Abstract
Suppression of dimethylarginine dimethylaminohydrolase (DDAH) activation is related to endothelial dysfunction in hyperlipidemia, and nonmuscle myosin regulatory light chain (nmMLC20) has been show to exert transcriptional function in regulation of gene expression. This study aims to explore whether the suppression of DDAH activation promotes endothelial injury under the condition of hyperlipidemia and whether nmMLC20 can regulate DDAH expression in a phosphorylation-dependent manner. The rats were fed with high-fat diet for 8 weeks to establish a hyperlipidemic model, which showed an increase in plasma lipids and endothelial injury, accompanied by an elevation in myosin light chain kinase (MLCK) activity, phosphorylated nmMLC20 (p-nmMLC20) level, and asymmetric dimethylarginine (ADMA) content as well as a reduction in DDAH2 expression, DDAH activity, and nitric oxide (NO) content. Next, human umbilical vein endothelial cells (HUVECs) were incubated with oxidized low-density lipoprotein (ox-LDL; 100 μg/mL) for 24 hours to establish a cellular injury model in vitro. Consistent with the finding in vivo, ox-LDL induced HUVECs injury (apoptosis and necrosis) concomitant with an increase in MLCK activity, p-nmMLC20 level (in total or nuclear proteins), and ADMA content as well as a reduction in DDAH2 expression, DDAH activity, and NO content; these phenomena were attenuated by MLCK inhibitor. Either in hyperlipidemic rats or in ox-LDL-treated HUVECs, there was not significant change in DDAH1 expression. Based on these observations, we conclude that the suppression of DDAH2 expression might account for, at least partially, the vascular endothelial dysfunction in hyperlipidemia, and nmMLC20 plays a role in suppression of DDAH2 expression in a phosphorylation-dependent manner.
Keywords
Introduction
It is well recognized that endothelial function is impaired in humans and animals with hyperlipidemia. Endothelial dysfunction is an early event for many of the cardiovascular diseases such as atherosclerosis, coronary heart disease, and hypertension. 1,2 In hyperlipidemia, the elevated levels of plasma lipids, particularly the oxidized low-density lipoprotein (ox-LDL), play a key role in promotion of vascular endothelial dysfunction. There is evidence that the detrimental effects of ox-LDL on endothelial dysfunction is related to the inhibition of nitric oxide (NO) generation, 3,4 but the exact mechanisms are not fully elucidated.
Asymmetric dimethylarginine (ADMA), an endogenous nitric oxide synthase (NOS) inhibitor, can inhibit the activity of NOS by competing with L-arginine to decrease NO generation. Thus, ADMA is emerging as a risk factor for endothelial dysfunction, and the elevation of ADMA level in patients with hyperlipidemia or hyperlipidemic animals might account for the decrease in NO production. 5,6 Asymmetric dimethylarginine is metabolized by dimethylarginine dimethylaminohydrolase (DDAH) to produce L-citrulline and dimethylamine. There are 2 isoforms, DDAH1 and DDAH2, for DDAH. 7 Accumulating evidence has demonstrated that the elevated plasma levels of ADMA in many cardiovascular diseases including hyperlipidemia are due to, at least partially, the decrease in DDAH activity. 8 –11 Although the activities of DDAH have been reported to be reduced in hyperlipidemic animals, it is not known whether the decrease in DDAH activities involves the suppression of DDAH gene expression.
Myosin regulatory light chain (MLC20) is a subunit of myosin with a molecular weight of 20 kD. The isoform of MLC20 in nonmuscle cells is named as nonmuscle MLC20 (nmMLC20). 12 Recent studies from other laboratories and ours have demonstrated that nmMLC20 could act as a transcription factor to modulate gene expression through a phosphorylation-dependent manner, which is mainly controlled by myosin light chain kinase (MLCK). 13,14 In rabbits fed by high-cholesterol diet, the MLCK activity and endothelial permeability in the aortas have been found to be dramatically elevated, which was attenuated by ML-7, a specific inhibitor of MLCK. 15 In the cultured human umbilical vein endothelial cells (HUVECs) incubated with ox-LDL, MLCK expression was upregulated in a time- and dose-dependent manner. 16 Based on these reports as well as the transcriptional function of nmMLC20, we speculate that nmMLC20 may involve the suppression of DDAH expression through raising the levels of phosphorylated nmMLC20 (p-nmMLC20) in patients with hyperlipidemia or hyperlipidemic animals, resulting in the decrease of DDAH activities while increase of ADMA levels.
The main purposes of this study were to explore the role of nmMLC20 in vascular endothelial dysfunction in hyperlipidemia and to elucidate the underlying mechanisms. Using a rat model of hyperlipidemia, we evaluated the status of endothelial function and the association of p-nmMLC20 with DDAH (DDAH1 and DDAH2) expression. Using a model of oxLDL-induced HUVECs injury as well as the MLCK inhibitor, we verified the downregulation of DDAH2 by ox-LDL and the function of nmMLC20 in suppression of DDAH2 expression in a phosphorylation-dependent manner.
