Abstract
Objectives:
Asymmetric dimethylarginine (ADMA) is an endogenous inhibitor of nitric oxide synthase. The breakdown of ADMA is mainly governed by the activity of dimethylarginine dimethylaminohydrolases (DDAHs). We investigated if genetic variation in the DDAH1 and DDAH2 genes were related to ADMA and
Methods:
In 1016 70-year-old participants of the population-based Prospective Investigation of the Vasculature in Uppsala Seniors (PIVUS) study (50% women), we measured endothelium-dependent vasodilation (EDV) using the invasive forearm technique with acetylcholine given in the brachial artery and the brachial artery ultrasound technique with measurement of flow-mediated dilatation (FMD). Plasma
Results:
Several of the genotypes in the DDAH1 gene were highly significantly related to ADMA levels (p = 10−7 at best), but not to the
Conclusion:
A close relationship was seen between SNPs in the DDAH1, but not DDAH2, gene and ADMA levels. However, variation in those genes was not related to measures of EDV in this elderly population.
Introduction
Ever since the discovery by Furchgott and co-workers in 1980 that the endothelium possesses vasoactive properties, and the later discovery that the main vasodilatory compound was nitric oxide (NO) derived from the amino acid
ADMA is generated during proteolysis of various proteins containing methylated arginine residues. The methylation step is carried out by enzymes denoted protein-arginine methyltransferases (PRMTs). 6
The breakdown of ADMA on the other hand is mainly governed by the activity of dimethylarginine dimethylaminohydrolase (DDAH), 7 and it has been estimated that more than 70% of ADMA is metabolized by DDAH. 7 Therefore, it has been suggested that therapeutic action targeting DDAH activity could be a way to improve vascular health. 8 Two isoforms of DDAH exist, but the relative role of these two forms of DDAH in the breakdown of ADMA and thereby putative effects on endothelium-dependent vasodilation has not been well characterized in man. 9
Today, flow-mediated vasodilation (FMD) of the brachial artery is the most commonly used method for the study of endothelium-dependent vasodilation (EDV) in humans.10,11 This method uses shear stress as the stimulus for NO production in a conduit artery. Apart from the measurement of FMD, EDV has also frequently been evaluated in the forearm vasculature by use of intra-brachial infusion of acetylcholine, mainly evaluating the function of the resistance arteries. 12 Both of these techniques have shown endothelial vasodilatory dysfunction to predict future cardiovascular events.13–15
We have previously shown that the
Methods
Subjects
Eligible were all individuals aged 70 years living in the community of Uppsala, Sweden. The subjects were chosen from the register of community living and were invited in a randomized order from the start of the study in April 2001 to the last included subject in June 2004. 17 The subjects received an invitation by letter within 1 month of their 70th birthday in order to standardize for age. Of the 2025 subjects invited, 1016 subjects were investigated, giving a participation rate of 50.1%. The study was approved by the Ethics Committee of the University of Uppsala and the participants gave informed consent.
Baseline investigation
The participants were asked to answer a questionnaire about their medical history, smoking habits and regular medication. All subjects were investigated in the morning after an overnight fast. No medication or smoking was allowed after midnight. After recordings of height, weight, abdominal and hip circumference, an arterial cannula was inserted in the brachial artery for blood sampling and later regional infusions of vasodilators. During the investigation, the subjects were supine in a quiet room maintained at a constant temperature. Blood pressure was measured by a calibrated mercury sphygmomanometer in the non-cannulated arm to the nearest mmHg after at least 30 minutes of rest and the average of three recordings was used. Lipid variables and fasting blood glucose were measured by standard laboratory techniques.
Clinical characteristics are given in Table 1. Approximately 10% of the cohort reported a history of coronary heart disease, 4% reported stroke and 12% diabetes mellitus. Almost half of the cohort reported use of any cardiovascular medication (45%), with antihypertensive medication being the most prevalent (31%). Fifteen percent reported use of statins, while use of insulin and oral antiglycemic drugs were reported by 2% and 6%, respectively.
Basic characteristics, major cardiovascular risk factors and measures of endothelium-dependent vasodilation in the total sample (n = 1016).
