Abstract
Peripheral artery disease (PAD) is a highly prevalent condition that frequently goes undetected and untreated. Socioeconomic factors associated with unrecognized PAD are not known. The ankle–brachial index (ABI) was calculated in 1656 study participants undergoing non-emergent coronary angiography with PAD defined as an ABI <0.9. Subjects were followed for mortality and cardiovascular outcomes. Compared to those without PAD, those with unrecognized PAD at enrollment were older, had higher rates of cardiovascular comorbidities, and had higher major adverse cardiovascular events (MACE) (p<0.03 for all). Among those enrolling without a reported history of PAD, there was a higher prevalence of PAD with decreasing income (p=0.004), education level (p<0.001), social isolation (p=0.027) and depression (p=0.034); 50% of these individuals reported symptoms suggestive of claudication. In conclusion, the prevalence of unrecognized PAD is high amongst a cohort of high-risk individuals referred for coronary angiography. A profile of lower socioeconomic status is associated with unrecognized PAD. These subjects will report symptoms suggestive of claudication and impaired walking ability when directly queried.
Introduction
PAD is a rapidly growing problem, which is estimated to affect over 200 million people worldwide.1,2 In the United States alone, approximately 10 million adults have PAD and as many as 29% of primary care individuals over the age of 70 years have been reported to have PAD. 3
Data from the Reduction of Atherothrombosis for Continued Health (REACH) registry showed that patients with PAD experience the highest rates of cardiovascular death and major cardiovascular events among all patients with established arterial disease. 4 In fact, individuals with PAD have a twofold greater risk of total mortality and stroke than those without PAD.5–7 Because PAD is recognized as a coronary heart disease (CHD) risk equivalent, guidelines put forth by the American Heart Association now advocate screening for PAD in at-risk individuals.8,9
Unfortunately, PAD frequently goes unrecognized. In the seminal PARTNERS study of primary care patients over 70 years old or over 50 years with the risk factors of diabetes and a history of smoking, over half of the patients identified as having PAD did not previously carry the diagnosis. Additionally, it has been shown that the public is unfamiliar with PAD relative to other cardiovascular conditions and therefore presumably less likely to discuss the condition with their physician and pursue treatment. Furthermore, surveys have shown that physicians feel unfamiliar with PAD demographics, screening and diagnostic approaches.10,11 Given that secondary prevention measures have been shown to improve outcomes in PAD, 12 cases where the diagnosis of PAD is missed may result in preventable morbidity and mortality.
Previous studies have compared individuals with unrecognized PAD to those that carry a PAD diagnosis, and a number of factors have been identified which diverge between these two groups.8,13,14 However, among the clinically relevant group of at-risk individuals not known to have disease, limited studies have evaluated risk factors that may aid in the identification of those with occult PAD, 13 who presumably do not receive appropriate secondary prevention. Accordingly, the aim of this study is to identify the clinical and socioeconomic factors associated with unrecognized PAD in order to discern potential barriers to PAD diagnosis in a high-risk cohort.
Methods
Study population
The Genetic Determinants of Peripheral Arterial Disease (GenePAD) is a study of individuals (n=1755) who underwent elective, non-emergent coronary angiogram at Stanford University and Mount Sinai Medical Center between 1 January 2004 and 1 March 2008, as previously described.15–17 Eligible individuals were greater than 40 years of age and received a referral for angiography secondary to suspected coronary artery disease (CAD) due to angina, shortness of breath or an abnormal stress test. Individuals were excluded from the study sample if they had a history of radiation therapy, organ transplant or viral disease such as HIV or hepatitis. The GenePAD study was approved by the Institutional Review Boards at Stanford University and Mount Sinai and registered at http://clinicaltrials.gov (NCT 00380185). Informed consent was taken from all individuals. The study was funded by the National Heart, Lung, and Blood Institute.
Inclusion criteria
Participants were included in the study sample if complete data on all relevant covariates including age, sex, race, smoking history, systolic blood pressure (SBP), use of anti-hypertensive and lipid-lowering medications, use of insulin or oral hypoglycemic agents, total cholesterol and ankle–brachial index (ABI) were available. Remaining eligible subjects with an ABI >1.4 18 were excluded (n=26). Using these criteria, 1656 subjects were identified.
PAD group classification
Before the coronary angiogram, posterior tibial, dorsalis pedis, and brachial artery systolic pressures were measured with a 5-MHz Doppler ultrasound. The ABI was calculated for each leg by dividing the higher of the posterior tibial or dorsalis pedis pressures by the higher of the left or right brachial systolic pressures. Individuals who carried a clinical history of PAD, determined if the patient indicated at enrollment either a history of ‘blockage or narrowing in the arteries of the legs’ or ‘surgery, angioplasty, or stenting of any of the arteries supplying blood to your legs to relieve blockage’, comprised the ‘Recognized PAD’ group. Among the at-risk group without a clinical history of PAD, the ‘Unrecognized PAD’ group comprised those found to have an ABI <0.9 at the time of enrollment. Those without a previous history of PAD found to have an ABI of ≥0.9 at the time of enrollment comprised the ‘No-PAD’ group.
