Abstract
Background:
Psychiatric comorbidity is associated with lower quality of life, more fatigue, and reduced adherence to disease-modifying therapy in multiple sclerosis (MS).
Objectives:
The objectives of this review are to estimate the incidence and prevalence of selected comorbid psychiatric disorders in MS and evaluate the quality of included studies.
Methods:
We searched the PubMed, PsychInfo, SCOPUS, and Web of Knowledge databases and reference lists of retrieved articles. Abstracts were screened for relevance by two independent reviewers, followed by full-text review. Data were abstracted by one reviewer, and verified by a second reviewer. Study quality was evaluated using a standardized tool. For population-based studies we assessed heterogeneity quantitatively using the I2 statistic, and conducted meta-analyses.
Results:
We included 118 studies in this review. Among population-based studies, the prevalence of anxiety was 21.9% (95% CI: 8.76%–35.0%), while it was 14.8% for alcohol abuse, 5.83% for bipolar disorder, 23.7% (95% CI: 17.4%–30.0%) for depression, 2.5% for substance abuse, and 4.3% (95% CI: 0%–10.3%) for psychosis.
Conclusion:
This review confirms that psychiatric comorbidity, particularly depression and anxiety, is common in MS. However, the incidence of psychiatric comorbidity remains understudied. Future comparisons across studies would be enhanced by developing a consistent approach to measuring psychiatric comorbidity, and reporting of age-, sex-, and ethnicity-specific estimates.
Keywords
Introduction
Psychiatric comorbidity is associated with lower quality of life, greater levels of fatigue, and reduced adherence to disease-modifying therapy in multiple sclerosis (MS).1,2 Therefore a clear understanding of the risk of developing these conditions (their incidence), and their prevalence in established MS populations is needed. Depression, and to a lesser extent, anxiety are recognized to affect the MS population more often than the general population. 3 However, many studies evaluating the prevalence of these conditions have been clinic-based, creating potential selection biases. Prevalence estimates for bipolar disorder and psychosis have been variable,4,5 and the literature is inconsistent with respect to the relative frequency of these conditions in the MS population versus the general population.
In this systematic review we aimed to estimate the incidence and prevalence of selected psychiatric comorbidity in MS, as well as to evaluate the quality of all studies reviewed. We expect that this work will improve our understanding of the gaps in the literature regarding psychiatric comorbidity and assist the design of future studies evaluating the impact of psychiatric comorbidity in MS.
Methods
This study was conducted as part of a larger review of the global incidence and prevalence of comorbidity in MS. 6 Herein we describe the findings for comorbid psychiatric disorders including anxiety, alcohol abuse, bipolar disorder, depression, drug abuse, personality disorders, and psychosis.
Briefly, the search strategy for psychiatric disorders examined the published literature and conference proceedings using PubMed, PsychInfo, SCOPUS, and Web of Knowledge for all years available through November 18, 2013 (Supplemental Appendix I). 6 We manually reviewed the reference lists of studies identified from the electronic searches. After identifying unique citations, two reviewers (RAM, NR) independently assessed abstract relevance. If either reviewer selected the abstract it underwent full-text review to determine if it met study inclusion criteria. Specifically, the study had to (i) include an MS population, (ii) report original data, (iii) specify the comorbidity or comorbidities of interest, (iv) report the incidence or prevalence of the comorbidity, and (v) be published in English. After the two reviewers independently completed the full-text review, disagreements were resolved by consensus.
One reviewer abstracted data using a standardized data collection form and the findings were verified by the second reviewer. The data collection form captured general study characteristics as well as incidence and prevalence estimates (see Marrie et al. 6 ). Each study was critically appraised using a standardized assessment tool developed for a systematic review of the incidence and prevalence of MS. 7 Quality scores were awarded based on yes or no responses to nine questions. 6 This process supported a qualitative assessment of study heterogeneity.
