Abstract
Smoking is an established risk factor for cardiovascular disease. It has also been shown to result in endothelial dysfunction as assessed by flow-mediated dilation (FMD) in response to reactive hyperemia (RH)-induced increases in shear stress. Handgrip exercise (HGEX) is an emerging alternative method to increase shear stress for FMD assessment (HGEX-FMD) and the purpose of this study was to identify the impact of smoking on HGEX-FMD in young healthy subjects. Brachial artery RH-FMD and HGEX-FMD (10-minute bout of HGEX) was assessed in eight smokers (S) and 14 non-smokers (NS) (age 21 ± 2 years). Brachial artery diameter and mean blood velocity were assessed with echo and Doppler ultrasound, respectively. Shear stress was estimated by shear rate (SR = brachial artery blood velocity/diameter). The SR stimulus did not differ between groups for either test (RH-FMD (SR area under the curve until peak diameter measurement), p = 0.897; HGEX-FMD (average SR over 10-minute exercise bout), p = 0.599). The RH-FMD magnitude was not significantly different between groups (S: 7.7 ± 2.2% vs NS: 7.9 ± 2.4%, p = 0.838); however, the HGEX-FMD magnitude was significantly impaired in smokers (S: 6.1 ± 3.4% vs NS: 9.6 ± 3.6%, p = 0.037). In conclusion, HGEX-FMD assessment detected vascular dysfunction in young healthy smokers while RH-FMD did not. This suggests that HGEX-FMD may be useful in the early detection of smoking-induced impairments in endothelial function. Further research is required to explore this phenomenon in other populations and to isolate underlying mechanisms.
Keywords
Introduction
The proper function of the endothelial cells that line the interior of the arteries is essential for the maintenance of vascular health.1,2 Endothelial dysfunction is indicated by an impaired dilatory response to an increase in blood flow-associated shear stress (flow-mediated dilation (FMD)).3,4 The most popular non-invasive FMD assessment methodology uses the release of a temporary limb occlusion (reactive hyperemia (RH)) to create a transient shear stress stimulus. 3 While RH-mediated FMD (RH-FMD) has proven useful in identifying endothelial dysfunction,5–9 the focus on this one technique has left the FMD response to other shear stress stimulus profiles understudied.
In contrast to RH, handgrip exercise (HGEX) results in a sustained, intensity-dependent increase in brachial artery shear stress.10,11 It has been demonstrated that dilation of the brachial artery during HGEX is stimulated by that shear stress and is not the result of a conducted vasodilatory signal.11,12 Assessment of FMD stimulated by HGEX-induced increases in shear stress (HGEX-FMD) may be valuable for several reasons. In daily living, FMD occurs during exercise and this may have functional relevance for perfusion.13–15 It is therefore important that the factors influencing exercise-induced FMD are well characterized. In addition, recent evidence suggests that HGEX-FMD may provide very sensitive detection of endothelial dysfunction in some populations,16,17 although this finding is not universal. 18
Cigarette smoking has long been identified as a significant and modifiable risk factor contributing to cardiovascular disease and atherosclerosis.19,20 The evidence to date indicates that a deleterious impact of smoking on vascular endothelial function is likely to play an important mechanistic role.7,21,22 Impaired RH-FMD in smokers versus non-smoking control subjects has been well documented;7,22–26 however, the impact of smoking on HGEX-FMD is unknown. The purpose of the present study was therefore to compare the brachial artery FMD of young healthy smokers and non-smokers in response to HGEX- and RH-mediated increases in shear stress. We hypothesized that the young smokers would have an impaired brachial artery RH- and HGEX-FMD response.
Methods
Subjects
Fourteen healthy non-smoking and eight healthy smoking males (age range 18–26 years) participated in the study. Smokers were required to be currently engaged in their smoking habit and to have smoked the equivalent of one pack year (20 cigarettes/day for 1 year). In one subject, a smoking history including cigarettes (< 1 pack year) in combination with other tobacco products was assumed to exceed this threshold. Health status was confirmed via a medical screening questionnaire for risk factors associated with endothelial dysfunction. The study procedures were approved by the Queen’s University Health Sciences Human Research Ethics board and all subjects completed a consent form approved by the same board.
On an initial visit, subjects were introduced to the equipment and the HGEX protocol and it was ensured that a clear image of the brachial artery, and a clear blood velocity signal, with no interference from adjacent veins, could be obtained. Subjects also completed a 7-day physical activity recall questionnaire.27,28 Prior to the experimental visit subjects were instructed to abstain from alcohol, caffeine, food and exercise for 12 hours and cigarettes for 8 hours prior to commencing the study. The experimental visit took place in the morning (starting between 7:30 am and 9:30 am) in a quiet, temperature-controlled room (~20°C).