Questionnaires
Complete Walking Impairment Questionnaire (WIQ) data were available in greater than 99% of these individuals for all three WIQ category scores. Subjects completed the WIQ at enrollment with a trained nurse or research assistant. The WIQ consists of three primary categories assessing walking distance, stair-climbing and walking speed, as previously described in the GenePAD study.19,20 Subjects were classified as having symptoms suggestive of claudication if they responded affirmatively (‘slight’, ‘some’, ‘much’, or ‘very much’) to Section A of Question 1 in the WIQ, which asked if the subject experienced ‘pain, aching or cramp in the calves or buttocks’. 21
The Beck Depression Inventory (BDI) 22 was added to the GenePAD enrollment materials partway through the enrollment period and was distributed consecutively to patients. Results were therefore only available for 446 individuals. The BDI is a 21-question self-reported measure of the severity of depressive symptoms with a score of 0 to 64, where normal ranges from 0 to 10 and scores of 11 or higher indicate potential clinical depression.
Outcomes
The outcomes of interest in this analysis were major cardiovascular events (MACE) including heart attack, stroke, heart failure, coronary bypass and percutaneous transluminal coronary angioplasty (PTCA). We additionally examined peripheral bypass procedure, death from any cause and death from cardiovascular causes including myocardial infarction, cardiac arrest, stroke, heart failure or aneurysm rupture. Mortality, cause of death and MACE were ascertained through phone or postal communication, medical record review and the Social Security Death Index. New mortalities were identified through 31 March 2012.
Covariates
Detailed information on all included covariates was obtained by a trained nurse or clinical research assistant at enrollment. Age, sex, race, smoking history, income, maximum education level, doctor visit frequency, claudication and social history were acquired by self-report, and SBP was measured. Income level was reported as household income per year and stratified as ≥$100,000, $50,000–99,999, $25,000–49,999 and <$25,000. Maximum education level was reported as any graduate school, any college, grades 9–12 and grades 1–8. Doctor visit frequency was categorized as the presence or absence of annual routine check-ups. The use of lipid-lowering and anti-hypertensive medications was evaluated by direct medication inventory. Diabetes status was classified as self-reported use of insulin or oral hypoglycemic agents. Total cholesterol levels were measured at the time of coronary angiography.
Statistical methods
Baseline patient characteristics were analyzed for the Unrecognized PAD and No-PAD groups and compared using a t-test or chi-squared test. For all survival analyses the follow-up time was defined as the period between the enrollment interview and the last confirmed follow-up or date of death.
Kaplan–Meier curves were constructed to examine the risk of all-cause mortality and MACE in the Unrecognized PAD and No-PAD groups with survival functions compared using a log-rank test. Cox proportional hazard models were used to calculate unadjusted and adjusted hazard ratios (HRs) to compare all-cause, cardiovascular mortality, MACE and peripheral revascularization risks. Adjusted models included the following covariates: age, sex, race, current smoking status, systolic blood pressure, use of anti-hypertensives, diabetes, and total cholesterol. Proportional-hazards assumptions were evaluated by Schoenfeld residuals tests.
The prevalence of PAD was examined across strata of income and education level, whether the patient lived alone or with others and whether the patient had a BDI score in the normal or abnormal range with p-values for trend or difference calculated using both unadjusted and age, sex and race adjusted logistic regression analysis.
Tests were considered significant if the two-sided p-value was <0.05. All analyses were performed using Stata version 12.0 (StataCorp, College Station, TX, USA). Study data were collected and managed using REDCap electronic data capture tools hosted at Stanford University. 23
Results
Baseline demographics
There were 207 subjects with unrecognized PAD amongst 1558 subjects without a prior diagnosis of PAD at enrollment. Only 98 subjects in the entire GenePAD cohort reported a history of PAD prior to enrollment ABI testing. The prevalence of unrecognized PAD among all individuals with PAD was 67.8% (207/305). There were 198 mortalities, 54 from cardiovascular causes, with 371 MACE, 322 coronary bypasses or PTCAs and 30 peripheral bypasses during a median follow-up period of 5.0 years (interquartile range 4.0–6.3).
A comparison of the demographics and cardiovascular risk factor profiles are presented in Table 1. The mean age was 71 years for the Unrecognized PAD group and 65 years for the No-PAD group (p<0.001). There were 13% (27/207) of the Unrecognized PAD group that had isolated PAD without CAD identified at angiography. Women made up 44% of the Unrecognized PAD group compared to 33% in the No-PAD group (p=0.001). The mean ABI for the Unrecognized PAD group was 0.70 (0.61–0.83) compared to an ABI of 1.07 (1.00–1.14) in the No-PAD group. The Unrecognized PAD group also had significantly higher mean SBP (145 vs 138 mmHg; p<0.001), higher anti-hypertensive medication use (86 vs 76%; p=0.002), higher total cholesterol (40 vs 37%; p=0.024), and a greater proportion of current smokers (15 vs 8%; p=0.003), diabetics (45 vs 27%; p<0.001) and African Americans (25 vs 12%; p<0.001). Additionally, the Unrecognized PAD group had similar access to routine medical care compared to the No-PAD group (78 vs 85%; p=0.613). There were no significant differences in baseline characteristics noted between the Unrecognized PAD group compared to the Recognized PAD group with the exception of higher enrollment ABI values (p<0.001).