Methods
Animals
Male Sprague-Dawley (SD) rats (120-130 g) were purchased from the Laboratory Animal Center, Xiang-Ya, School of Medicine, Central South University, China. All animals received humane care in compliance with the “Guide for the Care and Use of Laboratory Animals” published by the National Institutes of Health (NIH Publication, 8th edition, 2011), and the protocol was approved by the Central South University Veterinary Medicine Animal Care and Use Committee.
Experiments in Animals
SD rats were randomly divided into the control and the hyperlipidemia group (n = 9 per group). Animals in the control group or in the hyperlipidemia group were fed on standard diet or high-fat diet (including 66.7% basic diet, 10% milk powder, 10% lard, 10% refined sugar, 3% cholesterol, and 0.3% sodium cholate) for 8 weeks. At the end of 8 weeks, rats were weighed and blood samples from all animals (∼5 mL/per rat) were collected in a sterile tube containing the anticoagulant heparin for plasma isolation under the condition of anesthesia (sodium pentobarbital, 30 mg/kg, intraperitoneally) after overnight fasting.
Determination of Plasma Lipids
The levels of total cholesterol (TC), total triglycerides (TG), low-density lipoprotein (LDL), and high-density lipoprotein (HDL) in plasma were measured spectrophotometrically following the instructions provided by the supplier of diagnostic kits (Jiancheng Institute of Biotechnology, Nanjing, China). For measuring the plasma level of ox-LDL, a commercial ELISA kit was utilized (R&D Systems, Minneapolis, Minnesota).
Isolated Rat Aortic Ring Assay
After collection of the blood samples, the rat was killed under the condition of anesthesia. The thoracic aorta was quickly isolated for measurement of the vasodilator responses according to a protocol we described previously. 4 Briefly, the aortic ring in ∼3 mm length was suspended horizontally between 2 stainless steel wires and mounted in a 5 mL organ chamber filled with warmed and oxygenated Krebs solution. One of ring ends was connected to a force transducer. The aortic ring was stretched with 2 g of resting force and equilibrated for 60 minutes and then precontracted with KCl (60 mmol/L). After a maximal response to KCl was achieved, the ring was washed repeatedly with Krebs solution and equilibrated again for 30 minutes. To measure vasodilator responses, the ring was contracted with phenylephrine to 40% to 50% of its maximal contraction. Once the contraction stabilized, an accumulative concentration–response curve to acetylcholine (10−8 to 10−6 mol/L) was recorded. The concentration–response curve to sodium nitroprusside was performed in our preliminary experiments to exclude the effect of hyperlipidemia on smooth muscle function (data not shown).
Immunohistochemical Assay
To evaluate the endothelial injury of the aorta, the Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL)-based immunohistochemical staining was utilized. The aortic tissues were fixed in 4% paraformaldehyde and embedded in paraffin and then cut into 5-μm sections. The procedure for TUNEL staining was carried out following the manufacturer’s instructions (Roche, Nutley, New Jersey). Lack of terminal transferase in TUNEL reaction mixture served as a negative control. The aortic slices were observed under microscope at 200× magnification and photographed by a high-resolution digital camera (Nikon Eclipse 80i, Tokyo, Japan). Ten random high-power fields from each aortic sample were selected and blindly quantitated. The results were presented as TUNEL-positive cell numbers per mm2 of aortic tissues.
To evaluate the protein levels of DDAH (DDAH1 and DDAH2), p-nmMLC20, and nmMLC20 in endothelium of the aorta, immunohistochemical staining was performed. Briefly, the aorta slices were treated with 0.3% hydrogen peroxide to quench endogenous peroxidase activity and incubated with phosphate-buffered saline (PBS) containing 1% Triton X-100 and 1% normal rabbit serum for 2 hours. Then, the slices were incubated with the primary antibody against rat DDAH1 (Abcam, Cambridge, Massachusetts), DDAH2 (Abcam), p-nmMLC20 (Invitrogen, Thermo Fisher, Grand Island, New York), or nmMLC20 (Santa Cruz, California) for 12 hours followed by the second antibody for extra 2 hours. Diaminobenzidine was employed for color reaction to detect positive signal according to routine procedure. The slices were photographed by a digital camera coupled to a computer.
Experiments in Cell Cultures
The in vitro experiments were designed to verify the role of p-nmMLC20 in endothelial injury and the correlation between the p-nmMLC20 and DDAH2 expression. To mimic the condition of endothelial injury in hyperlipidemic rats, HUVECs (ATCC CRL-1730, Virginia) were incubated with ox-LDL to induce injury (apoptosis and necrosis). The cells were divided into 3 groups (6 individual experiments per group): the control group, HUVECs were cultured under normal conditions; the ox-LDL group, HUVECs were incubated with ox-LDL (100 μg/mL, Yiyuan Biotech, Guangzhou, China) for 24 hours; the ox-LDL plus ML-7 (a specific inhibitor of MLCK, dissolved in water, Sigma Aldrich, Shanghai, China) group, ML-7 was added to the culture medium (2 μmol/L, final concentration) before the ox-LDL treatment, and it was incubated with HUVECs till the end of ox-LDL treatment; 24 hours after ox-LDL treatment, the culture medium was collected for measurements of lactate dehydrogenase (LDH) release, ADMA, and NO contents, while the HUVECs were saved for Hoechst staining, measurements of DDAH, MLCK and caspase 3 activities, or real-time polymerase chain reaction (PCR) and Western blot, respectively.