Subject monitoring
Heart rate (HR) was monitored with a three-lead electrocardiogram for the duration of the study. Blood pressure (mean arterial pressure, MAP) was monitored throughout each trial via photoplethysmography (Finometer PRO; Finapres Medical Systems, Amsterdam, The Netherlands).
Brachial artery blood velocity and diameter measurements
The brachial artery blood velocity was measured with Doppler ultrasound operating at 4 MHz (GE Vivid i2; GE Medical Systems, London, ON, Canada). The Doppler shift frequency spectrum was analyzed using a Multigon 500P TCD (Multigon Industries, Yonkers, NY, USA). The corresponding voltage output was sampled continuously (Powerlab; AD Instruments, Colorado Springs, CO, USA) and stored (LabChart; AD Instruments) for later analysis. For reasons previously described, 29 all scans were performed at an insonation angle of 68°. The ultrasound probe was placed over the brachial artery to acquire an optimal image of the vessel and a clear arterial blood velocity signal. Once in position, a guide was adhered to the skin so that the same region of the vessel would be scanned in all trials. The brachial artery diameter was obtained using two-dimensional grey-scale ultrasound imaging technology operating at 12 MHz in B-mode (Vivid i2; GE Medical Systems). The images were recorded in .avi format on an external personal computer with commercially available software (Camtasia Studio; TechSmith, Okemos, MI, USA) for later analysis.
Experimental protocol
Subjects lay supine with both arms extended out to their sides. Ultrasound measurements were conducted on the left arm while blood pressure was measured on the right arm. Subjects participated in five trials during their experimental visit: three HGEX trials and two RH trials (whether RH or HGEX trials were performed first was counterbalanced between subjects). There were therefore three replicate HGEX trials and two replicate RH trials. Like trials were averaged to yield one HGEX and one RH response for each subject. This averaging approach was employed to obtain a better characterization of the subject’s response and has been reported previously.29,30 Three trials of HGEX were performed (rather than two) because these data were also a part of a separate investigation looking at FMD dynamics and three or more replicate trials facilitate that analysis. The stability of RH- and HGEX-FMD magnitude (no systematic increase or decrease) across multiple trials has been demonstrated previously.31,32
RH trials. An occlusion cuff was secured around the forearm just below the anticubital fossa. One minute of baseline was recorded after which the cuff was inflated to 250 mmHg for 5 minutes. Measurements resumed for the last minute of occlusion and continued for the 3 minutes following cuff release. The RH procedure was repeated for a second trial after a minimum of 10 minutes or until the artery diameter returned to baseline.
HGEX trials. During the initial visit subjects were asked to perform two isometric maximal voluntary contractions (MVC) using a handgrip dynamometer. Subjects then performed a series of contractions to identify the contraction intensity (%MVC) that elicited the blood flow velocity required to achieve an exercise target shear rate of 75 s−1. This target was selected because previous work has shown that it is associated with an exercise intensity that can be maintained for several minutes and that it elicits a significant FMD response.11,12 Shear rate (an estimate of shear stress without blood viscosity) was calculated as: mean blood velocity/vessel diameter. The blood velocity required to achieve the target shear rate was calculated for each subject as: required velocity = 75 s−1 × brachial artery diameter. To perform the calculation, the brachial artery diameter for each subject was estimated by manual caliper placement on the ultrasound image before the trials commenced.
All parameters were measured for 1 minute and then, to ensure a uniform baseline shear rate (and therefore a uniform change in shear rate from baseline to exercise) in all subjects, arterial compression11,29,33 was performed for 4 minutes. To achieve this, a researcher placed two fingers on the subject’s arm distal to the ultrasound probe and applied pressure to the brachial pulse. The compression force was adjusted based on continuous blood velocity output (an online moving average) in order to maintain the shear rate at 10 s−1. As previously described, 11 to achieve a rapid increase in shear rate upon compression release, during the last 15 seconds of the arterial compression, subjects performed several (ranging from two to four) contractions at the designated %MVC (the intensity expected to elicit a shear rate of 75 s−1 as determined during the initial visit). Upon compression release the subjects performed 10 minutes of HGEX. During the HGEX, isometric handgrip force feedback was displayed continuously for the subjects on a computer data acquisition system (Powerlab; AD Instruments). Subjects achieved the necessary %MVC (determined in initial visit) and duration for each contraction by displacing the force readout line to the desired level in time with a 2-second contraction / 3-second relaxation duty cycle metronome. Brachial artery blood velocity was monitored continuously throughout the trial, and experimenters coached subjects through minor increases and decreases in force production in order to maintain the required blood velocity. Three HGEX trials were completed for each participant and a minimum of 10 minutes or the time until the diameter returned to baseline separated the trials.
