Abstract
Background:
Comorbidity is an area of increasing interest in multiple sclerosis (MS).
Objective:
The objective of this review is to estimate the incidence and prevalence of comorbidity in people with MS and assess the quality of included studies.
Methods:
We searched the PubMed, SCOPUS, EMBASE and Web of Knowledge databases, conference proceedings, and reference lists of retrieved articles. Two reviewers independently screened abstracts. One reviewer abstracted data using a standardized form and the abstraction was verified by a second reviewer. We assessed study quality using a standardized approach. We quantitatively assessed population-based studies using the I2 statistic, and conducted random-effects meta-analyses.
Results:
We included 249 articles. Study designs were variable with respect to source populations, case definitions, methods of ascertainment and approaches to reporting findings. Prevalence was reported more frequently than incidence; estimates for prevalence and incidence varied substantially for all conditions. Heterogeneity was high.
Conclusion:
This review highlights substantial gaps in the epidemiological knowledge of comorbidity in MS worldwide. Little is known about comorbidity in Central or South America, Asia or Africa. Findings in North America and Europe are inconsistent. Future studies should report age-, sex- and ethnicity-specific estimates of incidence and prevalence, and standardize findings to a common population.
Introduction
Comorbidity is an area of increasing interest in multiple sclerosis (MS) as evidence emerges that comorbidity is associated with diagnostic delays, disability progression, health-related quality of life, and progression of lesion burden on magnetic resonance imaging (MRI).1–5 However, the reported prevalence of comorbidity in MS varies widely, depending on the number and type of conditions considered as well as the characteristics of the study population.6–8 Estimates of the incidence of comorbidity in MS are even more limited.9,10 Reliable and valid estimates of incidence and prevalence are important for evaluating changes in the frequency of comorbidity due to changing population demographics, or exposures. When evaluating possible excess risks of comorbidities such as cancer in clinical trials, it is critical to have age- and sex-specific incidence rates with which observed rates can be compared, as sample sizes in clinical trials are too small to draw accurate conclusions. Similarly, accurate estimates of incidence and prevalence support post-marketing pharmacovigilance efforts. Finally, as increasing numbers of studies are designed to assess the impact of comorbidity on MS outcomes, accurate incidence and prevalence estimates are also important for designing such studies.
The purpose of this systematic review was to estimate the incidence and prevalence of comorbidity in MS, determine the most common comorbidities in prevalent MS cohorts, and to evaluate the quality of all included studies. These findings will improve our understanding of the gaps in the literature, the most common comorbidities in MS, and facilitate future studies of the impact of comorbidity in MS.
Methods
Comorbidities of interest
We selected comorbidities for inclusion in this systematic review based on the consensus of five experts in clinical neurology, epidemiology and clinical trials who considered the frequency of the conditions in MS, whether they were known to have effects on the nervous system (e.g. diabetes), and whether they were relevant to pharmacovigilance considerations (clinical trials) and clinical decision making vis-à-vis management of MS. If a systematic review and meta-analysis of a comorbidity of potential interest was published within the last three years, the comorbidity was not reviewed (e.g. migraine). Therefore the comorbidities included in this review were: diabetes, hypertension, hyperlipidemia, ischemic heart disease, valvular disease, cardiac arrhythmias, congestive heart failure, cerebrovascular disease (stroke, transient ischemic attack), peripheral vascular disease, autoimmune disease (alopecia areata, ankylosing spondylitis, autoimmune thyroid disease, bullous pemphigoid, celiac disease, dermatomyositis, idiopathic thrombocytopenic purpura, inflammatory bowel disease, myasthenia gravis, pemphigus vulgaris, pernicious anemia, polymyositis, primary adrenocortical insufficiency, primary biliary cirrhosis, psoriasis, rheumatoid arthritis, Sjögren’s syndrome, systemic lupus erythematosus, systemic sclerosis, uveitis, vitiligo, Wegener’s granulomatosis), chronic lung disease (asthma, chronic obstructive pulmonary disease), gastrointestinal disease (gallbladder and biliary tract disease, gastroesophageal reflux disease, irritable bowel syndrome, liver disease, peptic ulcer disease), renal disease, visual disorders (cataracts, glaucoma, retinal disease), musculoskeletal disorders (crystal arthropathies, fibromyalgia, osteoarthritis), epilepsy, renal disease, cancer and psychiatric comorbidity (alcohol abuse, anxiety, bipolar disorder, depression, personality disorders, psychosis, substance abuse).
