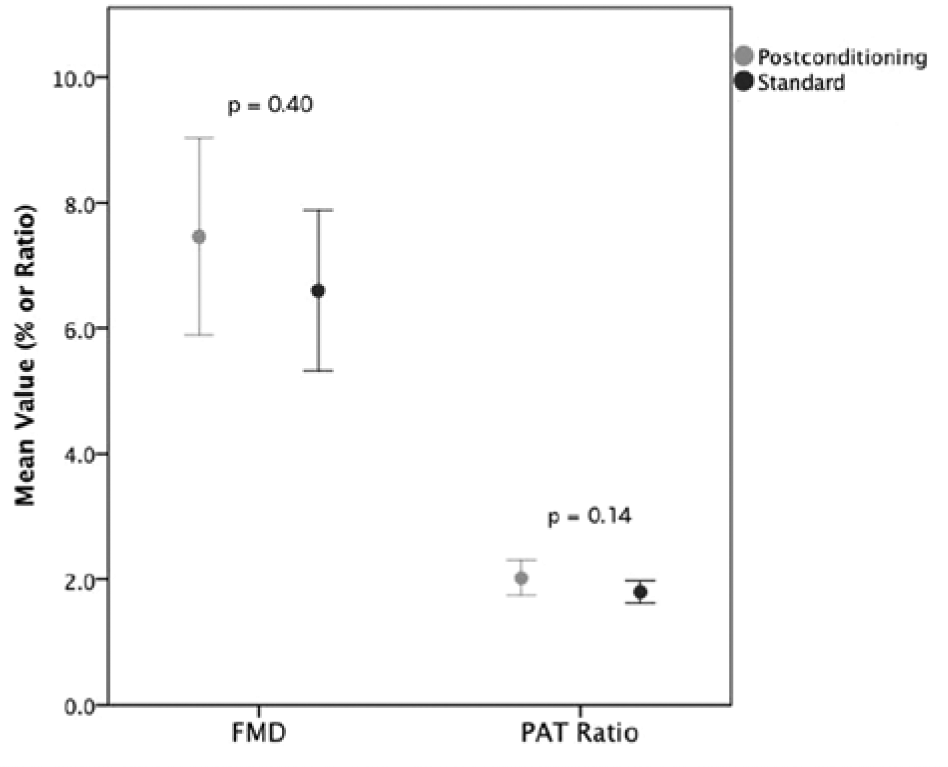
Average flow mediated dilatation and PAT ratio for the postconditioned and standard groups.
On multivariate regression analysis there was no significant interaction between the FMD response to IPC and age, gender, diabetes, hypertension, dyslipidemia, smoking, or family history of premature coronary artery disease.
There was no difference in peak hyperemic VTI between the IPC and standard groups (77.7 ± 26.2 cm versus 70.9 ± 30.2 cm respectively, p=0.28) (Figure 2). Similarly, the hyperemic flow ratio, shear stress ratio, and VTI ratio were not greater in the IPC group than in the standard group (Table 2). Furthermore, the hyperemic PAT ratio was not statistically greater in the IPC group than in the standard group (2.0 ± 0.9 versus 1.8 ± 0.6 respectively, p=0.14) (Figure 1).
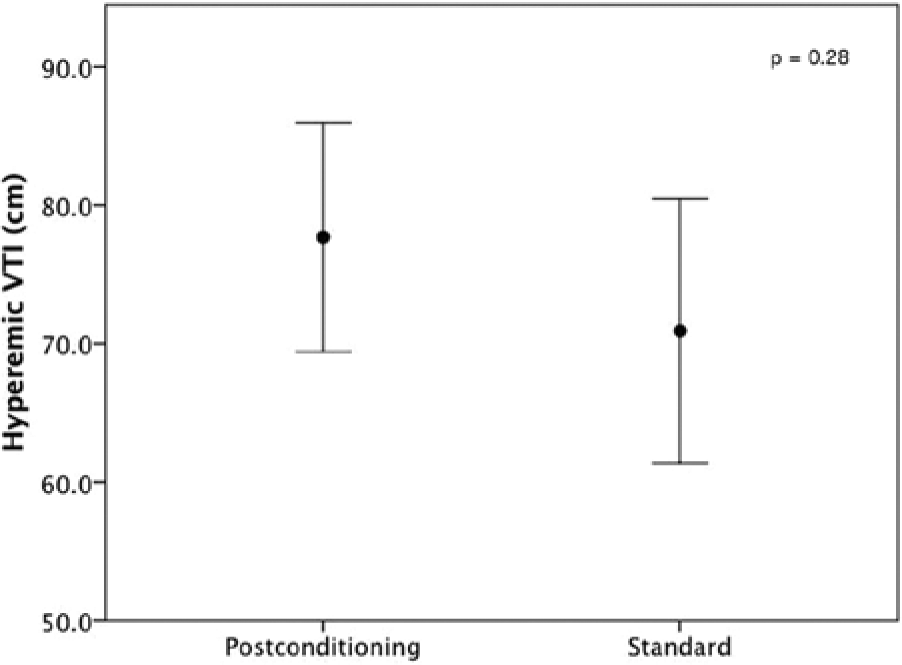
Average hyperemic velocity time integral (VTI) for the postconditioned and standard groups.