Statistical analysis
Using the I2 test we quantified study heterogeneity among population-based studies with the aim of estimating incidence or prevalence. Using a Microsoft Excel spreadsheet, we conducted random-effects meta-analyses. 8 For studies in which zero events were recorded we employed a continuity correction of 0.5. 9
Results
Psychiatric disorders
Search
The search identified 4047 unique citations (Supplemental Figure 1). After abstract screening and hand searching of reference lists, 317 articles met the criteria for full-text review, of which we excluded 199. A total of 118 studies were the subject of this review (Supplemental Tables 1-15).3,5,10–130
Study characteristics
The studies were conducted from 1953 to 2012, and most were conducted in Europe (58, 49.1%), followed by North America (43, 36.4%), Asia (10, 8.5%), Australia (four, 3.4%) and South America (one, 0.85%). Psychiatric disorders were identified predominantly using validated or unvalidated questionnaires (80, 67.8%), diagnostic interviews (16, 13.5%), administrative data (14, 11.9%), and medical records or clinical databases (four, 3.4%). Some studies used more than one type of data source. Common study limitations included the lack of a population-based design, failure to describe non-responders in survey-based studies, the failure to use validated criteria to assess the diagnosis of MS, and failure to report confidence intervals (CIs). Quality scores varied widely from one of nine to eight of eight overall, and from four of eight to eight of eight among population-based studies ((Supplemental Table 1, “Overview” manuscript). 6 The rest of the text will focus largely on findings from the population-based studies.
Anxiety
None of the studies reported the incidence of anxiety disorders, while 41 reported the prevalence of diagnosed anxiety disorders (13, 31.7%), symptoms of anxiety (26, 63.4%), or both (two) (Supplemental Table 2).3,14,16,18,19,24,26,27,30,32,34,37–39,43,50,56–80 Diagnosed anxiety disorders or previously undiagnosed anxiety disorders meeting diagnostic criteria were captured using structured diagnostic interviews, medical records review or self-reported diagnoses. Symptoms of anxiety were captured using validated instruments including the Hospital Anxiety and Depression Scale (HADS), Hamilton Anxiety Rating Scale, State-Trait Anxiety Inventory, and Beck Anxiety Inventory. Of these the most commonly used instrument was the HADS.
Considering all studies, the prevalence of diagnosed anxiety ranged from 1.24% to 36% (Supplemental Table 2). The prevalence of a self-reported diagnosis of anxiety at the time of MS symptom onset was 2.72%, while it increased to 6.23% at the time of MS diagnosis. 38 One study evaluated the prevalence of health anxiety as a construct distinct from generalized anxiety, reporting a prevalence of 26.4%. 67
Eight population-based studies with quality scores ranging from six of eight to eight of eight reported the prevalence of anxiety (generalized or unspecified) to range from 1.2% to 44.6% (Figure 1).3,16,56,59,65,74,80 Heterogeneity among these studies was substantial (I2 = 99.2), and the summary estimate was 21.9% (95% CI: 8.76%–35.0%). Recognizing that differences in study design, including the method of assessing anxiety, likely contributed to the high degree of heterogeneity, we conducted a sensitivity analysis in which we restricted the analysis to those using questionnaires to assess symptoms of anxiety. Among these five studies heterogeneity remained substantial (I2 = 88.6), and the summary estimate was 25.5% (95% CI: 16.7%–34.3%). We then further excluded the only one that did not use the HADS, but the observed heterogeneity persisted (I2 = 91.3) and the impact on the summary estimate was minimal (27.2%; 95% CI: 16.0%–38.4%). When we considered the three population-based studies that relied on diagnoses captured in administrative data, electronic medical records, or other medical records, heterogeneity remained substantial (I2 = 99.8) and the summary estimate was lower (15.4%; 95% CI: 0%–39.0%).