Search strategy and study selection
We performed a comprehensive literature search for each comorbidity using strategies developed by investigators with expertise in clinical neurology, clinical epidemiology and systematic review methodology with the support of a research librarian. We also compared our strategies to those used in other systematic reviews of the conditions of interest (e.g. diabetes prevalence in the general population). Specifically, we searched electronic databases of peer-reviewed literature including PubMed, EMBASE, SCOPUS and PsycINFO (for psychiatric comorbidities only) for all years available through November 20, 2013. We hand-searched the reference lists of original studies and reviews identified during the initial search to identify potentially relevant studies. We also searched conference proceedings using the Web of Knowledge. References were imported into Eppi-Reviewer 4 (eppi.ioe.ac.uk/cms/er4/). Search strategies are shown in the supplemental appendices of related manuscripts that describe detailed findings.11–16
After duplicate records were removed, abstracts were read by two independent reviewers (RAM, NR) to assess if they met the inclusion criteria. Eligible studies had to be (i) conducted in an MS population, (ii) include original data, (iii) specify the comorbidity or comorbidities of interest, (iv) report the incidence or prevalence of the comorbidity, and (v) be published in English. Case reports, intervention studies evaluating the incidence of comorbidity after exposure to treatment, and articles evaluating the frequency of MS in another chronic disease population were not reviewed. We specifically did not restrict the search to population-based studies as we anticipated that relatively few studies would meet these criteria, and the initial goal was to obtain a broad understanding of what work has been conducted to assess the epidemiology of comorbidity in MS (similar to a scoping review), including the source of common perceptions and assumptions. Abstracts selected by either reviewer at this stage went on to full text review. Two independent reviewers independently reviewed the full text of selected articles to determine if they met the inclusion criteria. Disagreements were resolved by consensus.
Data extraction and study quality
Data were extracted by one reviewer and verified by a second reviewer using standardized report forms (Appendix 1). Data captured included general study information (e.g. name, region where conducted, year of publication), characteristics of the study population, criteria used to define MS, case definition used for the comorbidity, methodological information (e.g. study design, source of the population, completeness of case ascertainment), and outcome (e.g. incidence, prevalence).
Each study was critically appraised using a standardized assessment tool designed for a prior systematic review of the incidence and prevalence of MS, 17 based on a scoring system suggested by Boyle. 18 We based quality scores on the responses to the following questions where each affirmative answer was accorded one point: 1) Was the target population clearly described? 2) Were cases ascertained either by survey of the entire population or by probability sampling? 3) Was the response rate >70%? 4) Were the non-responders clearly described? 5) Was the sample representative of the population? 6) Were data collection methods standardized? 7 and 8) Were validated diagnostic criteria or approaches used to assess the presence/absence of disease? To accommodate evaluation of diagnostic criteria for MS and for comorbidity, this was split into two questions. 9) Were the estimates of prevalence or incidence given with confidence intervals? As in our prior work, for studies based solely on administrative (health claims) data that were population based, reviewers were asked to mark “yes” for questions 3, 4, 5 and 6. For those studies using multiple sources of ascertainment, reviewers were asked to mark “not applicable” for question 4, and quality was scored out of 8. Finally, because standardization of estimates is important for comparing findings from study populations with differing age and sex structures, we added a question 10) Are standardized estimates reported? We reported this question separately as it was not part of the original tool. For papers that reported more than one comorbidity, quality scores could vary by comorbidity if, for example, confidence intervals were reported for prevalence estimates for one comorbidity but not another.
Statistical methods
Heterogeneity of the studies was assessed both qualitatively and quantitatively. Qualitative assessment included examining similarities and differences across study populations, designs, outcomes, and study quality for all included studies.