Blood lipids
At the end of the ultrasound data collection, a small sample of blood was taken by fingerprick for analysis of blood lipid levels using a Cholestech LDX System (Alere Inc., Ottawa, ON, Canada).
Data analysis
HR and MAP
HR and MAP were analyzed offline and compiled into 3-second average time bins using the data acquisition software program LabChart (AD Instruments) as previously described. 12 Hemodynamic data from the FMD trials are reported as 1-minute averages taken from baseline and the last minute of exercise (for HGEX-FMD) or the last minute of reactive hyperemia (for RH-FMD).
Brachial artery blood velocity
Blood velocity data were analyzed offline in 3-second average time bins using the data acquisition software program LabChart (AD Instruments) as previously described. 12
Brachial artery diameter
The brachial artery diameter was determined using automated edge-detection software (FMD/Blood Flow Acquisition and Analysis; Reed Electronics, Perth, WA, Australia) (an updated version of the software described in Woodman et al. 34 ). The diameter data were compiled into 3-second time bins. Missing data due to tracking error in individual trials was interpolated to facilitate calculation of the average response across repeated trials.
Shear rate
The shear rate, an estimate of shear stress without blood viscosity, was calculated as: mean blood velocity/vessel diameter, calculated from the 3-second average velocity and diameter time bins. For the HGEX-FMD test the shear rate was characterized as the average over the 10-minute exercise bout and is also graphically presented as 1-minute time bins. 11 For the RH trials, the peak shear rate and the area under the curve (AUC) of the shear rate stimulus from cuff release until the time of peak diameter measurement were reported.4,30
FMD
The RH-FMD was quantified by calculating the percent increase in diameter from baseline diameter measured prior to cuff inflation (1-minute average) to the peak 3-second average diameter post-occlusion cuff release. 4 The HGEX-FMD was quantified by calculating the percent increase in diameter from the last minute of arterial compression (baseline) to the average diameter in the last minute of exercise.11,12 We identified this as our key characterization of HGEX-FMD magnitude a priori, and it was chosen because typically end exercise yields the largest FMD magnitude.11,12 This approach is consistent with previous work.10–12,18 To examine the impact of exercise duration on HGEX-FMD we also report the average FMD at minutes 2, 4, 6 and 8 of exercise.
Statistical analysis
To compare the results for all measures, t-tests and analysis of variance (ANOVA) (factors: between subjects – group (smokers vs non-smokers) and within subjects – time) were used as appropriate. A significant interaction between time and group was identified for HGEX-FMD and this was examined via one-way repeated measures (RM) ANOVA (factor: time – minutes 2, 4, 6, 8 and 10 of exercise) within each group. A significant main effect of time within the RM ANOVA was further assessed with a Tukey post hoc test. All statistical analysis was completed using SigmaPlot 11 (Systat Software, Chicago, IL, USA) or IBM SPSS, Version 20 (SPSS Inc., Chicago, IL, USA). Data are presented as means ± SD.
Results
Baseline characteristics
The baseline characteristics of the subjects are displayed in Table 1. There were no significant between-group differences. In addition, brachial artery diameter in the last minute of cuff occlusion (RH-FMD test) did not differ from the baseline diameter (measured prior to occlusion) (p = 0.799) and this did not depend on smoking status (interaction between group and diameter, p = 0.899).
Baseline characteristics of smokers and control subjects.
