Abstract
Little is known about the risk factors for peripheral artery disease (PAD) among diabetic patients in Saudi Arabia. We therefore investigated the association of traditional and non-traditional PAD risk factors, as well as clinical markers, with the prevalence of PAD in 598 diabetic patients in Saudi Arabia. PAD was diagnosed as an ankle–brachial index (ABI) <0.9. Information on socio-demographic variables, smoking status, duration of diabetes, and medication were collected by questionnaire. Body weight, height, blood pressure and clinical markers were also measured. The prevalence of PAD in this population was 23.1%. Hypertension (OR (odds ratio) = 2.13, 95% CI: 1.29–3.52), obesity (OR=1.75, 95% CI: 1.13–2.73) and longer duration of diabetes (OR for ≥20 years vs 2–4 years = 3.30, 95% CI: 1.66–6.58) were independently and significantly associated with a higher prevalence of PAD. Participants with the highest quartile of fasting blood glucose and homocysteine levels had a 67% (OR=1.67, 95% CI: 0.89–3.31) and 78% (OR=1.78, 95% CI: 0.87–3.66) higher prevalence of PAD, respectively. Our study identified several important and largely modifiable risk factors for PAD in this Saudi population with diabetes. These findings underscored the importance of reducing cardiovascular risk factors in patients with diabetes.
Introduction
Patients with diabetes have a significantly higher risk of peripheral artery disease (PAD).1,2 In previous studies, the prevalence of PAD in patients with diabetes ranged from 20% to 30%.2,3 In patients with diabetes, PAD is associated with an elevated risk of fatal and non-fatal cardiovascular and cerebrovascular events, including myocardial infarction and stroke.4,5 Patients with diabetes and PAD are also at a higher risk of gangrene, ischemic ulceration and lower extremity amputation than diabetic patients without PAD. 6 Symptomatic PAD is usually characterized by intermittent claudication, adversely affecting patients’ quality of life, 7 resulting in functional impairment. 8 Given these severe prognostic events associated with PAD, the most effective measure to manage PAD is to identify individuals at a high risk earlier, thereby preventing the onset of PAD and avoiding its severe symptoms, such as limb-threatening ischemia. 3
Several traditional risk factors for PAD have been identified, including smoking, advanced age, dyslipidemia and hypertension. 2 Emerging evidence has suggested some non-traditional risk markers for PAD, such as HbA1c, 9 waist-to-hip ratio, 10 duration of diabetes, 11 fibrinogen, lipoprotein(a), C-reactive protein (CRP), 12 and carotid intima-media thickness. 13 However, the findings have been inconsistent. Relatively little is known about the risk factors of PAD in patients with diabetes, while the prognosis, progression and pathophysiology of PAD may differ between patients with and without diabetes.2,14
Owing to the dramatic changes in lifestyle and diet in past decades, by 2011 Saudi Arabia was ranked as the country with the seventh highest prevalence of adult diabetes (20.2%) in the world. 15 Considering the accompanying complications, such as PAD, with the high prevalence of diabetes, the diabetes epidemic threatens to overburden the health care system and create insurmountable public health challenges in Saudi Arabia. Furthermore, few studies have addressed the risk factors of PAD in an Arabic population, which could be different from those already reported in other populations. Therefore, identifying individuals with high PAD risk and thus preventing onset of PAD among patients with diabetes are of great public health importance in Saudi Arabia. 16
In the present study, we investigated the association of PAD risk factors, including traditional and non-traditional risk factors, as well as clinical markers, with the prevalence of PAD among patients with diabetes in Saudi Arabia.
Subjects and methods
This study was a cross-sectional, population-based epidemiologic analysis among patients with diagnosed diabetes. Potential participants from Jeddah Governance, Saudi Arabia were recruited by advertisements between June 2009 and May 2010. Participants aged 30 years or older, who had had diabetes for at least 2 years and did not have current diabetic ulcers, were eligible for this study. The final study population was comprised of 598 participants. The study was approved by the King Abdulaziz University Hospital (KAUH) Ethical Committee. Study methods and objectives were explained to all participants. Written consent was obtained from all participants. The diagnosis and type of diabetes mellitus was first self-reported by patients and then confirmed by physicians using medical records. A questionnaire was administered in person by a vascular specialist and a trained nurse to collect information on nationality, age, smoking status, personal income level, educational attainment, current medication use and duration of diabetes. Nationality was classified as Saudi Arabic and non-Saudi Arabic. Smoking status was classified as ever-smoker and non-smoker. Personal income level was categorized as <3000, 3000–10,000 and >10,000 Saudi riyal (SR) /month (1 USD = 3.75 SR). We categorized education attainment as illiterate, high school, and university. Duration of diabetes was categorized into 2–4, 5–9, 10–19 and ≥20 years.