Discussion
The present study demonstrates that myocardial IPC does not acutely improve peripheral endothelial function in patients undergoing PCI for STEMI. Several studies have explored the influence of IPC on endothelial function in human models of forearm ischemia-reperfusion (IR) injury with mixed results.16–18 In 11 healthy volunteers brachial FMD was measured before and after IR (20 minutes of arm ischemia followed by 20 minutes of reperfusion). 16 IR caused endothelial dysfunction (FMD 9.1 ± 1.2% pre-IR, 3.6 ± 0.7% post-IR, p < 0.001), which was prevented by three 10-second cycles (FMD 9.9 ± 1.7% pre-IR, 8.3 ± 1.4% post-IR, p = NS) or three 30-second cycles of post-conditioning (FMD 10.8 ± 1.7% pre-IR, 9.5 ± 1.5% post-IR, p = NS). No protection was observed when the 10-second post-conditioning stimulus was delayed for 1 minute after the onset of reperfusion. These findings were confirmed in 9 healthy volunteers. 17
However, in a small cross-over study of 10 healthy male volunteers post-conditioning did not limit the impaired FMD of the radial artery induced by 15 minutes of forearm ischemia. 18 The post-conditioning protocol used was 3 cycles of 10 seconds of reperfusion separated by 20 seconds of brachial re-occlusion.
Only one previous study has specifically explored the influence of myocardial IPC on endothelial function in STEMI patients. 19 In this single-center randomized study of 92 Chinese patients, the depressed brachial FMD was improved in those who had undergone coronary IPC. The IPC protocol was three cycles of 30-second reperfusion followed by 30-seconds of re-occlusion and FMD was assessed 1 day after PCI. The FMD in the standard, IPC, and healthy control group was 4.3 ± 3.7%, 9.8 ± 5.5% and 14.5 ± 3.4% respectively, p<0.05. These results are in contrast to our own and may reflect differences in endothelial function in our predominantly Caucasian population, the possible lower utilization of intensive medical therapy in the Chinese population, or that the endothelial dysfunction present 1 day after PCI has resolved at 3 days. No published studies could be identified that explored the influence of IPC on PAT.
An improvement in endothelial function may not have been detected in our study, in contrast to those conducted in healthy volunteers, because of the coexistence of comorbidities such as age, diabetes, hypertension, and dyslipidemia that already impair endothelial function; more substantial endothelial injury as present during STEMI; and active treatments that promote vascular health. The presence of dyslipidemia and atherosclerosis reduce nitric oxide (NO) production 20 and diabetes is known to impair endothelial function 21 and attenuate the demonstrated reduction in cardiovascular event rates with remote preconditioning prior to PCI. 22 Our patients’ medical therapy could influence vascular function. Statins may improve endothelial function via enhancement of endothelial nitric oxide synthase (eNOS) expression and activity. 23 Angiotensin converting enzyme inhibitors increase bradykinin concentrations, which improve vascular function through nitric oxide (NO) and prostaglandin (PGI2) synthesis. 24 The high adherence to intensive medical therapy in our study population may have lead to higher FMD values that were less likely to be influenced by IPC. Indeed, our FMD values in the standard group were 2.3% higher than in the Chinese control group. 19
It is apparent that the endothelium is a large organ in communication between diverse vascular beds, perhaps via humoral and autonomic nervous system pathways.25,26 This is highlighted by observations that cycles of brief ischemia and reperfusion to a remote organ may confer myocardial protection.27,28 Recent studies have shown that cycles of forearm ischaemia-reperfusion administered in STEMI patients prior to receiving primary PCI improved myocardial salvage index 29 and reduced infarct size. 30 In contrast remote IPC with arm ischemia did not protect against procedural troponin rise in elective PCI patients, 31 suggesting that there is a threshold volume of ischemia to be realized in the target organ before remote IPC confers benefit.
Our original published data, from which the present cohort derived, did not demonstrate a reduction in myocardial infarct size with IPC but did show a trend towards increased myocardial salvage index. 10 Furthermore, there was no improvement in peripheral endothelial function as assessed by FMD and PAT. Despite enthusiasm for IPC based on animal models and early surrogates of human infarct size, recent human studies utilizing cardiovascular magnetic resonance imaging have shown modest reductions,32,33 no impact,10,34 or potential increase35,36 in infarct size with IPC protocols. The variability in myocardial response to IPC is likely influenced by the IPC protocols used, the method of infarct size assessment, as well as the presence of clinical confounders.
Whilst remote IPC may influence myocardial responses to ischemia, we have shown that the converse is not seen and unlikely to occur in the absence of significant cardioprotection imparted by IPC. Even if myocardial IPC improved coronary endothelial function it is possible that the benefit generated in this small vascular territory is marginal and unable to be detected peripherally utilizing brachial ultrasound and PAT. A benefit may have been detected had we performed invasive quantification of coronary endothelial function, which is considered the gold standard technique, or utilized other more comprehensive measures of endothelial function that include platelet function or adhesion molecules.
Our study was not adequately powered to detect a small difference in FMD. To assist with future trial planning, we have calculated that the minimum total sample size required to detect the observed 0.84% as well as a 1% absolute difference in FMD with 80% power would be 437 and 316 patients respectively.
The lower resting brachial artery diameter in the IPC patients is likely due to chance as there is no plausible explanatory hypothesis. Further, this would serve to bias an FMD improvement in the IPC group, which was not observed.
Our present cohort, despite a recent large myocardial infarction, had values of FMD similar to those reported in patients with coronary artery disease13,14 and a PAT ratio similar to patients with multiple cardiovascular risk factors. 12 The presumed attenuation of endothelial dysfunction post infarction highlights the importance of adherence to current standard medical therapy.
Conclusion
Myocardial ischemic post-conditioning did not lead to significant improvements in endothelial function assessed by brachial ultrasound and peripheral arterial tonometry 3 days after primary PCI.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The Heart and Stroke Foundation of Canada supported this work.
