Abstract
Safety monitoring of disease-modifying therapies (DMTs) used to treat multiple sclerosis (MS) has largely overlooked the domain of gynecological health. This topical review aims to provide MS clinicians with an overview of the three categories of complications described to date, as well as risk mitigation strategies. These are increased risk of human papilloma virus (HPV) positivity and related cervical dysplasia/cancers; inflammatory and infectious vaginitis and susceptibility to bacterial vaginosis (BV); and herpesvirus infections, including genital Herpes Simplex Virus (HSV). Current knowledge may be biased due to limited studies and lack of gynecological focus in neurological encounters. Risk mitigation strategies include promoting HPV vaccination, following guidance for immune compromised individuals relating to cervical cancer screening and antiviral suppression, and proactive communication with patients about gynecological health when starting DMTs. Together, these might improve gynecological health and thereby quality of life in females with neuroinflammatory diseases.
Highlights
There is an unmet need to understand the effects of MS disease-modifying therapies (DMTs) on gynecological health
DMTs have been associated with HPV-related cervical pathology, vaginitis, and herpesvirus infections
Risk mitigation strategies could improve gynecological health and overall DMT safety profiles
Introduction
Disease-modifying therapies (DMTs) are highly effective in preventing neuroinflammatory attacks that arise in multiple sclerosis (MS) and thereby, axonal loss and clinical progression. However, with prolonged use, complications can arise including lymphopenia, hypogammaglobinemia, infections, and colitis. 1 One arena that remains inadequately explored is the potential impact of long-term use of DMTs on female gynecological health. 2 Since reproductive and vaginal health are important components of overall well-being, understanding the potential impact of MS DMTs on gynecological complications is critical. Indeed, gynecologic cancer and cancer screening are identified as unmet needs in MS not only by clinicians but by patients. 3
The goal of this topical review is to describe what is known about DMTs and gynecological health in women with MS. This is becoming more important with the widespread use of B-cell depleting therapies (BCDTs), with clinical trials and real-world studies demonstrating effective reduction of relapse risk and favorable safety and tolerability. 4 Recent data point to potential elevated risk of vaginal inflammation, gynecological infections, 5 and gynecological dysplasia and cancer. 6 One possible mediating factor for these associations is the impact of DMTs with immunosuppressive properties on vaginal mucosal immunity, resulting in vaginal dysbiosis (VD, i.e. imbalances in microbial communities, which are more commonly studied in the gut) and infections. 7 This review further emphasizes integrating such considerations into comprehensive MS management, optimizing treatment strategies to balance therapeutic benefits with reduction of gynecological risks. A roadmap for future research is proposed, with the goals of ultimately enhancing patient outcomes.
Methods
This topical review sought to provide an in-depth review of the current understanding of the effects of DMTs on the gynecological and sexual health of women with MS. A search was conducted between 1 November 2024 and 1 February 2025 using three scientific databases (PubMed, Scopus, and Google Scholar) to gather relevant data and evidence. Articles were selected based on their relevance to the effects of DMTs on the gynecological health of women with MS. Keywords for the search included terms such as “disease-modifying therapy,” “multiple sclerosis,” “gynecological health,” “vaginal inflammation,” “immunosuppressive treatments,” “vaginal dysbiosis,” and “gynecological infections,” “HPV,” and “cervical cancer.” No restrictions were placed on country or article type. Studies with clinical trials, reviews, or case reports based on reliable scientific evidence were prioritized. A full list of the literature reviewed is presented in Supplementary Appendix 1.