The participant’s body weight was measured to the nearest 0.1 kg by an electronic weighing scale (Seca, Birmingham, UK). Height was measured without shoes to the nearest 0.5 cm using a stadiometer (Seca). Body mass index (BMI) was calculated as weight (kg) divided by height (m) squared (kg/m2). Waist circumference was measured as the smallest circumference between the rib margin and iliac crest. The cut-off points of BMI recommended by the World Health Organization (WHO) were used to define obesity (≥30 kg/m2). Abdominal obesity was defined as waist circumference ≥102 cm for men and ≥88 cm for women. All participants’ blood pressures were measured by an electronic vital signs monitor (SureSignsVS3; Philips, Andover, MA, USA). Two consecutive readings of blood pressure were taken in participants’ right arms in a seated position after 5 minutes of rest. The mean of the two measures was used for analysis. In this study, hypertension was defined as systolic blood pressure (SBP) ≥140 mmHg and/or diastolic blood pressure (DBP) ≥90 mmHg according to the Seventh Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure guidelines, 17 and/or using blood pressure-lowering drugs. Using a bi-directional hand-held Doppler ultrasound instrument (Huntleigh Diagnostics, Cardiff, UK), one trained nurse, under the supervision of a vascular specialist, measured the participant’s two ankle blood pressures at the posterior tibial and dorsalis pedis arteries in both legs with the participant in a supine position. The leg-specific ankle–brachial index (ABI) was computed by taking the higher pressure of the two arteries at the ankle divided by the average of the brachial systolic blood pressure measurements or the higher of the two brachial values if the difference between the two brachial readings was ≥10 mmHg. 18 PAD was diagnosed as ABI <0.9 in either leg. Diagnosis of PAD followed the American College of Cardiology (ACC) and American Heart Association (AHA) Practice Guidelines for the management of patients with PAD. 19 In participants with ABI >1.4, the toe–brachial index (TBI) was calculated by dividing the systolic pressure of the great toe by that of the brachial artery. In this case, PAD was diagnosed as TBI <0.7 in either leg. 20
Participants were instructed to fast and abstain from vigorous exercise for 12 hours prior to medical examination and blood draw. Fasting venous blood samples (10 ml) were taken from the antecubital vein by laboratory staff and sent to KAUH’s accredited central laboratory for daily assay. Fasting blood glucose, HbA1c, lipid profile (total cholesterol, high-density lipoprotein (HDL), low-density lipoprotein (LDL), triglycerides (TGs)), homocysteine, creatinine, high-sensitivity CRP, serum urea nitrogen, hemoglobin, and complete blood count (CBC) were measured using standard methods.
Statistical analyses
The general characteristics of the study population were compared between PAD cases and non-PAD cases. Participants’ ABI and age were presented as mean and standard deviation. All the clinical biomarkers, including fasting blood glucose, HbA1c, total cholesterol, HDL, LDL, triglyceride, homocysteine, creatinine, CRP, serum urea nitrogen, hemoglobin, white blood cells, and platelets were presented as 25% percentile, median and 75% percentile. The comparison of continuous variables between PAD cases and non-PAD cases was fulfilled by the two-sample t-test. The dichotomous and the ordinal categorical variables were presented as number and percentage. The χ2 test was employed to compare the distribution of dichotomous variables, including sex, nationality, obesity, abdominal obesity, smoking status, diabetes type, hypertension, insulin use, oral hypoglycemic use, aspirin use, Plavix use, and statin use, between PAD cases and non-PAD cases. The Cochran–Armitage χ2 test for trend was applied to test the univariate association between ordinal categorical variables, including education, income and duration of diabetes, and PAD. The multivariate logistic model was used to investigate the association between risk factors and PAD. The first model estimated the odds ratio and its 95% confidence interval (CI) for each risk factor with adjustment for sex, age (continuous) and nationality (Saudi Arabia, non-Saudi Arabia). In this model, all the continuous variables were categorized into quartiles. To further explore significant independent risk factors for PAD, we used a backward selection algorithm beginning with a model that including all the potential risk factors listed in Table 1. In this model, sex and age were forced to be included and all the biomarkers were included as median values of their quartiles. A variable was ultimately retained in the final model if its p-value was ≤0.05. This model examined the association between each selected independent risk factor and prevalence of PAD with adjustment for other independent risk factors. Considering the potential mediating role of creatinine for the association between homocysteine and PAD, 21 a model including all the independent risk factors and creatinine was also presented. All statistical analyses were conducted using SAS 9.2 software (SAS Institute, Cary, NC, USA). All p-values were two-tailed (α = 0.05).
