Abstract
Background:
The Nine-Hole Peg Test (9HPT) is the golden standard to measure manual dexterity in people with multiple sclerosis (MS). However, administration requires trained personnel and dedicated time during a clinical visit.
Objectives:
The objective of this study is to validate a smartphone-based test for remote manual dexterity assessment, the ico
Methods:
A total of 65 MS and 81 healthy subjects were tested, and 20 healthy subjects were retested 2 weeks later.
Results:
The FDT significantly correlated with the 9HPT (dominant: ρ = 0.62, p < 0.001; non-dominant: ρ = 0.52, p < 0.001). MS subjects had significantly higher FDT scores than healthy subjects (dominant: p = 0.015; non-dominant: p = 0.013), which was not the case for the 9HPT. A significant correlation with age (dominant: ρ = 0.46, p < 0.001; non-dominant: ρ = 0.40, p = 0.002), Expanded Disability Status Scale (EDSS, dominant: ρ = 0.36, p = 0.005; non-dominant: ρ = 0.31, p = 0.024), and disease duration for the non-dominant hand (ρ = 0.31, p = 0.016) was observed. There was a good test–retest reliability in healthy subjects (dominant: r = 0.69, p = 0.001; non-dominant: r = 0.87, p < 0.001).
Conclusions:
The ico
Keywords
Introduction
Multiple sclerosis (MS) 1 is the most common inflammatory and neurodegenerative disease in young adults, affecting 2.8 million people worldwide. Between 60% and 75% of persons with multiple sclerosis (PwMS) with mild disability have an impaired manual dexterity, with an increasing prevalence with worsening disease. 2 This impairment is related to decreased strength, sensory dysfunction, and/or tremor and results in a decreased participation in social, home, and productive activities. 3
The Nine-Hole Peg Test (9HPT) is considered the golden standard for the assessment of manual dexterity in MS, both in a clinical and research context. 4 In this test, subjects have to place nine pegs into holes and subsequently remove them one by one. This test has a good convergent, discriminant, and ecological validity and test–retest reliability. 4 However, its administration requires clinic visits, material, and trained personnel, which not all clinical sites have the resources for. Furthermore, there are significant practice effects after repeated administration, resulting in a biased estimation of dexterity. 4 Finally, MS is characterized by important fluctuations of symptoms due to temperature changes 5 and fatigue. 6 The moment of a clinical visit, which in itself may cause stress and fatigue, might thus not be the most accurate representation of an individual’s function.
Since the COVID-19 pandemic, there has been an increasing interest in remote patient monitoring, which reduces the need for clinical visits, and thus costs and patient burden. Smartphone-based assessments are of particular interest due to their broad accessibility 7 and allow the patient to perform the evaluation at a time of their choosing. In this study, we developed a smartphone-based test for the assessment of manual dexterity in MS and assessed the concurrent validity and test–retest reliability of the Finger Dexterity Test (FDT), as well as its correlation with clinical parameters and its ability to differentiate between PwMS and healthy controls (HCs).
Methods
Study protocol and ethics
The study protocol, patient information, and informed consent were approved by the ethics committees at the University Hospital Brussels (B.U.N. 143201940335, 2019/173, 17 July 2019) and the National MS Center Melsbroek. Informed consent was obtained from all participants before inclusion. This study is part of a bigger validation study of a smartphone app that assesses both cognitive performance 8 and manual dexterity in PwMS, to be included into the icompanion app. 9
Recruitment
PwMS were recruited at the University Hospital Brussels and the National MS Center Melsbroek, whereas HCs were recruited via online advertising, leaflets, and acquaintances of the examiners. All subjects were native Dutch-speaking or bilingual. For PwMS, the only additional inclusion criterion was a confirmed diagnosis of MS based on the modified McDonald criteria, 10 whereas a subject was considered a HC if no neurological, psychiatric, or learning disorder was present. Exclusion criteria for PwMS were the presence of other neurological, psychiatric, or learning disorders, hospitalization in the context of a medical problem, and a relapse within the last month.
In total, 65 PwMS and 81 HCs—matched for age, sex, and education—were included in this study. Of those, 60 PwMS and 79 HCs completed the FDT for both the dominant and non-dominant hand. To assess test–retest reliability, 20 HCs were tested a second time 2–3 weeks later, on a similar moment to the first testing session (morning vs evening, weekday vs weekend). During this second session, only the smartphone-based tests were repeated. PwMS were tested in the hospital, while HCs were tested outside a hospital setting, all in the presence of an examiner.
Demographic and clinical information
The following demographic information was collected: age, sex, education level, subjective visual and manual problems (yes/no), current medication, recent cognitive assessment, and Edinburgh Handedness Inventory (EHI 11 ). In addition, the following information was collected for PwMS: Expanded Disability Status Scale score 12 (EDSS, assessed by a neurologist during clinical visit within the last 6 months), disease duration, and disease type.
FDT
All tests were performed on a preconfigured smartphone (Samsung Galaxy A10), on which the application was installed. In the FDT, the goal is to “fry” and subsequently “serve” a total of 10 eggs as quickly as possible. For each of the eggs (presented in the same horizontal position on the screen but with a shuffled list of 10 different y coordinates of the egg), a sequence of steps is performed. First, an egg is presented on the screen, which the subject has to break by pinching it between two fingers. In a second step, the subject needs to drag and release the egg to a frying pan, where it is cooked. Finally, the cooked egg is pinched and dragged to a plate, where it is served (see Figure 1 and Supplemental Figure S1). A set of instructions, including a video and a practice phase of 10 eggs, precedes the actual testing phase. The entire test, including both the practice and test phase, is performed first with the dominant hand and subsequently with the non-dominant hand. The final score is defined as the total time to “serve” 10 eggs, which is recorded by the application.
