Abstract
Background
Pyoderma gangrenosum (PG) is a rare neutrophilic dermatosis characterized by painful and ulcerating lesions on the skin. It rarely involves the face and is often difficult to diagnose. There are few cases reported in the literature of PG involving the face.
Aim
To share our experience with 5 patients in whom the final diagnosis was PG involving the face, and to review the literature.
Methods
We report a series of 5 patients with a final diagnosis of PG involving the face and reviewed relevant literature. We searched through PubMed and
EMBASE using keywords such as “face” and “pyoderma gangrenosum,” “blastomycosis-like pyoderma gangrenosum, vegetative pyoderma gangrenosum and granulomatous pyoderma gangrenosum.”
Results
We report 5 patients (4 females) with pyoderma gangrenosum involving the face. All 5 had a final diagnosis of superficial granulomatous PG. All cases presented with nonhealing facial ulcer most commonly on cheeks and a common histopathology of mixed inflammatory infiltrates, multinucleated giant cells, and plasma cells with some granulomatous inflammation.
Conclusions
PG can involve the face, and all 5 of our patients had the superficial granulomatous as the most common form.
Keywords
Introduction
Pyoderma gangrenosum (PG) is a rare neutrophilic dermatosis that presents as an inflammatory ulcerative disorder. 1,2 It most often affects the lower extremities but can involve other areas of the skin. However, involvement of the face has rarely been reported. We present 5 cases of unusual ulcerating and vegetating facial plaques mimicking halogenoderma and blastomycosis-like pyoderma with granulomatous histology. In all patients, the final diagnosis was superficial granulomatous PG.
Many consider PG a diagnosis of exclusion. 1,2 There are four major types of PG: bullous, ulcerative, pustular, and superficial granulomatous. 1 -3 Superficial granulomatous PG presents as a localized vegetative or ulcerative lesion located usually on the trunk. 4 The lesions in the localized vegetative subtype, and in contrast to the other subtypes, are more likely to be confined to the skin commonly with no systemic association. 5 This subtype is expected to be more responsive to a less aggressive antiinflammatory therapy, but this is not always the case. Controversy exists as to whether it is a variant of PG or a separate disorder. 4,5 Clinically, the lesions exhibit a raised, sometimes verrucous, well-defined plaque studded with small pustules. We present 5 cases of the superficial granulomatous or vegetative type presenting as facial ulcers in a distinct distribution.
Case Series
We report the characteristics (demographics, clinical description, histopathology, associated diseases, response to treatment) of 5 patients with a final diagnosis of PG involving the face from 2 academic medical centers in North America (United States and Canada). Consent was obtained from all patients.
Literature Review
We conducted a database search of PubMed and EMBASE for PG involving the face. Our search included keywords “face” AND “pyoderma gangrenosum” OR “blastomycosis-like pyoderma gangrenosum” OR “vegetative pyoderma gangrenosum” OR “granulomatous pyoderma gangrenosum.” We searched from the year 1980 to June 2018 and included published papers in the English language.
The 5 patients who received a diagnosis of pyoderma gangrenosum involving the face are summarized below in Table 1. Photographs of each of the patients’ facial PG and histopathology is presented in Figure 1. A summary of the literature describing patients presenting with pyoderma gangrenosum involving the face is provided in Table 2.