Characteristics of participants according to peripheral artery disease status.
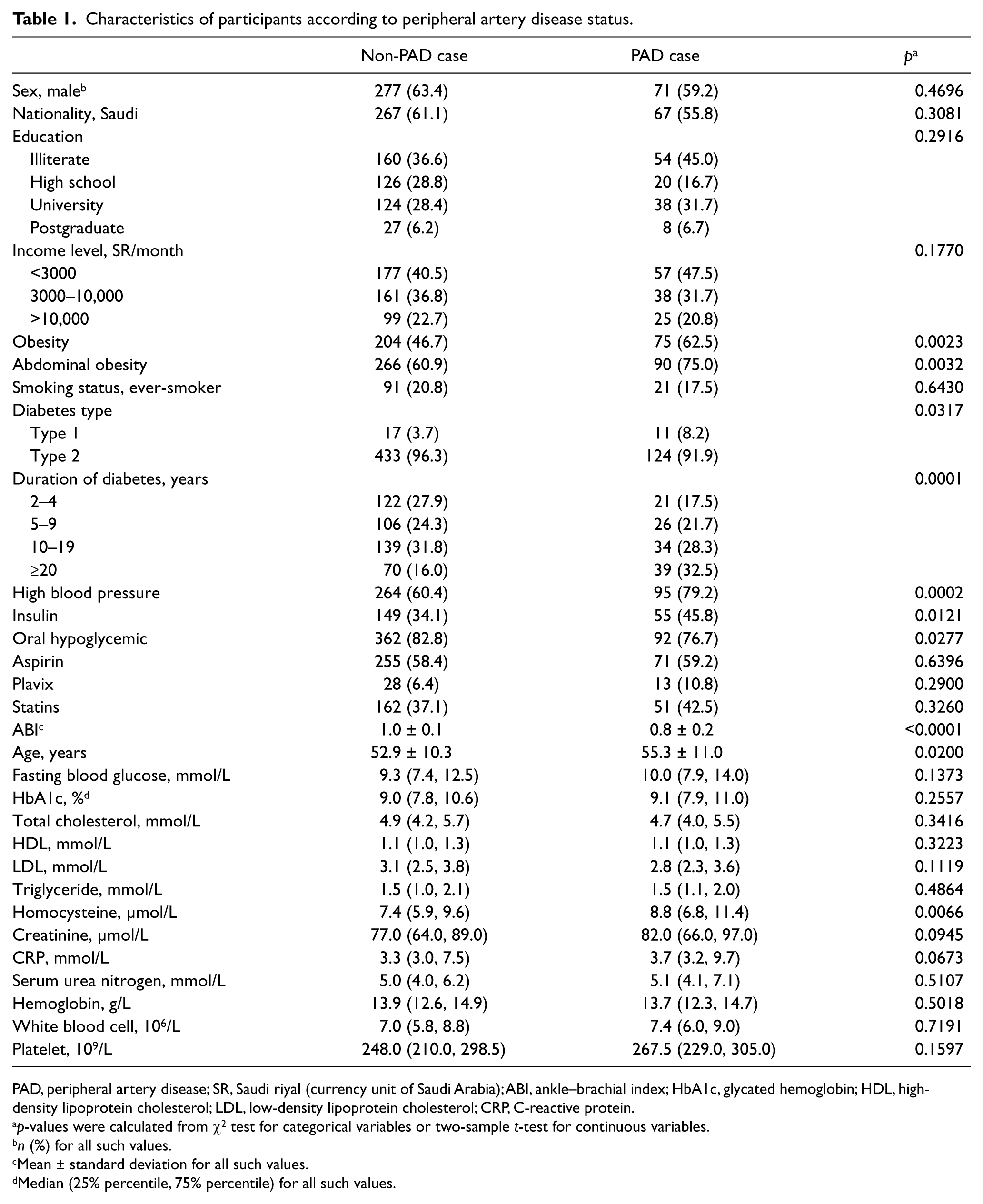
PAD, peripheral artery disease; SR, Saudi riyal (currency unit of Saudi Arabia); ABI, ankle–brachial index; HbA1c, glycated hemoglobin; HDL, high-density lipoprotein cholesterol; LDL, low-density lipoprotein cholesterol; CRP, C-reactive protein.