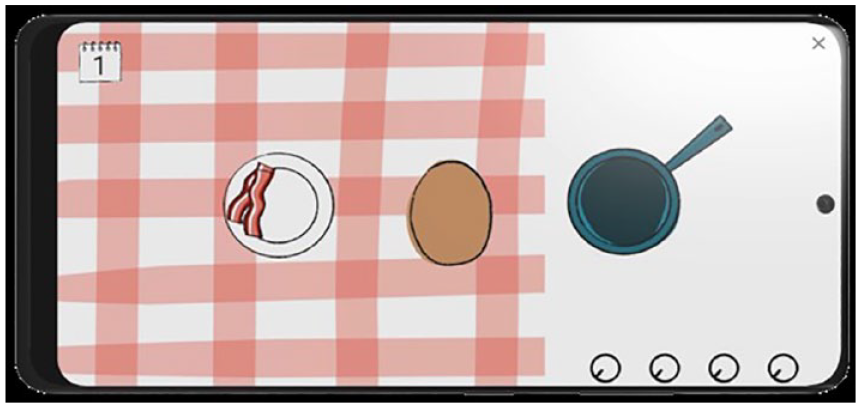
Finger Dexterity Test.
9HPT
The objective is to transfer nine plastic pegs from a plastic container into nine holes (phase 1) and subsequently back to the container (phase 2), one peg at a time, as quickly as possible. Subjects performed two trials with the dominant hand, followed by two trials with the non-dominant hand. If a peg fell on the table but was still reachable by the subject, the trial was continued. If the peg fell on the floor, the trial was discontinued and repeated from the start. The time to complete each trial was measured using a stopwatch. The score is the average time to complete the two trials for each hand. 4
Questionnaires
At the end of the testing session, subjects were asked to complete the Beck Depression Inventory (BDI 13 ) and the Fatigue Scale for Motor and Cognitive (FSMC 14 ) functions. Finally, the perceived burden of the performed tests was assessed on a seven-point scale. These questionnaires were completed independently by the subjects on paper, but they were able to ask questions or clarification if necessary.
Validation procedure and statistics
Group differences between PwMS and HC were assessed with a chi-square test for categorical variables and a Mann–Whitney U-test for continuous variables.
The validation criteria used in this study are based on the recommendations of Benedict et al. 15 Concurrent validity was assessed using the Spearman correlation between the FDT and the 9HPT. The difference in mean FDT score between PwMS and HC was investigated using a Mann–Whitney U-test. The correlation with clinical parameters was investigated by assessing the Spearman correlation between the FDT, on one hand, and age, education, disease duration, EDSS, BDI score, and FSMC score, on the other hand. Finally, the test–retest reliability was investigated in the subset of HC who performed a second testing session, by assessing the Pearson correlation (according to the recommendations of Benedict et al. 15 ) between the FDT score at the first and second sessions. Since our data were not normally distributed, we also assessed this using a Spearman correlation. Finally, we also assessed the intraclass correlation coefficient, which is a commonly used measure of test–retest reliability. 16 A two-sided test with a type I error probability of 0.05 was used for all analyses.
To interpret the magnitude of correlations, we used the classification of Portney and Watkins 17 , with a correlation coefficient <0.25, 0.25–0.50, 0.50–0.75, and >0.75 being considered as small, fair, moderate to good, and good to excellent, respectively. The interpretation of the intraclass correlation coefficient was based on the classification of Koo and Li 18 , with a coefficient <0.5, 0.5–0.75, 0.75–0.9, and >0.90 indicating a poor, moderate, good, and excellent reliability, respectively.
Per validation criterion, we performed an additional analysis without FDT outliers. Outliers were only excluded from the analyses if they met both of the following conditions: value below the 25th percentile minus 1.5 times the interquartile range or above the 75th percentile plus 1.5 times the interquartile range, and a valid reason for exclusion (i.e. if there was a lag of the application or interference of long fingernails). Furthermore, we repeated the above-mentioned analyses with a different scoring method for the FDT, namely, the number of eggs/second instead of the total time of serving 10 eggs, as suggested in Feys et al., 4 to reduce floor effects and to allow for a more normal distribution of the data. Finally, to correct performance on the FDT for age and sex, we performed a regression-based normalization separately for the dominant and the non-dominant hand, which can be found in Supplemental materials.
Results
Demographic
In Table 1, the group differences between PwMS and HC are depicted. A recent EDSS score was available for 60 PwMS. BDI and FSMC scores were available for 63 PwMS and for 80 and 74 HCs, respectively. PwMS had a higher score on the questionnaires for depression (BDI) and fatigue (FSMC). There was no significant difference between PwMS and HC on any of the other demographic and clinical parameters.
Demographic and clinical information.