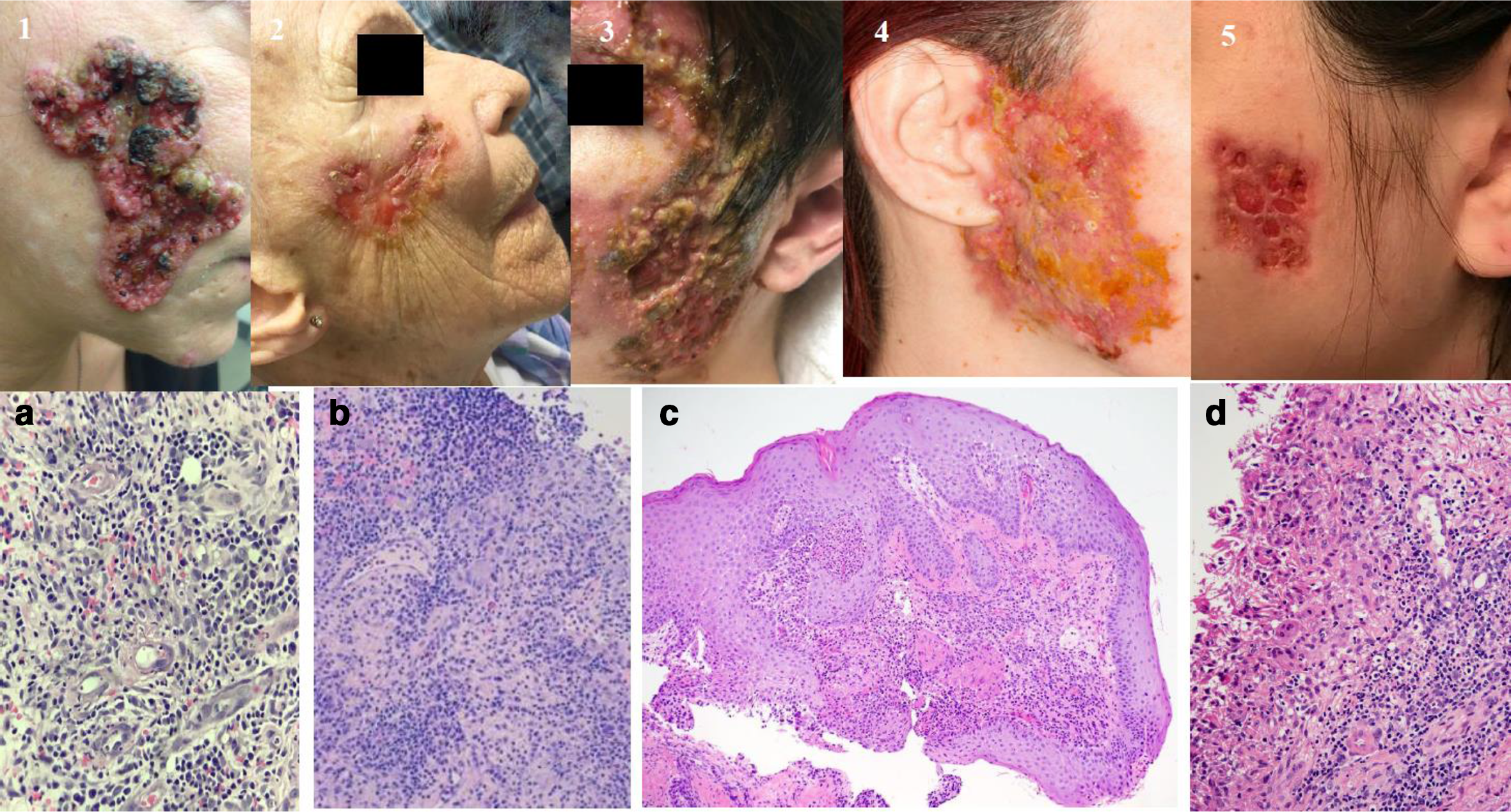
Lesions (1-5) and histopathology (a-d) of granulomatous pyoderma gangrenosum (GPG) of the face. Histopathology demonstrated dense mixed dermal infiltrate including lymphocytes, neutrophils, plasma cells, and histiocytes some of which are multinucleated.
Case Series of 5 Patients With Granulomatous Pyoderma Gangrenosum (GPG) of the Face.

Abbreviations: ACE = angiotensin converting enzyme; ANA = antinuclear antibody; ANCA = antineutrophil cytoplasmic antibody; AZA = Azathioprine; BID = twice daily; CRP = C reactive Protein; CT = computed tomography; DFA = direct fluorescent antibody; DIF = direct immunofluorescence; DVT = deep venous thrombosis ; Eos = eosinophils; ESR = Erythrocyte sedimentation rate; ESRD = End stage renal disease; GMS = Gomori’s methenamine silver stain; HSV = herpes simplex virus; IBD = inflammatory bowel disease; ILS = Intralesional steroids; F = female; Meds = Medications; MGUS = Monoclonal gammopathy of determined significance; MMF = Mycophenolate mofetil; OD = daily; PAS = Periodic acid Schiff; PCR = polymerase chain reaction; PEP = protein electrophoresis; plasc = plasma cells; PMH = past medical history; Pmn’s = polymorphonuclear neutrophils; RF = rheumatoid factor; UA = Urinalysis; US = ultrasonography; VZV = varicella-zoster virus; ZN = Ziehl-Neelsen.