p-values were calculated from χ2 test for categorical variables or two-sample t-test for continuous variables.
n (%) for all such values.
Mean ± standard deviation for all such values.
Median (25% percentile, 75% percentile) for all such values.
Results
Among the 598 patients with diabetes, 138 individuals were diagnosed with PAD by a low ABI; the prevalence of PAD in this population was 23.1%. The average ABI for participants with and without PAD were 1.0 ± 0.1 and 0.8 ± 0.2, respectively. The participants included 62.0% males; 59.5% were Saudi nationals. The mean age of the population was 53.4 ± 10.5 years. Of these patients, 95.3% were type 2 diabetes, 36.6% were under insulin treatment and 80.4% were using oral hypoglycemic medications. Compared to their counterparts without PAD, participants with PAD were older, more likely to be generally and abdominally obese, had a longer duration of diabetes and a higher prevalence of high blood pressure (Table 1). Participants with PAD had a significantly higher serum homocysteine level than those without PAD.
Table 2 shows the association between each risk factor and the prevalence of PAD after adjustment for age, sex and nationality. The prevalence of PAD was significantly higher in generally obese (OR=1.92, 95% CI: 1.26–2.91) and abdominally obese (OR=1.88, 95% CI: 1.18–2.99) participants. A significant association between higher prevalence of PAD and longer diabetes duration was also observed with a ptrend = 0.0007. Compared to type 2 diabetic patients, type 1 diabetic patients had a significantly higher prevalence of PAD (OR=3.52, 95% CI: 1.53–8.13). Participants with the duration of diabetes ≥20 years had a 3.08-fold prevalence of PAD (95% CI: 1.63–5.83) compared to those with 2–4 years’ history of diabetes. Hypertensive participants had a significantly higher prevalence of PAD (OR=2.18, 95% CI: 1.38–3.46) than normotensives. Participants under insulin treatment had a significantly higher prevalence of PAD (OR=1.68: 95% CI: 1.13–2.50). Among clinical markers, elevated levels of fasting blood glucose (ORQ4 vs Q1 = 1.88, 95% CI: 1.05–3.34, ptrend = 0.0189), HbA1c (ORQ4 vs Q1 = 1.68, 95% CI: 0.96–2.95, ptrend = 0.0256), homocysteine (ORQ4 vs Q1 = 2.23, 95% CI: 1.13–4.37, ptrend = 0.0024), creatinine (ORQ4 vs Q1 = 2.23, 95% CI: 1.16–4.27, ptrend = 0.0117), and CRP (ORQ4 vs Q1 = 1.88, 95% CI: 1.04–3.41, ptrend = 0.0109) were significantly associated with a higher prevalence of PAD.
Association between risk factors and prevalence of peripheral artery disease among individuals with diabetes. a

SR, Saudi riyal (currency unit of Saudi Arabia); HbA1c, glycated hemoglobin; HDL, high-density lipoprotein cholesterol; LDL, low-density lipoprotein cholesterol; CRP, C-reactive protein.
Logistic regression model adjusted for sex, age (continuous) and nationality (Saudi Arabia, non-Saudi Arabia).
For ordinal variables, the p-value was estimated from the linear trend test.
