Abstract
The genotype distribution of two gene polymorphisms, previously associated with peripheral artery disease (PAD), has been evaluated in a population of diabetic (DPAD) and non-diabetic (NDPAD) patients affected by symptomatic PAD (stages II–IV). A decreased frequency of the AA genotype of rs5498 (ICAM-1) was observed in the PAD subjects compared to controls but this result did not reach statistical significance (p=0.06 by chi-squared test). On the contrary, a significant increase in the frequency of the GG homozygous genotype of rs248793 (SRD5A1) was observed in the PAD patient group in comparison to controls (p=0.01). These data confirm that the GG genotype of rs248793 in the SRD5A1 gene is significantly associated with symptomatic PAD and show a trend towards a stronger association with the non-diabetic status.
Introduction
Peripheral artery disease (PAD) is the collective denomination of a group of disorders characterized by a reduction of blood flow to the limbs caused, in the vast majority of patients, by atherosclerosis of the corresponding arterial territory.
Several risk factors for PAD are the same as those identified for generalized atherosclerosis – smoking and diabetes mellitus are considered among the stronger ones.1–4 Indeed, diabetic patients show not only an earlier onset and a more rapid progression of PAD than non-diabetics, but also a different anatomical distribution of vascular pathology, with a greater severity of arterial disease in the profunda femoris and in all arterial segments below the knee. 5
PAD is considered as a polygenic and multifactorial disease, and both single-gene and genome-wide association studies have been performed in the attempt to identify susceptibility genes. Most of the gene polymorphisms associated with PAD exert proatherosclerotic and proatherothrombotic effects, although a few genes not directly linked to the atherosclerotic process have been also identified in association studies.6–9
A recent meta-analysis of genome-wide association study data from 21 population-based cohorts (more than 40,000 individuals, including 3409 participants with PAD) revealed only one genome wide significant association on chromosome 9p21 with the ankle–brachial index (ABI). 10 In another two-stage meta-analysis of ∼50,000 single nucleotide polymorphisms (SNPs) across ∼2100 cardiovascular candidate genes, in 21,547 individuals of European ancestry (1190 with ABI <0.9) from eight cohorts, none of the SNP associations for PAD were significant. 11 In both these large meta-analyses, the diagnosis of PAD was based on the detection of an ABI value below an established cut-off (<0.9), an easy diagnostic test that includes in a single category both symptomatic and asymptomatic PAD.12,13 Indeed, the prevalence of PAD, identified by an ABI <0.9, ranged from 4% to 12% in the eight cohorts examined by Wassel et al., 11 while the prevalence of patients with intermittent claudication was only 0.8–3.7%.
However, it is likely that genetic and environmental factors that are responsible for the onset of a subclinical form of atherosclerosis, such as asymptomatic PAD, do not fully overlap with genetic factors that determine the progression of the disease to a symptomatic stage. Indeed, previous studies evaluating symptomatic PAD have reported a significant association with genes involved in inflammation, coagulation, blood pressure regulation and lipid metabolism. Such studies are often limited by small sample size and lack of robust independent replication8,14–19and it is well known that genetic case–control association studies are affected by a large number of false-positive results due to several causes, among which population stratification is considered common. 20
With this in mind, in the present study, we re-evaluated single nucleotide polymorphisms in intercellular adhesion molecule-1 (ICAM-1) and steroid 5-α-reductase type I (SRD5A1) genes, previously associated with PAD,14,16,18 by analyzing their genotype distribution in a population of diabetic (DPAD) and non-diabetic (NDPAD) patients with symptomatic PAD (stages II–IV).
Materials and methods
Patients and diagnostic criteria
In this study we analyzed 218 caucasian symptomatic PAD patients (males 172, females 46; mean age of 69.5±10.7 years). Diagnosis was based on the presence of pain in the lower limbs due to walking (intermittent claudication), or previous diagnosis of PAD performed by angiography, or the use of vasodilating drugs (pentoxifilline, buflomedil chlorhydrate, anti-platelets drugs) for at least 6 months and a history of intermittent claudication. Finally, diagnosis was confirmed by the detection of an ABI ≤0.9. Only PAD patients without a clinical history of acute myocardial infarction or stroke were enrolled. A total of 130 diabetic patients were present in the PAD group (43 type 1 and 87 type 2).