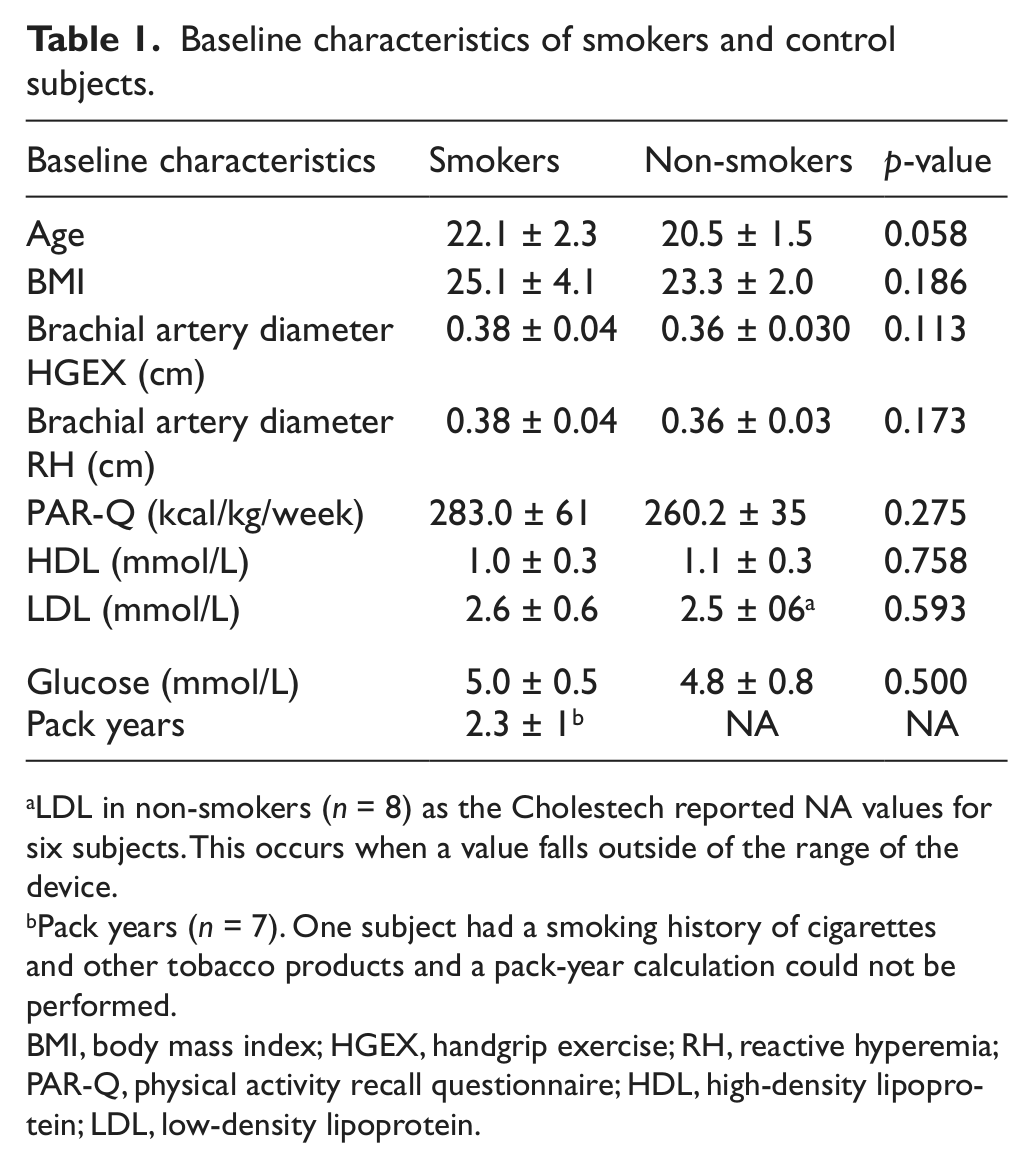
LDL in non-smokers (n = 8) as the Cholestech reported NA values for six subjects. This occurs when a value falls outside of the range of the device.
Pack years (n = 7). One subject had a smoking history of cigarettes and other tobacco products and a pack-year calculation could not be performed.
BMI, body mass index; HGEX, handgrip exercise; RH, reactive hyperemia; PAR-Q, physical activity recall questionnaire; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
HR and MAP
The HR and MAP data are reported in Table 2. HGEX caused a small but significant increase in HR and MAP from baseline (p < 0.001), although HR was significantly lower in the non-smokers (p = 0.025). There was no impact of smoking status on blood pressure (p = 0.613). In the RH protocol, there was a slight increase in HR and MAP from baseline to the last minute of hyperemia (p = 0.023 and p = 0.017). For HR the interaction between group and time almost reached significance (p = 0.052) and reflects the greater increase in HR from baseline to cuff release in the non-smokers. Similar to the HGEX test, smoking status had no impact on MAP (p = 0.740).
HR and MAP in smokers and non-smokers during the reactive hyperemia and handgrip exercise protocols.

The factor time has two levels: baseline and last minute of handgrip exercise or reactive hyperemia.
HR, heart rate; MAP, mean arterial pressure.
Shear rate
RH-FMD test. The baseline shear rate was significantly greater in the smokers compared to the non-smokers (36.1 ± 16.3 s−1 vs 21.0 ± 10.7 s−1, p = 0.016); however, the shear rate during the last minute of cuff inflation was not significantly different between groups (non-smokers: 8.2 ± 3.9 s−1; smokers: 6.6 ± 1.3 s−1, p = 0.258). Neither the AUC of the shear rate until the time of peak diameter measurement nor the peak shear rate were significantly different between smokers and non-smokers (AUC: 5165.2 ± 869.1 vs 5100.5 ± 1219.4, p = 0.897; peak shear rate: 186.7 ± 51.4 vs 166.8 ± 30.3, p = 0.264) (RH shear rate profile, Figure 1A).