Baseline study population characteristics.
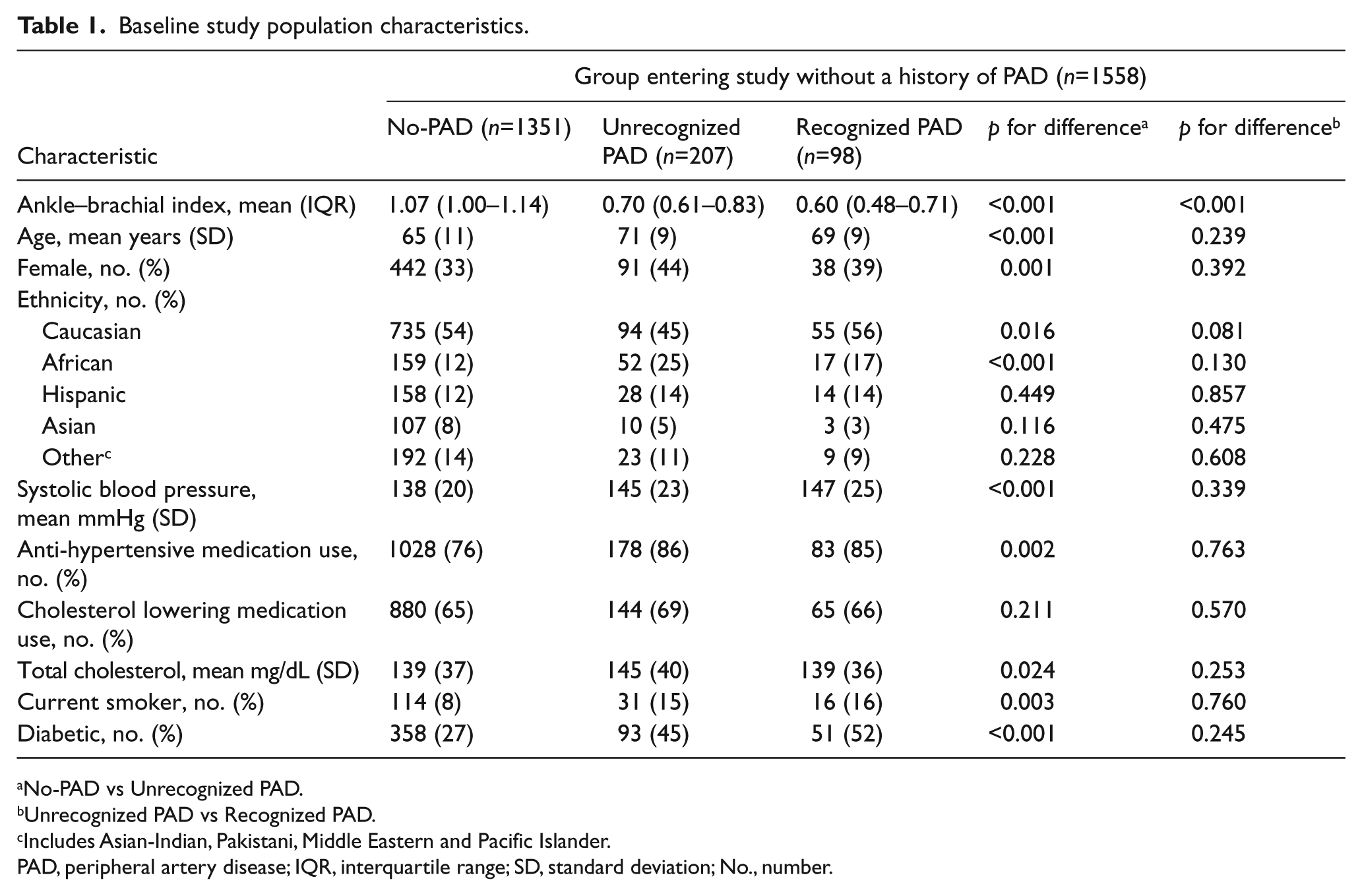
No-PAD vs Unrecognized PAD.
Unrecognized PAD vs Recognized PAD.
Includes Asian-Indian, Pakistani, Middle Eastern and Pacific Islander.
PAD, peripheral artery disease; IQR, interquartile range; SD, standard deviation; No., number.