The clinical stage of the enrolled patients was determined by the physicians of the Medical Angiology Unit. The symptomatic patients were considered in the second A stage if the referred free walking distance (FWD) was >250 m or in the second B stage if the FWD was <250 m. Patients suffering from pain at rest were considered in the third stage of Leriche’s classification and patients with severe ischemic clinic evidence (cyanosis, continuous pain and/or skin damage) were considered in the fourth stage.
A group of 239 subjects in apparent good health with no clinical history or laboratory findings of cardiovascular risk factors, arterial hypertension, diabetes or dyslipidemia (males 121, females 118; mean age of 54±12 years) were considered as a control group.
Our study was approved by the ethics committee of the ‘azienda ospedaliera di rilievo nazionale e di alta specializzazione Garibaldi’ (Catania, Italy). All procedures were conducted in accordance with the principles outlined in the Declaration of Helsinki.
DNA extraction
Genomic DNA (gDNA) was extracted from blood cells using the QIAamp DNA Blood Mini Kit (Qiagen, Milan, Italy) according to the manufacturer’s instructions. The concentration and the quality of the DNA was determined using a ND-1000 spectrophotometer (NanoDrop; Thermo Scientific, Waltham, MA, USA) as previously described.
SRD5A1, ICAM-1 genotype analysis
The SRD5A1 polymorphism (G/C, rs248793) was analyzed as previously described. 16
The ICAM-1 polymorphism (A/G, rs5498) was revealed by PCR amplification using the primer ICAM-1 1554 F5’ ATTCCCAGCAGACTCCAATG 3’and the ICAM-1 2017 R5’ AGGTGTAGCTGCATGGCATA3’and BstUI digestion (CG/CG; New England BioLabs, Ipswich, MA, USA). DNA (200 ng) was amplified in a reaction volume of 50 µl containing 50 pmoles of primers, PCR buffer 10×, 10 µM dNTP, MgCl2 50 mM, and 5 units of Taq polymerase (Invitrogen, Monza, Milan, Italy). Reactions were carried out at 95°C for 30 sec, followed by 35 cycles at 95°C for 1 min, 61°C for 1 min and 72°C for 1 min. The PCR product was digested using 2 units of BstUI at 60°C for 120 min. Digested products were analyzed using 1.6% agarose gel electrophoresis and ethidium bromide. When both alleles were lacking in the BstUI site only one band was detected (526 bp, lane AA), while two bands were detected in the presence of the BstUI site on both alleles (366 bp and 160 bp, lane AG). Three bands were revealed in the case of a heterozygous condition (526 bp, 366 bp, 160 bp).
A TaqMan Allelic Discrimination assay was used in order to confirm the genotyping results obtained by restriction analysis for the ICAM-1 polymorphism. Probes and primers for ICAM-1 (A/G, rs5498) polymorphism were chosen using the SNPbrowser™ software (www.allsnps.com/snpbrowser). The reaction was carried out according to manufacturer’s instructions (cod 4371355; Applied Biosystems, CA, USA). Each DNA sample was analyzed in triplicate.
Statistical analysis
The association between genotypes or alleles and disease and agreement of genotype distribution with the Hardy–Weinberg principle were evaluated by the Pearson’s chi-squared test. Statistical power calculation was performed by the software implemented by Skol et al. 21 under a multiplicative genetic model, assuming a 5% PAD prevalence 22 and a criterion for significance (alpha) at 0.05. On the basis of data from Flex A, et al. 14 for ICAM-1 rs5498 (ICAM-1 rs5498: risk allele G frequency 0.4 and GG genotype relative risk 3.7), a statistical power of 1.0 was calculated for a number of cases and controls comparable to that genotyped in the present study.