Seven Cases of Granulomatous Neutrophilic Dermatosis of the Face Reported in the Literature.
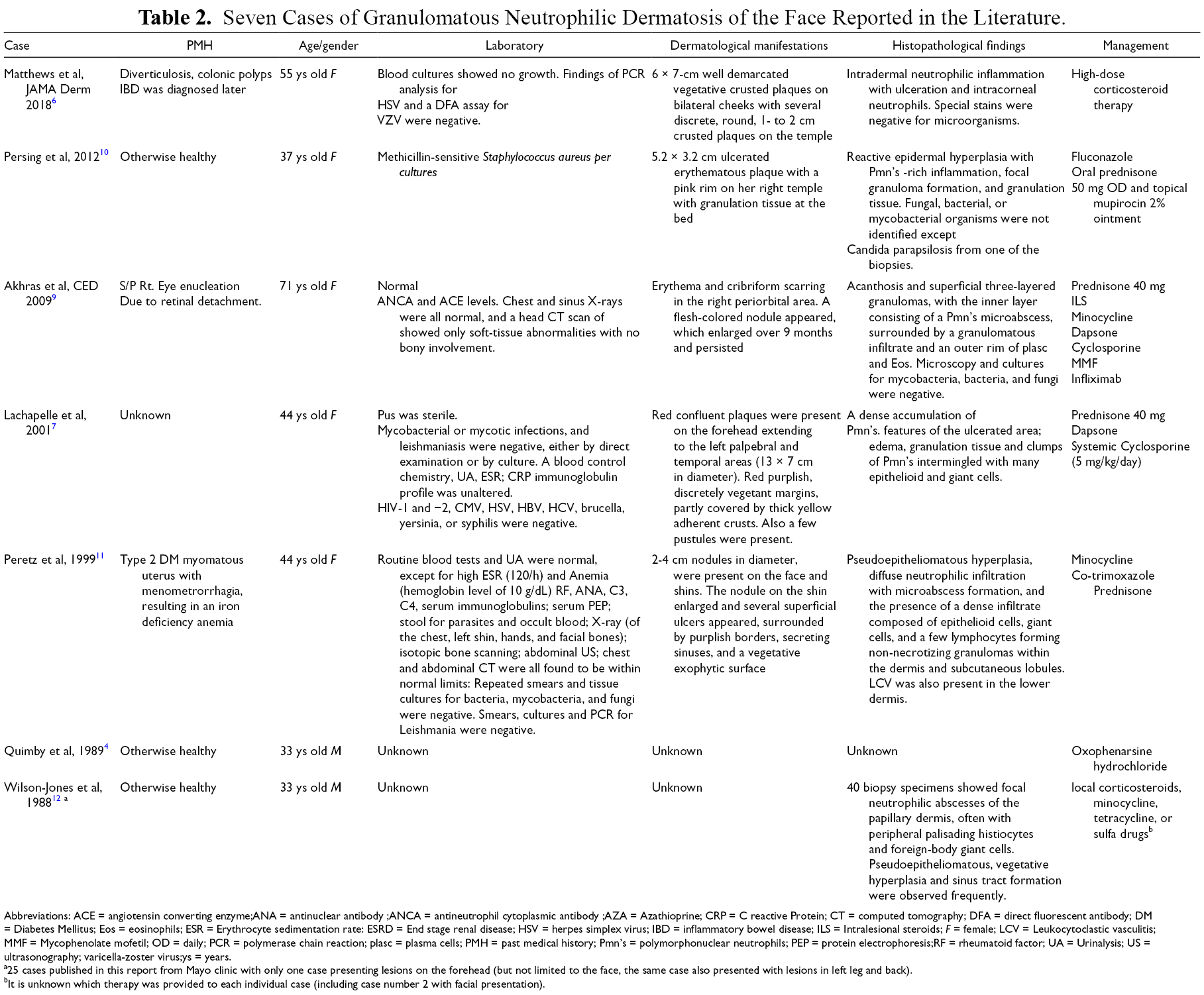
Abbreviations: ACE = angiotensin converting enzyme;ANA = antinuclear antibody ;ANCA = antineutrophil cytoplasmic antibody ;AZA = Azathioprine; CRP = C reactive Protein; CT = computed tomography; DFA = direct fluorescent antibody; DM = Diabetes Mellitus; Eos = eosinophils; ESR = Erythrocyte sedimentation rate: ESRD = End stage renal disease; HSV = herpes simplex virus; IBD = inflammatory bowel disease; ILS = Intralesional steroids; F = female; LCV = Leukocytoclastic vasculitis; MMF = Mycophenolate mofetil; OD = daily; PCR = polymerase chain reaction; plasc = plasma cells; PMH = past medical history; Pmn’s = polymorphonuclear neutrophils; PEP = protein electrophoresis;RF = rheumatoid factor; UA = Urinalysis; US = ultrasonography; varicella-zoster virus;ys = years.