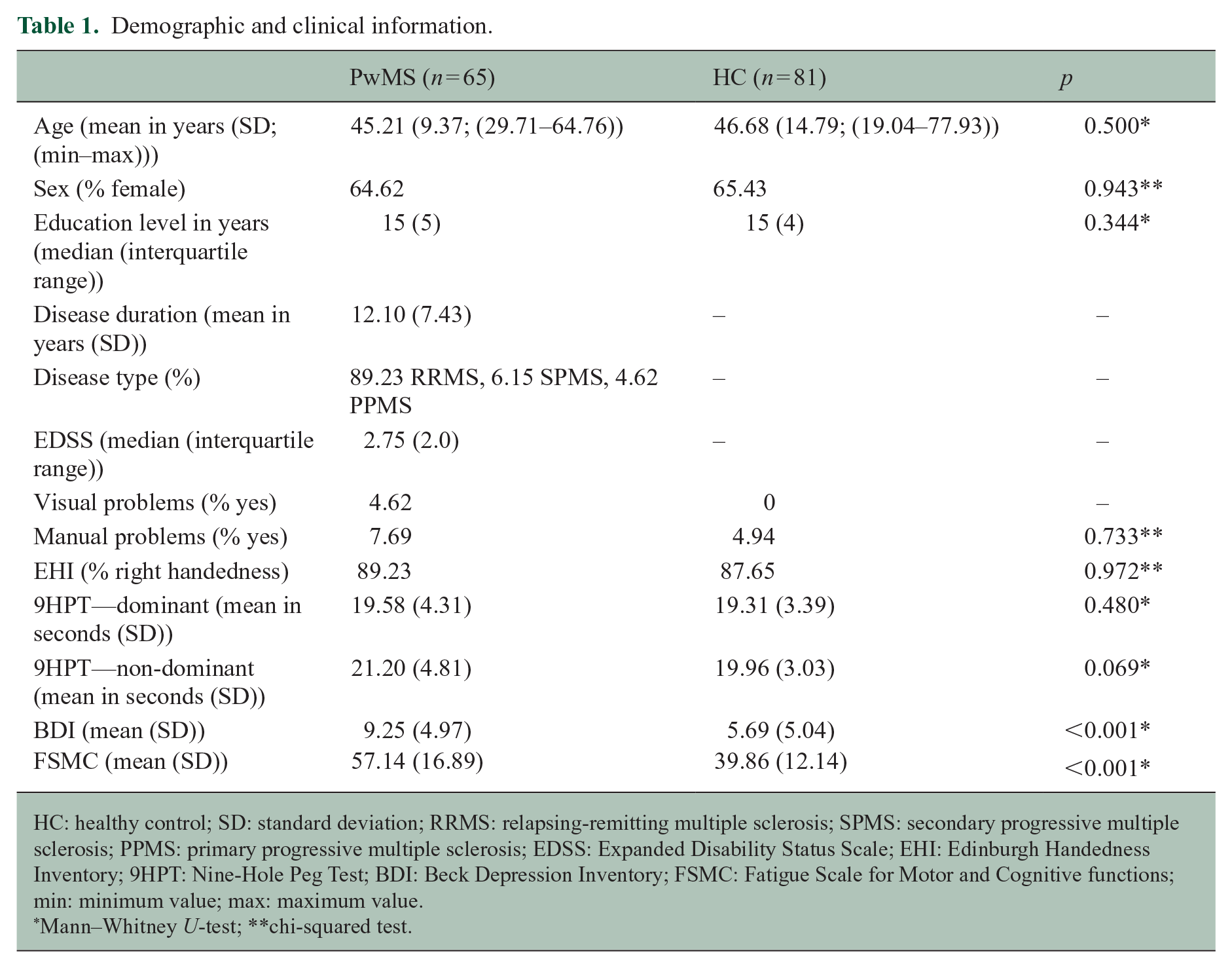
HC: healthy control; SD: standard deviation; RRMS: relapsing-remitting multiple sclerosis; SPMS: secondary progressive multiple sclerosis; PPMS: primary progressive multiple sclerosis; EDSS: Expanded Disability Status Scale; EHI: Edinburgh Handedness Inventory; 9HPT: Nine-Hole Peg Test; BDI: Beck Depression Inventory; FSMC: Fatigue Scale for Motor and Cognitive functions; min: minimum value; max: maximum value.
Mann–Whitney U-test; **chi-squared test.
Concurrent validity
There was a significant correlation between the 9HPT and FDT scores of the PwMS in the dominant and non-dominant hand (Spearman’s ρ (hereafter referred to as ρ) = 0.62, p < 0.001 and ρ = 0.52, p < 0.001, respectively), as well as the HC (dominant hand: ρ = 0.28, p = 0.012; non-dominant hand: ρ = 0.37, p = 0.001). When excluding outliers (MS: 2 dominant, 1 non-dominant; HC: two dominant hand, five non-dominant hand), the correlations with the 9HPT remained significant for the PwMS (dominant: ρ = 0.63, p < 0.001; non-dominant: ρ = 0.49, p < 0.001) and the HC (dominant: ρ = 0.30, p = 0.007; non-dominant: ρ = 0.35, p = 0.002). The association between these two variables is depicted in Figures 2 and 3. The depiction without outliers can be found in Supplemental materials (Supplemental Figures S2 and S3).
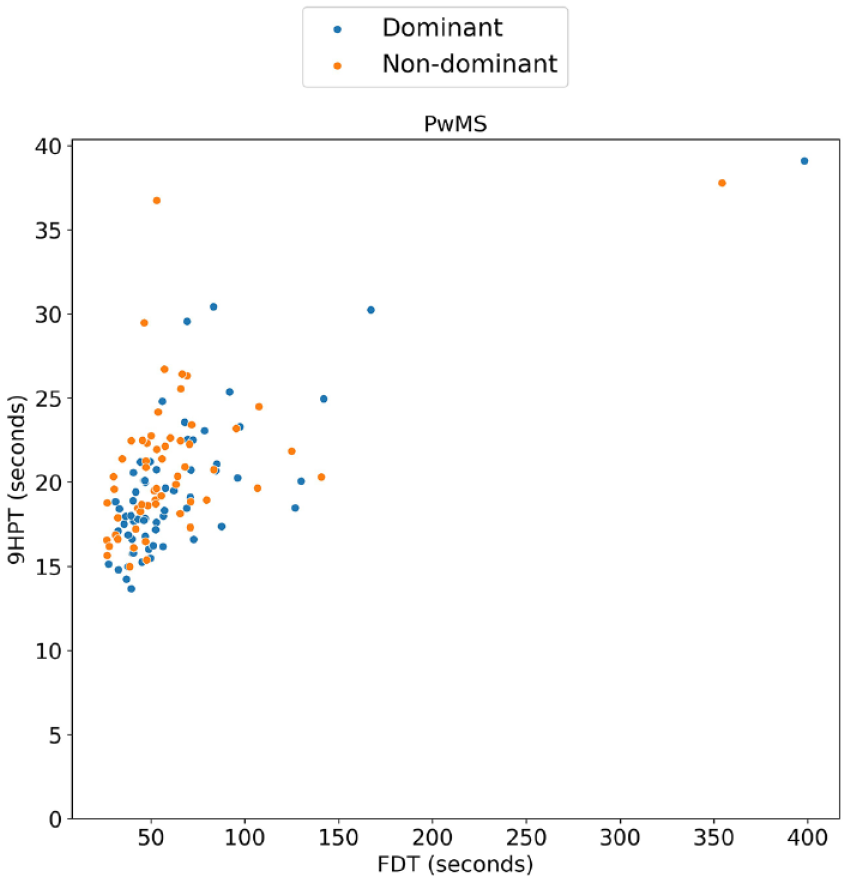
Concurrent validity in subjects with MS (PwMS)—scatterplot between Finger Dexterity Test (FDT) and Nine-Hole Peg Test (9HPT).