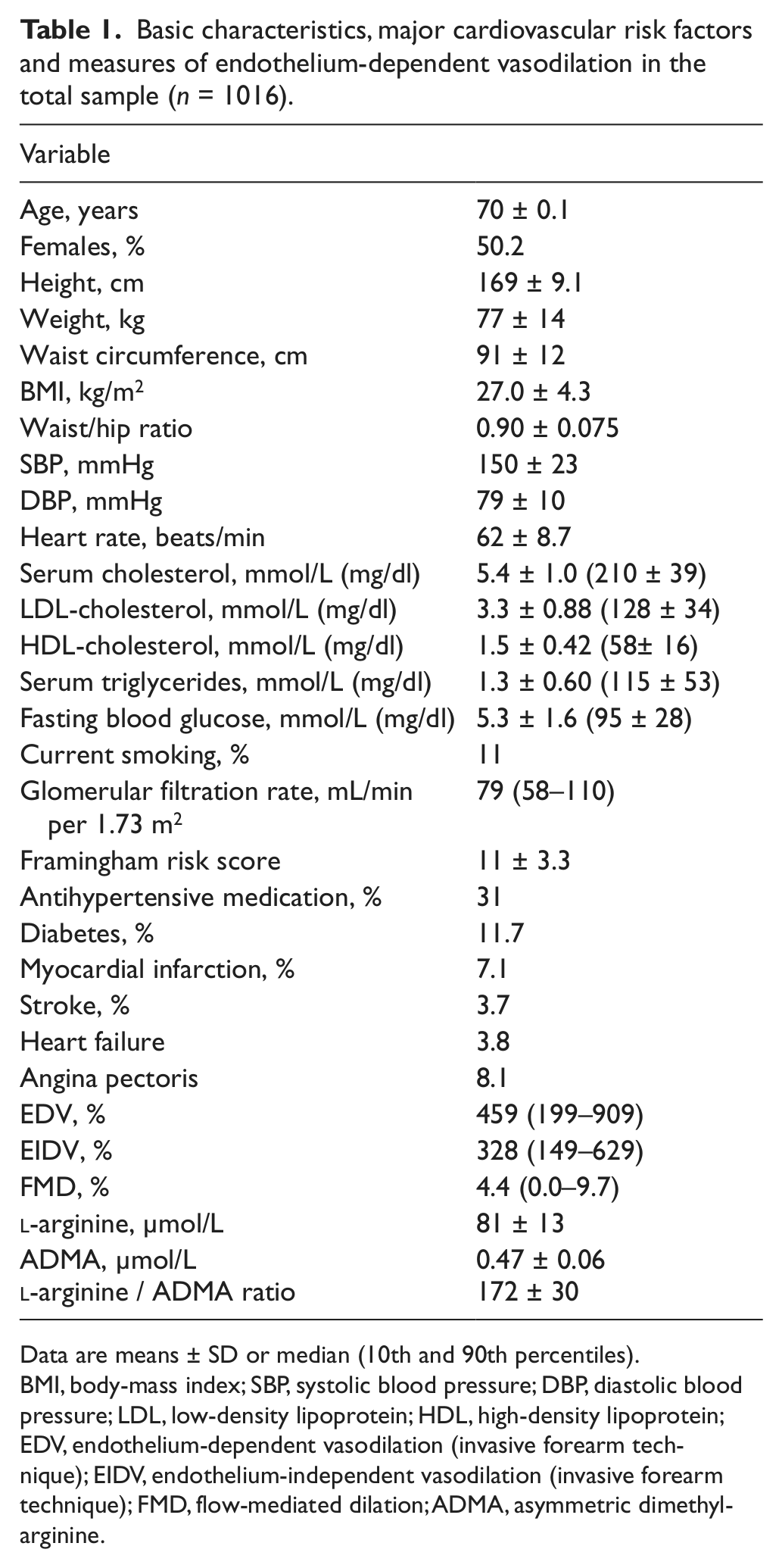
Data are means ± SD or median (10th and 90th percentiles).
BMI, body-mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; LDL, low-density lipoprotein; HDL, high-density lipoprotein; EDV, endothelium-dependent vasodilation (invasive forearm technique); EIDV, endothelium-independent vasodilation (invasive forearm technique); FMD, flow-mediated dilation; ADMA, asymmetric dimethylarginine.
As the participation rate in this cohort was only 50%, we carried out an evaluation of cardiovascular disorders and medications in 100 consecutive non-participants. The prevalences of cardiovascular drug intake, history of myocardial infarction, coronary revascularization, antihypertensive medication, statin use and insulin treatment were similar to those in the investigated sample, while the prevalences of diabetes, congestive heart failure and stroke tended to be higher among the non-participants (see ref. 17 for details).