We restricted quantitative assessment to population-based studies where the aim of the study was to estimate the incidence or prevalence of comorbidity, as is more typical of systematic reviews. Quantitative assessment used the I2 test; a chi-square test of homogeneity was performed to determine strength of evidence that heterogeneity is genuine. Unlike the Q statistic, which estimates the simple presence or absence of heterogeneity, this statistic describes the proportion of variation in point estimates due to heterogeneity of studies rather than to sampling error. The Q statistic is calculated as the weighted sum of squared differences between individual study effects (in this case based on the number of events (cases of comorbidity) in the study population (sample size)). The I2 test = [(Q – degrees of freedom)/Q]*100. It may be best considered a measure of inconsistency. It has the advantage that it can be compared between meta-analyses with different numbers of studies and different types of outcome data. 19 The authors who developed the statistic suggest that values of I2 of <25% are considered low, of 25%–50% are considered moderate, and >75% are considered high. 19 We considered meta-analysis for population-based studies where the stated purpose of the study was to evaluate the incidence or prevalence of comorbidity in MS. Because the observed degree of heterogeneity was high for all or nearly all conditions, we conducted random-effects, rather than fixed-effects, meta-analyses using a Microsoft Excel spreadsheet developed for this purpose. 20 For studies in which zero events were recorded, we employed a continuity correction of 0.5. 21 This approach is relatively robust under extreme distributional assumptions. 22 Given the inconsistency in reporting of study characteristics, we did not attempt meta-regression to further evaluate sources of heterogeneity.
Results
Herein we present an overview of the findings from the systematic review. For a more detailed presentation of findings related to individual comorbidities, please see the related publications.11–16
Searches
We conducted a series of searches for individual comorbidities (e.g. hypertension) or category of comorbidity (e.g. autoimmune disease). Across all comorbidities the searches identified a total of 7643 citations after duplicates were removed. After full-text review, 249 unique studies were the subject of this review (Supplemental Table 1).3,4,6–10,23–265 Several studies provided data for multiple comorbidities.
Study characteristics
The studies were conducted from 1905 to 2012, although the time period was not reported in several studies. Most studies were conducted in Europe (130, 52.2%) or North America (84, 33.7%) with only 24 (9.6%) conducted in Asia, seven (2.8%) in Australia or New Zealand, and four (1.6%) in South America. Data sources used included clinical databases or medical records review (85, 34.1%), administrative data (52, 20.9%), self-report (84, 33.7%) and interviews (28, 11.2%); some studies used multiple data sources (Supplemental Table 1).
Among studies using clinical databases, medical records or interviews, the diagnostic criteria for MS were reported in 53 studies. Most studies using administrative data specified the diagnostic codes for MS used; however, few studies specified that the administrative case definitions for MS had been validated against a reference standard. Specific diagnostic criteria for comorbidity were rarely reported, and the validity of administrative case definitions for comorbidity were infrequently assessed or described. Study quality was highly variable with quality scores ranging from 0/9 to 8/8 overall, and from 2/9 to 8/8 among population-based studies (Supplemental Table 2). Common limitations included failure to report the study time period, no indication of the diagnostic criteria or validity of the approach used to identify MS or comorbidity, lack of a population-based design, and failure to report age, sex- or race/ethnicity-specific estimates or to age-standardize findings.
Incidence
The most frequently studied comorbidities were epilepsy and cancer. The range of incidence estimates and the number of studies from which these estimates were drawn for each comorbidity are shown in Table 1. For most comorbidities incidence estimates varied widely. For meta-analysis we restricted the selection of studies to those that were population based and for which the goal was estimating the incidence of comorbidity. Even among these studies, heterogeneity of the estimates was substantial as measured by the I2 statistic. The summary estimates based on meta-analysis are shown in Table 1. Based on meta-analyses of population-based studies, the comorbidities with the highest incidence were hypertension, stroke and cancer; however, population-based studies were lacking for many conditions.
The incidence of comorbidity in multiple sclerosis in population-based studies.