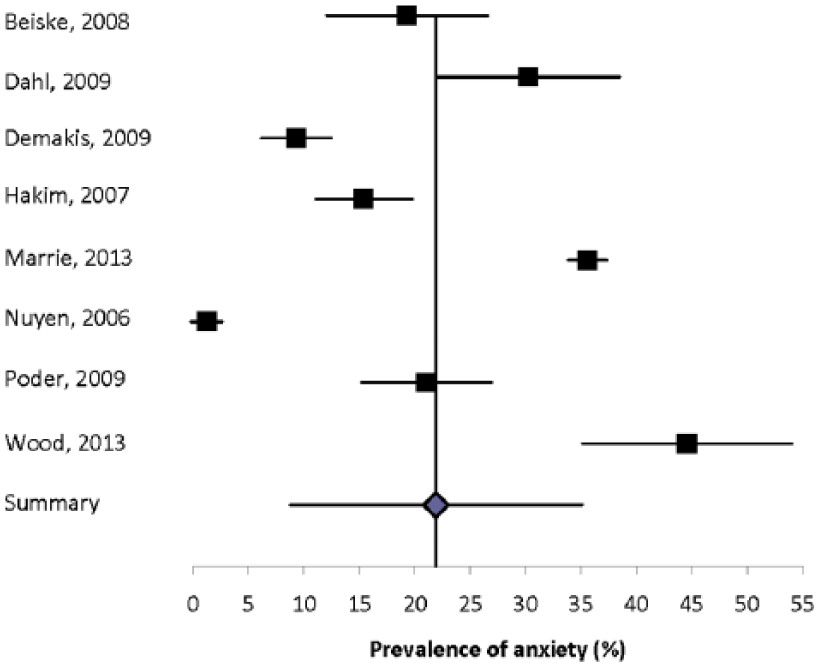
Forest plot of the prevalence of anxiety in multiple sclerosis in population-based studies.
Some studies evaluated subpopulations, including the pediatric MS population and individuals in nursing homes. Two studies reported the prevalence of anxiety in a pediatric MS population.26,79 In the Canadian study the prevalence was 31.0% by patient report, and 22.6% by parental report. In the Italian study the prevalence of panic disorders was 3.57%. 26 In one American study the prevalence of an anxiety disorder in nursing home residents with MS was 9.32%. 16 No other studies evaluated individuals with MS who did not live in the community.
Eight studies compared the prevalence of anxiety disorders or symptoms of anxiety in the MS population and a comparator population (Supplemental Table 2).3,24,38,56,58–60,79 Six of these studies found a higher prevalence of anxiety in the MS population, for both men and women. The sole study comparing specific anxiety disorders found that any anxiety disorder was more statistically significantly common in the general population, but obsessive compulsive disorder, panic disorder, simple phobia, agoraphobia, and social phobia were not, possibly because of the small sample size (n = 50 per group). 24
Alcohol abuse
None of the studies reported the incidence of alcohol abuse, while eight reported the prevalence of alcohol abuse, misuse or dependence (Supplemental Table 3).18,24,74,83–87 However, the definitions used for these states varied, at least in part, because of the differing methods used in these studies, including using screening tools to predict individuals at high risk of these conditions (four, 50%
Only three studies compared the prevalence of alcohol misuse in the MS population to a comparator population; they consistently reported a higher prevalence in the MS population (Supplemental Table 4).24,84,86
Bipolar disorder
None of the studies reported the incidence of bipolar disorder, while 12 studies reported the lifetime prevalence to range from 0% to 16.2% (Supplemental Table 5).3,15–17,21,24,26,30,32,38,81,82 Some studies distinguished between bipolar I and bipolar II disorders, but this was not universal. Only one study was truly population based, and estimated the prevalence of bipolar disorder using administrative data to be 5.83%. 3
One questionnaire-based study reported the prevalence of bipolar disorder at MS onset to be 0.50% and at MS diagnosis to be 0.98%. 39 Of the 12 studies, one Italian study reported the prevalence of bipolar disorder in the pediatric MS population to be 3.57%. 26
One study reported that the incidence of bipolar disorder was two-fold higher in the MS population during the four-year period after diagnosis as compared to the general population but did not report the absolute incidence in either the MS or comparator population. 81 All seven studies comparing the prevalence of bipolar disorder in the MS population to a comparator population found that bipolar disorder was more common in the MS population (Supplemental Table 6).3,15,17,21,24,38,82 Only one of these studies was population-based, reporting a 1.7-fold higher prevalence of bipolar disorder in the MS population than in age-, sex-, and regionally matched controls. 3
Depression
Two studies reported the incidence of depression (Supplemental Table 7),45,52 while 71 reported the prevalence (Supplemental Tables 8 and 9).3,10–38,41–59,63–67,70–80,93–130 The incidence of depression ranged from 4.0% in one year to 34.7% over a five-year period.45,52 The approaches used differed in the two studies. One study used a scale and the other administrative data, thus the findings are difficult to compare. Studies evaluating the prevalence of depression also varied in the methods used to identify depression, with some using formal diagnostic interviews, others using diagnoses abstracted from administrative databases or medical records, and still others using questionnaires (scales) aimed at detecting possible depression (Supplemental Table 9).