Evaluation of the Ox-LDL-induced HUVECs Injury
The HUVECs injury was determined by ox-LDL-induced cellular apoptosis and necrosis. Hoechst staining and caspase 3 activity was chosen for analysis of HUVECs apoptosis. The procedure for Hoechst staining was performed following the manufacturer’s instruction (Beyotime, Shanghai, China). Briefly, the HUVECs were fixed for 15 minutes in 4% paraformaldehyde, washed in PBS, air dried, and incubated at room temperature for 5 minutes with 1g/mL of Hoechst 33258, a bisbenzimide cell-permeant dye that fluorescent bright blue on binding to DNA. Stained cells were washed twice with PBS and imaged under a fluorescent microscope (excitation, 350 nm; emission, 460 nm). Twenty random high-power fields from each sample were chosen and blindly quantified. The number of apoptosis cells was presented as percentage of the total cells.
The procedure for measurement of caspase-3 activity was performed according to the manufacturer’s instructions (Beyotime, Shanghai, China). In brief, 10 μL of cell lysates was mixed with 90 μL of work solution containing caspase 3 substrate (Ac-DEVD-pNA), and then the mixture was incubated at 37°C for 60 minutes. The absorbance was recorded at 405 nm. The enzyme activity was presented as U/g protein, and 1 U was defined as the amount of enzyme required to cleave 1.0 nmol of Ac-DEVD-pNA per hour at 37°C.
The HUVECs necrosis was determined by the LDH release, which was measured with a coupled enzymatic reaction that resulted in the conversion of a tetrazolium salt into a red color formazan by diaphorase. The protocol for LDH release assay was performed according to the manufacturer’s instructions (Beyotime, Shanghai, China). In brief, 20 μL of culture medium was mixed with 60 μL of LDH work solution and incubated at 30°C for 25 minutes. Absorbance was recorded at 490 nm. Percentage of cell damage was calculated according to a formula provided by the kit supplier.
Measurement of the Activities of DDAH and MLCK
To measure the DDAH activity, the aortic tissue homogenate or HUVECs lysate (100 μL) was incubated with the work solution (4 mmol/L ADMA and 0.1 mol/L sodium phosphate buffer, 400 μL) for 2 hours at 37°C. The reaction was stopped by the addition of equal volume of 10% trichloroacetic acid, and the supernatant was boiled with diacetyl monoxime (0.8% [wt/vol] in 5% acetic acid) and antipyrine (0.5% [wt/vol] in 50% sulfuric acid). The amounts of L-citrulline formed were determined spectrophotometrically at 466 nm. As the blank, the enzyme preparations heated in a boiling water bath were subjected to the determination of the activity. The DDAH activity was presented as the amounts of L-citrulline (μmol) formation per min per gram protein of aortic tissue or HUVECs.
To measure the MLCK activity, an ELISA kit based on the principle of double-antibody sandwich technique was used (R&D Systems, Minnesota, USA). Briefly, 10 μL of aortic tissue homogenate or HUVECs lysate or culture medium together with 40 μL of sample dilution was added to microtiter plate well and incubated at 37°C for 30 minutes, followed by incubation with horseradish peroxidase (HRP)-conjugate reagent. The absorbance was read at 450 nm after adding stop solution. The MLCK activity of samples was calculated according to a standard curve made from the standard solutions provided by the supplier and presented as unit per gram protein of aortic tissue or HUVECs.
Determination of ADMA and NO Contents
The ADMA contents in the aortic tissues or culture medium were measured by high-performance liquid chromatography (HPLC) as we described previously. 4 Briefly, HPLC was carried out using a Shimadzu LC-6A liquid chromatograph with Shimadzu SCL-6A system controller and Shimadzu SIC-6A autosampler. o-Phthaldialdehyde adducts of methylated amino acids and internal standard ADMA produced by precolumn mixing were monitored using a model RF 530 fluorescence detector set at kex = 338 and kem = 425 nm on a Resolve C18 column. Samples were eluted from the column using a linear gradient containing mobile phase A composed of 0.05 mol/L (pH = 6.8) sodium acetate–methanol–tetrahydrofuran (81:18:1 v/v/v) and mobile phase B composed of 0.05 mmol/L sodium acetate–methanol–tetrahydrofuran (22:77:1 v/v/v) at a flow rate of 1 mL/min. The ADMA content in aortic tissues or culture medium was expressed as the amounts of ADAM (μmol) per gram protein of aortic tissue or per liter of culture medium.
The NO contents in the aortic tissues or culture medium were reflected indirectly by the contents of nitrite and nitrate. The procedure was performed as described previously. 17 Briefly, nitrate was converted to nitrite with aspergillus nitrite reductase, and the total nitrite was measured with the Griess reagent. The absorbance was determined at 540 nm with a spectrophotometer. The NO content in aortic tissues or culture medium was expressed as the amounts of nitrite and nitrate (μmol) formation per gram protein of aortic tissue or per liter of culture medium.