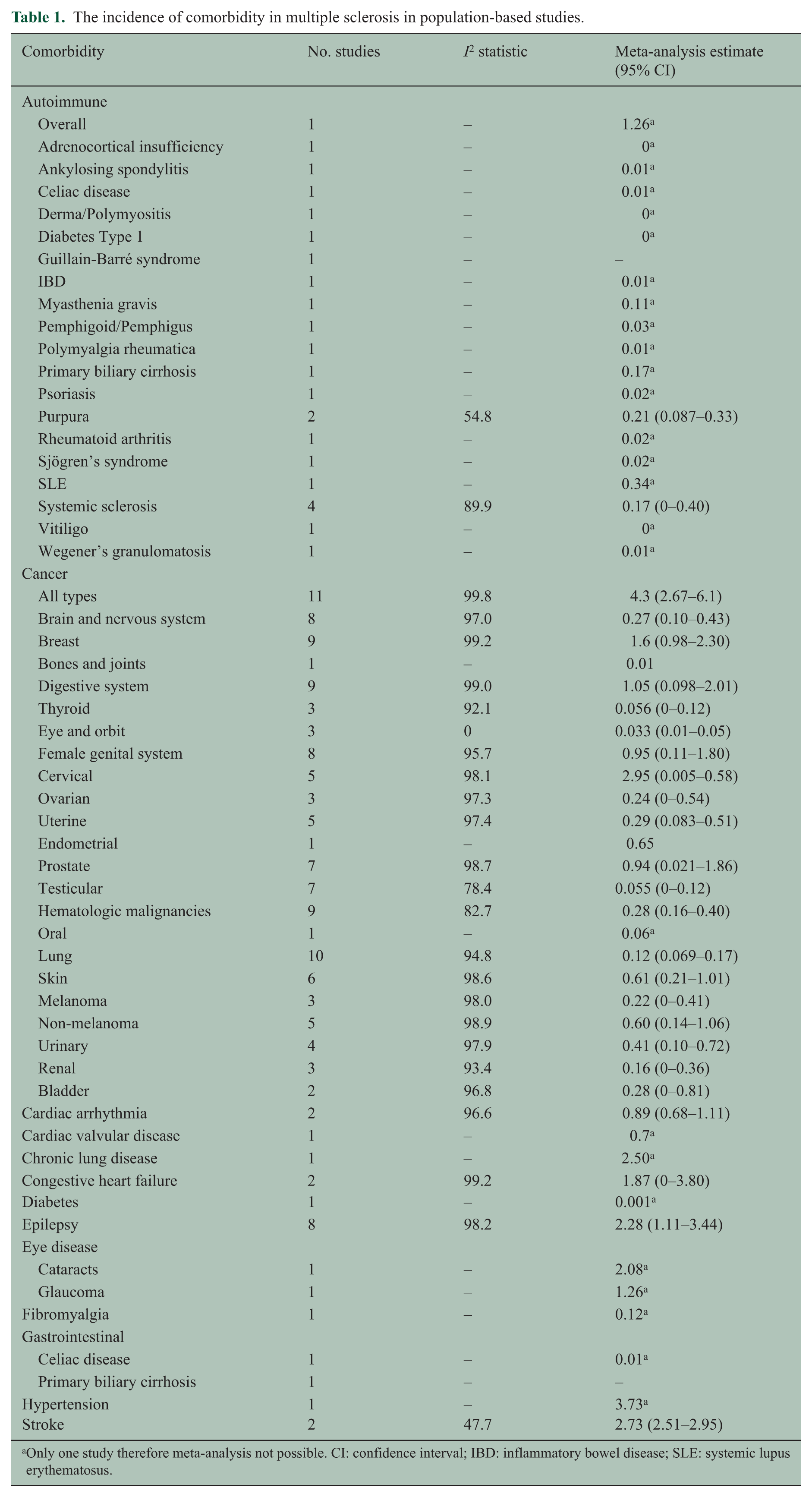
Only one study therefore meta-analysis not possible. CI: confidence interval; IBD: inflammatory bowel disease; SLE: systemic lupus erythematosus.
Prevalence
The most frequently studied comorbidities were psychiatric, autoimmune, cancer, lung disease and epilepsy. The prevalence estimates for each comorbidity evaluated are shown in Table 2. As we observed for estimates of incidence, for most comorbidities the estimates of prevalence varied widely. Even among population-based studies with the goal of estimating prevalence, heterogeneity of the estimates was substantial as measured by the I2 statistic. The summary estimates based on meta-analysis are shown in Table 2. Based on meta-analysis the five most prevalent comorbidities were depression, anxiety, hypertension, hyperlipidemia and chronic lung disease.