Among the studies using diagnostic interviews, some used the Structured Clinical Diagnostic Interview for the Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV) (SCID), while others used the Mini-International Neuropsychiatric Interview, the Composite International Diagnostic Interview (CIDI), or unstructured clinical interviews. Based on diagnoses conferred by interview or obtained from data sources other than questionnaires, the prevalence of depression ranged from 3.80% to 68.4% (Supplemental Table 8). Many of these studies focused on lifetime depression rather than current depression, but the distinction was not always explicit. Studies using diagnostic interviews noted that some individuals with MS met the diagnostic criteria for depression but had not been formally diagnosed. 41
Among studies using validated questionnaires aiming at detecting possible depression (Supplemental Table 9),20,28,41,45,50,56–59,63–67,70–80,93–130 the instruments used also varied widely including the Beck Depression Inventory, Center for Epidemiologic Studies Depression Scale, Depression, Anxiety and Stress Scale (DASS-21), Patient Health Questionnaire-9, the HADS, Montgomery Asberg Depression Rating Scale, Hamilton Rating Scale for Depression, and Zung’s Self Rating Depression Scale, as well as pediatric instruments and translated versions of these instruments. Of these instruments the most commonly used were the Beck Depression Inventory, followed by the HADS and the Center for Epidemiologic Studies Depression Scale. The performance of some of these scales has been evaluated in the MS population. Although performance has generally been adequate, false positives are a potential concern in these studies. Based on the use of questionnaires, the prevalence of depression ranged from 6.94% to 70.1% (Supplemental Table 9).
Fifteen population-based studies with quality scores ranging from four of eight to eight of eight to eight of nine evaluated the prevalence of depression using a mixture of methods, and reported estimates ranging from 4.98% to 58.9% (Figure 2).3,10,16,33,41,43,47,56,59,65,70,74,80,113,120 Heterogeneity was substantial (I2 = 97.3), and the summary estimate was 23.7% (95% CI: 17.4%–30.0%).
