Abstract
Background:
Hidradenitis suppurativa (HS) and pyoderma gangrenosum (PG) are chronic neutrophilic dermatoses characterized by dysregulated inflammation. Their coexistence in adults has been reported, but this has not been well documented in pediatric populations.
Objectives:
To characterize the overlap, clinical characteristics, and treatment options for pediatric patients with concurrent HS and PG.
Methods:
We conducted a retrospective case series of pediatric patients ages 3 to 17 with clinical diagnoses of HS and PG at The Hospital for Sick Children between January 2015 and January 2022. Demographic, clinical, laboratory, genetic, and treatment data were extracted from electronic medical records and analyzed descriptively.
Results:
Seven of 122 pediatric HS patients (5.7%) had concurrent PG. HS onset preceded PG in all cases, with mean onset ages of 11.6 and 16.1 years, respectively. The cohort had a slight male predominance (57%) and high rates of comorbidities, including obesity (71.4%) and acne conglobata (42.9%). Elevated erythrocyte sedimentation rate (ESR) (mean 56.9 mm/hour) was a significant predictor of HS severity (P = .027). Three patients had autoinflammatory gene variants, and 57.1% met criteria for PASH or PAPA syndrome. Cytokine profiling revealed elevated IL-1β and IL-6 in select cases. Three of 4 patients who underwent incision and drainage for HS developed PG at the surgical sites.
Conclusions:
Pediatric patients with HS-PG may represent a distinct, severe inflammatory subset. Further investigation is needed to define risk factors, inflammatory biomarkers, and optimal treatment strategies in this rare overlap population.
Keywords
Introduction
Hidradenitis suppurativa (HS) and pyoderma gangrenosum (PG) are chronic inflammatory dermatoses characterized by neutrophilic infiltration and dysregulated inflammation. 1 Although these can present on their own, coexistence has been reported in adults. 1 To date, little is known of comorbid HS and PG in the pediatric population.
HS manifests with recurrent painful nodules, abscesses, and draining tracts, and is associated with complications including scarring, contractures, and malodorous discharge, which results in psychosocial distress in pediatric patients. 2 It is uncommon and likely under-recognized in children, affecting 0.028% of pediatric patients, with a mean age of onset of 12.5 years.2,3 PG is even rarer, affecting only 0.00063% of the population, with pediatric patients accounting for <5% of reported cases.4,5 It presents with extremely painful, slow-healing ulcers that often heal with cribriform scarring.
The pathophysiology of HS and PG is not fully understood. However, emerging evidence suggests dysregulated innate immunity as a common key driver in their pathogenesis. Both are characterized by neutrophilic infiltration and are linked by PASH (PG, acne, HS) syndrome.4 -8 Notably, IL-1B-mediated inflammation is a hallmark of both conditions. 7
Herein, we report on the characteristics and treatment of 7 pediatric patients with clinical diagnoses of concurrent HS and PG.
Materials and Methods
This retrospective case series included pediatric patients aged 3 to 17 years with clinically diagnosed HS and PG treated at The Hospital for Sick Children (SickKids, Toronto, ON, USA), between January 1, 2015 and January 1, 2022. Institutional ethics approval was obtained, and data were extracted from electronic medical records (EPIC and ChartMaxx). Patients were de-identified to ensure confidentiality.
Inclusion criteria required documented assessment by dermatology, and subsequent retrospective photographic verification of lesions by 3 independent physician experts to minimize diagnostic variability. Patients were excluded if they were younger than 3 or older than 17 years or if clinical documentation was insufficient.
Demographic, clinical, and laboratory data were manually abstracted, including age of onset, comorbidities, family history, lesion distribution and morphology, treatment history, and inflammatory markers (ESR, C-reactive protein (CRP), fecal calprotectin). HS severity was retrospectively evaluated using the HS-Investigator Global Assessment (IGA) scale by 3 investigators based on available photographs at initial presentation and follow-up intervals. Statistical analyses were performed using Microsoft Excel, 365 (Microsoft).
Results
We identified 122 patients with a clinical diagnosis of HS, of whom 7 (5.7%) had concurrent PG. Of these, 4 (57%) were male and 3 (43%) were female. Racial distribution included 2 white, 3 multiethnic, 1 Asian, and 1 Hispanic. HS diagnosis preceded PG onset for all patients. The average age of onset for HS was 11.6 years (SD 3.7), and the average age of onset for PG was 16.1 years (SD 0.69). The mean interval between HS and PG onset was 4.6 years (SD 3.7).
The mean HS severity was an HS-IGA of 2.9 with minimal fluctuation over time (SD 0.30). HS-IGA scores increased from 2.21 (SD 0.99) around HS diagnosis to 3.07 (SD 0.86) around PG diagnosis. However, this change was not statistically significant according to the Wilcoxon signed-rank test (W = 10, P = .068).