The invasive forearm technique
Forearm blood flow (FBF) was measured by venous occlusion plethysmography (Elektromedicin, Sweden) with the strain-gauge technique. FBF was calculated from at least five consecutive recordings. An arterial cannula was placed in the brachial artery. Resting FBF was measured 30 minutes after cannula insertion, and local intra-arterial drug infusions were given during 5 minutes for each dose with a 20-minute washout period between the drugs. The infused dosages were 25 and 50 µg/minute for acetylcholine (Clin-Alpha, Switzerland) and 5 and 10 µg/minute for sodium nitroprusside (Nitropress; Abbot, UK). The drugs were given in a random order at a maximal rate of 1 mL/min. EDV with this technique was defined as FBF during infusion of acetylcholine minus resting FBF divided by resting FBF. Endothelium-independent vasodilation (EIDV) was defined as FBF during infusion of 10 µg/min of sodium nitroprusside minus resting FBF divided by resting FBF. Cannulation of the artery was not performed in the 3% of subjects who were on regular treatment with warfarin and in another 10% cannulation failed. The reproducibility (coefficient of variation, CV) for EDV and EIDV was 8–10%. 18
The brachial artery ultrasound technique
The brachial artery was assessed by external B-mode ultrasound imaging 2–3 cm above the elbow (Acuson XP128 with a 10-MHz linear transducer; Acuson, Mountain View, CA, USA), according to the International Brachial Artery Reactivity Task Force. 11 A cuff was placed below the elbow and inflated to a pressure at least 50 mmHg above systolic blood pressure for 5 minutes. FMD was defined as the maximal brachial artery diameter recorded between 30 and 90 seconds following cuff release minus diameter at rest divided by the diameter at rest, using electronic callipers for measurements. FMD was successfully evaluated in 97% of the participants. The reproducibility (CV) was 3% for baseline brachial artery diameter and 29% for FMD. 19
Each vasodilatory technique was performed and evaluated by one unique technician throughout the study who was not aware of other data. A more detailed description of the study sample and techniques has previously been presented. 17
Measurement of ADMA and l -arginine
EDTA-plasma samples were stored at −80°C until analysis. Plasma concentrations of ADMA and arginine were measured by high-performance liquid chromatography (HPLC) with fluorescence detection as previously described, 20 with modified chromatographic separation conditions. 21 Inter-assay coefficients of variation were < 3% for both ADMA and arginine.
Genotyping of DDAH1 and DDAH2
Single nucleotide polymorphisms (SNPs) were selected from the dbSNP database (www.ncbi.nlm.nih.gov/SNP/) to be evenly distributed over a region of about 10 kb upstream and 5 kb downstream from the two genes to be investigated to cover the major variation in SNPs with a minor allele frequency (MAF) > 5%.
The SNPs were genotyped at the SNP technology platform at Uppsala University (www.genotyping.se/) using the Golden Gate assay from Illumina, Inc.
Using the HapMap population of European ancestry (CEU) as a reference, for the 162 kb genomic segment encompassing the gene DDAH1, there were 168 SNPs with MAF exceeding 5%. The SNPs genotyped in this study could tag 158 of 168 SNPs (94%) in the region with an r2 > 0.8. Further, the 18 kb genomic segment encompassing the gene DDAH2 had 11 SNPs with a MAF exceeding 5%. The SNPs genotyped in this study could tag all 11 SNPs (100%) in the region with an r2 > 0.8.
The average call-rate per SNP was 98.97%, with a no-call rate < 95%. All SNPs were in Hardy–Weinberg equilibrium. A 99.99% reproducibility was seen (one duplicate conflict in 8602 duplicate tests) and a 100% consistent inheritance was found (0 inheritance conflicts in 576 inheritance tests).
Statistics
Non-normally distributed variables (i.e. EDV, EIDV, and FMD) were log-transformed in order to achieve a normal distribution. Relationships between the continuous variables and the genotypes were evaluated by linear regression analysis adjusting for gender. We also performed an additional adjustment for the major cardiovascular risk factors and diseases: age, gender, history of coronary heart disease, history of stroke, hypertension, diabetes, body mass index (BMI), high-density lipoprotein (HDL)- and low-density lipoprotein (LDL)-cholesterol and smoking. An allelic additive effect was assumed for the genotypes. Altogether, 330 tests were performed, giving a Bonferroni-adjusted p-value of 0.05/330 = 0.000151.