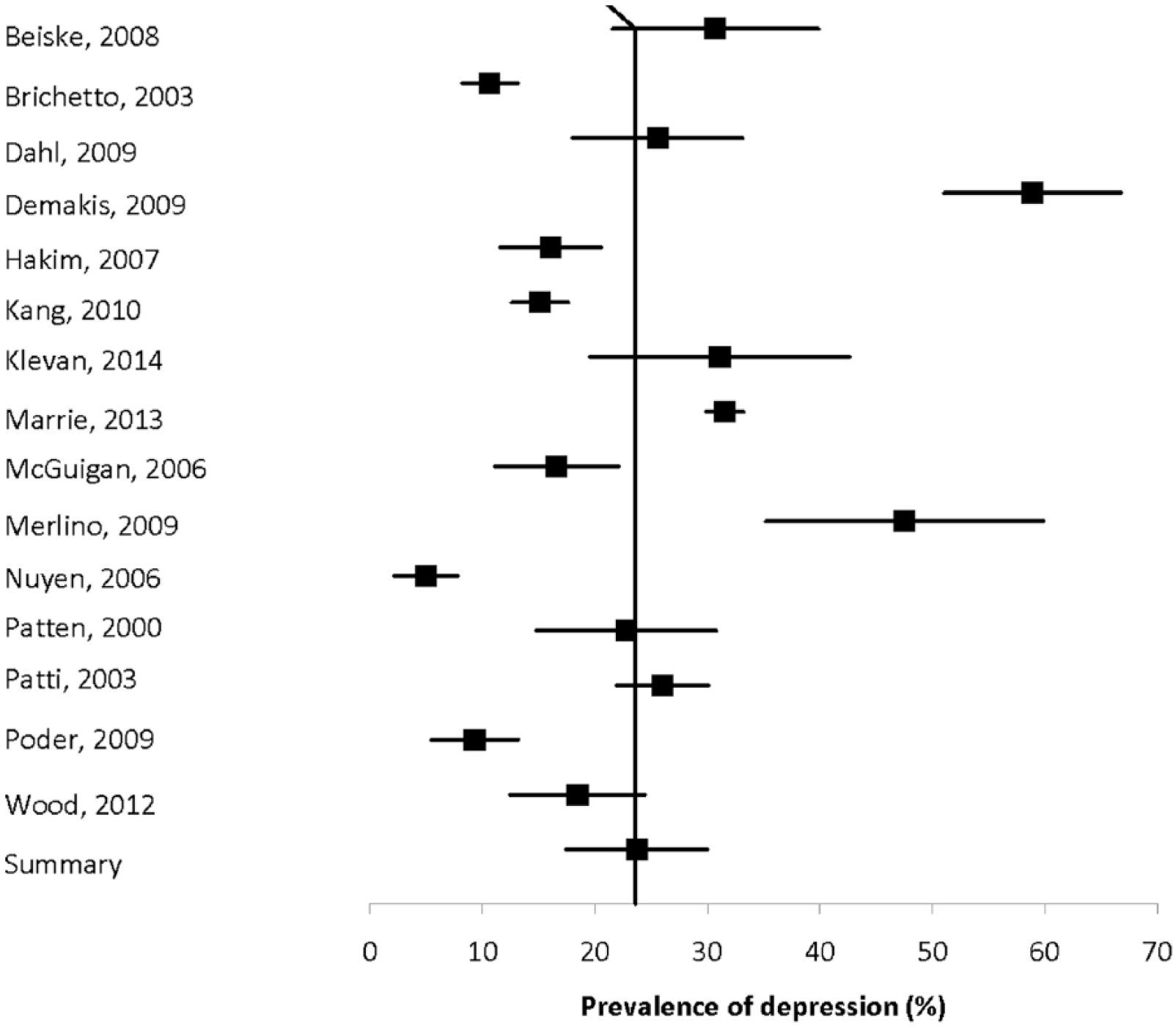
Forest plot of the prevalence of depression in multiple sclerosis in population-based studies.
Ten studies compared the prevalence of depression diagnoses conferred by interview or obtained from data sources other than depression scales in the MS population and comparator populations (Supplemental Table 10).3,15,17,21,24,33,38,46,52,55 All found that depression was more prevalent in the MS population. Seven studies compared the prevalence of depression in the MS population and a comparator population based on the use of validated scales (Supplemental Table 11). All found that depression was more prevalent in the MS population, although one study in the pediatric MS population found this difference based only on parental report.
Drug abuse
None of the studies reported the incidence of drug abuse while three studies reported the prevalence of drug abuse, again using variable definitions (Supplemental Table 12).18,43,83 Two studies reported the prevalence of drug abuse to range from 2.5% to 7.4%.43,83 The lower of the two estimates was from a population-based study in the Netherlands.
Personality disorders
In one study of 651 patients, the prevalence of personality disorders in an Israeli clinic population was 2.6% (Supplemental Table 13). 27 The authors did not classify the personality disorders by cluster.
Psychosis
None of the studies reported the absolute incidence of psychosis. Ten studies reported the prevalence of psychosis,3,5,16,17,27,30,32,33,38,43 but four of these studies were restricted to schizophrenia3,30,32,38 and one was restricted to brief psychotic disorders (Supplemental Table 14). 17 The prevalence of psychosis ranged from 0.41% to 7.46%, while the prevalence of schizophrenia ranged from 0% to 7.4%. The estimated prevalence of psychosis was markedly higher in the population-based study from Taiwan that used administrative data, as compared to other estimates in North America or Europe regardless of the methods used. The heterogeneity of the two population-based estimates for psychosis5,33 was substantial (I2 = 97.8) and the summary prevalence estimate was 4.3% (95% CI: 0%–10.3%) (Figure 3). The sole population-based estimate for schizophrenia was 0.93%.