Logistic regression models were implemented according to Hosmer and Lemeshow. 23
Results
The distribution of PAD patients according to Fontaine-Leriche’s stages is reported in Table 1A. The average age, sex distribution and % of subjects showing vascular risk factors in the non-diabetic (NDPAD) and diabetic PAD (DPAD) subgroups are reported in Table 1B. The anatomical distribution of the vascular pathology, as assessed by angiography, is reported in Table 1C (% of patients showing abnormalities in each vascular region). As expected, the diabetic subgroup show a higher percentage of patients affected in the tibial artery segments.
(A) Distribution of PAD patients according to Fontaine-Leriche’s stages and ABI data. (B) Demographic and clinical data of the non-diabetic and diabetic PAD subgroups. (C) Distribution of pathologic peripheral arteries in 76 NDPAD and 110 DPAD patients; percentage of patients showing abnormalities in the specified arterial segment in each subgroup.
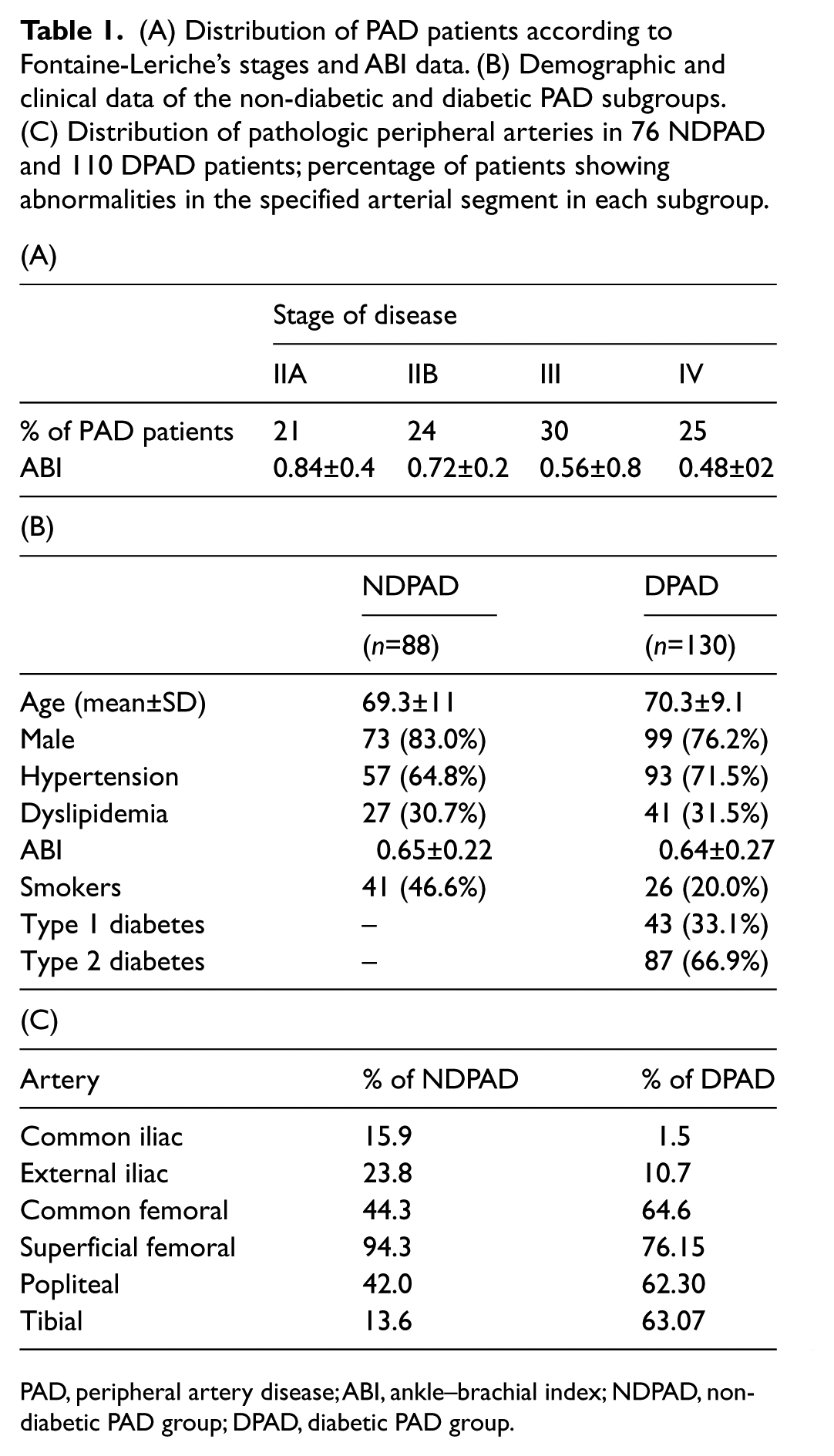
PAD, peripheral artery disease; ABI, ankle–brachial index; NDPAD, non-diabetic PAD group; DPAD, diabetic PAD group.