The prevalence of comorbidity in multiple sclerosis in population-based studies.
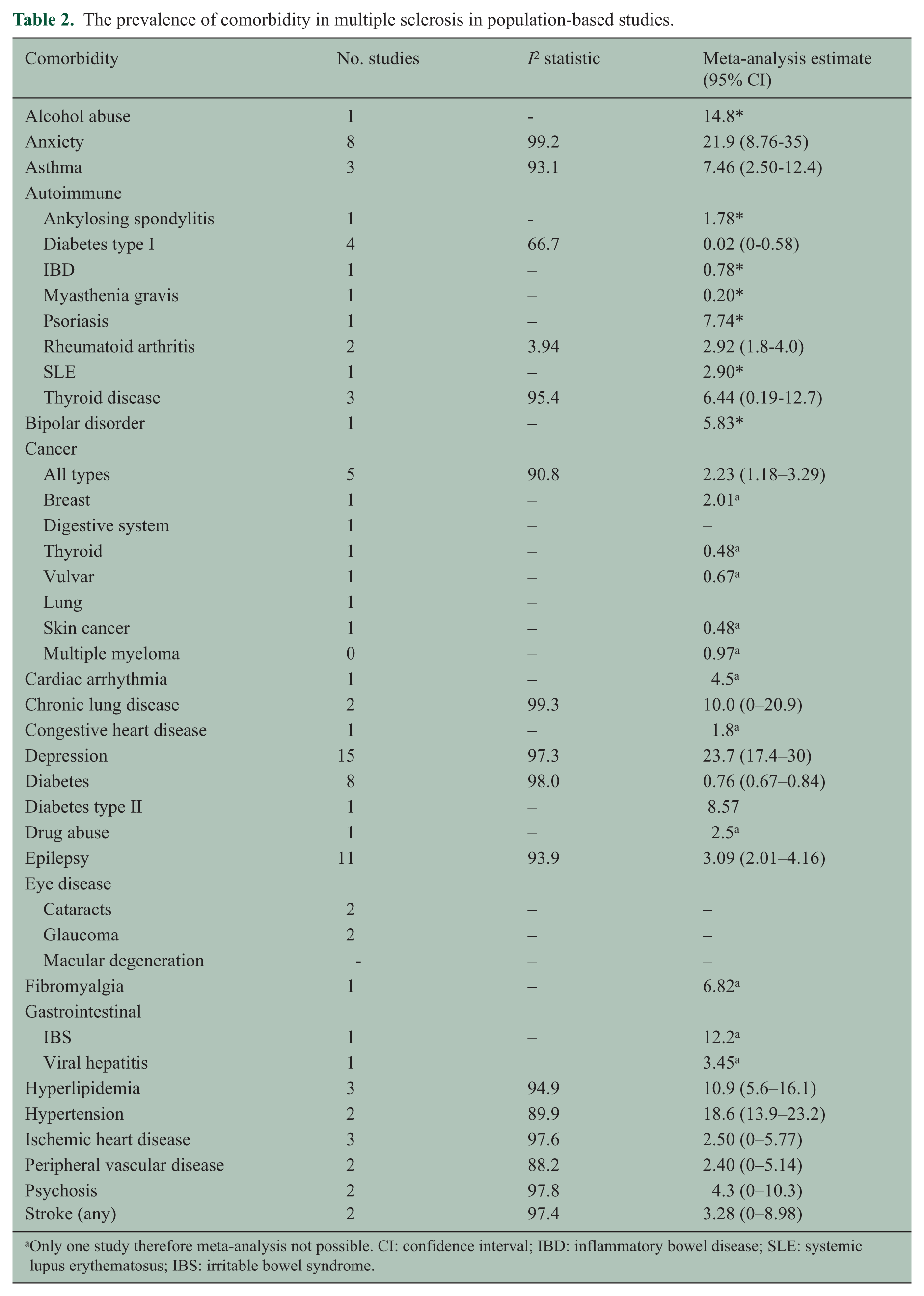
Only one study therefore meta-analysis not possible. CI: confidence interval; IBD: inflammatory bowel disease; SLE: systemic lupus erythematosus; IBS: irritable bowel syndrome.