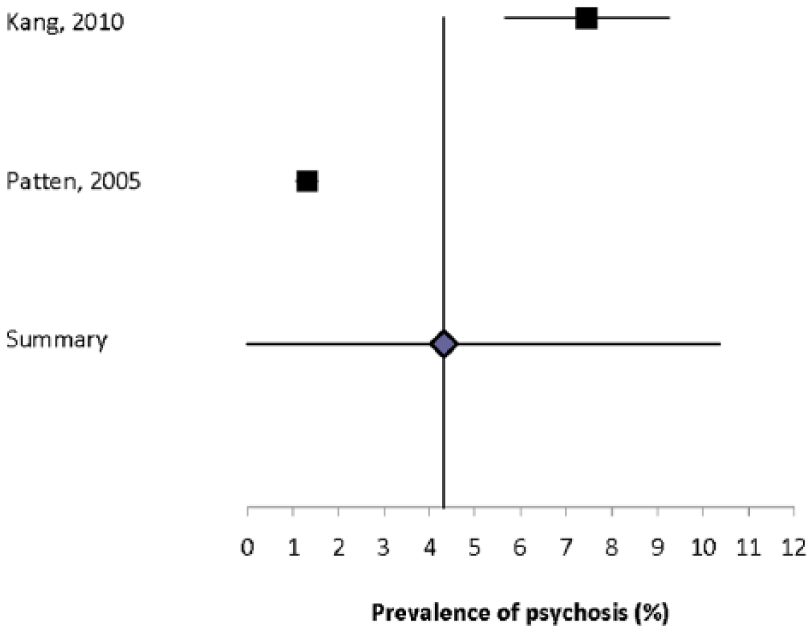
Forest plot of the prevalence of depression in multiple sclerosis in population-based studies.
One study reported the prevalence of schizophrenia to be 0.06% at MS symptom onset, and to be 0.08% at MS diagnosis. 39
One study reported that the incidence of psychosis did not differ in the MS population and the general population but did not report the absolute incidence of disease in either the MS or comparator population (Supplemental Table 15). 81 Five studies compared the prevalence of psychosis or schizophrenia in the MS population to another population.3,5,17,33,38 All three studies that used concurrent controls reported that the prevalence of psychosis was higher in the MS population than in the comparator population.5,17,33 The sole study that evaluated schizophrenia using concurrent controls did not identify a difference in the prevalence of schizophrenia between the populations. 3
Discussion
Psychiatric comorbidity has long been recognized as a concern in the MS population. This comprehensive systematic review confirms that psychiatric comorbidity is common in the MS population, and that this is particularly true for depression and anxiety, each of which affect more than 20% of the population. We identified nearly twice as many studies focused on depression as on anxiety, and far fewer studies devoted to other comorbidities such as bipolar disorder or alcohol abuse, despite their potential clinical importance. The prevalence of psychiatric comorbidity is high even at the time of MS diagnosis, and rises over the course of the disease. Further, depression, anxiety, and bipolar disorder occur substantially more often in the MS population than in the general population even in studies accounting to some degree for socioeconomic status (largely by considering education). Findings for psychosis are inconsistent and require further study.
Global variation in the burden of depression and anxiety is recognized in the general population,131,132 but differences in study design prevented us from comparing findings across regions in the present review. Studies were largely conducted in Central or Western Europe or select regions of North America. Most world regions are understudied and warrant future attention.
Study designs were heterogeneous with respect to data sources, populations, and definitions of psychiatric comorbidity. Even among population-based studies heterogeneity remained high. This was particularly striking for depression, which was variously identified using diagnostic interviews, medical records review, administrative databases, and questionnaires. More than nine different questionnaires were used. To improve comparability prevalence estimates of depression or other psychiatric comorbidities across populations, future studies should use a consistent approach. In particular, comparisons of the psychometric properties of questionnaires used in the MS population would be useful, with a view to selecting one preferred instrument. We also noted that few studies reported age, sex or ethnicity-specific estimates. None of the studies standardized estimates to a common population, again limiting comparability of findings across studies.
