Abstract
Wearable technology refers to any sensor worn on the person, making continuous and remote monitoring available to many people with chronic disease, including multiple sclerosis (MS). Daily monitoring seems an ideal solution either as an outcome measure or as an adjunct to support rater-based monitoring in both clinical and research settings. There has been an increase in solutions that are available, yet there is little consensus on the most appropriate solution to use in either MS research or clinical practice. We completed a scoping review (using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR) guidelines) to summarise the wearable solutions available in MS, to identify those approaches that could potentially be utilised in clinical trials, by evaluating the following: scalability, cost, patient adaptability and accuracy. We identified 35 unique products that measure gait, cognition, upper limb function, activity, mood and fatigue, with most of these solutions being phone applications.
Keywords
Introduction
Assessment of new treatments in multiple sclerosis (MS) studies necessitates effective, reliable and validated outcome measures. Most clinical MS outcome measures are rater dependent and are applied episodically. 1 Clinical trials in MS require sensitive outcome measures, that can detect small changes in disability or functional improvement on a frequent basis, which can then reliably reflect long-term changes. 2
With the advances in technology over the last few decades, it is now possible to explore methods of accurate, sensitive and objective continuous remote monitoring.3,4 The World Health Organization (WHO) defines mHealth as a medical and public health practice supported by mobile devices, such as mobile phones. 5 Wearable technology is defined as incorporating a microprocessor and an Internet connection. Wearable technology, otherwise often referred to as wearables, are mobile technology solutions that can be worn by the person, as accessories or even embedded in clothing, and often include passive or active tracking capabilities which can be used to assess health and well-being. 6 Our definition of wearable technology encompasses mHealth solutions because the growth of mobile networks (3G, 4G and 5G) has enabled the development of wearables. Wearables evolved from fitness activity trackers to wristwatches and the more robust mobile applications including Bluetooth headsets, smartwatches and smartphones. Common examples of these devices include the Fitbit® activity band and smartphone applications like MapMyRun®. Advancements in wearable technology and phone applications (‘apps’) enable continuous patient-based monitoring and provide feedback on daily life. The results of daily monitoring using wearable technology could be used either as an outcome measure or as an adjunct to support rater-based assessments. There has been an increase in solutions that are available for those who are diagnosed with chronic illness, especially in regard to neurological disease. 7 Yet, there is little consensus on the most appropriate solution to use in either MS research or clinical practice.
Studies have shown that patients, caregivers and health care professionals find value in using such devices, especially when they are less invasive in day-to-day situations and provide real-time feedback.8,9 Currently, there are two randomised controlled trials (RCTs)10,11 that have utilised wearable technology in people with multiple sclerosis (PwMS); however, there are several more RCTs currently on-going that utilise wearable technology, for example, the MD3001 (SPI2) trial. 12
The advantages and disadvantages of wearable technology are summarised in Table 1 above.
Potential advantages and disadvantages of wearable technology in trials.