The genotypes of the following SNPs were determined in 239 healthy controls and 218 symptomatic PAD patients: (1) rs5498 (A/G, K469E) is a polymorphism localized in exon 6 of the ICAM-1 gene that determines a single amino acid substitution of lysine (K) in glutamic acid (E) at position 469 of the ICAM protein and (2) rs248793 (G/C), a synonymous substitution localized in exon 1 of the SRD5A1 gene. Genotype distributions in the control group were in agreement with the Hardy–Weinberg equilibrium (Tables 2 and 3).
(A) Chi-squared analysis of the frequency of alleles and genotypes for ICAM-1 gene polymorphism rs5498 (A/G). (B) Chi-squared analysis of the frequency of alleles and genotypes of ICAM-1 gene polymorphism rs5498 (A/G) in PAD cases without or with type 1 and type 2 diabetes (NDPAD: n=88; DPAD: n=130) and controls (n=239).
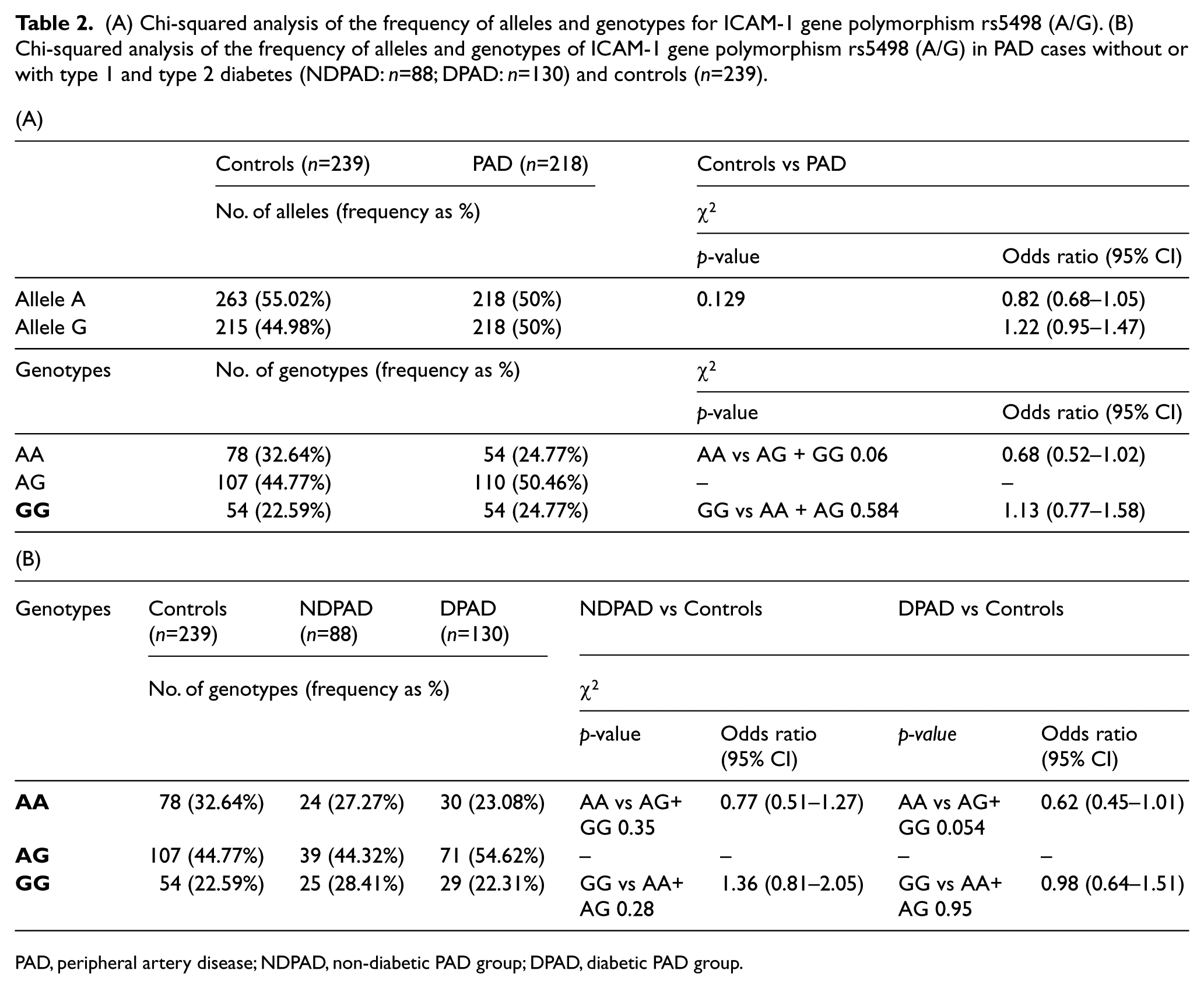
PAD, peripheral artery disease; NDPAD, non-diabetic PAD group; DPAD, diabetic PAD group.