The most prevalent autoimmune diseases were thyroid disease and psoriasis. Based on population-based studies, the cancers with the highest incidence in the MS population were cervical, breast and digestive system cancers.
Multiple studies compared the incidence or prevalence of comorbidities in the MS population to a comparator population. Findings were inconsistent overall, but suggest that meningiomas and possibly urinary system cancers, inflammatory bowel disease, irritable bowel syndrome, epilepsy, depression, anxiety, bipolar disorder, early cataracts, and restless legs syndrome were more common than expected in the MS population.
Discussion
We systematically reviewed the world literature regarding the incidence and prevalence of comorbidity in MS, evaluating a total of 249 unique manuscripts. Of these, only a small fraction were population-based studies aimed at estimating the prevalence of comorbidity, while even fewer such studies estimated the incidence of comorbidity. Further, many studies of incidence lacked a clearly defined denominator. The most frequently studied comorbidities were autoimmune disorders and cancer. Most of the studies evaluated were from (western) Europe and North America, chiefly Canada and the United States. A handful of studies were conducted in Australia or in Asia, principally in Taiwan. Data regarding the burden of comorbidity in MS populations of Central or South America and Africa were minimal.
Study quality was highly variable, and study designs were heterogeneous, rendering comparisons between studies difficult. To enhance the sensitivity of our search we opted to include all studies reporting comorbidity in MS, regardless of whether estimating the incidence or prevalence of comorbidity was the articulated goal. This likely exacerbated the problem of heterogeneity and identified lower-quality studies. However, we restricted quantitative analysis of the studies to those that were population based, where the articulated aim was to estimate the incidence or prevalence of comorbidity, and still observed substantial heterogeneity for nearly all conditions, despite the fact that most such studies had quality scores of ≥4/8 and very few (29 of 249) were published more than 20 years ago. This means that summary estimates should be considered cautiously and that the 95% confidence intervals are more relevant measures. We also observed several common study limitations. Many studies were not population based. Although population substructures vary from one world region to another, and the burden of comorbidity is well known to vary by age and sex in the general population, 266 age- and sex-specific estimates of the incidence or prevalence of comorbidity were rarely reported. Ethnicity-specific estimates were reported even less often. Further, estimates were rarely standardized to a common population such as the world population, making it difficult to determine if the prevalence of comorbidity really varies across geographic regions. Finally, the validity of the data sources used was not well established in many studies.
The data sources used to identify the MS populations and the comorbidity of interest included clinical databases and medical records, questionnaires, patient interview, and administrative (health claims) data. In clinical databases and medical records review the degree of standardization was often uncertain. Administrative data are of particular interest as a potential resource for ongoing surveillance of comorbidity in MS, including pharmacovigilance activities. In many countries with public, universally funded health care systems, administrative data are population based, accessible and cost effective for research. However, these data are collected for health system management; therefore, their validity for research must be assessed carefully. For example, a single diagnosis for a condition such as MS may merely indicate that a condition is being “ruled out.” Prior work in North America has identified the need for case definitions to include multiple health care contacts for MS before adequate specificity for MS is achieved.267,268 Findings are similar for other chronic diseases such as inflammatory bowel disease. 269 Administrative data may underestimate the presence of comorbidities in persons with multiple chronic conditions owing to biases in coding, or when physicians are limited in the number of diagnoses they can record per encounter.270,271 However, many of the studies evaluated did not validate their case definitions for MS, nor for comorbidity. Further, several studies were limited to hospital claims data, potentially underestimating the burden of conditions that are unlikely to require hospitalization, such as psoriasis or autoimmune thyroid disease. 172 Therefore, much additional methodological work is needed if administrative data are to be fully exploited for evaluating the incidence and prevalence of comorbidity in MS.