Real-Time PCR Analysis
Real-time PCR was used to determine DDAH1 and DDAH2 messenger RNA (mRNA) expression in HUVECs. β-actin served as a loading control. The primers for DDAH1, DDAH2, and β-actin were listed in Table 1. Real-time PCR was carried out according to the standard protocols. Briefly, 2 μg of total RNA from each sample was reversely transcribed to obtain the complementary DNAs (cDNAs). Then, 10 μL of reaction mixture containing 2 μL cDNA template, 5 μL SYBR Master mix, 0.20 μL ROX, 2.4 μL H2O, and 0.20 μL of each primer were amplified according to the following procedures: denaturing at 95°C for 10 minutes and 45 cycles of the amplification steps (denaturation at 95°C for 15 seconds, annealing and extension at 60°C for 1 minute). All amplification reactions were performed in triplicate, and the averages of the threshold cycles were used to interpolate curves using 7300 System SDS Software. Results were presented as the ratio of DDAH1 or DDAH2 mRNA to β-actin mRNA.
Primers for Real-Time PCR.

Abbreviations: DDAH, dimethylarginine dimethylaminohydrolase.
Western Blot Analysis
Aortic tissues or HUVECs were homogenized in ice-cool lysis buffer (250 mmol/L Tris-HCl, pH 6.8; 4% sodium dodecyl sulfate (SDS), 20% glycerol, and 1 Roche EDTA-free protease inhibitor cocktail) and sonicated for 1 minute, then centrifuged for 15 minutes at 15 000
Statistics
SPSS software (version 20) was chosen for statistical analysis. Data were expressed as means ± standard error of the mean. Differences in measured values between the 2 groups or among the multiple groups were analyzed by Student
Results
Body Weight and Plasma Lipid Profile
After 8 weeks of high-fat diet feeding, body weight of rats in the hyperlipidemia group was significantly increased compared to that in the control group (Table 2). In the hyperlipidemia group, plasma levels of lipids including TC, TG, LDL, and ox-LDL were dramatically elevated compared with that in the control group. On the contrary, plasma level of HDL in the hyperlipidemia group was markedly decreased compared with that in the control group.
Body Weight and Plasma Lipids.

Abbreviations: TC, total cholesterol; TG, total triglycerides; LDL, low-density lipoprotein; HDL, high-density lipoprotein; ox-LDL, oxidized low-density lipoprotein.
a
Endothelial Dysfunction in Hyperlipidemic Rats
To evaluate endothelial function, rat aortas were isolated to measure the vasodilator responses to acetylcholine. In presence of phenylephrine, acetylcholine (10−8 to 10−6 mol/L) caused a concentration-dependent relaxation in the isolated aorta from hyperlipidemic or control rats. In comparison to control rats, vasodilator responses to acetylcholine were significantly decreased in the hyperlipidemic rats (Figure 1A).

Endothelial dysfunction in hyperlipidemic rats. A, Representative traces for vasodilator responses of the isolated rat aortas to acetylcholine (left) and the dose–effect curve of vasodilator responses to acetylcholine (right). B, Representative images of TUNEL-based immunohistochemical staining in rat aortas (left, the TUNEL-positive cells were indicated by arrows) and quantified results of TUNEL-positive cells (right). All values are expressed as mean ± SEM (n = 9 in each group). **
In agreement with the results of the isolated aorta experiments, there was few TUNEL-positive cells within the endothelium of the aorta from control rats but TUNEL-positive cells (densely stained) were found in the aorta from hyperlipidemic rats, showing a significant increase in endothelial apoptotic cells in hyperlipidemic rats (Figure 1B).
Downregulation of DDAH2 Expression and DDAH Activity in Aorta From Hyperlipidemic Rats
Immunohistochemical staining showed that DDAH2 is present in intima and media of the rat aorta. Compare to the control rats, the signal of DDAH2 staining in the endothelium of aorta from the hyperlipidemic rats was much weaker (Figure 2A, left panel). Consistently, Western blot showed a significant downregulation of DDAH2 protein expression (Figure 2A, right panel). However, there was no significant difference in the signal of DDAH1 staining between the hyperlipidemic rats and the control rats (Figure 2B, left panel), which was confirmed by Western blot (Figure 2B, right panel). Consistent with the downregulation of DDAH2 protein expression, DDAH activity in the aorta from the hyperlipidemic rats was dramatically decreased compared to that of the control rats concomitant with the elevated ADMA content and the reduced NO content (Figure 2C–E).

