Abstract
Background
Ustekinumab is an interleukin 12/23 inhibitor, approved by the US-FDA for the management of moderate-to-severe plaque psoriasis and psoriatic arthropathy.
Purpose
This review aims to describe the dermatological implications and applications of ustekinumab.
Methodology
PubMed and Google Scholar were searched for scholarly articles related to ustekinumab and its utility in dermatology using the search terms “Ustekinumab” AND “Psoriasis” AND “dermatological diseases”. Studies utilising Ustekinumab in psoriasis and other dermatological indications were analysed and formulated into a systematic review discussing the utility of the drug in psoriasis, as well as other dermatological conditions.
Results
Ustekinumab is a valuable biologic agent for the management of psoriasis and psoriatic arthropathy, as well as other dermatological disorders like hidradenitis suppurativa, lichen planus, pityriasis rubra pilaris, Behcet’s disease, lupus erythematosus, alopecia areata and pyoderma gangrenosum.
Conclusion
Ustekinumab’s usage is not limited to psoriasis. Its benefit extends to many more dermatological conditions. It is considered to be the biologic of choice in childhood psoriasis and adult psoriatic patients with concurrent Crohn’s disease. Besides, it has an acceptable safety profile.
Introduction
Ustekinumab is a human monoclonal antibody generated from transgenic mice using techniques of genetic engineering. It targets and blocks the p40 subunit, shared by interleukin (IL)-12 and 23, thereby inhibiting ensuing signaling, differentiation, and cytokine production, pivotal in inflammatory diseases. 1 Currently, ustekinumab is approved by the US Food and Drug Administration for the treatment of moderate-to-severe plaque psoriasis in adults and children (≥6 years of age) and active psoriatic arthropathy (PsA). 2 Apart from these approved indications, ustekinumab has been used “off-label” for a number of inflammatory dermatological disorders. 3 However, most publications stating the off-label utility of ustekinumab are confined mainly to case reports and small case series, with a lack of systematic reviews. In this appraisal, aside from discussing ustekinumab’s role in psoriasis, its application in various other inflammatory dermatoses will be emphasized.
Methodology
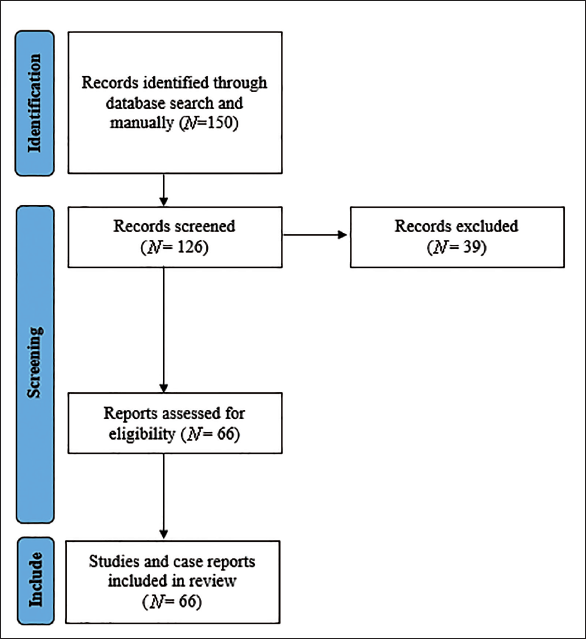
We searched PubMed and Google Scholar with the keywords “Ustekinumab” AND “Psoriasis” AND “Psoriatic arthropathy” AND “Hidradenitis Suppurativa” AND “Lichen planus” AND “Pityriasis rubra pilaris” AND “Behcet’s disease” AND “Lupus erythematosus” AND “Alopecia areata” AND “Atopic dermatitis” AND “Pyoderma gangrenosum.” A total of 150 articles were evaluated from 2006–2023. Of them, 126 articles were included in the review. Sixty-six articles contained studies, case series, and case reports that were employed for final analysis. The search strategy is illustrated in Figure 1.
Schematic of the Search Strategy.