Table 3 shows the independent risk factors selected by a backward stepwise logistic model. High blood pressure (OR=2.13, 95% CI: 1.29–3.52), obesity (OR=1.75, 95% CI: 1.13–2.73) and longer duration of diabetes (OR≥20 vs 2–4 years = 3.30, 95% CI: 1.66–6.58) were associated with a significantly higher prevalence of PAD. Every 10-year increment of diabetes duration was associated with a 63% higher prevalence of PAD (OR=1.63, 95% CI: 1.15–2.31). Of the clinical markers, elevated fasting blood glucose and homocysteine levels were associated with a significantly higher prevalence of PAD. Participants with the highest quartile of fasting blood glucose and homocysteine levels had a 67% (OR=1.67, 95% CI: 0.89–3.31) and a 78% (OR=1.78, 95% CI: 0.87–3.66) higher prevalence of PAD, respectively. A one unit increment of fasting blood glucose (1 mmol/L) and homocysteine (1 μmol/L) corresponded to a 7% (OR=1.07, 95% CI: 1.00–1.13) and a 5% (OR=1.05, 95% CI: 1.00–1.11) higher prevalence of PAD, respectively. After further adjustment for creatinine level, the ORs associated with homocysteine level were slightly attenuated, but the overall trend remained similar.
Backward multivariate logistic regression of risk factors associated with peripheral artery disease. a
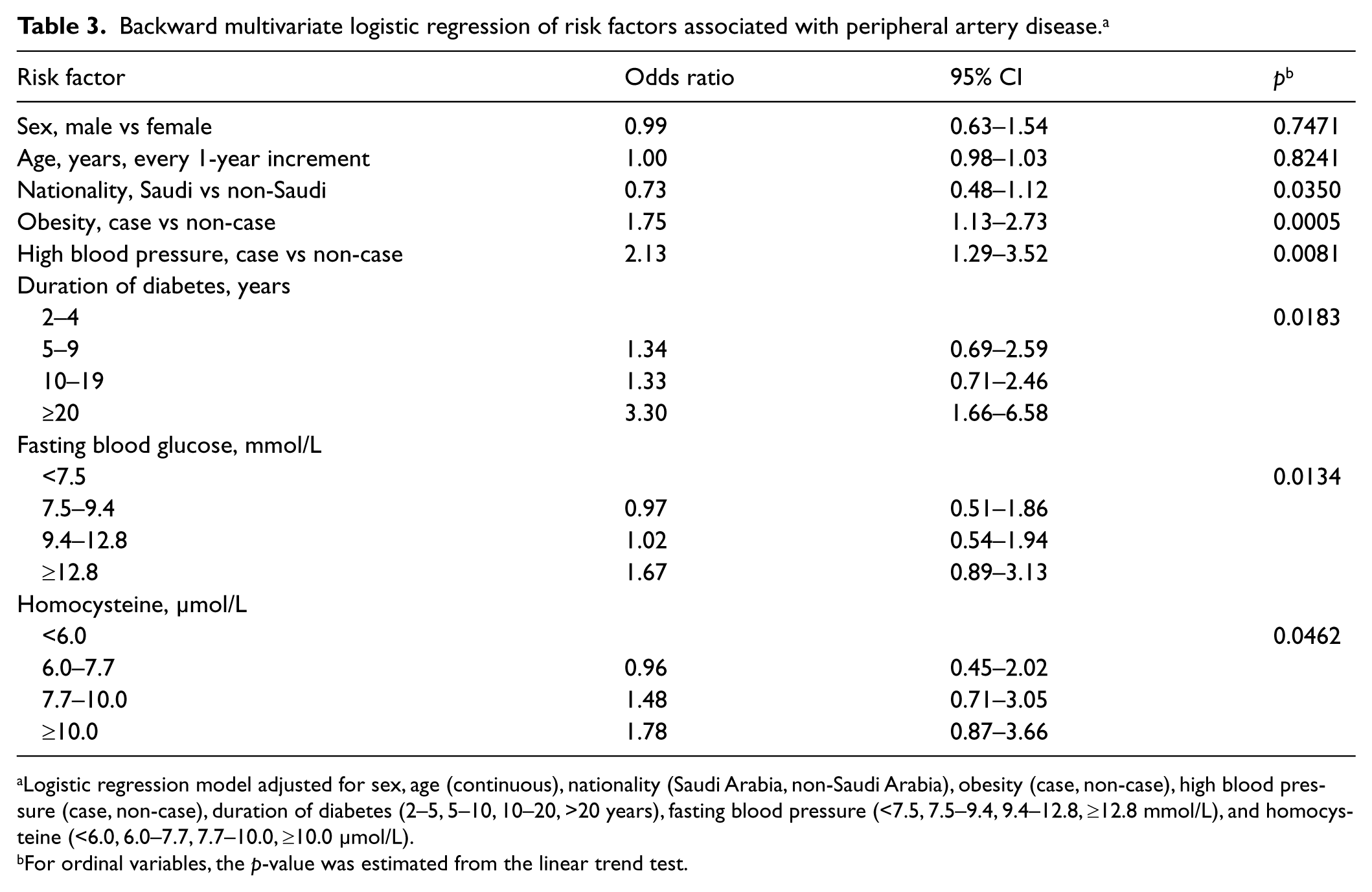
Logistic regression model adjusted for sex, age (continuous), nationality (Saudi Arabia, non-Saudi Arabia), obesity (case, non-case), high blood pressure (case, non-case), duration of diabetes (2–5, 5–10, 10–20, >20 years), fasting blood pressure (<7.5, 7.5–9.4, 9.4–12.8, ≥12.8 mmol/L), and homocysteine (<6.0, 6.0–7.7, 7.7–10.0, ≥10.0 μmol/L).