Suppression of DDAH2 expression in hyperlipidemic rats. Diaminobenzidine was used in immunohistochemical staining of proteins (brown color). A, Representative images of immunohistochemical staining for DDAH2 in rat aortas (left panel, the staining of endothelium was indicated by the arrow) and Western blot analysis for aortic tissues (right panel: top, representative images; bottom, ratio of optical density between the protein bands of DDAH2 and β-actin, n = 6 per group). B, Representative images of immunohistochemical staining for DDAH1 in rat aortas (left panel, the staining of endothelium was indicated by the arrow) and Western blot analysis for aortic tissues (right panel: top, representative images; bottom, ratio of optical density between the protein bands of DDAH1 and β-actin, n = 6 per group). C, DDAH activities in rat aortas (n = 9 per group). D, ADMA contents in rat aortas (n = 9 per group). E, NO contents in rat aortas (n = 9 per group). All values are expressed as mean ± SEM. *
Elevation of MLCK Activity and p-nmMLC20 Level in Aorta From Hyperlipidemic Rats
As shown in Figure 3A, MLCK activity in aorta from the hyperlipidemic rats was significantly increased compared to that of the control rats. In agreement with the changes of MLCK activity, the signal of p-nmMLC20 staining in the endothelium of aorta from the hyperlipidemic rats was much stronger than that of the control rats (Figure 3B, left panel). Consistently, Western blot showed an elevated level of p-nmMLC20 (Figure 3B, right panel). However, there was no significant difference in the signal of nmMLC20 staining between the hyperlipidemic rats and the control rats (Figure 3C, left panel), which was confirmed by Western blot (Figure 3C, right panel).

Upregulation of MLCK activity and p-nmMLC20 level in hyperlipidemic rats. Diaminobenzidine was used in immunohistochemical staining of proteins (brown color). A, MLCK activities in rat aortas (n = 9 per group). B, Representative images of immunohistochemical staining for p-nmMLC20 (left panel, the staining of endothelium was indicated by the arrow) and Western blot analysis for aortic tissues (right panel: top, representative images; bottom, ratio of optical density between the protein bands of p-nmMLC20 and β-actin, n = 6 per group). C, Representative images of immunohistochemical staining for nmMLC20 (left panel, the staining of endothelium was indicated by the arrow) and Western blot analysis for aortic tissues (right panel: top, representative images; bottom, ratio of optical density between the protein bands of nmMLC20 and β-actin, n = 6 per group). All values are expressed as mean ± SEM. **
Inhibition of MLCK Attenuated Ox-LDL-Induced Endothelial Cell Injury In Vitro
As displayed in Figure 4, incubation of HUVECs with ox-LDL (100 μg/mL) for 24 hours was able to induce cellular apoptosis and necrosis, reflecting by an increase in Hoechst dye-positive cells (A and B), caspase-3 activity (C), and LDH release (D). Administration of MLCK inhibitor (ML-7) dramatically attenuated ox-LDL-induced HUVECs injury.