Outcome
Kaplan–Meier survival curves for all-cause mortality and MACE are presented in Figure 1. The Unrecognized PAD group had significantly worse survival from all-cause mortality compared to the No-PAD group (p=0.015) and significantly better survival compared to the Recognized PAD group (p<0.001). Regarding MACE, the Unrecognized PAD group had worse survival compared to the No-PAD group (p<0.001) and did not significantly differ from the Recognized PAD group (p=0.724).
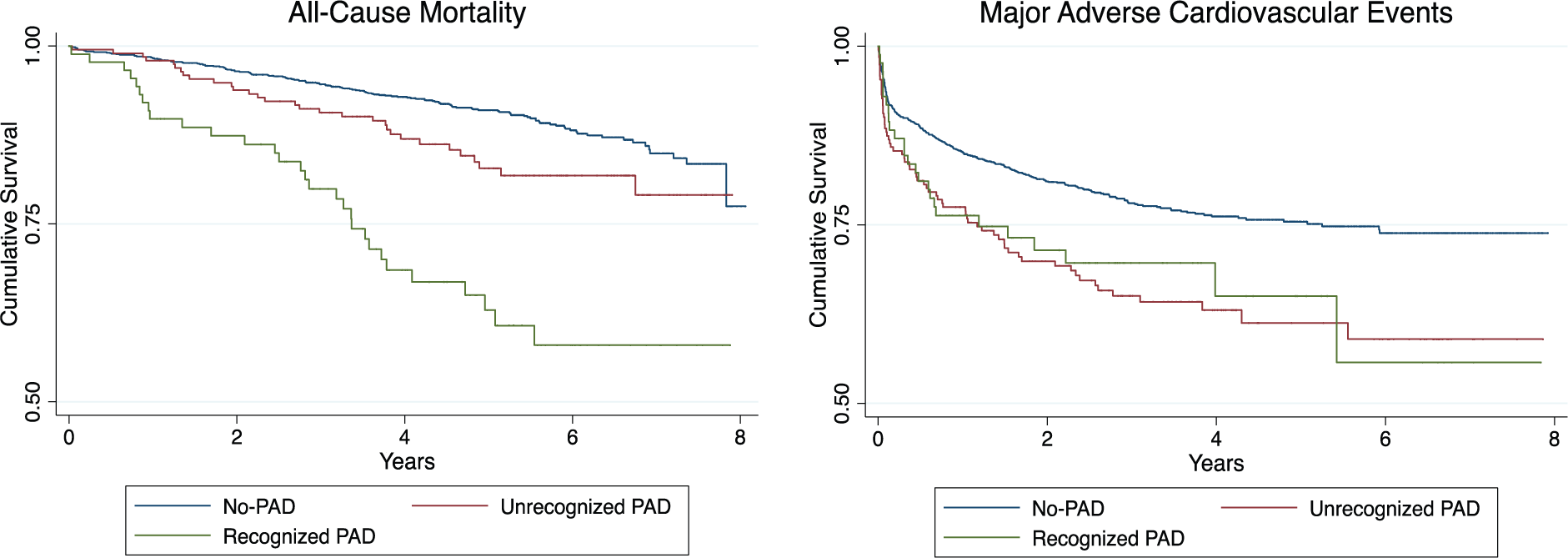
Kaplan-Meier curves showing cumulative survival in patients with No-PAD, Unrecognized PAD, and Recognized PAD.
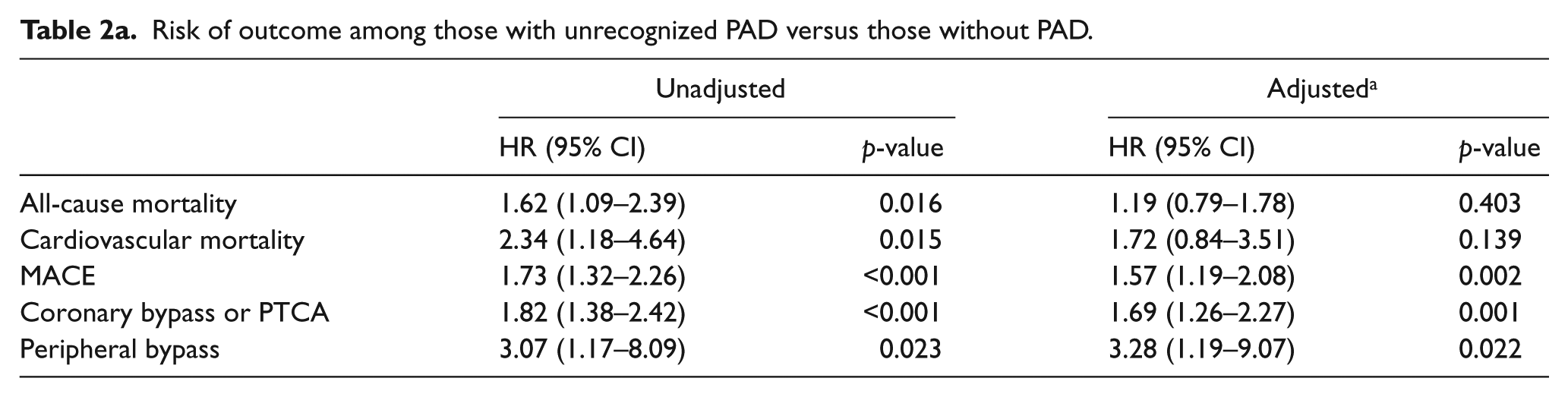
In adjusted regression analyses (Table 2), the Unrecognized PAD group had a significantly higher risk of MACE (p=0.002), coronary bypass or PTCA (p=0.001) and peripheral bypass (p=0.022) compared to the No-PAD group. However, compared to those in the Recognized PAD group, the Unrecognized PAD group had a significantly lower risk of all-cause mortality (p<0.001), cardiovascular mortality (p=0.028) and peripheral bypass (p=0.002).