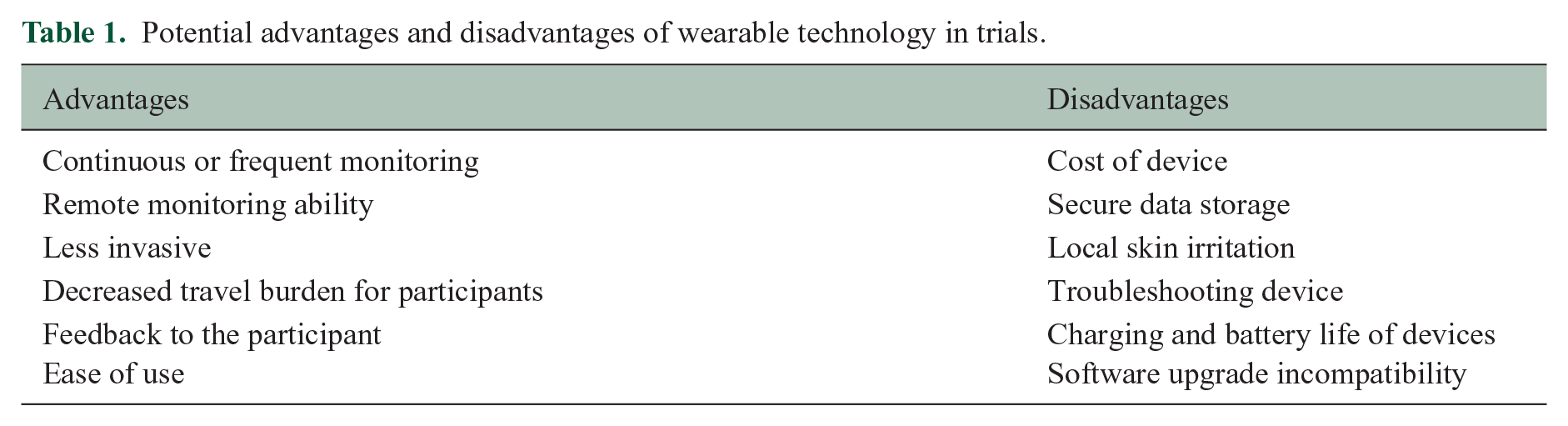
Software upgrades could be one of the biggest limitations to wearables produced by commercial entities. The upgrade also applies to device algorithms as manufacturers attempt to improve both parameter estimation and user satisfaction. This leads to a change in not only the appearance and behaviour of the device but also the algorithms used in logging and reporting the data. 13 Maintaining a solution through a trial without a software update is difficult, and if an update is done, it could significantly change any wearable-related outcomes.
The rationale for conducting this scoping review was to understand each solution and its utility in MS. This review was commissioned by the UK MS Society on behalf of the Outcome Measures Working Group (part of the Expert Consortium on Progression in MS Clinical Trials, a UK MS Society initiative). We reviewed wearable technology solutions with a particular interest in their potential to detect changes in function for PwMS in a more reliable and accurate manner, and their suitability for use in a UK-wide multi-centre platform trial.
Objective
The objective was to identify all validated wearable solutions for PwMS and determine suitability for use in a UK-wide multi-centre platform trial by considering the following factors: reproducibility in MS populations, feasibility (including cost), patient adaptability and prior use in an RCT.
Methods
We used a scoping review approach which aims to map the key concepts underpinning a research area, especially where an area has not been reviewed comprehensively before (Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR)). 14
Our search strategy utilised subject heading searches: ‘Multiple Sclerosis’ and ‘wearable electronic devices’, as well as keywords ‘wearable technology’ and ‘electronic devices’. The literature search was conducted using MEDLINE (via PubMed) and Embase (via OVID) databases. This search included articles published from database inception to 30 May 2019. Additional searches looked at authors who have frequently published with different devices as well as forward and backward citation tracking of included papers. The scoping review followed the PRISMA-ScR guidelines. 15
When examining suitability of a wearable device for use in a UK-wide multi-centre platform trial, we considered several factors: reproducibility (defined as the number of studies examining the solution in MS), feasibility (including cost), patient adaptability and prior use in an RCT. The results of this are shown in Figure 2 and Table 2.
Summary of unique solutions and their general characteristics.