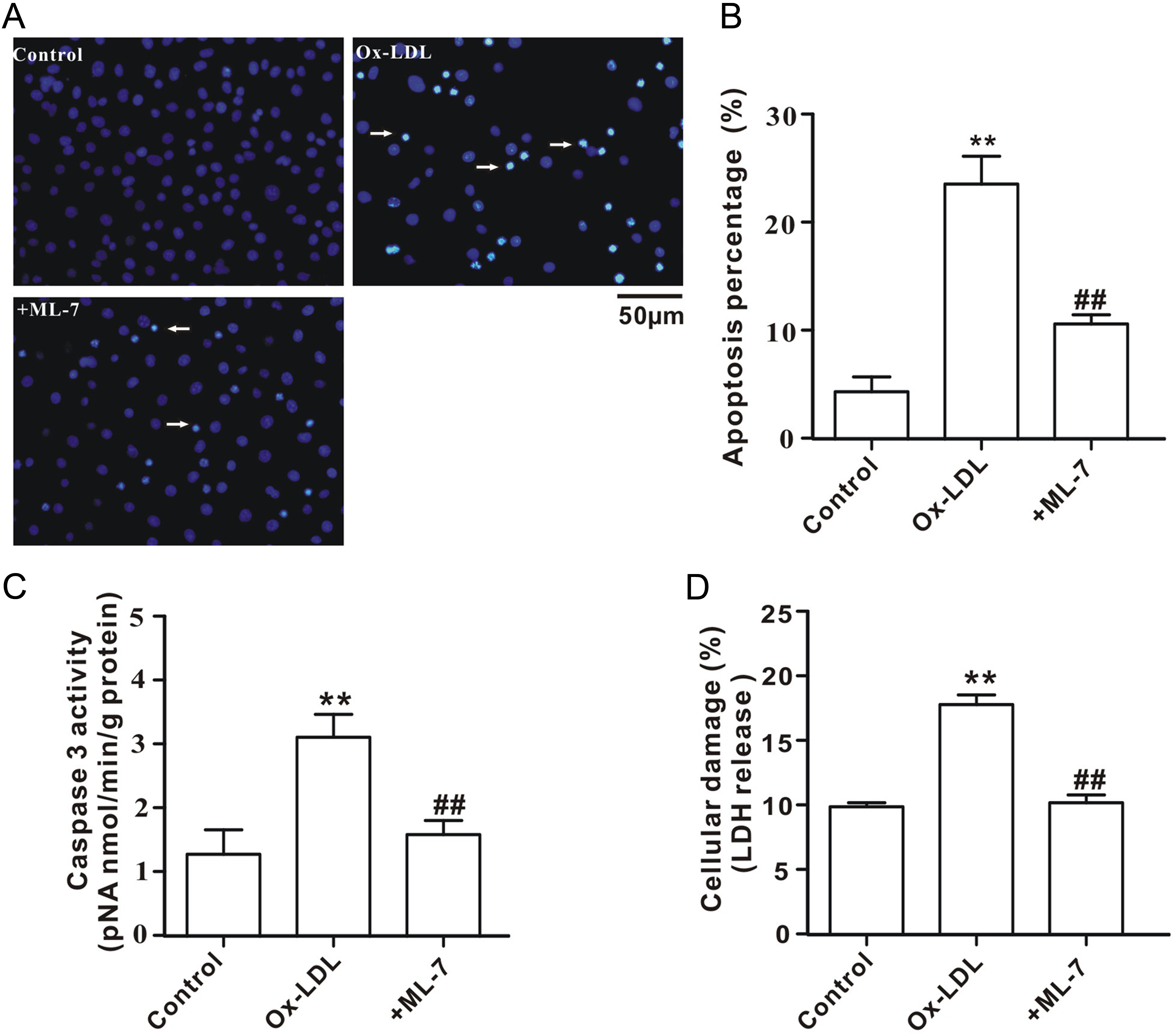
Attenuation of ox-LDL-induced HUVECs injury by MLCK inhibitor. A, Representative image of Hoechst staining. The apoptotic cells are indicated by arrows. B, Percentage of apoptotic cells per total number of HUVECs. C, Caspase-3 activity in HUVECs. D, LDH release from HUVECs. All values are expressed as mean ± SEM (n = 6 in each group). **
Effect of MLCK Inhibitor on DDAH Expression and Activity
Consistent with the findings in aorta from the hyperlipidemic rats, DDAH2 mRNA (Figure 5A, left panel) and protein (Figure 5B) expression in HUVECs was significantly downregulated following ox-LDL treatment, which was reversed by MLCK inhibitor (ML-7). However, there was no significant difference in DDAH1 mRNA (Figure 5A, right panel) and protein expression (Figure 5C) between the ox-LDL-treated HUVECs and the control cells. In agreement with the downregulation of DDAH2 in ox-LDL-treated HUVECs, DDAH activity was dramatically decreased concomitant with the elevated ADMA content and the reduced NO content (Figure 5D–F); these effects were attenuated in the presence of MLCK inhibitor.

MLCK inhibitor blocks DDAH2 downregulation in ox-LDL-treated HUVECs. A, DDAH2 and DDAH1 mRNA expression. B, DDAH2 protein expression. Top, representative images of Western blot. Bottom, ratio of optical density between DDAH2 and β-actin. C, DDAH1 protein expression. Top, representative images of Western blot; bottom, ratio of optical density between DDAH1 and β-actin. D, DDAH activities in HUVECs. E, ADMA concentrations in culture medium. F, NO contents in culture medium. All values are expressed as mean ± SEM (n = 6 in each group). **
Effect of ML-7 on MLCK Activity and p-nmMLC20 Level
Consistent with the results in aorta from the hyperlipidemic rats, MLCK activities in culture medium or in HUVECs (Figure 6A) were markedly elevated following ox-LDL treatment concomitant with the increased levels of p-nmMLC20 but not nmMLC20 (Figure 6B). Interestingly, the levels of p-nmMLC20 (but not nmMLC20) in the nuclear extracts from the ox-LDL-treated HUVECs were significantly elevated compared to that from the control cells (Figure 6C). The above-mentioned phenomena were dramatically attenuated by ML-7, a MLCK inhibitor.
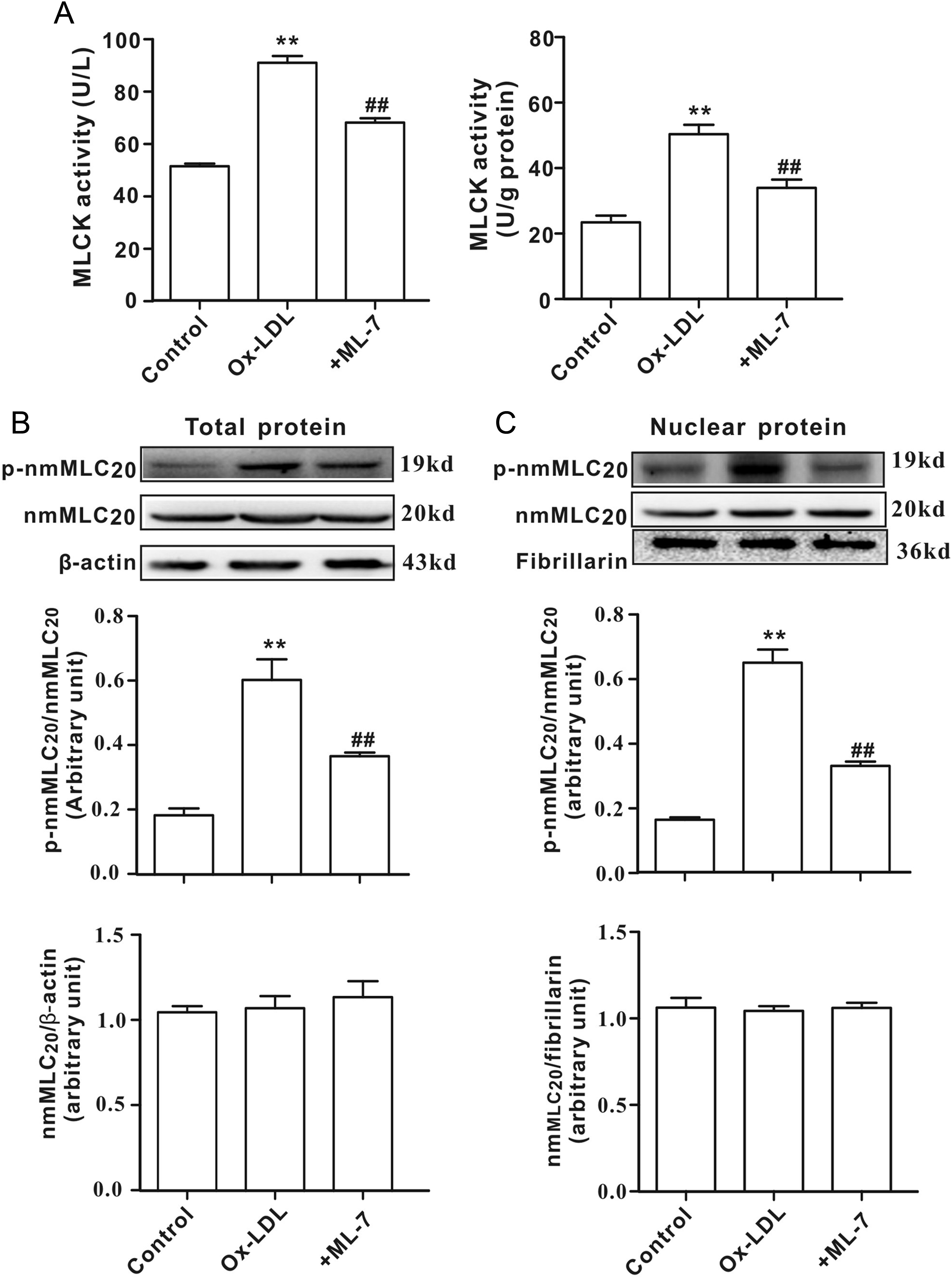
MLCK inhibitor decreases nmMLC20 phosphorylation in ox-LDL-treated HUVECs. A, MLCK activities in culture medium (left panel) or HUVECs (right panel). B, The protein levels of p-nmMLC20 or nmMLC20 in total proteins. Top, representative images of Western blot; middle, ratio of optical density between p-nmMLC20 and nmMLC20; bottom, ratio of optical density between nmMLC20 and β-actin. C, The protein levels of p-nmMLC20 or nmMLC20 in nuclear proteins. Top, representative images of Western blot; middle, ratio of optical density between p-nmMLC20 and nmMLC20; bottom, ratio of optical density between nmMLC20 and fibrillarin. All values are expressed as mean ± SEM (n = 6 in each group). **