(A) Chi-squared analysis of the frequency of alleles and genotypes of SRD5A1 gene polymorphism rs248793 (G/C). (B) Chi-squared statistical analysis of the frequency of alleles and genotypes of SRD5A1 gene polymorphism rs248793 (C/G, allele A/B) in PAD cases without or with type 1 and type 2 diabetes (NDPAD: n=88 and DPAD: n=130) and controls (n=239).
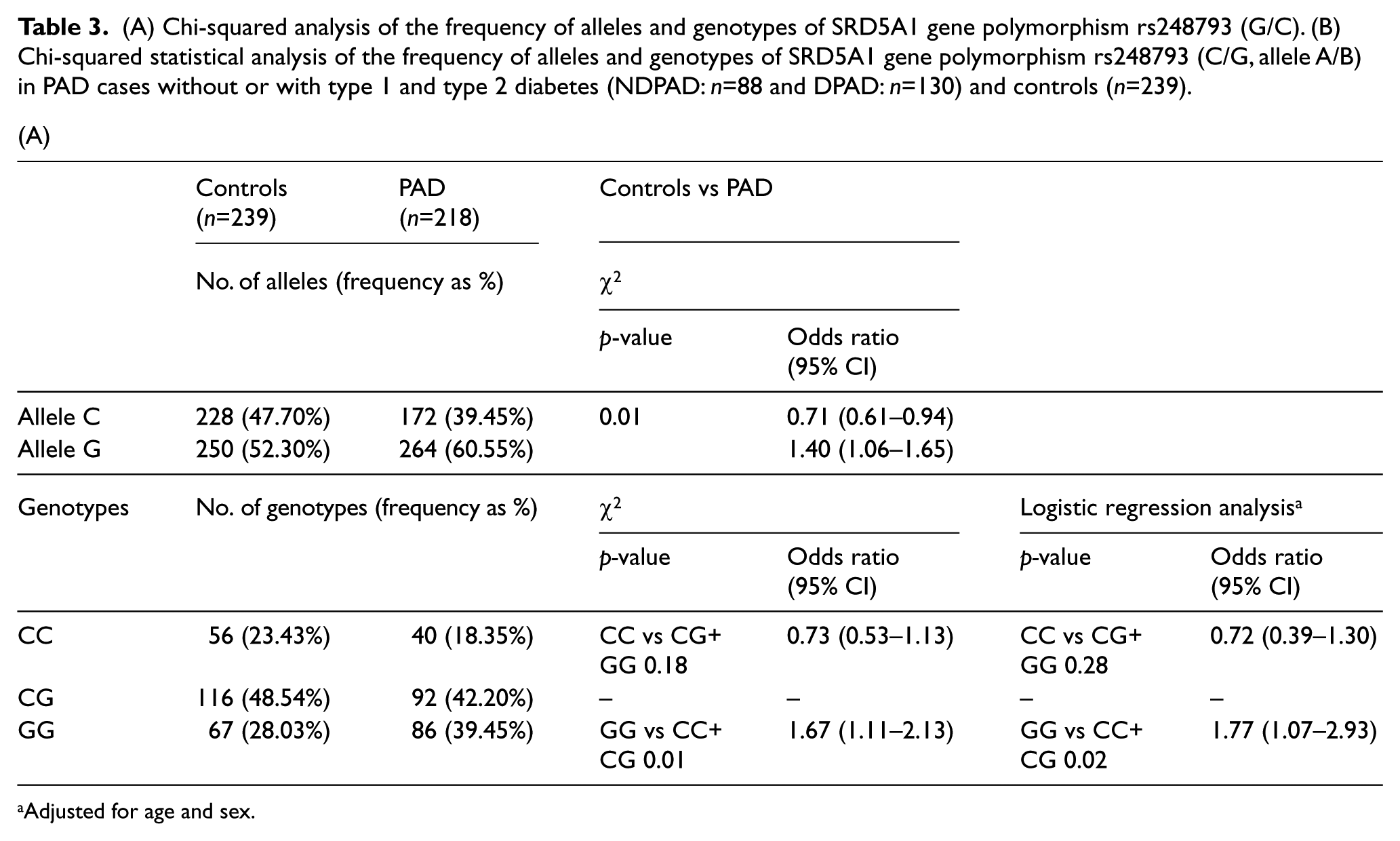
Adjusted for age and sex.
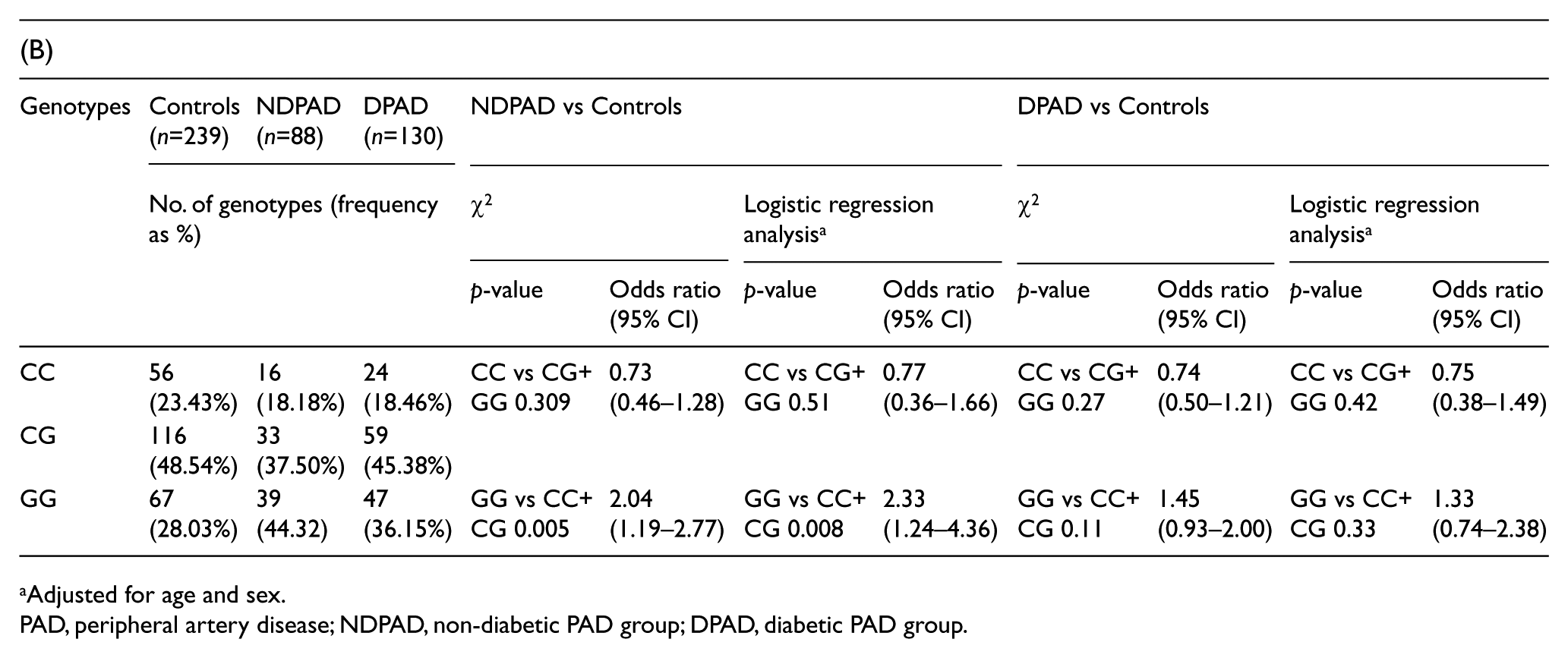
Adjusted for age and sex.