Risk of outcome among those with unrecognized PAD versus those without PAD.
Risk of outcome among those with unrecognized PAD versus those with recognized PAD.
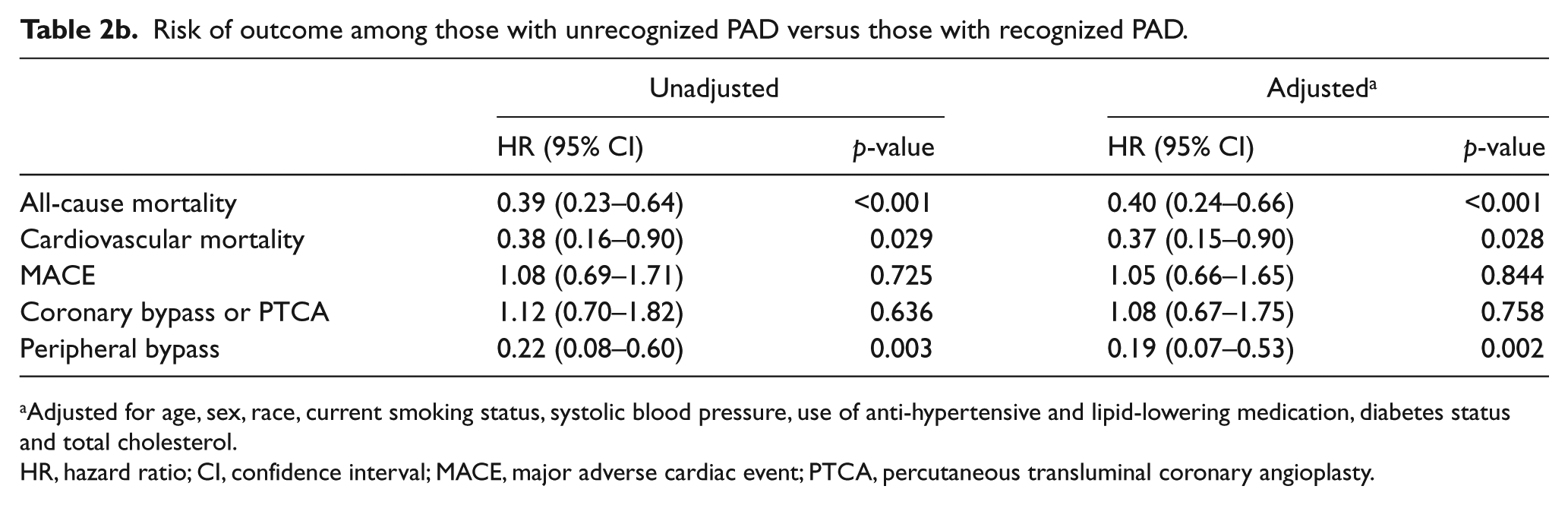
Adjusted for age, sex, race, current smoking status, systolic blood pressure, use of anti-hypertensive and lipid-lowering medication, diabetes status and total cholesterol.
HR, hazard ratio; CI, confidence interval; MACE, major adverse cardiac event; PTCA, percutaneous transluminal coronary angioplasty.
Socioeconomic risk factors
Table 3 shows the relationship of socioeconomic factors with the prevalence of unrecognized PAD. After adjustment for sex, race, and age, income level and maximum education level were both inversely proportional to the prevalence of unrecognized PAD among those without a previous PAD diagnosis at enrollment. The prevalence of unrecognized PAD was greater among individuals living alone compared to those living with others (19 vs 11.9%; p=0.027) and in those with abnormal BDI scores compared to those in the normal range (21.5 vs 10.8%; p=0.034). Additionally, regression analysis examining the BDI as a continuous variable found a significantly increased risk of having unrecognized PAD with increasing BDI scores, even after adjustment for age, sex and race (odds ratio=1.06; 95% CI, 1.01–1.11; p=0.020). The prevalence of unrecognized PAD among those found to have PAD at enrollment did not differ across socioeconomic factors.
Socioeconomic factors impacting the prevalence of unrecognized PAD.