Discussion
In the present study, by using the hyperlipidemic rats and ox-LDL-induced HUVECs injury model in vitro, we investigated the roles of nmMLC20 and DDAH in vascular endothelial dysfunction as well as the potential mechanisms for suppression of DDAH2 expression. Our results from animal and cell experiments showed that endothelial injury was obvious in hyperlipidemic rats or after ox-LDL treatment in vitro concomitant with the activated MLCK, the elevated p-nmMLC20 level, the suppressed DDAH2 (expression and activity) and the increased ADMA contents. Inhibition of MLCK (by ML-7) dramatically decreased the p-nmMLC20 level, accompanied by the enhanced DDAH2 activation (expression and activity), the reduced ADMA contents in HUVECs, and the ameliorated HUVECs injury. Either in hyperlipidemic rats or in ox-LDL-induced HUVECs injury model, there was no significant change in DDAH1 expression. To the best of our knowledge, this is the first study to provide evidence that vascular endothelial dysfunction in hyperlipidemic rats involves the suppression of DDAH2 expression and nmMLC20 plays a role in suppression of DDAH2 expression in a phosphorylation-dependent manner.
Hyperlipidemia, also called hyperlipoproteinemia or hyperlipidaemia, is a disorder of lipid metabolism involving abnormally elevated levels of lipids (such as cholesterol and triglycerides) and lipoproteins (such as LDL) in the blood. In this study, 8 weeks of high-fat diet feeding led to markedly elevated plasma levels of cholesterol, triglycerides, LDL, and ox-LDL in rats, whereas the HDL level (the good cholesterol) was decreased, indicating that the rat model of hyperlipidemia was successful. Although elevation of the plasma lipids and lipoproteins can promote atherosclerosis or atherosclerosis-related diseases through multiple mechanisms, hyperlipidemia-, particularly ox-LDL-induced, endothelial injury is thought as the first step in initiation of atherosclerosis development. 18,19 In the present study, we have found that the number of TUNEL-positive cells in the endothelium of aorta was obviously increased in the hyperlipidemic rats concomitant with a decrease in the vasodilator responses to acetylcholine, revealing the endothelial injury under condition of hyperlipidemia. Incubation of HUVECs with ox-LDL could also induce cellular injury in vitro, further confirming our results in vivo. It is worth to point out that there is no significant change in relaxation function of smooth muscle under the condition of hyperipidemia because administration of sodium nitroprusside promotes an identical dose-dependent relaxation in isolated aortic rings from both the hyperlipidemic rats and controls. 20,21 We obtained similar results in our preliminary experiments (data not shown), which exclude an impairment of smooth muscle function in hyperlipidemic aorta.