PAD, peripheral artery disease; NDPAD, non-diabetic PAD group; DPAD, diabetic PAD group.
Single nucleotide polymorphism rs5498 (A/G, K469E) of the ICAM-1 gene
A decreased frequency of the AA genotype of rs5498 (ICAM-1) was observed in the PAD subjects compared to controls but these results were not statistically significant (p=0.06 by chi-squared test, Table 2A). No differences were observed even after age and sex adjustment by a logistic regression model.
After stratification of PAD patients according to diabetic status, the decreased frequency of the AA genotype in DPAD patients in comparison to controls did not reach a significant value (p=0.054, Table 2B).
The lack of association with diabetic status was also confirmed by a logistic regression model that directly compared NDPAD versus DPAD (case-only study) 24 after adjustment for age, sex, smoking habit, dyslipidemia and hypertension.
Single nucleotide polymorphism rs248793 (G/C) of the SRD5A1 gene
A significant increase of the frequency of the GG homozygous genotype of rs248793 (SRD5A1) was observed in the PAD patient group in comparison to controls (p=0.01 by chi-squared test, Table 3A), confirming previous results. 16 After adjusting for age and sex, by logistic regression analysis, the GG genotype was still significantly associated with PAD (OR=1.77, 95% CI=1.07–2.93, p=0.02).
When patients were subdivided according to diabetic status, the strength of association of the GG genotype was higher with NDPAD (OR=2.04, significantly different from controls with p=0.005) than with DPAD (OR=1.45, not significantly different from controls, p=0.11) (Table 3B). Indeed, the previously reported lack of association between diabetes and rs24879325,26 allowed the use of a single healthy group of controls for both PAD subgroups.
Logistic regression analysis restricted to NDPAD (n=88) and controls (n=239) confirmed that, after adjusting for age and sex, the GG genotype was significantly associated with PAD (OR=2.33, 95% CI=1.24–4.36, p=0.008).
When the association with diabetic status was tested by a logistic regression model, adjusted for age, sex, smoking habit, dyslipidemia and hypertension, that directly compared NDPAD versus DPAD (case-only study), using diabetic status as the outcome variable and gene polymorphism as the descriptor variable, the association of the GG genotype with the non-diabetic status (OR=1.46) did not reach the statistical significance level (p=0.2; 95% CI=0.81–2.63).
Since the NDPAD group contained a higher percentage of smokers (Table 1A), the hypothesis of an association of the GG genotype with smoking habit was also tested by a logistic regression model that compared smoker PAD patients with non-smoker PAD patients. After adjusting for age, sex, dyslipidemia, hypertension and diabetic status, no association of the GG genotype with smoking habit was revealed among PAD patients (OR=0.97; 95% CI=0.50–1.88).