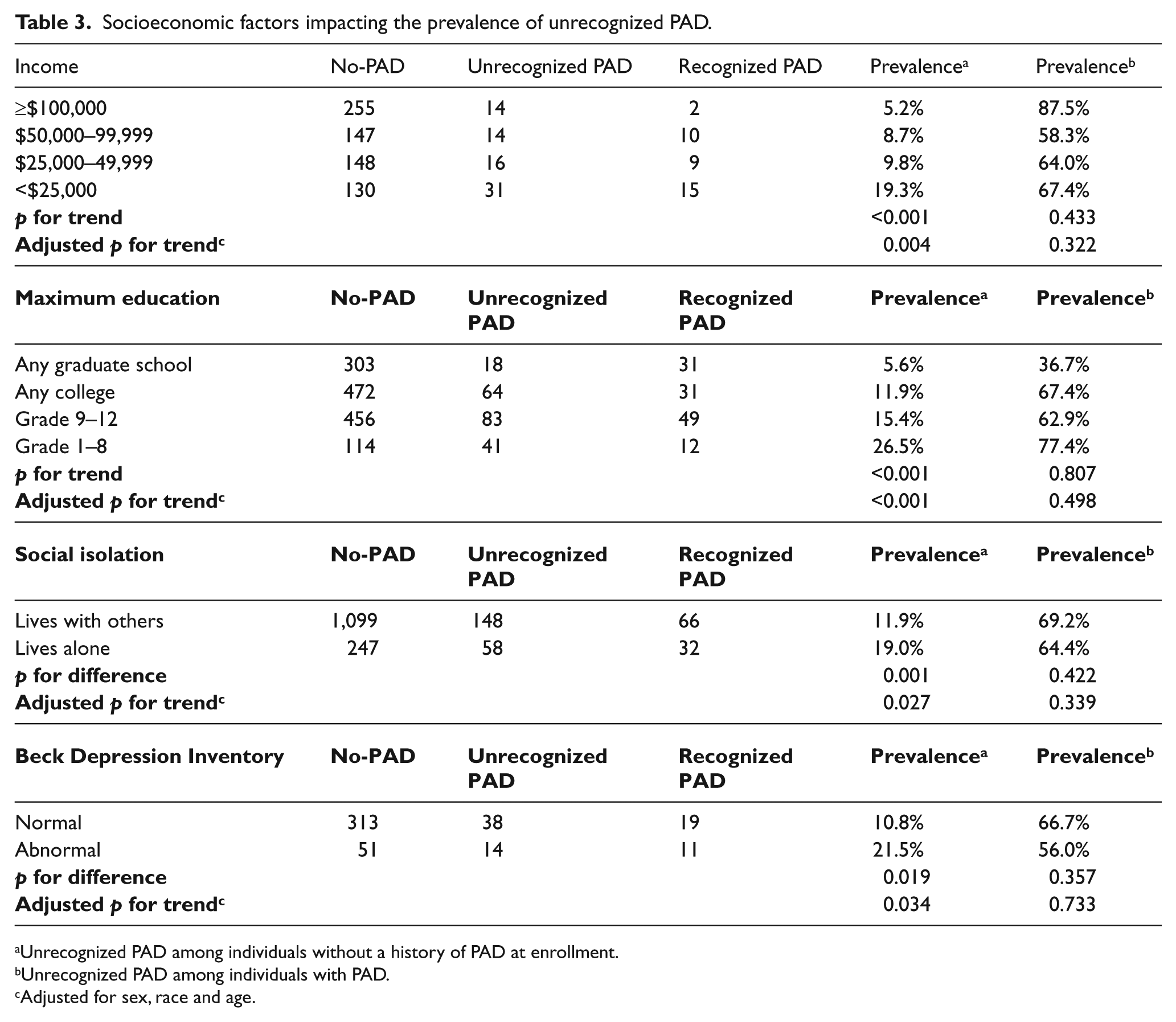
Unrecognized PAD among individuals without a history of PAD at enrollment.
Unrecognized PAD among individuals with PAD.
Adjusted for sex, race and age.
Walking impairment questionnaire
Finally, individuals with unrecognized PAD demonstrated significant walking impairment compared to the group without PAD (Table 4), as reflected by lower WIQ category scores for walking distance (49 vs 75; p<0.001), stair-climbing (36 vs 58; p<0.001) and walking speed (32 vs 56; p<0.001). However, those in the Unrecognized PAD group had significantly better WIQ scores compared to the Recognized PAD group (p<0.02). Importantly, symptoms suggestive of claudication, including pain, aching, or cramping of the calves or buttocks, were present in 50% of individuals with unrecognized PAD, which was significantly greater than in those without PAD (p<0.001).
Claudication symptoms and Walking Impairment Questionnaire scores.