Results
Immune regulation and the vaginal mucosa: overview
Physical and immunological barriers in vagina
The first-line physical defense for the female reproductive tract is an ectocervix and vagina lined with stratified non-keratinized squamous epithelium, which provides a protective barrier, further supported by chemical and biological barriers. An intact mucosa contains immune factors including cytokines, complement, antibodies, and antimicrobial peptides. 8 Immune cells within the vaginal mucosa provide layers of innate (e.g. macrophages, natural killer (NK) cells, dendritic cells) and adaptive (CD4+ helper T cells, CD8+ cytotoxic T cells and CD19 B cells) immunity. 6 B cells help maintain mucosal immunity by producing antibodies such as secretory IgA (sIgA) and IgM to protect against microbial colonization and inflammation, as well as IL-10 to regulate immune responses. 9 Dysfunction in any component of this system is associated with heightened risk of herpes virus infections and cervical cancer linked to human papillomavirus (HPV). 10
Vaginal microbiome
Role in immunity
Overall, maintaining a minimally diverse, lactobacilli-dominated vaginal bacterial microbiome (VBM) appears essential to promoting a healthy vaginal ecosystem and supporting host-microbial interactions through production of bacteriocins, hydrogen peroxide, and lactic acid. 11 VD is characterized by a reduction in protective Lactobacillus species and greater microbial diversity. The presence of BV-associated species can raise vaginal pH, reduce antimicrobial defenses, and promote pathogenic growth, leading to BV, Chlamydia trachomatis, and vulvovaginal candidiasis. 12 Changes in the VBM have been linked to gynecological sequelae including susceptibility to trichomoniasis, HPV, and genital herpes simplex virus-2 (HSV-2). 13
Factors influencing the vaginal microbiome
Numerous factors influence VBM composition, emphasizing the complex interplay between behavioral, environmental, and physiological factors in maintaining vaginal health and preventing dysbiosis. These include age, reproductive stage, exogenous estrogens, local exposures (hygiene or sexual practices), and smoking. Broad-spectrum antibiotics like metronidazole can disrupt the vaginal microbiome by depleting Lactobacillus, allowing opportunistic microbes to thrive. Finally, immune-modulating therapies, such as DMTs for MS, can have variable effects on local mucosal tolerance mechanisms. potentially exacerbating underlying inflammatory disorders or other complications within the female reproductive tract. 14
MS DMTs, immune regulation, and gynecological sequelae
DMTs target the immune system through varied mechanisms of action, and their impact on T cells, B cells, and NK cells/subsets varies considerably, therefore, they could over time influence immune cells relevant to vaginal and mucosal health in different ways. For example, BCDTs could over time by lower secretory IgA and disrupt vaginal microbiota. 15 Sphingosine-1-receptor (S1P) modulators impair lymphocyte trafficking, leading to systemic immunosuppression that could reduce vaginal immune surveillance and compromise microbiome stability. 16 Natalizumab, by inhibiting α4-integrin, prevents immune cell migration, potentially altering mucosal homeostasis and microbial diversity. 17 Overall, further research is needed to clarify potential mechanisms underlying DMT-induced vaginal microbiome shifts and their clinical implications.
Specific gynecological outcomes
Cervical cancer and dysplasia
HPV enters the basal keratinocytes through micro-abrasions, replicating during epithelial migration, and releases virions at the surface, evading systemic immunity. This implies that any effects of DMTs on HPV clearance would be mediated by local effects on immune surveillance, 18 thereby decreasing HPV clearance and increasing the likelihood of cervical dysplasia and cancer. Recent studies have raised concerns regarding the incidence of refractory HPV infections in patients treated with DMTs,19,20 and this topic is particularly important given that researchers and patients alike identify gynecological cancers as a research gap in MS. 3
To contextualize a potential link between DMTs and cervical cancer, it is important to consider the timescale as well as potential confounders. Cervical carcinogenesis is a prolonged process, and the 2- to 4-year follow-up in typical MS cohorts may be insufficient to fully capture the long-term oncogenic risks of high-efficacy immunotherapies. Furthermore, the risk of cervical cancer in women with MS could be influenced by diagnostic neglect, whereby more disabled women are underrepresented in cancer prevention programs, and may be less likely to receive recommended cancer screening.21,22 Findings from a nationwide cohort study, of no MS-associated risk of female genital organ cancer until after 1996, suggests this could be due to the introduction of DMTs. 23 Regarding specific DMTs, to date, the data are mixed. This is the case overall for associations between natalizumab and cervical abnormalities24,25 with larger studies, including a Swedish registry and the AFFIRM trial, reporting no significant increased risk of cervical cancer; one case of cervical carcinoma in situ was reported in AFFIRM. 26 Glatiramer acetate and interferon-beta have not been linked to cervical cancer risk in MS, 27 while fingolimod and cladribine have been linked to increased cervical abnormalities. 6
An Australian multicenter cohort study (1998–2019) demonstrated higher incidence of abnormal cervical screening tests (CSTs), including both low- and high-grade squamous intraepithelial lesions (LSIL-HSIL), among women with MS exposed to moderate-high efficacy DMTs (in this context, fingolimod, cladribine, dimethyl fumarate, natalizumab, alemtuzumab and BCDTs), relative to non-exposed individuals. 28 This pattern aligns with other immunocompromised populations (HIV, solid organ transplant recipients), where persistent HPV infections heighten the risk of cervical pre-cancer and progression to malignancy.29,30 Despite these concerns, current evidence regarding the relationship between BCDT exposure in MS and cervical abnormalities remains inconclusive. A Swedish cohort study evaluating the incidence of cervical intraepithelial neoplasia grade 3 (CIN3) in individuals receiving natalizumab, fingolimod, and rituximab, identified a borderline significant elevation in risk for fingolimod (HR: 1.63, 95% CI: 0.94–2.82), but not the other DMTs. 26
Ongoing post-marketing studies, such as the Verismo study evaluating malignancy incidence in ocrelizumab-treated patients, are critical for understanding the long-term safety profile of DMTs. The findings from these studies may inform future patient care strategies, including enhanced cancer screening protocols and individualized risk-based treatment decisions. 31
Further longitudinal investigations with extended follow-up periods are warranted to elucidate whether BCDT use in MS predisposes patients to an HPV-associated cervical abnormalities and malignancies.