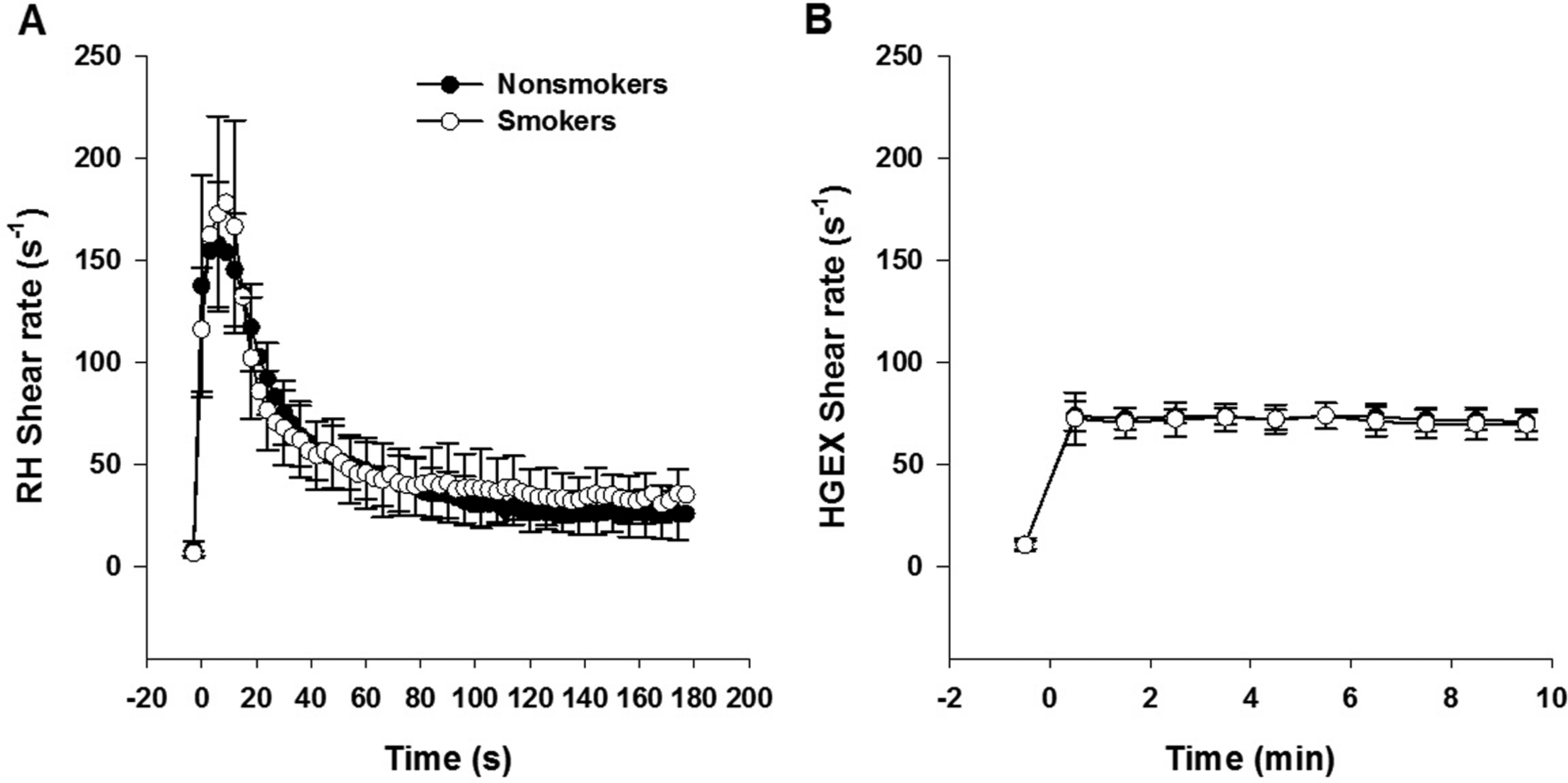
(A) Shear rate profile upon occlusion cuff release in the RH-FMD test. Each data point represents a 3-second average. (B) Shear rate profile during the HGEX-FMD test. Each data point represents a 1-minute average. The ‘–1’ time point denotes the last minute of arterial compression. There were no significant between-group differences. Error bars represent ± SD. RH, reactive hyperemia; HGEX, handgrip exercise.