Results
DDAH genotypes and l -arginine and ADMA levels
Several of the genotypes in the DDAH1 gene were significantly related to ADMA levels (rs11161606, rs12132677, rs13373844, rs233099, rs233109, rs3768227, rs11161614, rs12140935, rs3768226, rs527762, rs582145, rs6669293, rs669173, rs7521189), but not to the
Relationship between SNPs in the DDAH1 gene and circulating levels of ADMA,
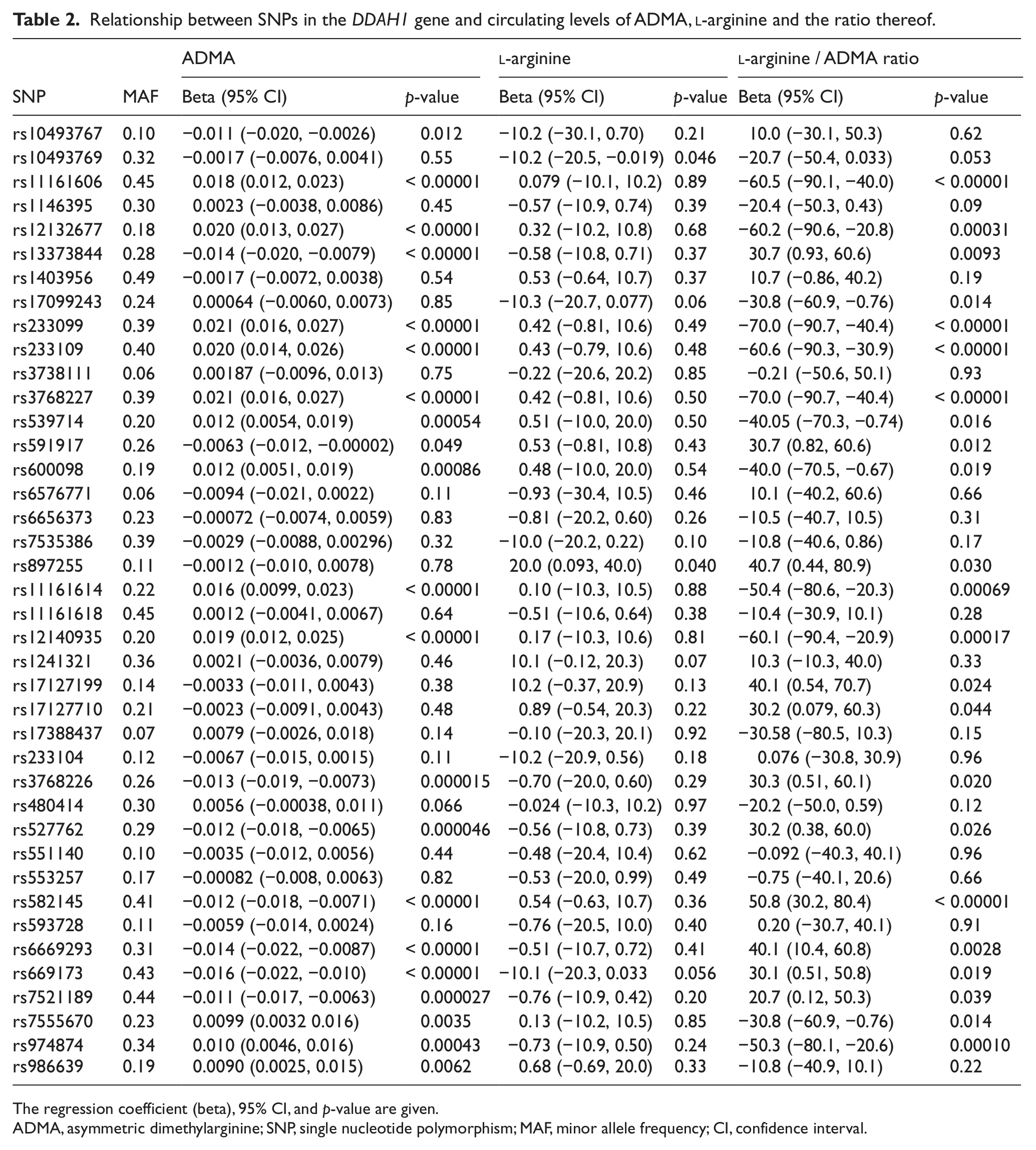
The regression coefficient (beta), 95% CI, and p-value are given.
ADMA, asymmetric dimethylarginine; SNP, single nucleotide polymorphism; MAF, minor allele frequency; CI, confidence interval.
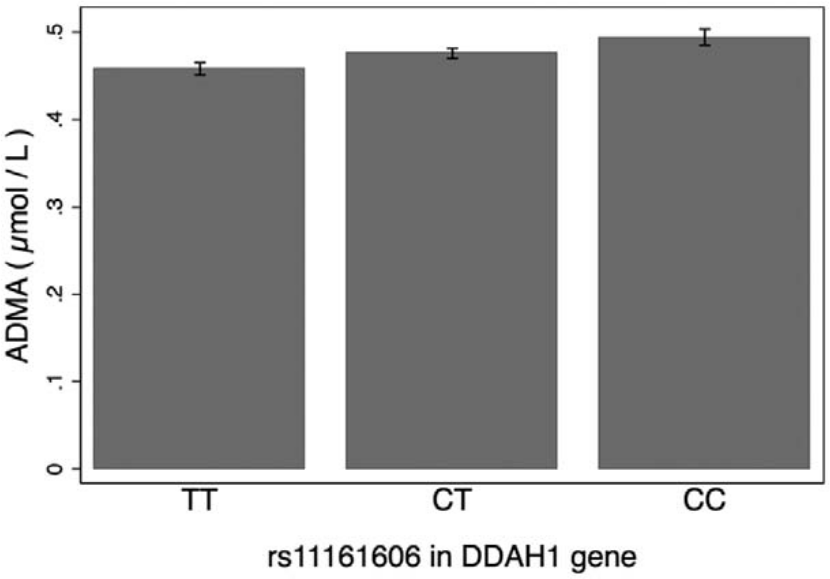
Means and 95% confidence intervals (CIs) for ADMA levels in one of the SNPs in the DDAH1 gene (p < 0.00001 for differences between groups). (n = 315 for TT, n = 473 for CT and n = 209 for CC.)