For ordinal variables, the p-value was estimated from the linear trend test.
Discussion
In this large Saudi population with diabetes, we found that hypertension, longer duration of diabetes, elevated levels of fasting blood glucose and homocysteine, as well as obesity were independently associated with a higher prevalence of PAD. In addition, insulin therapy, abdominal obesity and elevated levels of HbA1c, creatinine, and CRP were also related to a significantly higher prevalence of PAD, but these associations became non-significant after multivariate adjustment.
The prevalence of PAD in this study population was 23.1%, which is similar to those reported in other Middle East populations. In a previous study on 331 Bahrain participants with an average age of 54.2 ± 8.3 years in a primary care setting, a PAD prevalence of 20.2% was observed. 22 In a multi-ethnic study in several Middle East countries, including Egypt, Lebanon, and Jordan, among 3989 participants with diabetes in an outpatient medical setting with an average age of 55.8 ± 12.6 years, a relatively lower PAD prevalence of 13.4% was reported compared with our finding. 23 This could be explained by the relatively higher BMI (31.2 ± 6.8 kg/m2) and longer duration of diabetes (16.8 ± 4.8 years for type 1 diabetes, 11.2 ± 6.4 years for type 2 diabetes) in our study population compared to this multi-ethnic study population (BMI, 29.4 ± 5.1 kg/m2; duration of diabetes, 13.4 ± 8.2 years for type 1, 9.6 ± 7.0 years for type 2).
Hypertension, cigarette smoking, and dyslipidemia are recognized as modifiable risk factors for PAD. 2 In this current study, elevated blood pressure was associated with a more than twofold prevalence of PAD, which is consistent with findings from a nationally representative sample in the USA 24 and confirms high blood pressure as a risk factor for PAD. 25 Our study observed a non-significantly higher prevalence of PAD among ever-smokers. A relatively small number of PAD cases (n=48) and the large variation of smoking status (current smoker, ex-smoker and different quantities of cigarette smoking) within ever-smokers might have limited our power to detect this association. Our study did not confirm the association between dyslipidemia and higher prevalence of PAD. Because of the coexistence of dyslipidemia and diabetes, our study population had a very high prevalence of dyslipidemia and lipid-lowering drug use (89.8%) and thus lipid levels were likely to have been heavily influenced by the treatment.