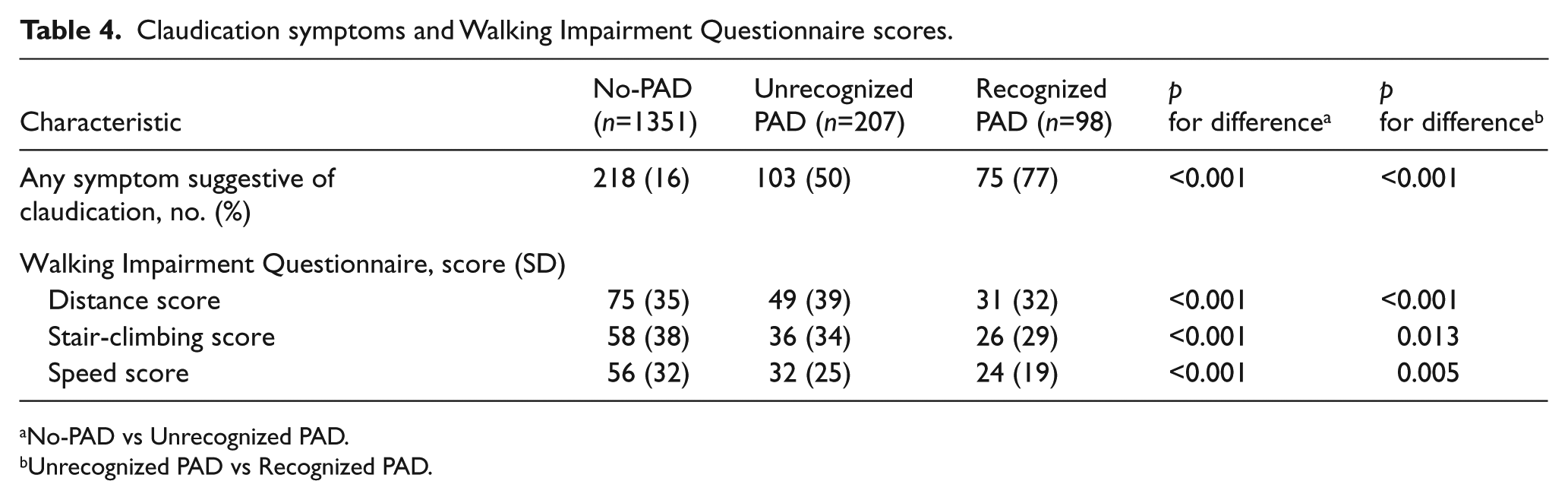
No-PAD vs Unrecognized PAD.
Unrecognized PAD vs Recognized PAD.
Discussion
The purpose of this study was to identify the factors that may obscure the diagnosis of PAD amongst high-risk individuals not previously diagnosed with this disease. We anticipate that an awareness of these barriers might facilitate the earlier identification of PAD patients, and therefore promote the initiation of therapies known to save life and limb. 12
As one might expect, we found that subjects with unrecognized PAD had high rates of classical PAD risk factors. Perhaps unsurprisingly, this group also had higher rates of future major adverse cardiovascular events than those individuals without PAD. These adverse outcomes may be compounded by less aggressive medical care, as evidenced by the fact that this group does not receive sufficient anti-hypertensive therapy and frequently is not prescribed a lipid-lowering agent, despite the common comorbidity of CAD. Additionally, we also observed the presence of several socioeconomic risk factors that were associated with unrecognized PAD. These included differences in income, education level, depression and social isolation, each of which could constitute additional barriers to physician recognition of PAD, beyond the known demographic risk factors of sex and race. 24 Taken together, it appears that there are several quantifiable clinical and socioeconomic factors associated with unrecognized PAD among at-risk individuals that could be used to identify those with unidentified disease, who otherwise might not receive important secondary prevention measures.
PAD screening studies performed in the primary care setting have shown that PAD goes undiagnosed 55% of the time. In the current study, which was performed in the catheterization laboratory setting, we observed an even higher rate (67.8% of all PAD patients in the GenePAD study were previously unrecognized), which is surprising considering a cardiovascular specialist had evaluated these patients. Adoption of an aggressive PAD screening of patients undergoing diagnostic coronary angiography for ischemic heart disease should be considered based on these data. This low rate of diagnosis has been ascribed to the fact that those with PAD may be asymptomatic or can present with a broad range of atypical symptoms. 25 These explanations are not reassuring, however, since asymptomatic PAD patients can experience mortality nearly as high as symptomatic PAD patients. 6 In addition, overlooking the diagnosis of PAD in our study cohort can lead to a higher risk for peri-procedural complications. Prior studies have reported a high risk of procedural failure and a significant risk of major vascular complications amongst PAD patients undergoing angiography.26,27 It is therefore worrisome that over two-thirds of PAD patients referred for coronary angiography are referred to the catheterization laboratory without an appropriate diagnosis of this comorbidity, where a lack of awareness can lead to tangible increases in peri-procedural risk. Particularly because utilization of a radial artery access approach may result in fewer vascular complications in subjects with PAD, 26 we believe that knowledge about this comorbid condition should be sought prior to any percutaneous intervention.