We did not include studies in languages other than English; however, we identified few such articles during the study identification process, suggesting that including non-English articles would not have substantially altered our findings. We did not formally evaluate publication bias, which is typically assessed graphically using funnel plots. Based on simulation studies, funnel plots and methods to correct for observed bias do not perform well in heterogeneous meta-analyses as observed here. 272 Although we quantified heterogeneity, we did not quantitatively evaluate factors contributing to that heterogeneity due to inconsistent reporting across studies. Further, we did not explicitly consider the impact of treatment on the risk of comorbidity although this may already play a role with some comorbidities, and likely will play a greater role in the future as more therapies emerge. In many of the studies evaluated some participants were likely exposed to disease-modifying therapies although this was discussed infrequently. This requires explicit evaluation in future studies.
Despite growing interest in the impact of comorbidity on outcomes in MS,5,90 and the emergence of therapies that increase the risk of comorbidity or that may be relatively contraindicated in the presence of comorbidity, 273 relatively little high-quality information is available regarding the incidence or prevalence of comorbidity in MS at any point in the disease course. Future work should develop and validate population-based data sources for surveillance of comorbidity worldwide. When estimates of the incidence and prevalence of comorbidity are reported, they should include age- and sex-specific estimates, and ideally ethnicity-specific estimates as well. Further, estimates should be standardized to the world population to facilitate comparisons across regions. Ideally, such efforts would be coordinated across jurisdictions so that sources of methodologic heterogeneity are minimized and results could be compared or pooled if sufficiently similar.
Footnotes
Acknowledgements
Thanks to Tania Gottschalk, BA, MEd, MSc (Librarian, University of Manitoba), who provided assistance regarding the development of the search strategies for this review. This study was conducted under the auspices of the International Advisory Committee on Clinical Trials of New Drugs in Multiple Sclerosis, whose members include Jeffrey Cohen, MD (Cleveland Clinic Foundation, Cleveland, OH, United States), Laura J. Balcer, MD, MSCE (NYU Langone Medical Center, New York City, NY, United States), Brenda Banwell, MD (The Children’s Hospital of Philadelphia, Philadelphia, PA, United States), Michel Clanet, MD (Federation de Neurologie, Toulouse, France), Giancarlo Comi, MD (University Vita-Salute San Raffaele, Milan, Italy), Gary R. Cutter, PhD (University of Alabama at Birmingham, Birmingham, AL, United States), Andrew D. Goodman, MD (University of Rochester Medical Center, Rochester, NY, United States), Hans-Peter Hartung, MD (Heinrich-Heine-University, Duesseldorf, Germany), Bernhard Hemmer, MD (Technical University of Munich, Munich, Germany), Catherine Lubetzki, MD, PhD (Fédération des maladies du système nerveux et INSERM 71, Paris, France), Fred D. Lublin, MD (Mount Sinai School of Medicine, New York, NY, United States), Ruth Ann Marrie, MD, PhD (Health Sciences Centre, Winnipeg, Canada), Aaron Miller, MD (Mount Sinai School of Medicine, New York, NY, United States), David H. Miller, MD (University College London, London, United Kingdom), Xavier Montalban, MD (Hospital Universitari Vall d’Hebron, Barcelona, Spain), Paul O’Connor, MD (St Michael’s Hospital, Toronto, Canada), Daniel Pelletier, MD (Yale University School of Medicine, New Haven, CT, United States), Stephen C. Reingold, PhD (Scientific & Clinical Review Assoc, LLC, Salisbury, CT, United States), Alex Rovira Cañellas, MD (Hospital Universitari Vall d’Hebron, Barcelona, Spain), Per Soelberg Sørensen, MD, DMSc (Copenhagen University Hospital, Copenhagen, Denmark), Maria Pia Sormani, PhD (University of Genoa, Genoa, Italy), Olaf Stuve, MD, PhD (University of Texas Health Sciences Center, Dallas, TX, United States), Alan J. Thompson, MD (University College London, London, United Kingdom), Maria Trojano, MD (University of Bari, Bari, Italy), Bernard Uitdehaag, MD, PhD (VU University Medical Center, Amsterdam, The Netherlands), Emmaunelle Waubant, MD, PhD (University of California-San Francisco, San Francisco, CA, United States), Jerry S. Wolinsky, MD (University of Texas HSC, Houston, TX, United States)
Conflicts of interest
Ruth Ann Marrie receives research funding from: Canadian Institutes of Health Research, Public Health Agency of Canada, Manitoba Health Research Council, Health Sciences Centre Foundation, Multiple Sclerosis Society of Canada, Multiple Sclerosis Scientific Foundation, Rx & D Health Research Foundation, and has conducted clinical trials funded by Bayer Inc and Sanofi-Aventis.