The association of psychiatric comorbidity and MS may reflect several factors. First, in the general population, bidirectional relationships exist among depression, anxiety, and immune function. 133 Depression, for example, may occur in response to immunological and inflammatory changes. 134 Second, structural brain abnormalities as measured by brain atrophy and brain lesions are associated with depression in MS. 135 In some patients psychosis has been attributed to brain lesions in the temporal lobe. 136 Third, depression or anxiety may constitute a general response to chronic illness. Fourth, disease-modifying and symptomatic therapies used to manage MS may cause depression or anxiety. For example, corticosteroids may cause transient depression, mania or psychosis;137,138 while the pivotal trials of interferon-beta for MS raised concern about this therapy causing depression. 135 Finally, psychosocial risk factors also play a role in psychiatric comorbidity in MS, and some of these factors may also be associated with MS. 47
We only reviewed studies published in English; however, only 16 publications were excluded because of this, suggesting that this was unlikely to have introduced significant bias. Although diagnostic criteria for MS have changed over time, and the advent of MRI in the 1980s improved diagnosis, we opted to include studies irrespective of time period to gain a more comprehensive view of the world literature. This may have contributed to the high degree of heterogeneity observed; however, this persisted even when we restricted our review to population-based studies and conducted sensitivity analyses.
Psychiatric comorbidity, particularly depression and anxiety, are common in MS. However, the incidence of these and other psychiatric disorders remain under-studied, and study designs were highly heterogeneous. Future comparisons across studies would be enhanced by developing a consistent approach to measuring psychiatric comorbidity, reporting of age-, sex-, and ethnicity-specific estimates, and standardization of estimates to a common (world) population.
Footnotes
Acknowledgements
Thanks to Tania Gottschalk, BA, MEd, MSc (Librarian, University of Manitoba), who provided assistance regarding the development of the search strategies for this review. This study was conducted under the auspices of the International Advisory Committee on Clinical Trials of New Drugs in Multiple Sclerosis whose members include Jeffrey Cohen, MD (Cleveland Clinic Foundation, Cleveland, OH, United States), Laura J. Balcer, MD, MSCE (NYU Langone Medical Center, New York City, NY, United States), Brenda Banwell, MD (The Children’s Hospital of Philadelphia, Philadelphia, PA, United States), Michel Clanet, MD (Federation de Neurologie, Toulouse, France), Giancarlo Comi, MD (University Vita-Salute San Raffaele, Milan, Italy), Gary R. Cutter, PhD (University of Alabama at Birmingham, Birmingham, AL, United States), Andrew D. Goodman, MD (University of Rochester Medical Center, Rochester, NY, United States), Hans-Peter Hartung, MD (Heinrich-Heine-University, Duesseldorf, Germany), Bernhard Hemmer, MD (Technical University of Munich, Munich, Germany), Catherine Lubetzki, MD, PhD (Fédération des maladies du système nerveux et INSERM 71, Paris, France), Fred D. Lublin, MD (Mount Sinai School of Medicine, New York, NY, United States), Ruth Ann Marrie, MD, PhD (Health Sciences Centre, Winnipeg, Canada), Aaron Miller, MD (Mount Sinai School of Medicine, New York, NY, United States), David H. Miller, MD (University College London, London, United Kingdom), Xavier Montalban, MD (Hospital Universitari Vall d’Hebron, Barcelona, Spain), Paul O’Connor, MD (St Michael’s Hospital, Toronto, Canada), Daniel Pelletier, MD (Yale University School of Medicine, New Haven, CT, United States), Stephen C. Reingold, PhD (Scientific & Clinical Review Assoc, LLC, Salisbury, CT, United States), Alex Rovira Cañellas, MD (Hospital Universitari Vall d’Hebron, Barcelona, Spain), Per Soelberg Sørensen, MD, DMSc (Copenhagen University Hospital, Copenhagen, Denmark), Maria Pia Sormani, PhD (University of Genoa, Genoa, Italy), Olaf Stuve, MD, PhD (University of Texas Health Sciences Center, Dallas, TX, United States), Alan J. Thompson, MD (University College London, London, United Kingdom), Maria Trojano, MD (University of Bari, Bari, Italy), Bernard Uitdehaag, MD, PhD (VU University Medical Center, Amsterdam, Netherlands), Emmaunelle Waubant, MD, PhD (University of California-San Francisco, San Francisco, CA, United States), and Jerry S. Wolinsky, MD (University of Texas HSC, Houston, TX, United States).