HGEX-FMD. The shear rate during the last minute of arterial compression was close to the target of 10 s−1 and not significantly different between groups (smokers: 10.6 ± 2.0 s−1 vs non-smokers: 10.8 ± 3.2 s−1, p = 0.872). The average shear rate during the 10-minute exercise bout was close to the target of 75 s−1 and not significantly different between groups (smokers: 71.4 ± 6.9 s−1 vs non-smokers: 72.6 ± 4.3 s−1, p = 0.599) (HGEX shear rate profile, Figure 1B). The %MVC performed to achieve this shear rate was also not significantly different between groups (smokers: 27 ± 11%, non-smokers: 25 ± 6%, p = 0.430).
FMD
Time to peak diameter (61.8 ± 29.2 s vs 54.75 ± 17.9 s, p = 0.483) and RH-FMD magnitude were not significantly different between groups (p = 0.838) (Figure 2A). However, HGEX-FMD magnitude at the end of exercise was significantly lower in smokers versus non-smokers (p = 0.037) (Figure 2B). The HGEX-FMD over the 10-minute exercise bout is shown in Figure 2C. In ANOVA analysis, there was a significant interaction between time and group (p = 0.05). Post hoc analysis within each group identified that HGEX-FMD increased significantly between 2 and 10 minutes of exercise in non-smokers (p < 0.001), but not in smokers (p = 0.458). The RH-FMD and HGEX-FMD responses were not significantly correlated in either group (data not shown).
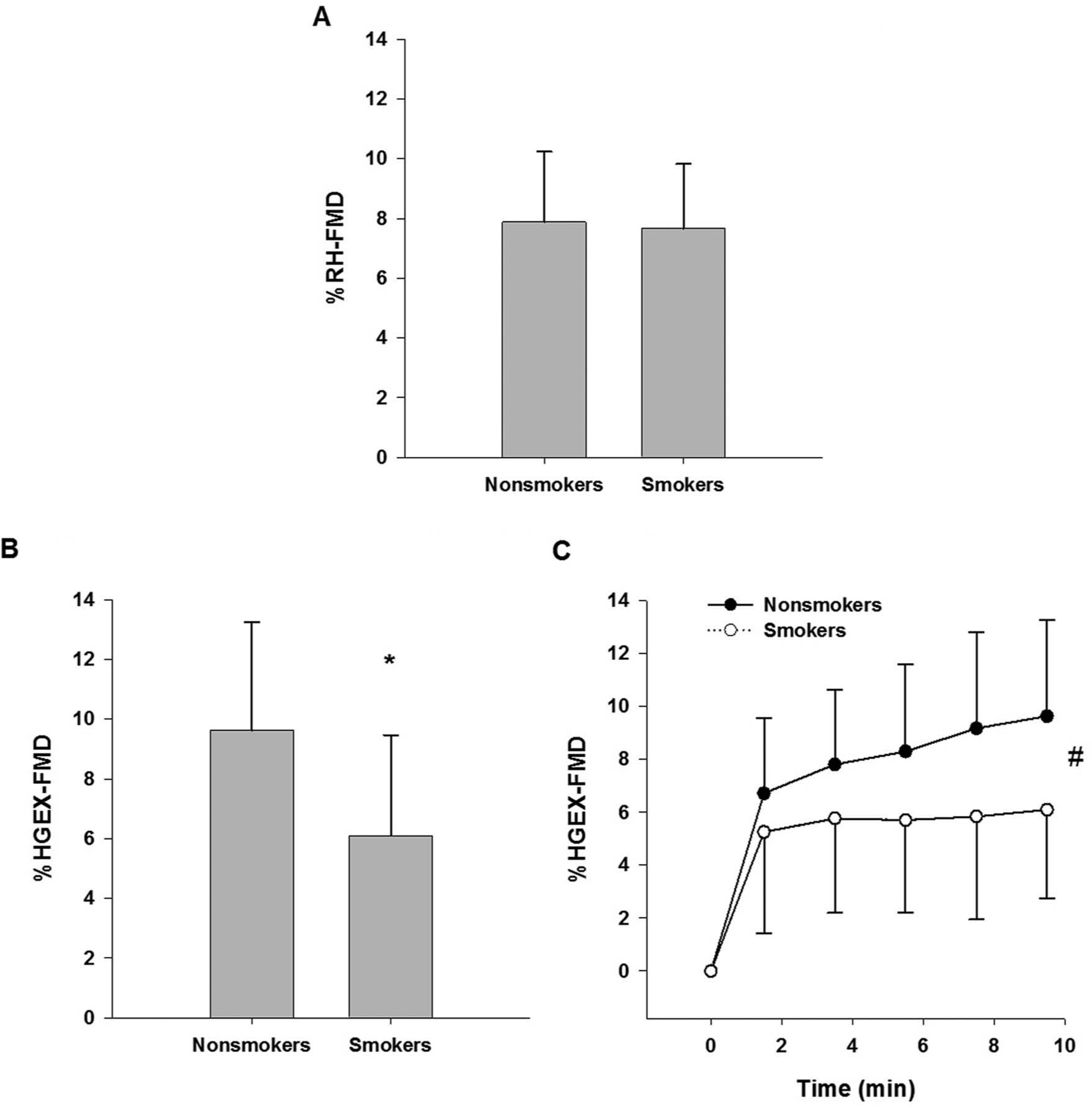
(A) %RH-FMD calculated from baseline (prior to occlusion) to the peak diameter during reactive hyperemia. Group difference p = 0.838; 95% confidence interval for %RH-FMD group difference: –2.3 to 1.9%. (B) %HGEX-FMD from baseline to the average diameter in the last minute of exercise. Group difference p = 0.037; 95% confidence interval for %HGEX-FMD group difference: –6.8 to −0.2%. *Significantly different from non-smokers (p = 0.037). (C) %HGEX-FMD over time in smokers and non-smokers. #There was a significant interaction between group and time such that HGEX-FMD increased over time in non-smokers (p < 0.001) but not in smokers (p = 0.458). Error bars represent ± SD. RH, reactive hyperemia; FMD, flow-mediated vasodilation; HGEX, handgrip exercise.
Discussion
This study was designed to compare the brachial artery FMD of young healthy smokers and non-smokers in response to HGEX- and RH-mediated increases in shear stress. The key novel finding was that young smokers had an impaired HGEX-FMD, but not an impaired RH-FMD response. In addition, HGEX-FMD plateaued after the second minute of exercise in smokers, but continued to increase from minutes 2 to 10 in non-smokers. Given the well-established link between smoking and vascular health, this suggests that HGEX-FMD may be an effective tool to detect changes in endothelial function early in the pathogenic process.
RH-FMD and smoking