All of the reported highly significant relationships between the different DDAH1 SNPs and ADMA levels were still highly significant following adjustment for age, gender, history of coronary heart disease, history of stroke, hypertension, diabetes, BMI, HDL- and LDL-cholesterol and smoking.
All SNPs in the DDAH1 gene that were highly significantly related to the ADMA levels were entered as independent variables (together with gender) in a backward stepwise linear regression model with ADMA as the dependent variable. Of the initial 16 SNPs included in the model, only three remained significant following the backward stepwise procedure (rs233109, rs12140935 and rs6669293) and were not in high linkage disequilibrium (LD) (r2 < 0.25 for the pairwise correlations).
No relationships between the genotypes in the DDAH2 gene and ADMA or
Relationship between SNPs in the DDAH2 gene and circulating levels of ADMA,
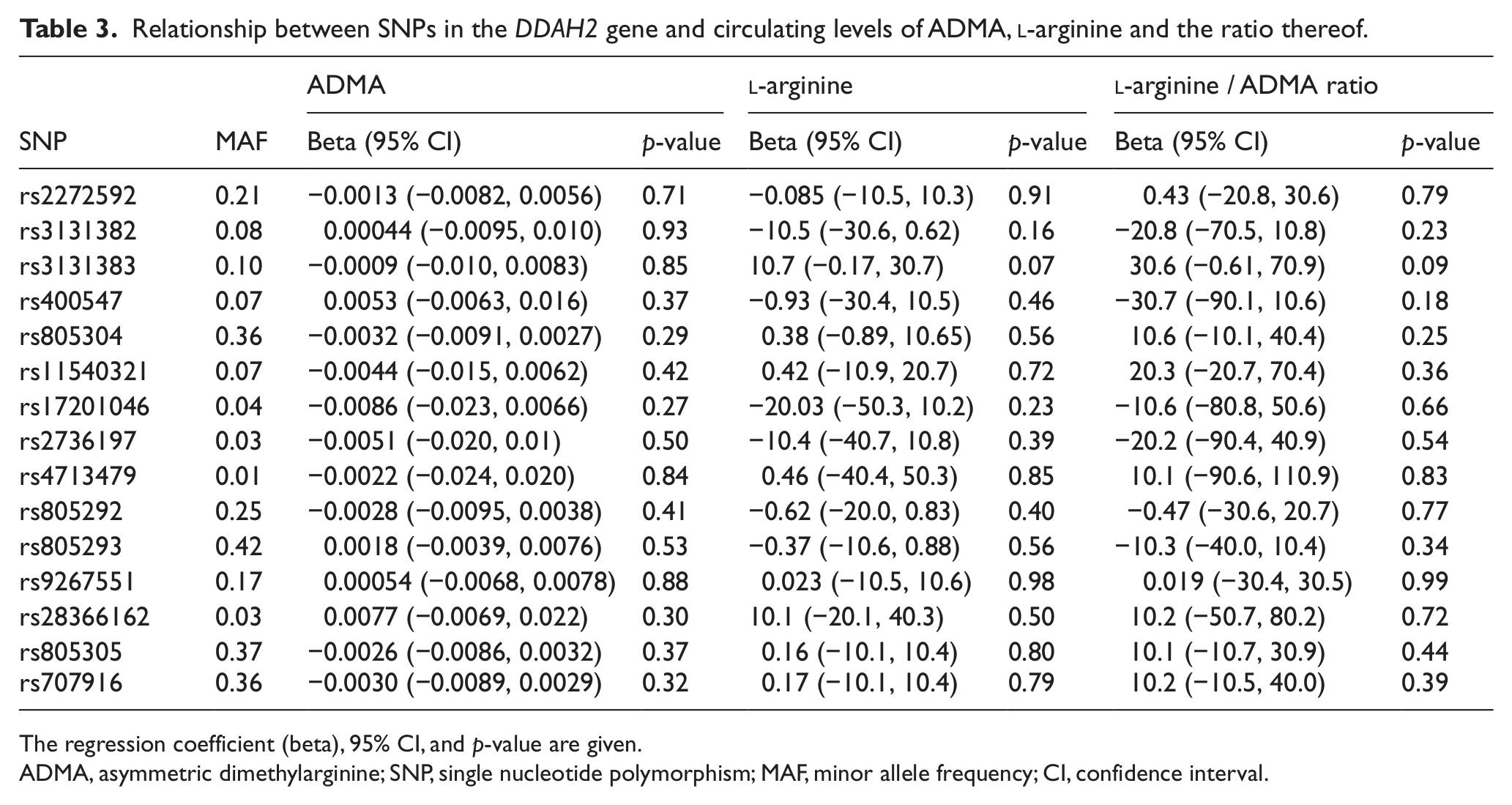
The regression coefficient (beta), 95% CI, and p-value are given.