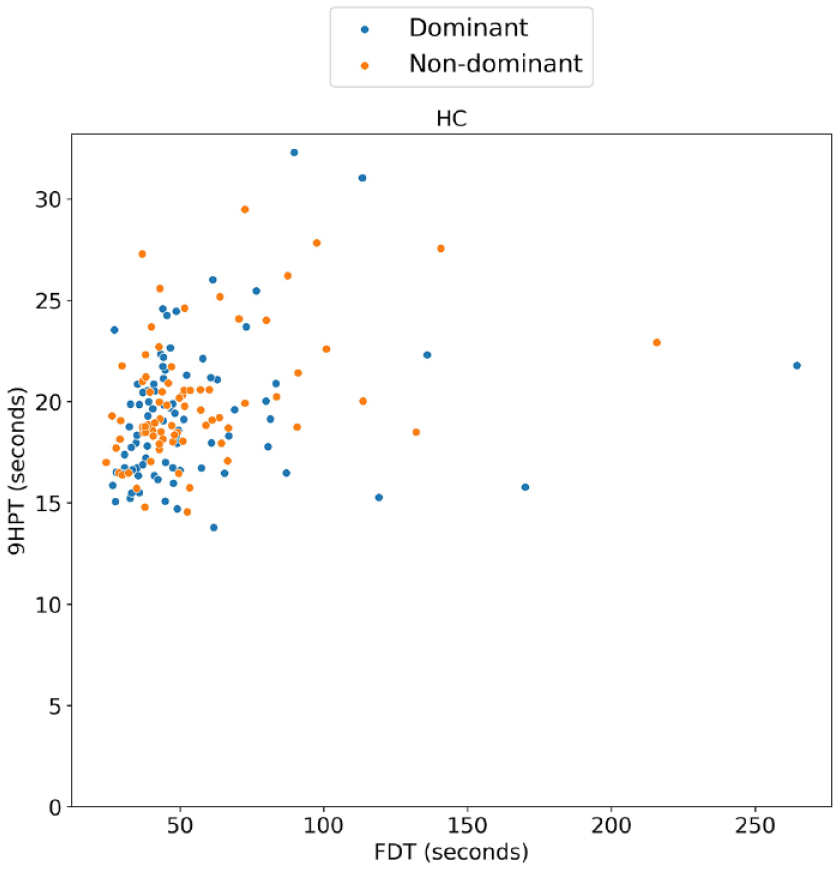
Concurrent validity in healthy control subjects (HC)—scatterplot between Finger Dexterity Test (FDT) and Nine-Hole Peg Test (9HPT).
Capacity to differentiate between MS and HC
There was a significant difference in FDT between PwMS and HC, both in the dominant and non-dominant hand. The difference between the two groups is depicted in Figures 4 and 5 and Table 2. When removing FDT outliers (MS: two dominant hand, one non-dominant hand; HC: two dominant hand, five non-dominant hand), the group difference remained significant for both hands (dominant: p = 0.014; non-dominant: p = 0.004). The depiction without outliers can be found in Supplemental materials (Supplemental Figures S4 and S5).
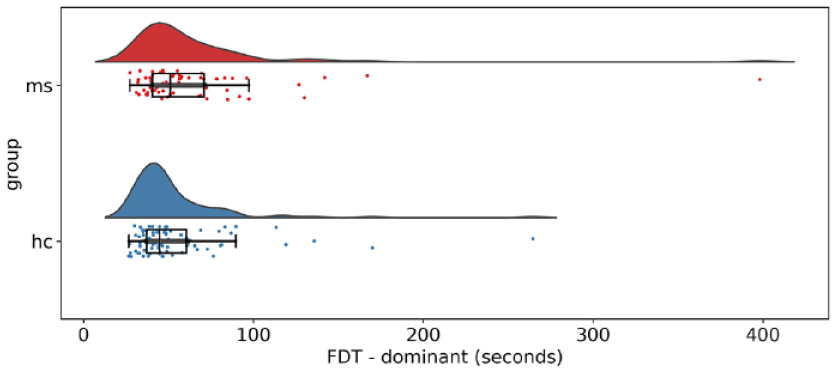
Group difference of Finger Dexterity Test (FDT) between subjects with MS and healthy controls, dominant hand.
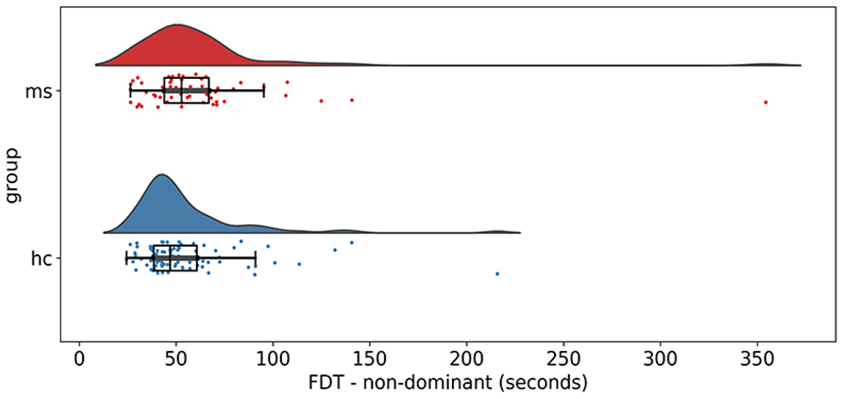
Group difference of Finger Dexterity Test (FDT) between subjects with MS and healthy controls, non-dominant hand.