Autoinflammatory genetic panels were completed for 5 patients, 3 of whom had abnormal findings, all classified as variants of unknown significance (VUS; 1 VUS for NOD-2, 1 VUS in ADA-2, and 1 VUS in mediterranean fever (MEFV)). One patient had a negative autoinflammatory panel. Patient demographics and HS-IGA are summarized in Table S1.
The most common comorbidities were obesity (71.4%), anxiety (57.1%), acne conglobata (42.9%), and depression (42.9%). Additionally, 4 patients met clinical criteria for PASH syndrome, and 1 patient met criteria for PAPASH syndrome (pyogenic arthritis, PG, acne, and HS). Three of these 5 patients had autoinflammatory panel testing, but PSTPIP1 variants were not identified. Detailed frequency of comorbidities is summarized in Table S2.
Overall, inflammatory markers were elevated with an average ESR of 56.9 mm/hour and CRP of 21.7 mg/dL. In a linear regression model, ESR was a statistically significant predictor of IGA (multiple R = 0.81, standard error = 0.58, P = .027, 95% CI: 0.0037-0.0399). However, CRP was not a statistically significant predictor of IGA (multiple R = 0.50, standard error = 0.85, P = .26, 95% CI: −0.0182 to 0.0543). Moreover, cytokine profiling was completed for 2 patients and revealed elevated IL-6 in both cases, with 1 patient additionally showing elevated CXCL-9 and IL-1B. Refer to Table S3 and Figure 1 for detailed inflammatory markers.
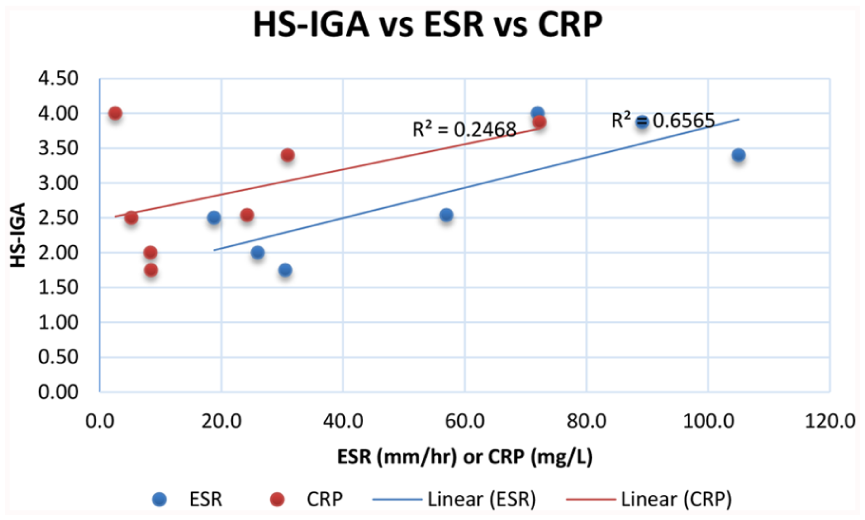
Scatter plot showing correlation between average HS-IGA and average of inflammatory markers. ESR (blue) and CRP (orange). HS-IGA, hidradenitis suppurativa-Investigator Global Assessment.
All patients received multimodal therapy. Prior to PG diagnosis, biologic therapies included adalimumab (n = 6), secukinumab (n = 2), infliximab (n = 1), and ustekinumab (n = 1). Systemic steroids (n = 2), IV ertapenem (n = 2), and incision and drainage (I&D; n = 4) were also documented. None of the patients underwent deroofing. Following PG diagnosis, 3 patients had biologic therapies switched to a different class with the aim of better disease control [secukinumab (n = 2), infliximab (n = 1)] and 1 patient was started on biologic (adalimumab). Additional interventions, listed in Table S4, included I&D (n = 3), IV ertapenem (n = 2), systemic steroids (n = 2), and cyclosporine (n = 1).
Four patients underwent I&D for HS prior to PG onset. Of these, 3 had clear documentation of I&D sites, and 2 of 3 patients (67%) subsequently developed PG at or near procedural sites within 6 months. One additional patient had a surgical excision that was complicated by wound dehiscence and subsequently developed PG at the surgical site 5 months later.
Discussion
We report on a novel pediatric case series of concurrent HS and PG. In our cohort, 5.7% of HS patients had comorbid PG, which is significantly higher than the 0.2% previously reported in adult HS populations, and which could reflect the complexity of patients seen at our quaternary center. 9 HS preceded PG onset in all cases, with an average HS and PG onset of 11.6 and 16.1 years, respectively, aligning with prior reports.2,10 Our cohort showed a slight male predominance at 57%, which contrasts with previous reports of female predominance in adult HS (75%-86%) and PG (75%). These demographic discrepancies may be due to the small sample size, the aforementioned referral bias, or under-recognition of PG in prior HS studies.