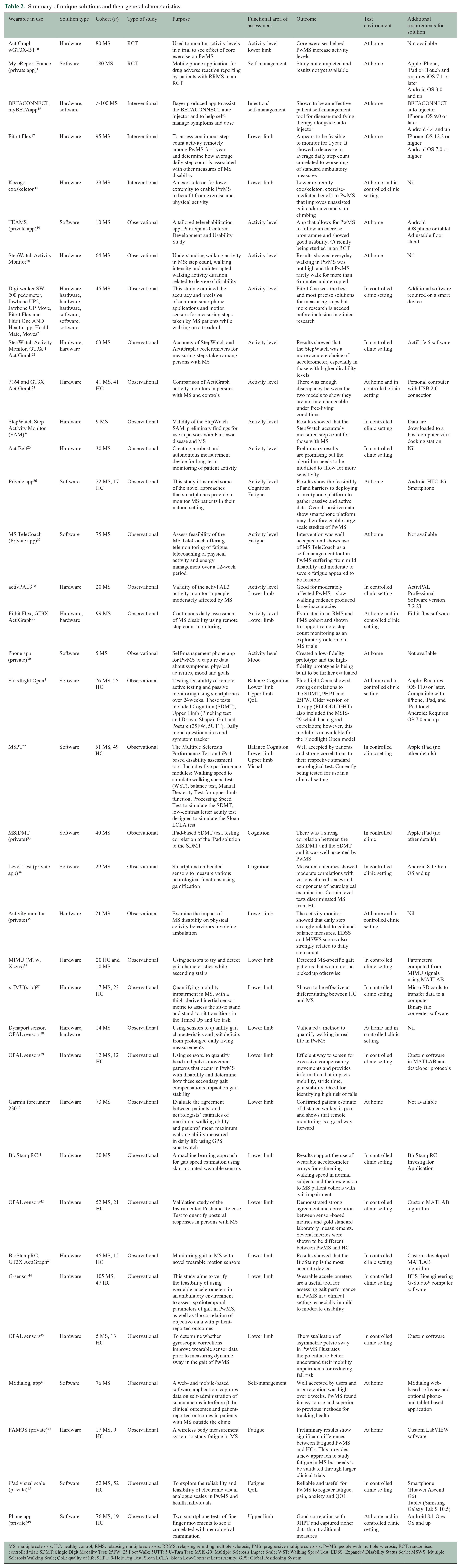
MS: multiple sclerosis; HC: healthy control; RMS: relapsing multiple sclerosis; RRMS: relapsing remitting multiple sclerosis; PMS: progressive multiple sclerosis; PwMS: people with multiple sclerosis; RCT: randomised controlled trial; SDMT: Single Digit Modality Test; 25FW: 25 Foot Walk; 5UTT: 5 U-Turn Test; MSIS-29: Multiple Sclerosis Impact Scale; WST: Walking Speed Test; EDSS: Expanded Disability Status Scale; MSWS: Multiple Sclerosis Walking Scale; QoL: quality of life; 9HPT: 9-Hole Peg Test; Sloan LCLA: Sloan Low-Contrast Letter Acuity; GPS: Global Positioning System.
Study eligibility criteria
The inclusion criteria were defined as (1) primary research studies that used wearable technology in a cohort of PwMS of all ages (adult or paediatric), (2) studies from any geographical location and (3) reported in the English language. Exclusion criteria were defined as (1) wearable solutions intended for another health condition, (2) non-wearable solutions, (3) non-primary research such as narrative reviews and (4) abstracts that did not have full-text available.
After screening titles and abstracts, duplicates were removed and the full text of each paper was assessed for eligibility according to the criteria stated above.
Data extraction
The data to be extracted for each article were determined in consultation with the second author (G.P.) and a data extraction form was created. Descriptive characteristics were extracted where available for (1) wearable device, (2) cohort, (3) type of study, (4) purpose, (5) functional area of assessment and (6) outcome.
Developers of the wearable technology were separated into the following categories:
Health care–related agency: Hospitals, clinics or government organisations directly related to health care
Pharmaceutical company: entities with commercial purposes to research, develop, market or distribute drugs in the context of health care
Educational organisation: any educational organisations such as universities, colleges, libraries or schools not directly related to health care
Small and medium enterprises: start-ups, software developing companies or any other private organisation that identified themselves as an enterprise and not individuals
Results
The searches yielded 1880 potentially relevant articles. Removing duplicates and applying the eligibility criteria resulted in a total of 35 unique MS wearable technology solutions, which included 3 unique solutions that were yielded from conferences and scientific meetings. Figure 1 describes the PRISMA flow diagram.