Clinical Uses
Plaque Psoriasis
Psoriasis is a chronic inflammatory disorder with predominant cutaneous features and systemic involvement, affecting 1%–3% of the global population, with plaque psoriasis accounting for 90% of the cases.4, 5 According to the new psoriasis model, the IL-23/Th17 axis contributes significantly to the development of psoriasis. 6 Activation of Th17 by IL-23 and subsequent generation of proinflammatory cytokines like IL-17, tumor necrosis factor-alpha (TNF-α), IL-26, and IL-29 majorly promote the recruitment of inflammatory cells, with eventual epidermal hyperplasia. 7 Ustekinumab blocks IL-23 and subsequently targets upstream cytokines, unlike downstream cytokines (viz., TNF-α and IL-17) targeted by other biological agents, enabling ustekinumab to be dosed at lesser frequencies when compared to other classes of biological drugs. 8 Moreover, blockade of IL-23 also plummets levels of IL-17 and IL-22, furthering ustekinumab’s anti-inflammatory effects. 9
The efficacy and safety of ustekinumab in plaque psoriasis has been illustrated through four large phase III randomized placebo-controlled trials (PHOENIX 1/2, PEARL, and LOTUS) and one phase III randomized active comparator trial (ACCEPT).10–14 In both PHOENIX studies, Psoriasis Area Severity Index (PASI) 75 responses at week 12 were observed in 66.4%–75.7% of ustekinumab-treated patients, which was strikingly greater than the placebo group (3%–4%). Besides, with ongoing treatment, improvements were sustained, and the incidence of adverse effects did not reveal anything alarming.10, 11 The PEARL and LOTUS studies correspondingly delineated statistically significant improvements in PASI 75 in the ustekinumab group when compared to placebo at week 12 (p < 0.001), which were maintained through week 36.12, 13 Furthermore, in the ACCEPT trial, PASI 75 was observed in 67.5%–73.8% of patients treated with ustekinumab and 56.8% of patients in the etanercept group, with comparable safety profiles in both groups. 14 Interestingly, consistent data regarding the efficacy and safety of ustekinumab has been noted through 5 years of follow-up, with dose escalation (90 mg every 8 weeks) resulting in a greater reduction of psoriasis in those patients unresponsive to the lower dosing (45 mg). 15 However, following cessation of ustekinumab therapy, exacerbations of psoriasis occur in most patients. Unfortunately, currently, there is no available data supporting the long-term use of ustekinumab, mandating further research in this field. 16
Though superior to etanercept in plaque psoriasis, PASI 90 values from randomized controlled trials (RCTs) have displayed ustekinumab’s inferiority to secukinumab (57.6% vs. 79%, respectively) and risankizumab (42% vs. 75.3%, respectively).17, 18 Nevertheless, ustekinumab does have value in the management of plaque psoriasis and has been associated with rapid and clinically significant amelioration of moderate-to-severe plaque psoriasis, with apparent results as early as 2 weeks following treatment initiation and maximum responses obtainable by 24 weeks of sustained therapy. Besides, it is the preferred biological in pediatric psoriasis and in adult patients with Crohn’s disease (CD). Furthermore, its widely spaced dosing schedule at weeks 0, 4, and then every 12 weeks, offers an added advantage over the older biological agents that require more frequent administration.10, 11, 15, 19
PsA
PsA is a chronic inflammatory arthritis observed in around 20% of psoriasis patients, associated with diverse clinical features. Although there are numerous therapeutic agents for PsA, discovering newer agents to treat PsA is readily embraced, owing to its refractoriness to various treatments. 20 Ustekinumab’s efficacy in PsA has been substantiated by two phase III randomized placebo-controlled trials (PSUMMIT 1/2) and one randomized controlled active comparator trial (ECLIPSA).21–23 The PSUMMIT trials (at 24 weeks) delineated an American College of Rheumatology-20 score in 42.4%–49.5% of participants receiving ustekinumab, which was almost double when compared to the placebo group (20.2%–22.8%),21, 22 and in the ECLIPSA study (at 24 weeks), the primary endpoint (defined as the Spondyloarthritis Research Consortium of Canada (SPARCC) index of 0) was demonstrated in 73.9% of subjects receiving ustekinumab and 41.7% of patients on TNF-α blockers. 23 Besides, enthesitis also demonstrated a favorable response to ustekinumab, along with a decline in radiological disease progression.21–23 This can be explained by the central role of the proinflammatory cytokine IL-23 in the development of enthesitis, with IL-23 blockade silencing the inflammatory effects of IL-23-sensitive cells in the entheses, and subsequent blockade of downstream inflammatory mediators like IL-17, IL-22, and TNF-α.24, 25 However, synovitis in PsA does not show much response to ustekinumab, as synovitis in PsA is less sensitive to IL-12/23 inhibition. 25
Interestingly, systematic reviews and meta-analyses have propounded the beneficial effects of combining ustekinumab with etanercept, adalimumab, golimumab, and certolizumab in recalcitrant cases of PsA and psoriasis with dose adjustments (ustekinumab being administered as 90 mg/12 weekly in these special scenarios). This integrated approach can be of particular value in the management of patients presenting with both PsA and psoriasis unresponsive to biological monotherapy. As various biologics act differently on cutaneous psoriasis and PsA (owing to the different pathogenesis), this concoction could have a role to play. However, the risk of infection and cardiovascular disease cannot be undermined while initiating this approach. Although there have been reports without encountering any adverse effects, flares of herpes zoster, retro-tonsillar abscess, erysipelas, and bacterial pneumonia have been confronted.26–28
Pustular Psoriasis
Pustular psoriasis comprises a clinically heterogeneous variant of psoriasis with both generalized and localized presentations. Generalized pustular psoriasis (GPP) usually has an acute or even fulminating course, but subacute forms are also commonly encountered. Localized forms of pustular psoriasis are usually confined to the hands and feet and tend to have a chronic discourse.