Group difference of the Finger Dexterity Test (FDT) between PwMS and HC, reported as mean values with standard deviations.
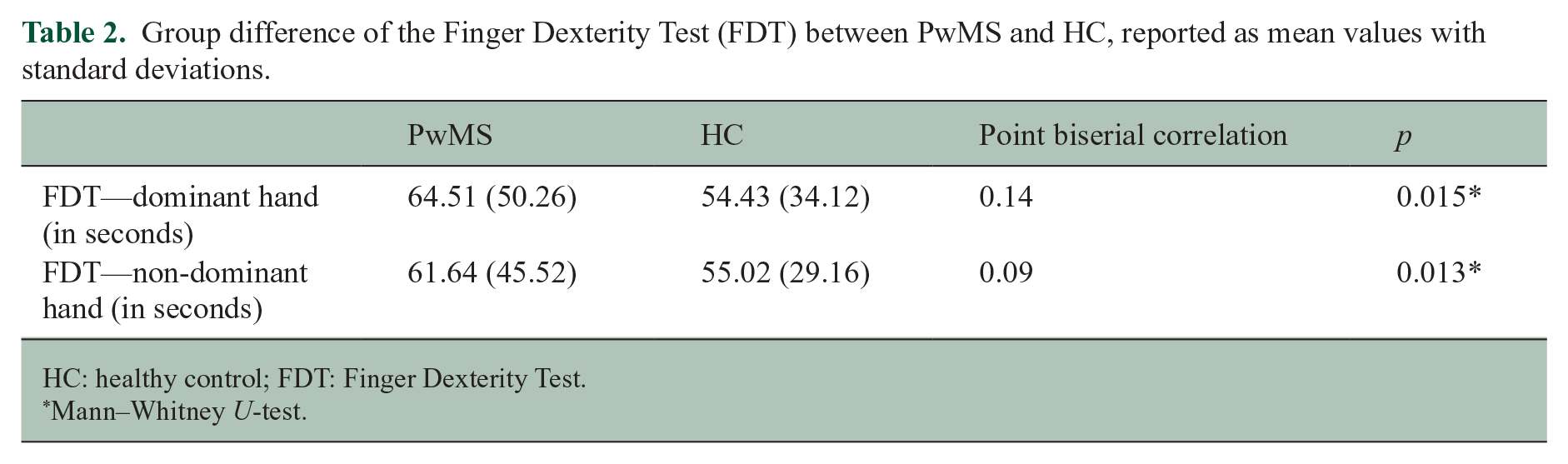
HC: healthy control; FDT: Finger Dexterity Test.
Mann–Whitney U-test.
Correlation with clinical parameters
There was a significant correlation with age in the dominant and non-dominant hand in PwMS (ρ = 0.46, p < 0.001 and ρ = 0.40, p = 0.002, respectively) and HC (ρ = 0.42, p < 0.001 and ρ = 0.41, p < 0.001, respectively). The correlation with EDSS was also significant for both hands (ρ = 0.36, p = 0.005 and ρ = 0.31, p = 0.024). Finally, the correlation with disease duration was only significant for the non-dominant hand (ρ = 0.31, p = 0.016). When removing FDT outliers (two dominant hand, one non-dominant hand), all correlations remained significant (age PwMS: dominant ρ = 0.50, p < 0.001 and non-dominant: ρ = 0.40, p = 0.002; age HC: dominant ρ = 0.39, p < 0.001 and non-dominant: ρ = 0.40, p < 0.001; EDSS: dominant ρ = 0.39, p = 0.002 and non-dominant ρ = 0.28, p = 0.047; disease duration: non-dominant ρ = 0.29, p = 0.032). Other correlations with education and measures of depression (BDI) and fatigue (FSMC) were not significant in PwMS. In HC, there was a significant correlation with FSMC in the non-dominant hand (ρ = −0.29, p = 0.015), which remained significant after removing FDT outliers (ρ = −0.31, p = 0.009), and a correlation with education in the dominant hand (ρ = −0.22, p = 0.047), which was no longer significant after removing outliers. The associations with age, EDSS, and disease duration in the PwMS are depicted in Figures 6–8.
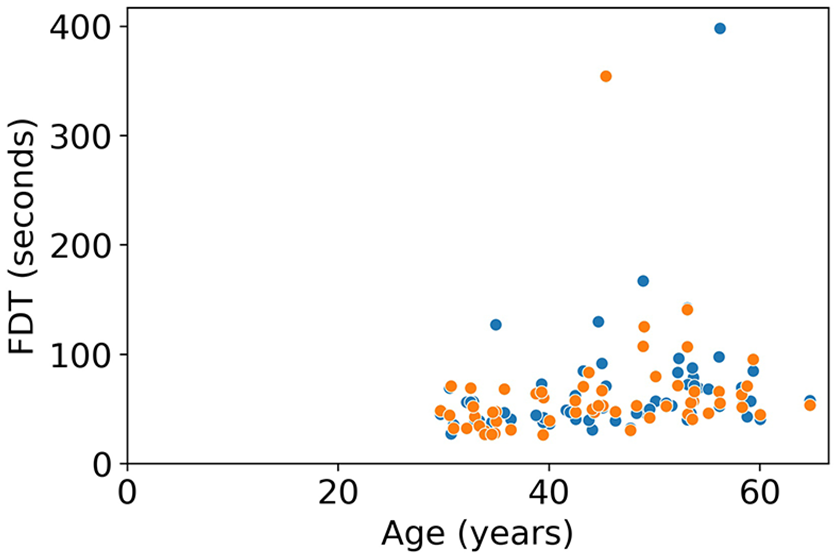
Ecological validity—scatterplot between Finger Dexterity Test (FDT) and age in PwMS.