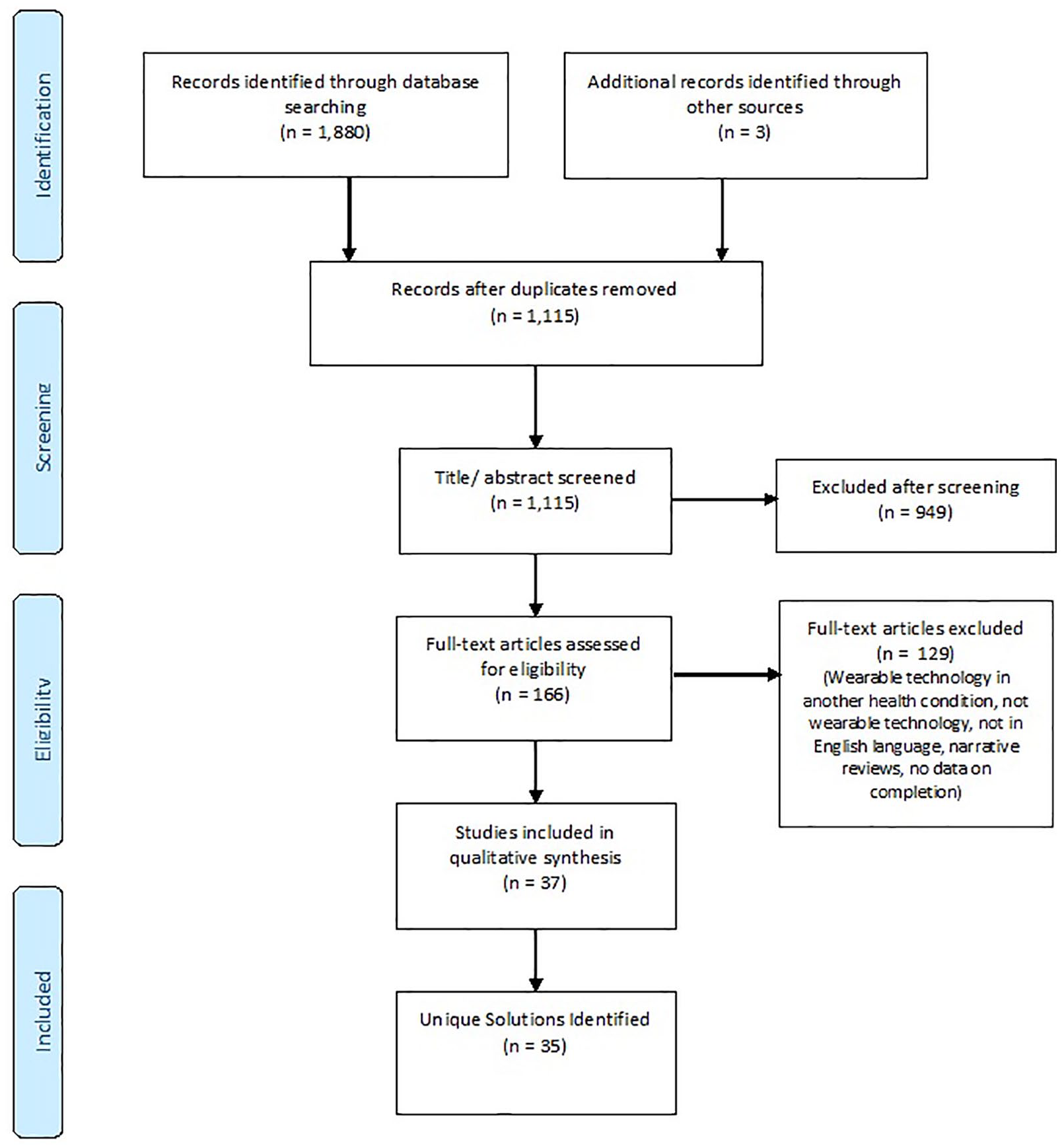
PRISMA flow diagram. 53
The list of the included wearable technology solutions and the frequency with which they appeared in validated studies is shown in Figure 2. A majority of the solutions that were used in studies in PwMS were applications (apps), accelerometers and activity monitors. The older studies predominantly focused on measuring activity, walking or gait since activity monitors, accelerometers and gyroscopes were the most readily available and advanced technology at the time. This result is not unexpected because the nature of MS disease progression would require sensors focused on assessments based on activity and function, both easily derived from accelerometers. Within the solutions available for monitoring activity levels, there is great variability between the outcomes available. Certain solutions provide a basic step count for example, Fitbit, whereas others provide additional metrics such as stride length and gait characteristics. It is important to determine the key outcome measures of interest to best guide which wearable to use. Included are four unique app solutions for cognition, which were all created more recently, as apps are becoming an easier wearable technology to develop and deploy. There were a handful of wearables that focused on fatigue, mood, quality of life (QoL) and self-management.