Risk mitigation strategies
An intentional and comprehensive approach is required to reduce the risk of HPV activation and cervical dysplasia/cancer in women with MS on DMTs that alter immune function and reduce surveillance against HPV, particularly those affecting lymphocyte presence and function, and with longer-term use of DMTs. 32 To achieve this, collaboration between neurologists, gynecologists, and primary care providers is key to educate patients about risks of HPV-related complications and encourage vaccines prior to DMT initiation. 33 The current Centers for Disease Control and Prevention (CDC) guidelines recommend three doses of HPV vaccination for individuals up to age 26 and ages 27–45 if deemed beneficial, noting that many eligible individuals may have missed the initial 2006 vaccination rollout. 34 Improving vaccination rates could ultimately reduce HPV-related disease burden in this at-risk demographic. Immunocompromised women are expected to benefit from vaccination, even if they have previously been exposed to the virus, as the vaccine-induced antibodies can prevent new infections, reinfections with eliminated HPV types, and the spread or reactivation of the virus within the genital tract. 35
Incorporating routine cervical cancer screenings, like Pap smears, into standard care alongside vaccination is crucial for early detection of HPV-related abnormalities. Due to the heightened likelihood of abnormal Pap smear results progressing to precancerous or cancerous conditions, follow-up colposcopy can further evaluate abnormal screening findings. 36 Guidelines vary by country and society; but more frequent HPV, cytological, or co-testing is recommended by the American Society for Colposcopy and Cervical Pathology in individuals with immunosuppression. 33 Proactive counseling, vaccination and increased screening for atypia could substantially reduce the risk of cervical cancer associated with immune suppression (Figure 1). 33
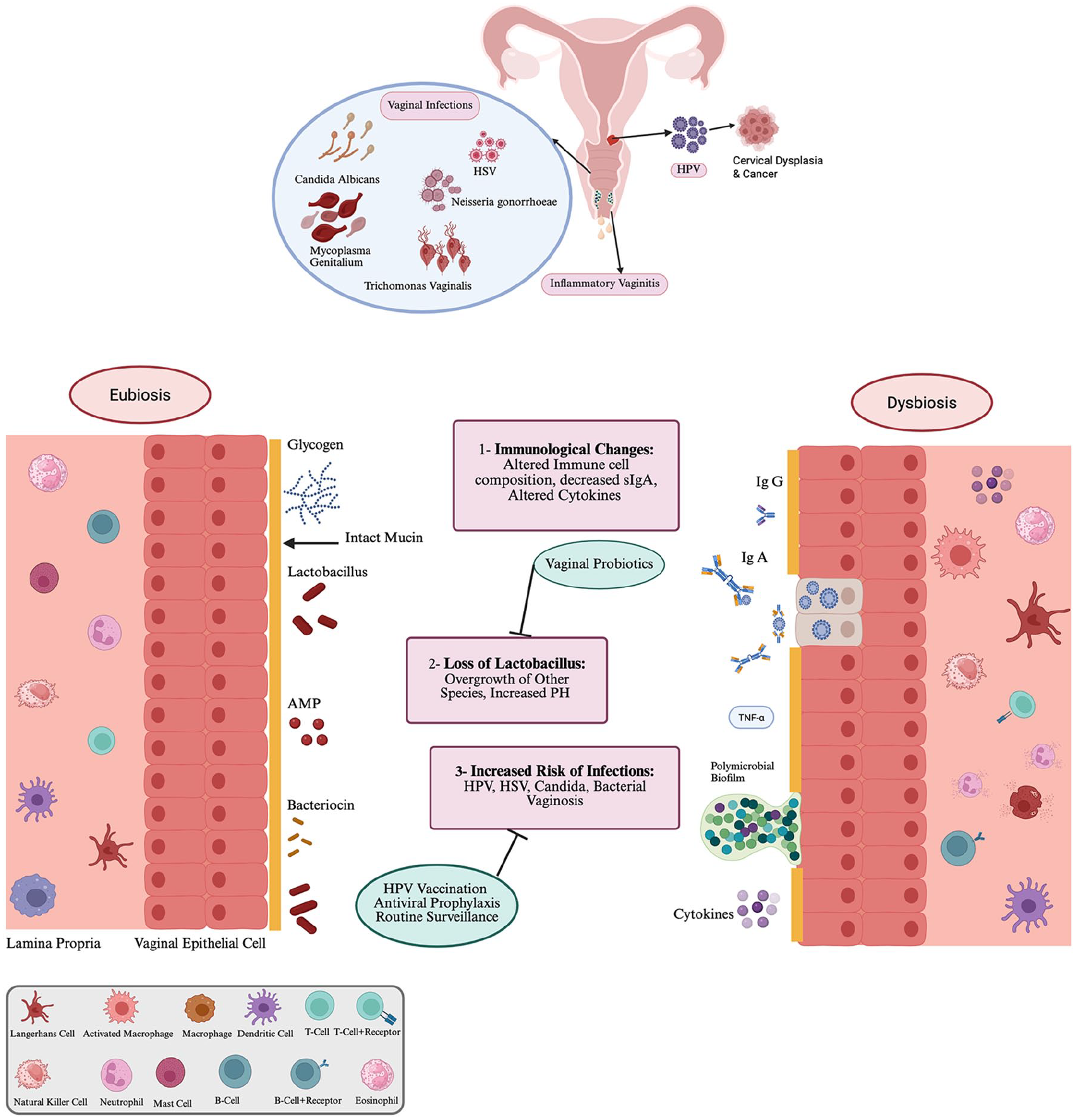
Overview of gynecological complications associated with Disease-Modifying Therapies for Multiple Sclerosis. Panel A. Three major categories of complications are (1) HPV infection and cervical dysplasia. (2) Infectious and inflammatory vaginitis and (3) HSV infection. Panel B. Overview of putative mechanisms leading to vaginal dysbiosis, and its sequelae. These figures were created using BioRender.com and is included with permission under a BioRender Publication License.