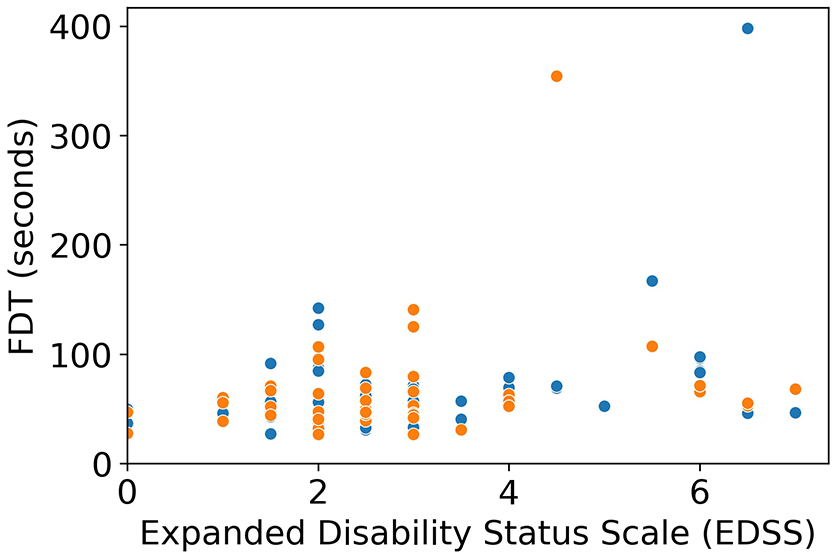
Ecological validity—scatterplot between Finger Dexterity Test (FDT) and EDSS.
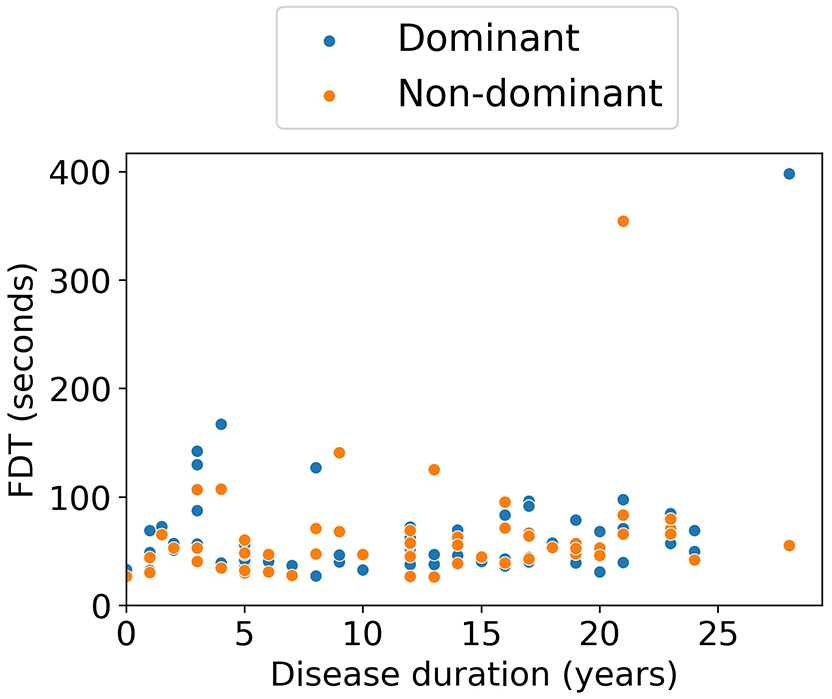
Ecological validity—scatterplot between Finger Dexterity Test (FDT) and disease duration.
Test–retest reliability
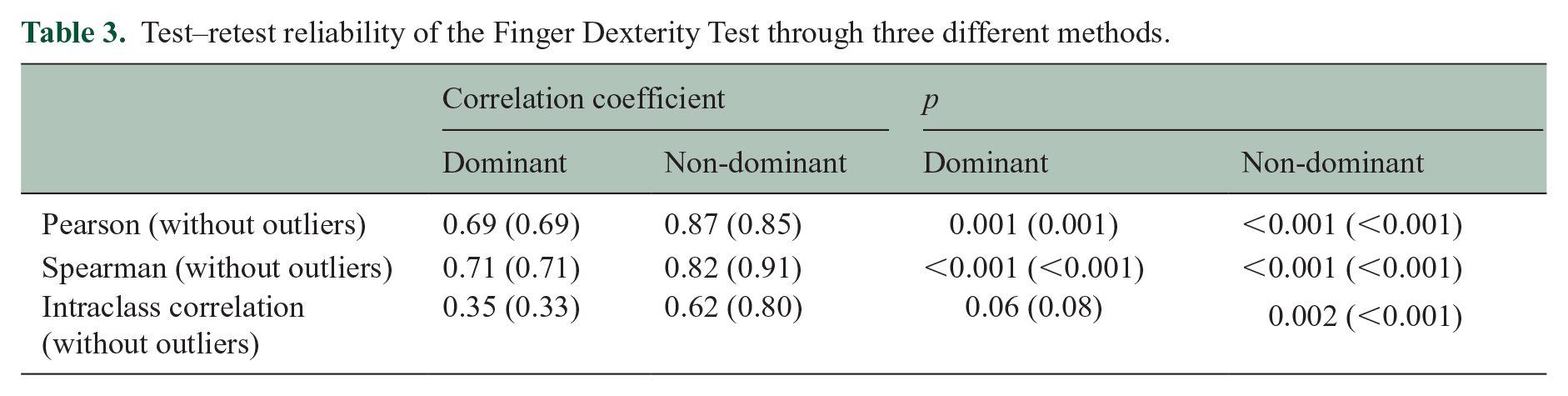
The test–retest reliability as assessed by the Pearson correlation, the Spearman correlation, and intraclass correlation can be found in Table 3. The correlations found after removing FDT outliers (0 for dominant hand, 5 for non-dominant hand) are also listed there. The association between the first and second testing session is depicted in Figure 9.