The duration of diabetes, even though not a modifiable risk factor, is of great importance for early identification and management of PAD. Previous studies have consistently reported a positive association between duration of diabetes and PAD. In the Health Professional Follow-up Study, 11 Al-Delaimy et al. reported a relative risk (RR) of 3.63 (95% CI: 2.23–5.88), 2.55 (95% CI: 1.50–4.32) and 4.53 (95% CI: 2.39–8.58) for developing PAD among patients with 6–10, 11–25 and >25 years of diabetes compared to those with <5 years’ history, respectively. Our study also found a strong and independent association between longer duration of diabetes and higher prevalence of PAD. The observed significantly higher prevalence of PAD in type 1 diabetics in the univariate analysis could also be explained by the usually longer duration of diabetes in type 1 diabetics compared to type 2 diabetics. However, it is worth noting that the 95% CI of the OR when comparing type 1 with type 2 diabetics is relatively wide, which could be due to the small sample size of type 1 diabetics and thus warrants caution in interpretation. Long-term exposure to metabolic disturbances associated with diabetes could enhance and accelerate atherosclerotic narrowing of the blood vessel lumen, which may be one of the underlying mechanisms of PAD. 26 In addition, the HbA1c level, a measurement for hemoglobin’s long-term exposure to plasma glucose and an indirect indicator of diabetes duration, was also positively associated with prevalence of PAD in our study. In the Atherosclerosis Risk in Communities (ARIC) study, the third tertile of the HbA1c level was associated with a RR of 4.56 (95% CI: 1.86–11.18) for PAD-related hospitalization when compared with the first tertile. 9 It is interesting to note that in our study, the positive association between HbA1c and PAD became non-significant after multivariate adjustment, whereas the association of fasting plasma glucose and PAD remained significant in the multivariate model. Finally, our study found a 2.13-fold prevalence of PAD in insulin users compared to non-users. A similar association between insulin therapy and PAD has been reported repeatedly in previous studies.10,27,28 Insulin treatment tends to be associated with a longer duration and greater severity of diabetes.10,29
Our study observed a significant positive association between obesity and PAD. Based on the National Health and Nutrition Examination Survey (NHANES) data, Ylitalo et al. reported a 3.10-fold OR of PAD (95% CI: 1.84–5.22) among participants with BMI ≥30 kg/m2 compared to BMI <30 kg/m2. 24 The ARIC study found that patients with diabetes in the highest quartile of waist-to-hip ratio had a 1.60-fold risk ratio of PAD (95% CI: 1.10–2.33) compared to the lowest quartile. 10 Systematic inflammation and vascular resistance caused by disturbance of adipocytokines may partially explain the mechanism underlying the association between excess adiposity and PAD, even though more studies are warranted to elucidate the underlying mechanisms.
We found a significant positive association between fasting blood glucose levels and PAD in patients with diabetes. As suggested by a previous study, hyperglycemia has a more significant effect on micro-vascular rather than macro-vascular endpoints. 2 Therefore, it is not surprising that fasting blood glucose was significantly and independently related to PAD in our study, which is consistent with a previous study. 30 This finding was also supported by results from patients with atherosclerosis. 30 Previous studies have suggested that the plasma concentration of homocysteine, an indicator of oxidative stress related to the pathogenesis of atherothrombosis, was elevated in patients with coronary and cerebral diseases.31,32 Several previous studies have also observed a positive association between elevated homocysteine levels and PAD.12,33 Our study found an independent positive association between homocysteine concentration and PAD; patients in the highest quartile of homocysteine level presented an OR of 3.37 for PAD compared to the lowest quartile. It was suggested that renal function could be a potential moderator for the association between homocysteine and PAD because homocysteine level has been associated with renal decline and a certain common pathological mechanism underlies both PAD and renal decline. 21 In our analysis, further adjustment for creatinine, an indicator of renal function, slightly attenuated the association, indicating a possible mediating role of renal function. However, possible collinearity or residual confounding could not be ruled out and further studies are warranted to clarify the pathological pathways. Furthermore, the homocysteine level was sensitive to B vitamin intake, 32 and thus future studies should adjust for plasma B vitamin levels. A large body of evidence has suggested that CRP, an inflammatory cytokine, was an independent marker of PAD.10,12,28,30,34 However, in our study, CRP was significantly associated with PAD in univariate analysis but became non-significant after multivariate adjustment.
To our knowledge, this is the first study on risk factors of PAD among the Saudi population with diabetes. The strengths of this study include a large sample size, detailed assessment of PAD, and comprehensive measures of clinical and biochemical markers. Several limitations of this study warrant attention. First, the cross-sectional nature of this study design limits the inference of causal relationships between risk factors and PAD. Therefore, our findings need to be confirmed in prospective studies. Second, because we used advertisements to recruit participants, eligible participants with severe risk factors might not have been able or willing to participate in this study. In addition, participants with known foot ulcers were excluded from the study population. Thus, findings from this study may not be extrapolated to the broader population with diabetes.
In conclusion, our study identified several important and largely modifiable risk factors for PAD in this Saudi population with diabetes, including hypertension, obesity, plasma concentrations of fasting glucose and homocysteine. These findings underscore the importance of reducing cardiovascular risk factors in patients with diabetes for the prevention of PAD. They also highlight the potential to identify patients with diabetes at high risk of PAD based on a combination of common risk factors and clinical markers. Prospective studies are warranted to further address the prediction of PAD risk in Saudi populations with diabetes.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Mohammed Hussein Al-Amoudi Chair for Diabetic Foot Research, King Abdulaziz University, Jeddah, Saudi Arabia.