The majority of studies have reported impaired RH-FMD in smokers versus non-smoking control subjects7,22,23,26,35,36 and smoking cessation has been shown to result in an improvement in endothelial function. 37 However, in agreement with the present findings, not all studies examining young healthy smokers have observed an impaired RH-FMD response.38,39 This suggests that in young healthy subjects, with a short history of smoking, endothelial dysfunction may not be present or may not be sufficiently severe to manifest as an impaired RH-FMD response. This is supported by the observation of a dose–response relationship between smoking exposure and endothelial dysfunction. 24 Although it did not reach significance, the peak shear rate and shear rate AUC until the time of peak diameter measurement were slightly higher in the smokers. It is possible that if the stimulus had been identical between groups a trend for lower RH-FMD in the smokers may have been more apparent. However, a similar RH stimulus difference was observed between trials in a within-subject comparison in healthy males and it had no impact on RH-FMD response magnitude. This suggests that the slightly higher stimulus magnitude observed in the smokers was not masking significant RH-FMD impairment.
Low-flow mediated constriction (LFMC) during cuff occlusion is gaining recognition as an index of vascular health;40,41 however, in the present study, regardless of smoking status brachial artery diameter did not differ between baseline and the last minute of cuff occlusion. Brachial artery responses to cuff occlusion are known to be variable 42 and an absence of brachial artery LFMC has been reported previously in healthy subjects. 43 Future research including radial artery measurements and subjects with a longer smoking history are required to fully characterize LFMC and its potential as an index of vascular health in smokers.
HGEX-FMD and the detection of endothelial dysfunction
In contrast with RH-FMD, HGEX-FMD at the end of the exercise bout was significantly impaired in smokers versus non-smokers (Figure 2B). The HGEX shear rate was well matched between groups and therefore stimulus differences do not explain this observation. The increase in HR and MAP with exercise was typical11,12 and similar between groups. Although the HR was slightly higher in the smokers (5 bpm) this is unlikely to explain the difference in HGEX-FMD, especially given that MAP (which would be a better indicator of greater sympathetically mediated vasoconstriction) was similar or lower in smokers.
The impaired HGEX-FMD in smokers is in agreement with the observation of Gaenzer et al., 23 that femoral artery dilation during submaximal cycling exercise was impaired in smoking versus non-smoking subjects. However, in that study the subjects were older (~39 years) with a longer smoking history and the RH-FMD of the smoking subjects was also impaired. Stoner et al. 39 examined FMD in response to RH, RH + heating, heating alone and HGEX-induced increases in shear stress to create a blood velocity–diameter relationship. These authors did not observe an effect of smoking on the relationship and the results are difficult to compare to the present study because the FMD responses to HGEX were not examined separately.