Test–retest reliability of the Finger Dexterity Test through three different methods.
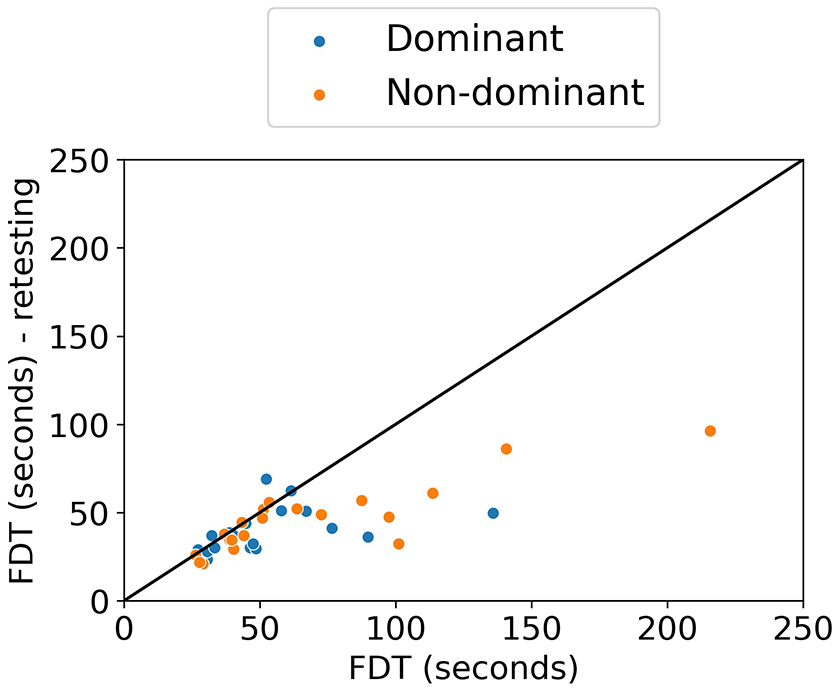
Test–retest reliability of the Finger Dexterity Test (FDT) in healthy controls: scatterplot between Finger Dexterity Test (FDT) scores of the first and second testing session, with identity line (x = y).
Alternative scoring method
Finally, we used an alternative scoring method, namely, the number of eggs/second instead of the total time of serving 10 eggs. All above-mentioned correlations remain similar with this method, but the group difference between PwMS and HC is no longer significant for the non-dominant hand (p = 0.015 for the dominant hand and p = 0.31 for the non-dominant hand).
Discussion
In this study, we established the validity of the smartphone-based icompanion FDT. The test met all validation criteria: there was a moderate-to-good concurrent validity with the 9HPT and an ability to distinguish between PwMS and HC. Furthermore, there was a weak-to-moderate correlation with age, EDSS, and disease duration in MS subjects and a good-to-excellent test–retest reliability in HC when assessed using a Pearson and Spearman correlation. The intraclass correlation coefficient is, however, much weaker, and in the dominant hand, this correlation is not significant. This might be explained by the difference in means between the FDT scores of the first and second testing sessions (see Supplemental Figures S6 and S7), since intraclass correlation coefficient also takes this difference into account. 16
Of note, there was no significant difference between PwMS and HC for the 9HPT, which is the golden standard for the assessment of manual dexterity in MS. 4 This may be explained by the frequent assessments carried out in the tertiary centers where the MS subjects were recruited. Alternatively, the included subjects had a relatively mild degree of disability (median EDSS of 2.75, with only around 11% having a progressive disease course) and may thus not yet show impairment of manual dexterity on the 9HPT. In any case, this warrants caution when using manual dexterity assessments frequently in clinical practice.
Several other smartphone- and tablet-based manual dexterity tests for PwMS already exist (see Table 4). However, the FDT measures maintained grip, as opposed to most other tests, which assess either pinching, drawing, or tapping speed.19–23 The FDT, on the contrary, contains pinching, a drag/transfer movement while maintaining this pinch, and speed of the execution. The aspect of maintained grip is also an important part of the 9HPT, which is the golden standard for clinical and research follow-up of manual dexterity in MS. 4 Since the FDT requires several aspects of dexterity to successfully complete the task, the FDT more closely resembles real-life situations. It is therefore representative of impairments that people with MS will encounter in their daily life, underscoring its value as a quick screening tool to follow-up manual dexterity problems and flag issues early on. Although tests of specific dexterity skills are equally important, we deem them to be useful for a more rigorous mapping of manual dexterity problems. The Manual Dexterity Test of the Multiple Sclerosis Performance Test (MSPT) also assesses the multiple aspects of dexterity, as it is a tablet-based version of the 9HPT. 24 However, it requires a tablet as well as a specific apparatus with holes, pegs, and a tray to place on the tablet, which limits its applicability in home situations. Another advantage of the FDT is that it will be integrated in a holistic patient application, 9 which will assess several other functionalities such as patient-reported outcome measures (PROMs) and cognitive tests. 8 This will increase adherence, as the app holistically assesses disease impact in MS. The implementation of the FDT into daily practice poses many advantages. First, manual dexterity assessment can be carried out independently by the patient at home as a regular screening for (the progression of) manual dexterity problems. This reduces the need for testing during clinical visits, which reduces the duration of the visit and the need for trained personnel. Moreover, patients can take the frequent symptom fluctuations5,6 better into account and perform the test at the patient’s preferred moment. Furthermore, the test is rater-independent and allows for the patient to take a more active role in their own clinical follow-up. Finally, mobile applications have the potential to allow assessment of multiple aspects of the test performance, such as speed or difficulty of different aspects of the test (e.g. pinch vs drag movement) and fatigability (i.e. reduced task performance after sustained effort 25 ). The investigation of these specific parameters is an important next step in the research of digital manual dexterity assessment tools, which may replace paper–pencil tests in the near future.