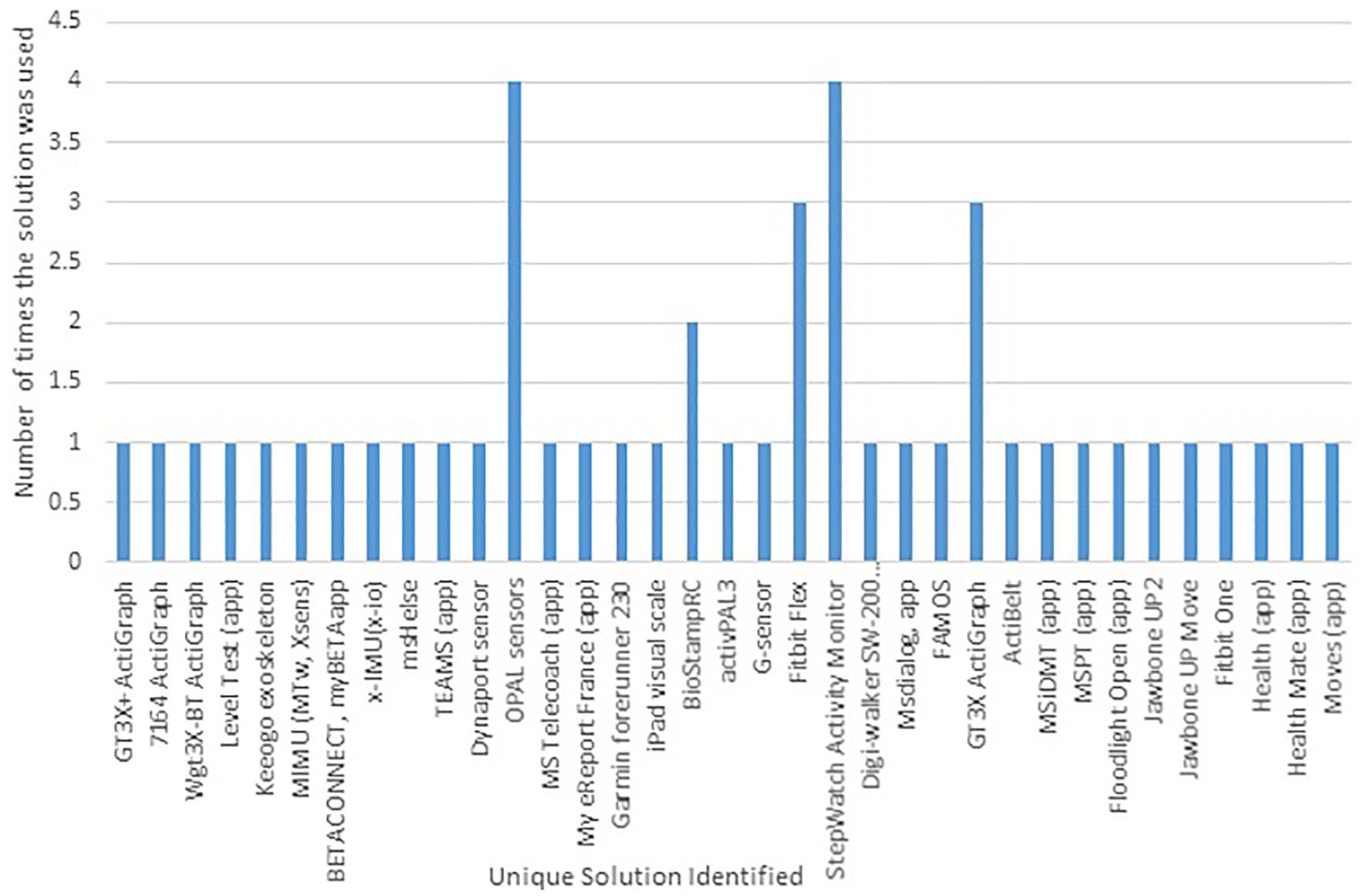
Unique devices and frequency of appearance.