The present finding of impaired HGEX- but not RH-FMD in smokers is in agreement with other recent observations identifying that FMD following exercise-induced/sustained increases in shear stress may provide a more sensitive index of FMD impairment in some populations. Grzelak et al. 16 performed a brief HGEX protocol and Bellien and colleagues 17 employed hand warming to evoke a moderate, sustained shear stimulus, similar to that evoked by HGEX. Both authors found that differences in FMD magnitude between healthy control subjects versus ‘at risk’ populations (risk factors: age and type I diabetes) were greater with these protocols versus RH-FMD. Taken together these data suggest that FMD following a sustained stimulus may be impaired before, or with a smaller perturbation than RH-FMD. If this is the case there may be clinical research applications for HGEX-FMD in identifying early stages of endothelial dysfunction. However, in contrast to these studies, Mullen et al. 44 found that while RH-FMD was impaired in a population with hypercholesterolemia, FMD stimulated by sustained increases in shear stress due to distal acetylcholine infusion was not. Further study is required to elucidate the mechanisms underlying these disparate results and any population specificity in the ability of HGEX-FMD to detect dysfunction.
The HGEX-FMD of the non-smokers increased over time (minutes 2–10) while that of the smokers did not (Figure 2C). This resulted in a widening of the smoker–non-smoker gap in HGEX-FMD magnitude with increasing exercise duration. Previous work in healthy non-smoking subjects has also identified a progressive increase in artery diameter with a stable prolonged shear stress stimulus.11,29,31,45 Pyke and colleagues 29 created a range of sustained brachial artery shear stress magnitudes using forearm heating and arterial compression and identified that the magnitude of the continued dilation after the initial rapid dilation phase was proportional to the shear stress magnitude. 29 This suggests that the progressive dilation is stimulated by the shear stress.
There is both animal and human evidence to suggest that distinct mechanisms are involved in initiating versus maintaining/continuing FMD,29,44,46,47 and it has been suggested previously that there may be a sequential recruitment of mechanisms when the shear stress stimulus is prolonged. 47 Taken together the present data suggest that smoking may have a larger early impact on FMD maintenance mechanisms. The specific nature of ‘initiation’ versus ‘maintenance’ mechanisms in humans is unclear and may be complicated.48,49 However, some evidence suggests that the contribution of nitric oxide is more pronounced initially44,50,51 and that other vasodilators become involved with a prolonged stimulus.44,47,52 The current findings of intact RH-FMD and impaired continued HGEX-FMD over time in young smokers could indicate that non-NO mechanisms are the first to be impaired by smoking. However, there is evidence that in older smokers with a longer smoking history, impaired NO bioavailability plays a significant role in RH-FMD impairment.22,36 The nature of FMD mechanisms over the course of a sustained, stable shear stress stimulus and the interaction between smoking and the mechanisms responsible for HGEX-FMD specifically require further research.
Limitations
We did not measure blood viscosity and therefore while shear rate was matched between smokers and non-smokers in the HGEX-FMD test, shear stress may have differed. However, it has been identified that smokers tend to have slightly higher viscosity versus non-smokers.53,54 Therefore, if anything, smokers may have had a greater HGEX shear stress stimulus. A greater stimulus in smokers with a lower HGEX-FMD would not alter our primary conclusion that HGEX-FMD is impaired in smokers. In addition, the sample size for smoking subjects was small and they were fairly homogeneous in age and smoking exposure. HGEX-FMD testing will need to occur in a larger population of smokers with a wide range of smoking history in order to fully establish the sensitivity of this test in detecting endothelial dysfunction.
Conclusion
This study reports the first direct comparison of RH- and HGEX-FMD in healthy young smokers and non-smokers. It was found that HGEX-, but not RH-FMD was impaired in the smokers. This suggests that HGEX-FMD may provide a sensitive test for detecting endothelial dysfunction early in the pathological process. From a functional perspective, impaired FMD responses to exercise-induced increases in shear stress may have negative implications for perfusion.13,15,26,55 Further study is required to elucidate the mechanisms underlying the sensitivity of HGEX-FMD, its functional significance and whether it is consistent in a range of populations.
Footnotes
Conflict of interest
The authors have no conflicts of interest to report.
Funding
This study was funded by a Natural Sciences and Engineering Research Council of Canada (NSERC) Discovery Grant and Canada Foundation for Innovation and Ontario Ministry of Research and Innovation Leaders Opportunity Funding to KE Pyke. BB Findlay was supported by an NSERC undergraduate research award. IC Szijgyarto was supported by a Ministry of Research Innovation Early Researcher Award to KE Pyke.