List of other digital manual dexterity tests in MS.

MS: multiple sclerosis; MSPT: Multiple Sclerosis Performance Test.
There are also limitations inherent to smartphone-based assessments that the patient carries out without supervision. First, differences in the execution of the test (phone in hand vs on table, pinch between thumb and second digit vs second and third digit) cannot be verified, as well as influencing factors such as distraction and long fingernails (capacitive touchscreens do not register touch commands performed with a fingernail because it is non-conductive 26 ). To mitigate this, patients will be able to report any issues after completion of the test. Second, experience with interacting with digital technology such as smartphone apps can have an impact on the test performance, especially in older individuals, but this is difficult to assess. Furthermore, as with all neuroperformance tests, particularly cognitive and manual dexterity tests,23,27,28 it is important to take practice effects into account. We tried to minimize this using a shuffled list of 10 different y coordinates of the egg. In this way, the subject cannot exactly anticipate where the egg will appear on the screen. However, this is likely not enough to eliminate practice effects. Another solution to reduce this risk would be to set a limit to the test frequency. However, more research is needed to determine the most opportune testing interval. Another limitation is that testing was carried out on the same model of smartphone for all subjects. It is unclear how variations in screen size and touchscreen technology could influence test performance. Finally, the application lagged for some subjects (MS: two subjects for the dominant hand, three for the non-dominant hand; HC: seven subjects for the dominant hand, eight for the non-dominant hand), for which we were unable to identify the underlying cause. Excluding the subjects for whom the score was an outlier and for whom the application lagged did, however, not significantly influence the results.
An external validation study is planned in the future, where subjects will carry out the testing at home, with their own mobile device. Furthermore, investigating the association of the FDT to questionnaires assessing upper limb function in daily activities, such as the Arm Function in Multiple Sclerosis Questionnaire (AMSQ 29 ), should be carried out to assess the ecological validity.
Conclusion
In conclusion, the FDT is a valid and reliable tool to assess manual dexterity in MS and could overcome practical issues with the current golden standard of assessing manual dexterity in daily practice.
Supplemental Material
sj-docx-1-msj-10.1177_13524585231216007 – Supplemental material for The Finger Dexterity Test: Validation study of a smartphone-based manual dexterity assessment
Supplemental material, sj-docx-1-msj-10.1177_13524585231216007 for The Finger Dexterity Test: Validation study of a smartphone-based manual dexterity assessment by Delphine Van Laethem, Stijn Denissen, Lars Costers, Annabel Descamps, Johan Baijot, Ann Van Remoortel, Annick Van Merhaegen-Wieleman, Marie B D’hooghe, Miguel D’Haeseleer, Dirk Smeets, Diana M Sima, Jeroen Van Schependom and Guy Nagels in Multiple Sclerosis Journal
Supplemental Material
sj-gif-2-msj-10.1177_13524585231216007 – Supplemental material for The Finger Dexterity Test: Validation study of a smartphone-based manual dexterity assessment
Supplemental material, sj-gif-2-msj-10.1177_13524585231216007 for The Finger Dexterity Test: Validation study of a smartphone-based manual dexterity assessment by Delphine Van Laethem, Stijn Denissen, Lars Costers, Annabel Descamps, Johan Baijot, Ann Van Remoortel, Annick Van Merhaegen-Wieleman, Marie B D’hooghe, Miguel D’Haeseleer, Dirk Smeets, Diana M Sima, Jeroen Van Schependom and Guy Nagels in Multiple Sclerosis Journal
Footnotes
Acknowledgements
The authors thank all subjects who were willing to participate in the study for their invested time and effort. Furthermore, the authors thank Florine Wöhler and Eva Keytsman for their invaluable help in subject testing, as well as Steve De Backer for his invaluable help in the creation of the application.
Data availability statement
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D.V.L. is funded by a Fonds Wetenschappelijk Onderzoek (FWO) PhD fellowship (1SD5322N, ![]() ). S.D. prepares a PhD with ico
). S.D. prepares a PhD with ico
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: D.V.L. is funded by an FWO Flanders PhD fellowship (1SD5322N, ![]() ). S.D. is funded by a personal industrial PhD grant (Baekeland, HBC.2019.2579) appointed by Flanders Innovation and Entrepreneurship. G.N. is a senior clinical research fellow of the FWO Flanders (1805620N). This study is in part funded through the CLAIMS project, which is supported by the Innovative Health Initiative Joint Undertaking (JU) under grant agreement no. 101112153. The JU receives support from the European Union’s Horizon Europe research and innovation program and COCIR, EFPIA, EuropaBio, MedTech Europe, Vaccines Europe, AB Science SA, and icometrix NV.
). S.D. is funded by a personal industrial PhD grant (Baekeland, HBC.2019.2579) appointed by Flanders Innovation and Entrepreneurship. G.N. is a senior clinical research fellow of the FWO Flanders (1805620N). This study is in part funded through the CLAIMS project, which is supported by the Innovative Health Initiative Joint Undertaking (JU) under grant agreement no. 101112153. The JU receives support from the European Union’s Horizon Europe research and innovation program and COCIR, EFPIA, EuropaBio, MedTech Europe, Vaccines Europe, AB Science SA, and icometrix NV.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