Obesity (71.4%) was the most common comorbidity, followed by acne conglobata (42.9%) and Crohn’s disease (28.6%). Among female patients, 66.7% had PCOS. These conditions align with established HS- and PG-associated comorbidities and may contribute to systemic inflammation.2,7,10 -12 This is further supported by the elevation of inflammatory markers, and ESR being a statistically significant predictor of HS-IGA severity (P = .027). Although these findings support the role of systemic inflammation in HS-PG, further studies are required to evaluate whether ESR could serve as a future biomarker for disease activity in this overlap population.
Three of 5 patients who had genetic autoinflammatory panel testing had variants of uncertain significance in ADA2, NOD-2, and MEFV. Pathogenic variants of these genes have been implicated in immune dysregulation and autoinflammatory conditions, including PG, PAPA, and PASH syndromes.8,13,14 This may explain the high prevalence of PASH/PAPASH syndromes in our cohort (57.1%) despite their rarity in the general population. Additionally, NOD-2 and MEFV are involved in inflammasome function, IL-1β B production, and neutrophil recruitment. Two patients in our cohort had elevated IL-1β B, IL-6, and CXCL-9, which have been implicated in HS and PG pathogenesis.14,15 These findings support the known role of neutrophils in HS and PG pathogenesis, and underscore the need for further genetic and immunologic studies to elucidate the shared pathophysiology in pediatric HS-PG.
Treatment strategies varied, with adalimumab being the most common therapy used. Most patients required trials of multiple biologics, reflecting the refractory nature of comorbid HS and PG. Additionally, several patients underwent I&D for HS (none underwent deroofing), but a subset developed PG at or around the surgical site within months, raising a question of pathergy as a contributing factor. This is consistent with previous reports of PG development post-surgical procedures, potentially through exacerbation of the pre-existing inflammatory state in HS patients.16 -19 Given our limited sample size, further research is needed to clarify the potential role of I&D-related trauma and pathergy in triggering PG development.
We have noted features including elevated inflammatory markers and the potential role of pathergy in PG development. However, given the limitations of this study, including the small sample size, retrospective nature, and incomplete genetic and cytokine data, the generalizability is limited. Future studies would help better define risk factors and biomarkers that may predict PG development in pediatric HS. Overall, increased recognition of pediatric overlap of HS-PG is needed to prompt earlier recognition and more appropriate targeted care for affected patients.
Supplemental Material
sj-docx-1-cms-10.1177_12034754251386763 – Supplemental material for Concurrent Hidradenitis Suppurativa and Pyoderma Gangrenosum in a Pediatric Cohort: A Retrospective Case Series
Supplemental material, sj-docx-1-cms-10.1177_12034754251386763 for Concurrent Hidradenitis Suppurativa and Pyoderma Gangrenosum in a Pediatric Cohort: A Retrospective Case Series by Vince Wu, Irene Lara-Corrales, Cathryn Sibbald and Rebecca Levy in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-2-cms-10.1177_12034754251386763 – Supplemental material for Concurrent Hidradenitis Suppurativa and Pyoderma Gangrenosum in a Pediatric Cohort: A Retrospective Case Series
Supplemental material, sj-docx-2-cms-10.1177_12034754251386763 for Concurrent Hidradenitis Suppurativa and Pyoderma Gangrenosum in a Pediatric Cohort: A Retrospective Case Series by Vince Wu, Irene Lara-Corrales, Cathryn Sibbald and Rebecca Levy in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-3-cms-10.1177_12034754251386763 – Supplemental material for Concurrent Hidradenitis Suppurativa and Pyoderma Gangrenosum in a Pediatric Cohort: A Retrospective Case Series
Supplemental material, sj-docx-3-cms-10.1177_12034754251386763 for Concurrent Hidradenitis Suppurativa and Pyoderma Gangrenosum in a Pediatric Cohort: A Retrospective Case Series by Vince Wu, Irene Lara-Corrales, Cathryn Sibbald and Rebecca Levy in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-4-cms-10.1177_12034754251386763 – Supplemental material for Concurrent Hidradenitis Suppurativa and Pyoderma Gangrenosum in a Pediatric Cohort: A Retrospective Case Series
Supplemental material, sj-docx-4-cms-10.1177_12034754251386763 for Concurrent Hidradenitis Suppurativa and Pyoderma Gangrenosum in a Pediatric Cohort: A Retrospective Case Series by Vince Wu, Irene Lara-Corrales, Cathryn Sibbald and Rebecca Levy in Journal of Cutaneous Medicine and Surgery
Footnotes
Acknowledgements
None.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: 2024 Emerging Investigator Research Grant from the Pediatric Dermatology Research Alliance (PeDRA).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