Inflammatory vaginitis and bacterial vaginosis
A growing body of work is linking the use of MS DMTs with a variety of vaginal inflammatory and infectious disorders, particularly implicating B-cell depletion. Unfortunately, the absence of systematic data collection limits accurate risk assessment, making current findings subject to bias.
Inflammatory vaginitis
Vulvovaginal pyoderma gangrenosum (PG) presents as painful vulvar or perianal ulcerations, with biopsies typically revealing neutrophilic dermatoses thought to result from an abnormal immune response or disrupted neutrophil chemotaxis. PG has been reported in women with hematological and rheumatologically conditions receiving rituximab,37,38 and some severe cases of PG associated with B-cell depletion are documented in women with MS, as well.39,40
Desquamative inflammatory vaginitis (DIV) is more common in women with autoimmune disorders and those receiving specific immunomodulatory therapies. There are some case reports of severe and recurrent DIV following treatment with rituximab, ocrelizumab, and natalizumab in MS.41–43 Another study noted no significant difference in inflammatory vaginitis rates between women with MS on BCDTs vs other DMTs, but vaginitis was common in both groups. 44 At present, it is not possible to distinguish whether these findings reflect treatment effects or an autoinflammatory process, given the higher autoimmunity risk in MS patients. The clinical impact of inflammatory vaginitis (IV) requires consideration. Indeed, it can have detrimental effects on quality of life and reproductive health, as detailed in a case series of three women with MS receiving BCDT, all of whom changed DMTs due to persistent and refractory IV symptoms. 41
Infectious vaginitis
A real-world study of women with MS reported an elevated risk of yeast vaginitis and bacterial vaginosis (BV) compared to the general population. In addition, rituximab use was associated with an increased risk of these conditions. 7 The available data are hindered by small sample sizes, confounding factors, and underdiagnosis and mismanagement.