The mechanisms responsible for endothelial injury caused by hyperlipidemia, particularly ox-LDL, are extensively studied, but the details still need to be elucidated. Reports from other laboratories and ours have repeatedly demonstrated suggests that DDAH/ADMA pathway plays a key role in regulation of vascular endothelial functions under physiological and pathological conditions. 8,9,22,23 As an endogenous inhibitor of NOS, ADMA is emerging as a biomarker for endothelial dysfunction. 24 In this study, we have found that the plasma levels of ADMA were significantly elevated in the hyperlipidemic rats concomitant with endothelial injury. Consistently, the ADMA levels were also elevated in culture medium after the incubation of HUVECs with ox-LDL. These results confirmed a positive correlation between ADMA and endothelial injury. Since DDAH is the key enzymes responsible for the clearance of ADMA, it is reasonable to predict that DDAH activity and/or DDAH expression was suppressed under the condition of hyperlipidemia. Our results clearly showed that DDAH2 expression was obviously downregulated in hyperlipidemic rats or in ox-LDL-treated HUVECs, accompanied by the elevation of ADMA levels, confirming our hypothesis. Interestingly, there was no significant DDAH1 expression in the hyperlipidemic rats or ox-LDL-treated HUVECs, suggesting that DDAH1 plays a minor role in regulation of ADMA level under the condition of hyperlipidemia.
Although the reduction of DDAH activity is a major reason resulting in the accumulation of ADMA under multiple pathological conditions such as hypertension, 25 atherosclerosis and hyperlipidemia, 11,26 the mechanisms responsible for DDAH suppression, particularly at the gene level, remain largely unknown. Recently, MLC20 or nmMLC20 has been reported to function as a transcriptional factor to modulate gene expressions through a phosphorylation-dependent manner. 13,14 MLCK, an enzyme to phosphorylate MLC20/nmMLC20, has been found to be activated in the hyperlipidemic animals or ox-LDL-treated endothelial cells. 15,16 We thus predict that there might be a correlation between MLCK/nmMLC20 pathway and DDAH2 expression in endothelial cells. In the present study, our results showed that the MLCK activity and the p-nmMLC20 level in aorta from the hyperlipidemic rats or in ox-LDL-treated HUVECs were markedly increased concomitant with the suppression of DDAH2 expression and DDAH activity. Block the phosphorylation of nmMLC20 by MLCK inhibitor (ML-7) could attenuate the suppression of DDAH2 expression in ox-LDL-treated HUVECs. To further corroborate the function of nmMLC20 in regulation of DDAH2 expression, we examined the p-nmMLC20 level in the nuclear proteins of HUVECs. As expected, the changes in nuclear p-nmMLC20 level were inversely correlated with DDAH2 expression, supporting a potential role of p-nmMLC20 in suppression of DDAH2 transcription. Nevertheless, more experiments are necessary before making a firm conclusion.
In summary, the results presented in this study demonstrate for the first time that suppression of DDAH2 expression might be responsible for, at least partially, vascular endothelial dysfunction in hyperlipidemia, and nmMLC20 plays a role in suppression of DDAH2 expression in a phosphorylation-dependent manner. Thus, inhibition of nmMLC20 phosphorylation might provide a novel strategy to protect endothelial function in hyperlipidemia.
Limitations of the Study
There are 2 major limitations that need to be acknowledged and addressed regarding the present study. First, since no intervention was utilized in the animal study, thus the cause and effect relationship between p-nmMLC20 and endothelial dysfunction could not be established in vivo although it was confirmed in the cultured endothelial cells. To verify the relationship between p-nmMLC20 and endothelial dysfunction in vivo, it is necessary to do more experiments by using the MLCK inhibitors (such as ML-7) or MLCK knockout animals in future studies. Secondly, although we performed Western blot to quantify the protein levels of DDAH1, DDAH2, p-nmMLC20, and nmMLC20 in the aortic tissues, it could not fully reflect the levels of these proteins in endothelium of the aorta.
Footnotes
Authors’ Note
Yan Wu and Jie-Jie Zhang contributed equally to this work.
Author Contributions
Yan Wu, Jie-Jie Zhang, and Lian-Sheng Li performed the animal experiments. Ting-Bo Li and Wei-Qi Liu participated in cell experiments. Xiu-Ju Luo, Jun-Lin Jiang, Qi-Lin Ma, Zhi-Chun Yang, and Jun Peng were responsible for design of the experiments, co-ordination of the project, and preparation of the manuscript. All authors reviewed and approved submission of the revised manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Major Research Plan of the National Natural Science Foundation of China (No. 91439104 to Jun Peng), National Natural Science Foundation of China (No. 81373409 to Jun Peng, No. 81370250 to Qi-Lin Ma, No. 81373408 to Jun-Lin Jiang), Natural Science Foundation of Hunan Province, China (No. 13JJ2008 to Jun Peng), and Doctoral Fund of the Ministry of Education of China (No. 20120162110056 to Jun Peng).