A summary of the general characteristics of the unique wearable technology solutions found is shown in Table 2.
There is significant variability in the per-unit cost of each product, and the decision as to which wearable to use depends largely on the study budget and outcomes of interest. Costs may vary significantly when using a physical wearable sensor compared to a smart-device application. Aside from per-unit cost, other considerations include repair or replacement of faulty devices, annual maintenance charges, software package costs, return of devices, charging capabilities and collection of data (postage vs. remote upload). Physical wearable sensors risk being ‘phased out’ and being replaced with newer models that have not been tested in an MS population. Applications may alleviate this problem by sending out software updates, which the user can download. Users could however face problems if this update exceeds the smart device support capability.
There does seem to be a shift towards developing more validated wearable technology solutions for MS and focusing on health care adoption to make sure that dissemination of the solutions is more successful and reaches a wider population. This is seen by the increase in the number of publications related to the subject of wearable technology in MS. In addition, as a result of an increased number of solutions being validated, wearable technologies are now becoming more utilised in RCTs. At the time of writing this review, we had identified two RCTs that utilise wearable technology; however, we are aware of at least one other RCT which was published using wearable technology in MS. 50 We believe that this study did not appear in our literature search, because the term ‘wearables’ or ‘mHealth’ is not used in the paper nor is it referred to in the keywords or subject headings. We acknowledge that our search terms may have excluded other studies involving wearable technology, in MS. We are also aware of several other RCTs that are currently being run, that employ wearable technology such as the SPI2 and TEAMS studies.51,52
While doing this review, we identified 10 (27%) solutions that we classed as ‘private’ solutions as they had been created, tested and not available to the public, as shown in Table 2. This seems to have happened for various reasons, including not having the necessary resources to further validate or improve the solution or not having enough resources to gain regulatory approval. All the identified solutions that are private and unavailable were created by health care or educational organisations. Ideally, independent validation prior to clinical or research use seems appropriate; however, this may not be feasible due to on-going costs. When comparing this to the solutions created or funded by pharmaceutical companies, for example, the FLOODLIGHT app and the MSPT, it was shown that 98% of the solutions created by pharmaceutical companies were successfully implemented and disseminated, as they had enough resources to manage the on-going cost and effort required to gain regulatory approval and market the products.
Wearables are gaining importance in MS; however, there are many lessons to be learnt from its use in other chronic neurological disorders, such as Parkinson’s disease. 54 While the promise of unsupervised assessments is alluring, and could save time and cost, using these devices in clinical and research settings is far from seamless due to several issues. Many wearable devices have not been validated or approved for clinical use in people with Parkinson’s disease. In addition, as gold standards are variable and sometimes scarce, unsupervised patient monitoring also brings new challenges (e.g. using a patients’ diary to validate devices capturing motor fluctuations).55,56
Limiting factors to consider when developing wearable technology are adherence and usability by both the participant and the researcher. PwMS have shown a high level of acceptance when using smartphone applications (apps), although this may wear off as the disease progresses due to increased disability (e.g. decreased hand dexterity). 8 Factors to consider when designing a solution are convenience, placement of the wearable device, appearance of the sensor and feedback of results to the PwMS. Patient feedback is extremely important in keeping PwMS interested and engaged in their own health. Many solutions have opted for patient-friendly readouts, while more complex data and parameters are available to the respective clinicians and researchers. 57 With regard to usability by the researcher, issues to consider include troubleshooting hardware and software, technical support and ease of implementation with the participant.