ADMA, asymmetric dimethylarginine; SNP, single nucleotide polymorphism; MAF, minor allele frequency; CI, confidence interval.
DDAH genotypes and vasoreactivity
None of the DDAH1 genotypes being closely related to ADMA levels were significantly related to EDV or FMD (Table 4). Neither were any of the DDAH2 genotypes significantly related to any of the measurements of vasoreactivity (Table 5).
Relationships between SNPs in the DDAH1 gene and EDV, EIDV and FMD.
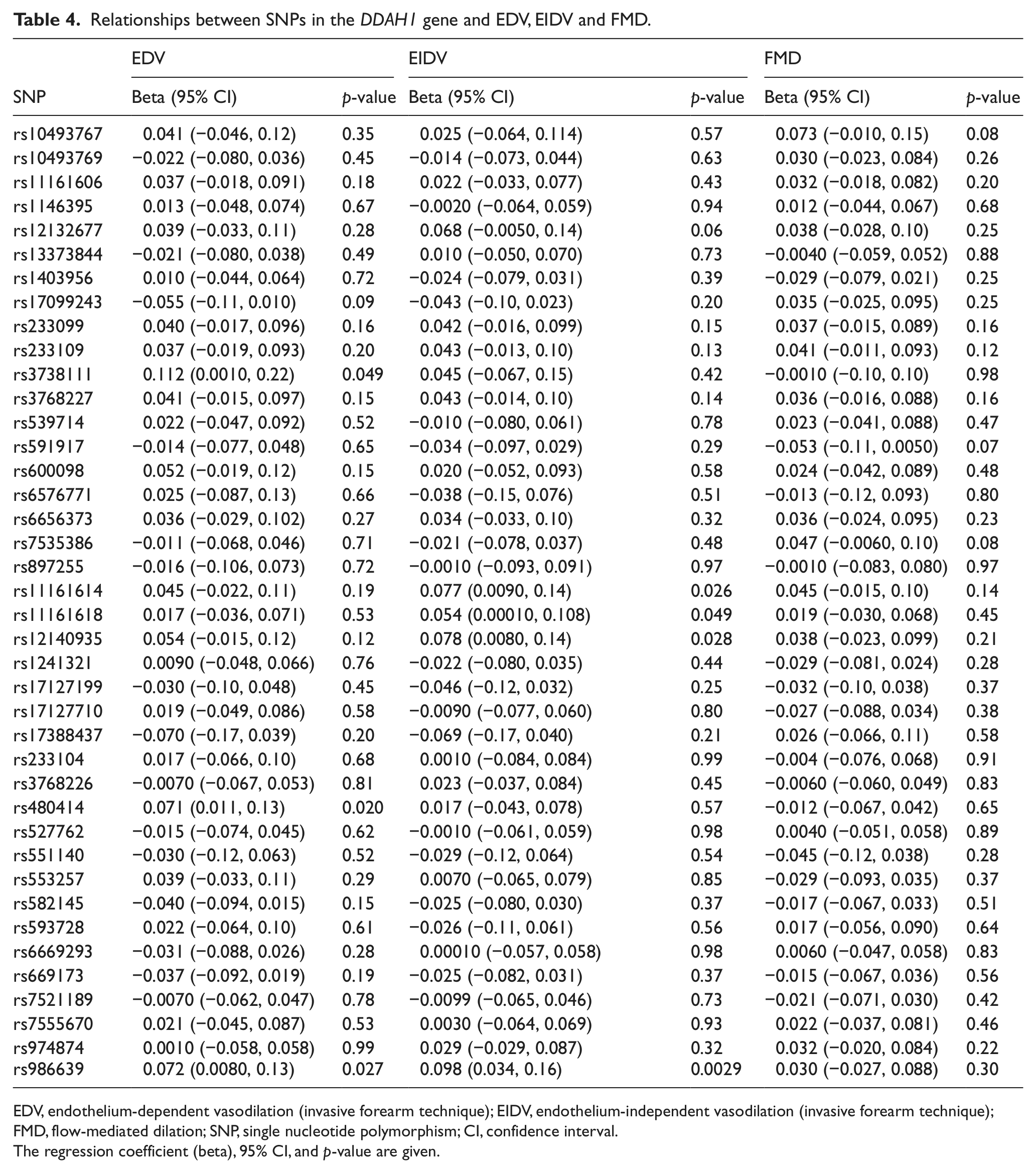
EDV, endothelium-dependent vasodilation (invasive forearm technique); EIDV, endothelium-independent vasodilation (invasive forearm technique); FMD, flow-mediated dilation; SNP, single nucleotide polymorphism; CI, confidence interval.