To characterize those patients with PAD who go unrecognized, we compared several clinical variables between those with unrecognized PAD and those without PAD. Previously, other groups have performed similar analyses in an attempt to identify barriers to diagnosis amongst high-risk individuals. For example, Moussa and colleagues reported that subjects undergoing coronary angiography who had undiagnosed PAD had a higher burden of CAD and more cardiovascular risk factors than subjects without PAD. 13 These results are in keeping with the findings reported here, and are consistent with the propensity for atherosclerosis to affect multiple vascular beds. 28
Presently, we found strong independent associations of unrecognized PAD with low income, low education, social isolation, and depression. Previously, a lower awareness of PAD has been identified as a barrier to diagnosis among people of lower educational and income status. Our study describes additional socioeconomic factors, which also may impede the appropriate identification of this disease. First, the social isolation of single living can be a barrier to receiving care. In the absence of interactions with caregivers, the isolated person may neglect his or her symptoms.29,30 Second, a patient’s level of education might lead to under-diagnosis because, in general, it may affect one’s ability to obtain, understand, or utilize information about the diagnosis. 31 The public is unaware of PAD in general, 32 and it may be that less educated individuals are even more unfamiliar with this condition and its presentation. Third, financial factors can influence a patient’s utilization of healthcare, particularly if they choose not to pursue the workup of a condition which is frequently only mildly distressing.10,33 Last, a patient’s mental status can impair a timely diagnosis. Psychologically, a person who is depressed may have functional impairment and be non-compliant, 34 while physiologically, depression may exacerbate endothelial dysfunction, circulating catecholamine levels, and platelet reactivity. 35 Future study is indicated to confirm the socioeconomic associations identified here.
An important finding from this study was that subjects with unrecognized PAD and subjects without PAD reported similar access to routine medical care. Accordingly, we assume that these socioeconomic factors may represent an independent barrier to recognition which is not explained by a simple disparity in contact with primary care practitioners. Though not investigated here, factors such as the presence and quality of coverage, 36 frequency of language barriers, 37 and compliance with physician recommendations may vary according to socioeconomic status and may contribute to delay PAD recognition. 38 Studies have shown that patients who are uninsured, underinsured, or have Medicaid receive a lower quality of hospital care 36 and may have worse surgical outcomes and mortality.39,40 Future studies are needed to understand the role socioeconomic factors play on both sides of the physician–patient interaction.
A final piece of dogma that was addressed in this report was the concept that subjects with early PAD either do not experience or under-report claudication symptoms. Previously, it was reported that only 11% of ambulatory PAD subjects report ‘classic’ claudication. 41 These findings have been used to partially explain the high rates of PAD under-diagnosis observed in subsequent studies. 3 Presently, we found that fully 50% of subjects with unrecognized PAD will report symptoms which are suggestive of claudication when directly asked. While some of these complaints may not be ischemic in nature, they could be used as a screening tool to trigger additional queries which could lead to the identification of vascular disease. Further, this group will report significantly lower values on each of the three WIQ survey categories, clearly indicating that they subjectively sense an evolving difficulty with ambulation. Although we and others continue to focus on novel biomarkers and more sensitive screening tests to help identify those at-risk individuals with PAD,16,19,42 it is important to have a heightened awareness of the high prevalence of unrecognized PAD and note the power of a review of systems in the general at-risk population. We would advocate for a greater awareness of the psychological and socioeconomic barriers to PAD identification, as it may be that individuals of a lower socioeconomic status may benefit from targeted screening.
Limitations
This study was conducted in a cohort of high-risk individuals at two academic medical centers referred for coronary angiography, and may not be generalizable to a broader patient cohort or stable outpatients. Additional limitations of this study include the determination of a clinical history of PAD, which was either self-reported or ascertained by the interviewer at the time of GenePAD data collection. Patients, therefore, may not be fully aware of their own diagnosis and could have incorrectly been considered to be unrecognized. Additionally, we define symptoms suggestive of claudication according to the WIQ rather than other commonly used questionnaires such as the Rose Questionnaire, and non-vascular conditions such as arthritis or radiculopathy could have therefore been misconstrued as claudication and inappropriately reported as such. The prevalence of unrecognized PAD in this study may be underestimated due to the use of rest rather than exercise ABI.
Conclusion
Among patients referred for coronary angiography, the prevalence of unrecognized PAD is high despite prior evaluation by a cardiovascular specialist and the presence of symptoms suggestive of claudication. Traditional cardiovascular risk factors are highly prevalent in this cohort and subjects with unrecognized PAD are at elevated risk of future cardiovascular events. Screening ABIs in a high-risk cardiovascular cohort could be useful to identify additional subjects with occult PAD and reinforce the need for anti-atherosclerotic therapy. Unrecognized PAD is associated with lower income, lower education level, social isolation and depression. Greater awareness of the socioeconomic factors associated with unrecognized PAD may decrease PAD-associated morbidity and mortality by facilitating earlier recognition and treatment.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by a grant from the National Heart, Lung, and Blood Institute (K12HL087746 to JPC).