a25 cases published in this report from Mayo clinic with only one case presenting lesions on the forehead (but not limited to the face, the same case also presented with lesions in left leg and back).
bIt is unknown which therapy was provided to each individual case (including case number 2 with facial presentation).
Discussion
We summarize 5 patients who presented with ulcerating and vegetating facial plaques and received a final diagnosis of PG. Intriguingly, the findings were consistent with the superficial granulomatous form of PG in all cases. On clinicopathologic correlation, the presentation mimicked halogenoderma with heavy neutrophilic and granulomatous histology. An associated systemic disorder was found in a minority of patients, as case number 3 was associated with IBD and case 5 with monoclonal gammopathy of undetermined significance (MGUS). These share similarities to those described by Matthews et al. 6 Some histopathological findings in our cases can be seen in the granulomatous variant of PG, including the presence of neutrophils and plasma cells. Multinucleated giant cells are more typically found in patients with a background of IBD. 6
Superficial granulomatous PG is an uncommon variant of PG presenting as painful distinct ulcers and only rarely reported on the face. 7 This clinical presentation on the face is unique, as only few reports were found. 6 -12 The clinical and histopathological findings may differ somewhat from that of the classic PG. Only few dozen cases of superficial granulomatous PG have been published in the literature, of which only six cases with facial involvement; however, only four reports were in the English language. 13 With additional recent case report from this year, 6 a total of 7 reports are summarized in Table 1.
Although PG has a wide range of presentation, the mean age in some reports has been reported to be between 50 and 63 years. 14 However, it seems that the cases of superficial PG may be represented at an earlier age, as seen in our case series and in previous reports (Tables 1–2). Moreover, in our sample study, 40% (2/5) of the cases were pediatric cases (cases 3 and 5). The gender preponderance was in accordance with the reported literature of PG, more commonly represented in the female population than in the male population, both in our cases and in the previously reported cases.
To emphasize the rarity of the facial presentation, in a literature review reported by Langan et al, of 46 cases of granulomatous PG, 52% of lesions were located on the trunk, 31% on the extremities, only 9% presented on the face, 5% in the groin, and 2% on the scalp. 13 In this case series, only 18% of the cases had underlying systemic disease. 13 In accordance with their findings, most of our cases in the series had no underlying disease. It is possible that blastomycosis-like PG is related to the vegetative PG subtype, and that subtype is not associated with any systemic disease. 8 Nevertheless, we were able to demonstrate one case as having an underlying hematological malignancy that was only diagnosed after our initial investigation.
Blastomycosis-like pyoderma has some clinicopathological resemblance to the presentation of superficial PG but mandates the growth of at least one pathogenic organism from the culture of a tissue-biopsy specimen. While our cases all clinically similar to infection but in all cases the culture and special tissue staining were negative. Moreover, empirical therapies with either antibiotics or antifungal medications were futile. In contrast to this, Persing et al (Table 2) reported a case with 2 revealed sources of infection, 10 and this raises a question whether this is indeed a case of PG or truly pyoderma as some resemblances do appear. Our cases are unique, since they may resemble the blastomycosis-like pyoderma, but are not truly infectious. Therefore, they are not pyoderma per se, rather pyoderma-like.
Consistent with other previous reports of superficial PG, this was difficult to treat. As indicated by our cases, 1,2,4,5 and in the previously reported cases, combined regimen may be required. 7,9 We believe that our series represent a poorly characterized, unique, and neglected group of facial dermatoses of the face, namely superficial granulomatous PG.
Our article suffers from some limitations, small sample size, and recall bias. Further prospective evaluations are required to better assess these unusual presentations. However, it is possible that more cases will be revealed as soon as the awareness rises for this uncommon entity. In conclusion, superficial PG of the face is an uncommon clinicopathological diagnosis. The aim of our report was to draw the attention to this rare clinicopathologic variant.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