The regression coefficient (beta), 95% CI, and p-value are given.
Relationships between SNPs in the DDAH2 gene and EDV, EIDV and FMD.
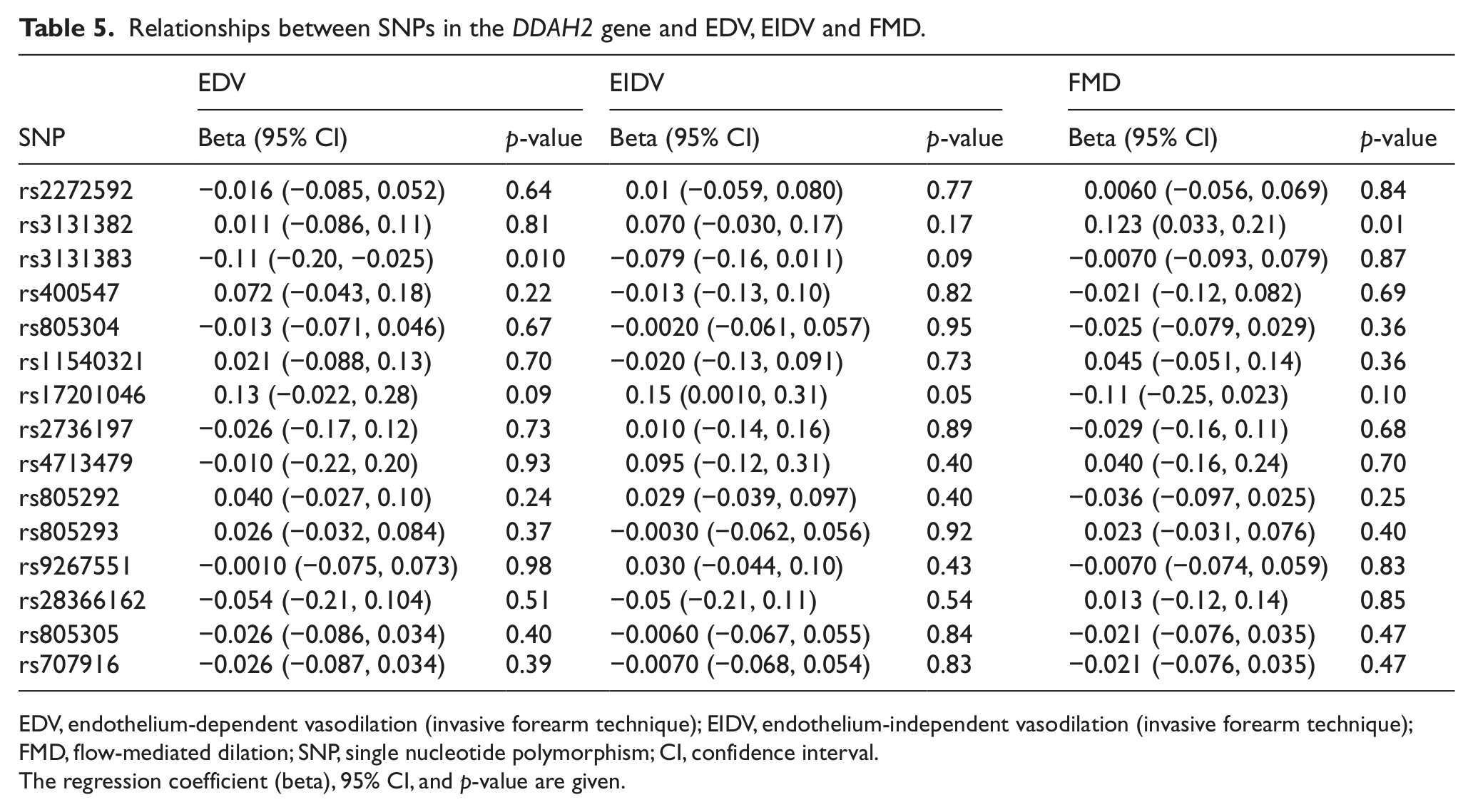
EDV, endothelium-dependent vasodilation (invasive forearm technique); EIDV, endothelium-independent vasodilation (invasive forearm technique); FMD, flow-mediated dilation; SNP, single nucleotide polymorphism; CI, confidence interval.
The regression coefficient (beta), 95% CI, and p-value are given.
No significant relationships between DDAH1 or DDAH2 SNPs and EDV were disclosed, even when we excluded subjects with hypertension, diabetes or cardiovascular disease from the analysis, either all together or only the specific diseases.
Discussion
The present study showed that several DDAH1 genotypes were closely related to ADMA levels, but not to measures of EDV in an elderly population. Variation in the DDAH2 gene was neither related to ADMA levels, nor to vasoreactivity.