Risk mitigation strategies
A proactive approach is required to manage the potential risk and sequelae of inflammatory and infectious vaginitis in patients receiving DMTs. This is especially important because patients may not associate vaginal symptoms with their MS management, and neurologists may not routinely enquire about patients’ gynecological health. Prior to initiating DMTs, counseling should address potential effects on vaginal health, empowering patients to recognize symptoms and seek medical attention when necessary. Clinicians should remain vigilant for signs of vaginal infections in MS patients receiving DMTs, incorporating discussions on vaginal health into safety monitoring. Symptoms like abnormal discharge, discomfort, or pruritus should prompt early consult with the patient’s primary care clinician or gynecologist to minimize diagnostic delays and consider interventions targeting the alleviation of dysbiosis and associated symptoms. Treatments may include antibiotics (CDC,) 45 and when appropriate, antifungal treatments, Lactobacillus probiotics, topical steroids, or intravaginal hormonal therapies for hormonal imbalances or atrophic vaginitis. 46 Managing additional risk factors like smoking and stress should also be considered to support vaginal health.
Viral infections
Most MS therapies are associated with an elevated risk of viral infections, including herpes simplex virus (HSV) reactivation. 47 Alemtuzumab and fingolimod, in particular, have been associated with markedly elevated rates of HSV infections, some of which were severe enough to require hospitalization.48,49 Ocrelizumab appears to increase the risk of HSV, although most cases were mild to moderate and none resulted in long-term complications. 50 Other DMTs for MS, such as teriflunomide, dimethyl fumarate, and mitoxantrone, do not show a clear association with an increased frequency or severity of HSV.
Risk mitigation strategies
Given the potential for DMTs used in MS to increase the risk of HSV infections, it is important to implement strong preventive strategies to reduce the risk of viral reactivation and associated complications. CDC-recommended strategies may include screening for prior viral exposure, and use of long-term low-dose preventive antivirals such as acyclovir, valacyclovir, and famciclovir in individuals who have experienced recurrence 51 —with specific dose and duration of antiviral prophylaxis tailored based on clinical context and individual risk factors. For patients on DMTs who experience viral reactivation, early recognition, prompt initiation of antiviral therapy, and vigilant monitoring are essential for controlling outbreaks and preventing severe complications and to ensure timely intervention and optimize patient recovery.
Discussion
The significant advances in MS treatment in recent years have been accompanied by emerging concerns about how DMTs might alter key regulatory mechanisms of gynecological health: innate immunity, the microbiome, and/or mucosal histology. This topical review identified three areas of concern: HPV activation and cervical dysplasia/cancer, inflammatory and infectious vaginitis, and HSV infections (Figure 1). Overall, studies were sparse, and some were likely affected by methodological limitations or bias. Furthermore, mechanistic investigations are lacking, particularly on the effect of DMTs on the vaginal microbiome. This scientific gap is particularly important given that the vaginal and gut microbiome represents key dimensions of materno-fetal immunoregulation, 52 alongside placental and breastmilk transfer of immunoglobulins. 53 Therefore, future studies should evaluate how maternal immune status might influence the development of microbial reservoirs and immune function in newborns.
From a research standpoint, incorporating routine gynecological assessments and microbiome analyses in MS clinical trials could provide valuable insights into the long-term impact of immunomodulatory therapies on reproductive health. Real-world studies could evaluate the role of DMT switch after symptoms, and the prophylactic role of vaginal probiotics, on gynecological outcomes. From a clinical practice standpoint, monitoring (e.g. through a brief gynecological health screen) and addressing potential gynecological impacts of DMTs could minimize risks of their use. Approaches are summarized in Table 1. Overall, clinicians should recognize gynecological complications as potential sequelae of long-term immune suppression, and adopt a multidisciplinary approach that integrates gynecological counseling and screening as integral to the MS therapeutic strategy. By doing so, they could mitigate gynecological risks, optimize patient outcomes, and enhance overall well-being.
Overview of gynecological complications associated with disease-modifying therapies used for the treatment of multiple sclerosis.