Another limiting factor, from a UK perspective, was the approach from regulatory bodies. Stricter guidelines determined what was seen as a medical device or classed as an observational tool. New guidelines which have recently come into effect, such as the new Evidence Standards Framework by the National Institute for Health and Care Excellence (NICE) in March 2019 and the Regulation (EU) 2017/745 by the European Parliament (May 2020), will also work to create stricter guidance for patient safety, security monitoring and data security. Many solutions that are available elsewhere have struggled to implement themselves outside of research in the United Kingdom due to these guidelines.
Limitations
Although we used a detailed process to search and document the currently validated solutions in MS, there were several limitations. The nature of this scoping report was web search based and, thus, relied on university subscription to journals to access the papers, although only 5 of 1115 titles screened could not be accessed in this way. The inclusion of only English language papers may also be considered a limitation.
Conclusion
In the coming years, we can expect to see more sensitive and comprehensive measures being developed, with the idea of using wearable solutions perhaps as the gold standard to measure outcome measures in studies and clinical practice. However, at present, guidelines on what wearable technology should be used in clinical practice and research are absent, and this is an area that will require considerable attention and stringency.
While doing the review, we came across many unvalidated solutions available for PwMS across a range of outcome measures, most of them being phone or iPad solutions. We classed a solution as ‘unvalidated’ if there was an inability to demonstrate test–retest reliability and/or failure to demonstrate difference between healthy controls and PwMS. In comparison, the validated solutions are rather limited, but are most advanced particularly in measurements of gait (characteristics) and balance. These solutions often provide the greatest accuracy and acceptance rate, as gait is one of the earliest outcome measures explored in MS wearable solutions.
Also with the advances in mobile technology, more solutions are focusing on utilising common wearables such as smartphones, smartwatches and tablets, to increase accessibility and minimise costs to the user. Looking forward, there is also a change occurring from single-measure solutions to multi-measure and multi-sensor solutions, such as the Floodlight Open app, which utilises multiple sensors within a smartphone to remotely measure gait, cognition and upper limb function.
As development in wearable technology in MS is still on-going, we can expect to see newer solutions focusing on other areas with technology advancements that allow for more upper body and cognitive measures. There is a dearth of validated solutions available for fatigue, mood and pain.
The future of wearable technology in MS therefore looks promising with the potential to become a primary, co-primary or adjunctive monitoring tool in research and clinical practice.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: S.A. does not have any disclosures. G.P. has received consultancy sums from the MS Society and the Eastern Academic Health Science Network and paid employment from the National Institute for Health Research (NIHR) East of England Applied Research Collaboration. E.G. does not have any disclosures. F.B. is a board member for Neurology, Brain, Radiology and Multiple Sclerosis Journal (MSJ) and a section editor for Neuroradiology. He has also received personal fees from Springer, Bayer, Biogen, IXICO Ltd, GeNeuro and Roche as well as grants from Novartis, TEVA, Merck, Biogen, IMI-EU, GE Healthcare, UK MS Society, Dutch Foundation MS Research, NWO and NIHR, outside the submitted work. In the last 3 years, J.C. has received support from the Efficacy and Evaluation (EME) Programme, a Medical Research Council (MRC) and NIHR partnership and the Health Technology Assessment (HTA) Programme (NIHR), the UK MS Society, the US National MS Society and the Rosetrees Trust. He has been a local principal investigator for a trial in MS funded by the Canadian MS society. A local principal investigator for commercial trials funded by Actelion, Biogen, Novartis and Roche; has received an investigator grant from Novartis; and has taken part in advisory boards/consultancy for Azadyne, Biogen, Celgene, MedDay, Merck and Roche.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by the MS Society and the Expert Consortium for Progression in MS Clinical Trials: Outcome Measures Working Group.