Genetic variation in the DDAH genes and ADMA levels
It has been estimated that more than 70% of the clearance of ADMA is due to DDAH activity.
7
It has been estimated in experimental studies that the apparent rate of ADMA metabolism is 70 times higher for DDAH1 compared to DDAH2.9,22 Using gene silencing techniques, disruption of DDAH1 gene activity induced a 50% increase in ADMA levels, while similar silencing of DDAH2 did not have any effects.
23
These data were supported by another laboratory.
9
Plasma and tissue ADMA and
The present study supports the view that DDAH1 is more important than DDAH2 in the metabolism of ADMA also in humans. Few other studies have investigated the role of genetic variation of the DDAH1 and DDAH2 genes and ADMA levels in humans. Recently, a study in diabetics showed SNPs in both of these genes to be related to ADMA levels. 25 In contrast, preliminary data from another study could not confirm the role of DDAH2 regarding ADMA levels. 26 Differences in SNPs investigate, or differences in populations resulting in different ADMA levels might be of importance to explain, the discrepancies regarding the results relating to the role of DDAH2 in humans.
Many of the SNPs analyzed in the DDAH1 gene were significantly related to ADMA levels. In most cases this was due to a high LD. However, three of these SNPs were not in high LD and were independently of each other related to ADMA levels, suggesting that these SNPs, or other SNPs in high LD with those, could cause different functional changes in the DDAH1 gene.
Genetic variation in the DDAH genes and vasoreactivity
In the gene silencing experiments described above, it was also seen that while DDAH1 silencing induced a major effect on ADMA levels, no effect on EDV was seen. On the contrary, silencing of DDHA2 did not affect ADMA levels, but attenuated EDV. 23 However, in experiments using electron paramagnetic resonance (EPR) spin trapping to determine NO production from the endothelium, silencing of both DDAH1 and DDAH2 induced a reduction in NO. 9
Overexpression of DDAH1 or DDAH2 in human umbilical vein endothelial cells increased DDAH activity, reduced ADMA concentrations and increased NO production. Similarly, overexpression of DDAH1 or DDAH2 in DDAH1 knock-out mice carotid vessels increased NO production and enhanced acetylcholine (ACh) relaxation. 27 Overexpression of human DDAH-1 in transgenic mice markedly reduced ADMA levels and increased acetylcholine-mediated vasodilation. The attenuation of acetylcholine-mediated vasodilation by exogenous ADMA was not seen in these transgenic mice when compared to wild-type mice. 28 Mice with selective knock-out of DDAH1 in the endothelium exhibited significantly attenuated acetylcholine-induced NO production and vessel relaxation in isolated aortic rings. 29
The present study found no relationship between SNPs in either DDAH1 or DDAA2 and vasoreactivity. In the case of DDAH1 this was seen despite a close relationship with ADMA levels. This supports previous experimental studies questioning the role of ADMA in EDV. To the best of our knowledge, no other studies have evaluated DDAH genotypes in relation to EDV in humans.
In elderly experimental animals, especially in resistance arteries, NO production is reduced and a yet unidentified endothelial hyperpolarizing might be a major player in acetylcholine-mediated vasodilation. 30 Also, Prostacyclin (PGI2) might play a more important role for vasodilation than NO in this elderly cohort. 31 This might explain why DDAH1 polymorphisms are related to ADMA levels, but not to EDV.
Limitations of the study
The present sample is limited to Caucasians aged 70 years. Caution should therefore be made to draw conclusions to other ethnic and age groups. The PIVUS study had a moderate participation rate. However, an analysis of non-participants showed the present sample to be fairly representative of the total population regarding most cardiovascular disorders and drug intake.
In the Hoorn Study, a large community-based study among men and women aged 50–75 years, the mean (SD) ADMA concentration, measured with the same method, was 0.50 (0.06) μmol/L. 32 In the present study, the mean ADMA concentration was slightly lower, but with a strikingly similar narrow distribution. Despite the large number of subjects and the very high precision of the HPLC method, this narrow concentration distribution may have limited our capacity to detect a possible true association between ADMA and genetic variation. 33
The measurements of EDV were slightly lower than seen in younger subjects. 17
It cannot be excluded that none of the SNPs for DDAH2 included in the present study are of functional importance and therefore the magnitude of DDAH2 activity on ADMA metabolism or endothelium-dependent vasodilation could not be properly investigated.
In conclusion, a close relationship was seen between SNPs in the DDAH1, but not DDAH2, gene and ADMA levels. However, variation in those genes was not related to measures of EDV in this elderly population.
Footnotes
Acknowledgements
The outstanding work at the endothelium laboratory performed by Nilla Fors, Jan Hall, Kerstin Marttala and Anna Stenborg in the collection and processing of the data and the skillful analysis of ADMA and arginine by Sigrid de Jong is highly acknowledged.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