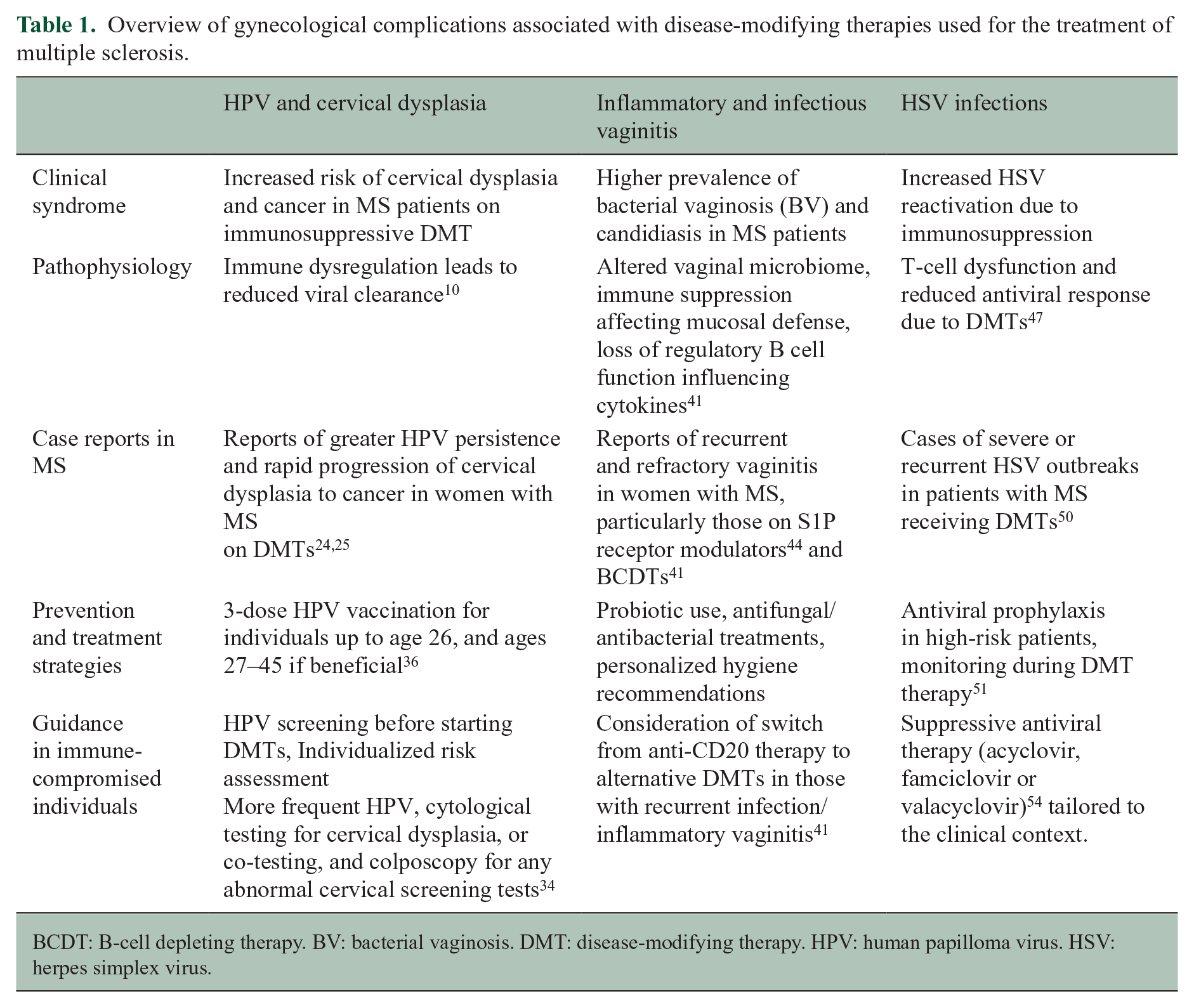
BCDT: B-cell depleting therapy. BV: bacterial vaginosis. DMT: disease-modifying therapy. HPV: human papilloma virus. HSV: herpes simplex virus.
Supplemental Material
sj-docx-1-msj-10.1177_13524585251346371 – Supplemental material for Gynecological health: A missing link in comprehensive treatment monitoring for multiple sclerosis
Supplemental material, sj-docx-1-msj-10.1177_13524585251346371 for Gynecological health: A missing link in comprehensive treatment monitoring for multiple sclerosis by Melika Arab Bafrani, Viviana Rios, Min Ji Kim, Ayushi Balan and Riley Bove in Multiple Sclerosis Journal
Footnotes
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.A.B., V.R., M.J.K, and A.B. report no conflicts.
R. B. has received research support from Biogen, Eli Lilly, Novartis and Roche Genentech. She has received personal fees for advisory boards and consulting from Alexion, Amgen, Cadenza, EMD Serono, Sanofi and TG Therapeutics.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: RB is funded by the National Multiple Sclerosis Society Harry Weaver Award.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