Olaf Stuve is an associate editor of JAMA Neurology, and he serves on the editorial boards of Multiple Sclerosis, Clinical and Experimental Immunology, and Therapeutic Advances in Neurological Disorders. He has participated in data and safety monitoring committees for Pfizer and Sanofi. Dr Stuve has received grant support from Teva Pharmaceuticals.
Jeffrey Cohen reports personal compensation for consulting from EMD Serono, Genentech, Genzyme, Innate Immunotherapeutics, Novartis, and Vaccinex. He receives research support paid to his institution from Biogen Idec, Consortium of MS Centers, US Department of Defense, Genzyme, US National Institutes of Health, National MS Society, Novartis, Receptos, Synthon, Teva, and Vaccinex.
Per Soelberg Sørensen has received personal compensation for serving on scientific advisory boards, steering committees, independent data monitoring boards in clinical trials, or speaking at scientific meetings from Biogen Idec, Merck Serono, Novartis, Genmab, Teva, GlaxoSmithKline, Genzyme, Bayer Schering, Sanofi-Aventis and MedDay Pharmaceuticals. His research unit has received research support from Biogen Idec, Merck Serono, Teva, Sanofi-Aventis, Novartis, RoFAR, Roche, and Genzyme.
Gary Cutter has served on scientific advisory boards for and/or received funding for travel from Innate Immunity, Klein-Buendel Incorporated, Genzyme, Medimmune, Novartis, Nuron Biotech, Spiniflex Pharmaceuticals, Somahlution, and Teva Pharma-ceuticals; receives royalties from publishing Evaluation of Health Promotion and Disease Prevention (The McGraw Hill Companies, 1984); has received honoraria from GlaxoSmithKline, Novartis, Advanced Health Media Inc, Biogen Idec, EMD Serono Inc, EDJ Associates Inc, the National Heart, Lung, and Blood Institute, National Institute of Neurological Diseases and Stroke, National Marrow Donor Program, Consortium of Multiple Sclerosis Centers, Mt. Sinai School of Medicine, and Teva Pharmaceuticals; and has served on independent data and safety monitoring committees for Apotek, Ascendis, Biogen-Idec, Cleveland Clinic, Glaxo SmithKline Pharmaceuticals, Gilead Pharmaceuticals, Modigenetech/Prolor, Merck/Ono Pharmaceuticals, Merck, Neuren, PCT Bio, Teva Pharmaceuticals, Vivus, NHLBI (Protocol Review Committee), NINDS, NMSS, and NICHD (OPRU oversight committee).
Stephen Reingold reports personal consulting fees from the National Multiple Sclerosis Society (NMSS) and the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS), during the conduct of this work; and over the past three years, personal consulting fees from Bayer HealthCare, Biogen Idec, Coronado Biosciences Inc, the Cleveland Clinic Foundation, Eli Lilly & Company, from EMD Serono and Merck Serono, Genentech, F. Hoffmann-LaRoche, Ironwood Pharmaceuticals Inc, ISIS Pharmaceuticals Inc, Medimmune Inc, Novartis Pharmaceuticals Corporation, Observatoire Français de la Sclérosis en Plaques, Opexa Therapeutics, Sanofi-Aventis, SK Biopharmaceuticals, Synthon Pharmaceuticals Inc, Teva Pharmaceutical Industries, and the Fondation pour l’aide à la Recherche sur la Sclérosis en Plaques, for activities outside of the submitted work.
Maria Trojano has served on scientific advisory boards for Biogen Idec, Novartis and Merck Serono; has received speaker honoraria from Biogen-Idec, Sanofi Aventis, Merck-Serono, Teva and Novartis; and has received research grants from Biogen-Idec, Merck-Serono, and Novartis.
Nadia Reider has nothing to declare.
Funding
This study was supported (in part) by the National Multiple Sclerosis Society and a Don Paty Career Development Award from the MS Society of Canada.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