Discussion
ICAM-1 is a member of the immunoglobulin superfamily of adhesion molecules that plays an important role in the adhesion of circulating leukocytes to the blood vessel wall and in their transendothelial migration to the vascular intima, one of the earliest steps in the pathogenesis of atherosclerosis.27,28 Indeed, a reduction in atherosclerosis in mice deficient in ICAM-1 29 and upregulation of endothelial ICAM-1 at atherosclerosis-prone sites in the ApoE-deficient mouse has been reported. 30
The genetic polymorphism rs5498 in exon 6 of the ICAM-1 gene results in the substitution of lysine to glutamate (K469E), and the early hypothesis on the possible pro-atherogenic role of this polymorphism focused on this amino acid change.14,18 Another possible functional consequence is suggested by the observation that this SNP is located three base pairs upstream of a splice donor site that produces an alternatively spliced form of ICAM-1 that lacks a transmembrane and intracellular domain (ICAM-1-S). It has been demonstrated that carriers of the G-allele (GG/GA genotypes) may have more difficulty in splicing ICAM-1-S compared to the A-allele (AA genotype), 31 thus producing less ICAM-1-S and more pro-atherogenic ICAM-1. 32 However, in the present study, we failed to observe a significant increase of the frequency of the GG genotype in the symptomatic PAD group. Our results might still have residual confounding given the predominance of older men in the PAD group compared to controls, despite adjusting for age and sex.
Unlike the ICAM-1 polymorphism study, the analysis of the SRD5A1 gene polymorphism rs248793 cannot be considered a replication study because the examined cohort is overlapping with that analyzed in a previous study. 16 However, a higher number of patients and controls has been recruited and the obtained data confirm the association of rs248793 with symptomatic PAD.
It is not possible to exclude that another genetic variant in linkage disequilibrium with rs248793 is responsible for the observed effect. Indeed, several SNPs are present in the same region and we have previously reported that a SNP located in intron 1 of the SRD5A1 gene, 5 kb downstream of rs248793, showed an identical association to PAD. 16 The protein encoded by the SRD5A1 gene is one of the steroid 5-α-reductase isoenzymes that catalyses the conversion of testosterone (T) to the bioactive potent androgen dihydrotestosterone (DHT) and represents a relevant regulator of local actions of T. Although rs248793 produces a synonymous substitution in exon 1 that does not change the amino acid sequence, individuals bearing the homozygous CC genotype of rs248793 had a significantly higher serum DHT/T ratio, an index of conversion of T to DHT, than that found in the other genotype groups. 26 Therefore, it is possible that the increased frequency of the GG genotype observed in PAD is correlated with a decreased 5-α-reductase enzyme activity. In agreement with such a hypothesis, it has been recently published that lower T or DHT levels are associated with symptoms of intermittent claudication in older men. 33 SRD5A1 is expressed in endothelial cells and is regulated by angiogenetic and anti-angiogenetic factors, 34 and treatment with DHT stimulates key angiogenic events both in vitro and in vivo. 35 Indeed, a large amount of scientific literature is dedicated to the role of androgens on vascular pathology and it is biologically plausible that a functional variant of this enzyme can influence the progression of the atherosclerotic process underlying PAD. 36
A decrease of DHT levels and 5-α-reductase expression occurs in the sciatic nerve in experimental models of diabetes. 37 If diabetic status itself produced a strong impairment of SRD5A1 function, the small difference in expression or function of this enzyme due to genetic polymorphisms would be irrelevant in its presence. This hypothesis would explain the trend towards a modification of the association between SRD5A1 polymorphism and PAD by diabetic status, observed in the present study. At the current state of knowledge, the above-described mechanism, responsible for interaction between SRD5A1 and diabetes, is fully speculative and requires experimental evidence on the possible angioprotective role of SRD5A1 and its alterations in diabetes. Future research should be addressed to ascertain a link between polymorphism of the SRD5A1 gene, its expression and the serum DHT/T ratio in diabetic and non-diabetic subjects.
Footnotes
Declaration of conflicting interest
The authors have no conflicts of interest to declare.
Funding
The authors gratefully acknowledge the financial support of the Assessorato Regionale alla Sanità, Regione Sicilia (Italy), Research Project 2007.