Conflicts of interest
Ruth Ann Marrie receives research funding from: Canadian Institutes of Health Research, Public Health Agency of Canada, Manitoba Health Research Council, Health Sciences Centre Foundation, Multiple Sclerosis Society of Canada, Multiple Sclerosis Scientific Foundation, Rx & D Health Research Foundation, and has conducted clinical trials funded by Sanofi-Aventis.
Nadia Reider has nothing to declare.
Olaf Stuve is an associate editor of JAMA Neurology, and he serves on the editorial boards of the Multiple Sclerosis Journal, Clinical and Experimental Immunology, and Therapeutic Advances in Neurological Disorders. He has participated in data and safety monitoring committees for Pfizer and Sanofi. Dr Stuve has received grant support from Teva Pharmaceuticals.
Jeffrey Cohen reports personal compensation for consulting from EMD Serono, Genentech, Genzyme, Innate Immunotherapeutics, Novartis, and Vaccinex. He receives research support paid to his institution from Biogen Idec, Consortium of MS Centers, US Department of Defense, Genzyme, US National Institutes of Health, National MS Society, Novartis, Receptos, Synthon, Teva, and Vaccinex.
Per Soelberg Sørensen has received personal compensation for serving on scientific advisory boards, steering committees, independent data monitoring boards in clinical trials, or speaking at scientific meetings from Biogen Idec, Merck Serono, Novartis, Genmab, TEVA, GSK, Genzyme, Bayer Schering, Sanofi-aventis and MedDay Pharmaceuticals. His research unit has received research support from Biogen Idec, Merck Serono, Teva, Sanofi-Aventis, Novartis, RoFAR, Roche, and Genzyme.
Maria Trojano has served on scientific Advisory Boards for Biogen Idec, Novartis and Merck Serono; has received speaker honoraria from Biogen-Idec, Sanofi-Aventis, Merck-Serono, Teva and Novartis; has received research grants from Biogen-Idec, Merck-Serono, and Novartis.
Gary Cutter has served on scientific advisory boards for and/or received funding for travel from Innate immunity, Klein-Buendel Incorporated, Genzyme, Medimmune, Novartis, Nuron Biotech, Spiniflex Pharmaceuticals, Somahlution, Teva Pharmaceuticals; receives royalties from publishing Evaluation of Health Promotion and Disease Prevention (The McGraw Hill Companies, 1984); has received honoraria from GlaxoSmithKline, Novartis, Advanced Health Media Inc, Biogen Idec, EMD Serono Inc, EDJ Associates Inc, the National Heart, Lung, and Blood Institute, National Institute of Neurological Diseases and Stroke, National Marrow Donor Program, Consortium of Multiple Sclerosis Centers; Mt. Sinai School of Medicine, and Teva Pharmaceuticals; and has served on independent data and safety monitoring committees for Apotek, Ascendis, Biogen-Idec, Cleveland Clinic, Glaxo Smith Klein Pharmaceuticals, Gilead Pharmaceuticals, Modigenetech/Prolor, Merck/Ono Pharmaceuticals, Merck, Neuren, PCT Bio, Teva, Vivus, NHLBI (Protocol Review Committee), NINDS, NMSS, and NICHD (OPRU oversight committee).
Stephen Reingold reports personal consulting fees from the National Multiple Sclerosis Society (NMSS) and the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS), during the conduct of this work; and over the past three years, personal consulting fees from Bayer HealthCare, Biogen Idec, Coronado Biosciences Inc, the Cleveland Clinic Foundation, Eli Lilly & Company, from EMD Serono and Merck Serono, Genentech, F. Hoffmann-LaRoche, Ironwood Pharmaceuticals Inc, ISIS Pharmaceuticals Inc, Medimmune Inc, Novartis Pharmaceuticals Corporation, Observatoire Français de la Sclérosis en Plaques, Opexa Therapeutics, Sanofi-Aventis, SK Biopharmaceuticals, Synthon Pharmaceuticals Inc, Teva Pharmaceutical Industries, and Fondation pour l’aide à la Recherche sur la Sclérosis en Plaques, for activities outside of the submitted work.
Funding
This work was supported (in part) by the National Multiple Sclerosis Society (US) and a Don Paty Career Development Award from the MS Society of Canada.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