GPP
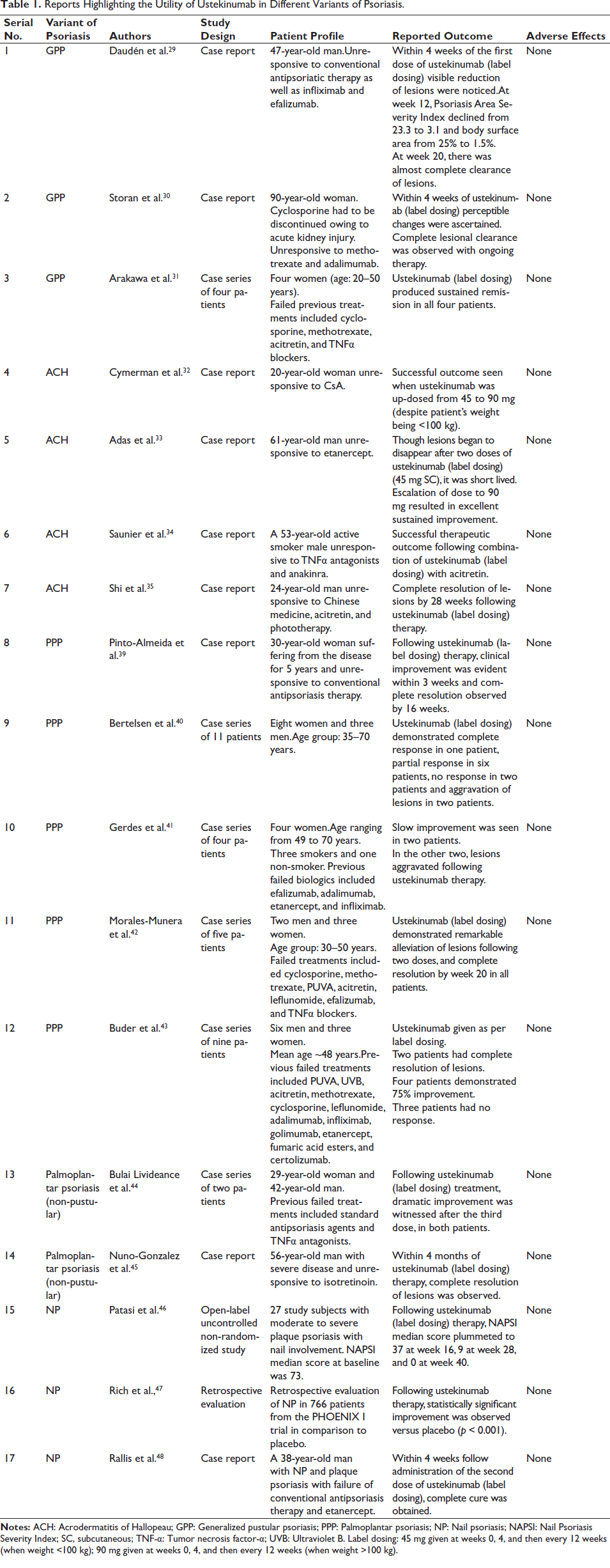
In GPP, there is a high risk of serious complications if appropriate treatment is not promptly instituted. Currently, there are two case reports and one case series demonstrating the successful management of GPP with ustekinumab, details of which have been outlined in Table 1.29–31
Reports Highlighting the Utility of Ustekinumab in Different Variants of Psoriasis.
Acrodermatitis Continua of Hallopeau
In acrodermatitis continua of Hallopeau (ACH), ustekinumab has proven to be highly advantageous, as represented in four case reports (Table 1).32–35 Apart from its conventional dosing, ustekinumab dose escalation (from 45 to 90 mg, even if body weight is <100 kg) in those cases of ACH where therapeutic responses are short-lived can be attempted.32, 33 Additionally, combining acitretin with ustekinumab in recalcitrant ACH is associated with a synergistic clinical outcome. 34 Generally, by 28 weeks of treatment with ustekinumab, acceptable lesional clearance is observed in ACH, with marked improvement in the Dermatology Life Quality Index (DLQI). 35
Palmoplantar Pustulosis
Palmoplantar pustulosis (PPP) is a localized variant of pustular psoriasis characterized by sterile pustules along with erythematous-desquamative lesions involving the palms and soles. The responsiveness of PPP to ustekinumab has been mixed, with profitable outcomes on the one hand and poor to paradoxical responses on the other. The positive effects of ustekinumab in PPP have been linked to the blockade of IL-23, which in turn prevents IL-17 secretion from Th17 cells, with the consequent impediment of neutrophil influx and neutrophil-mediated inflammatory responses. 36 On the contrary, poor or paradoxical responses associated with ustekinumab have been attributed to stimulation of interferon-α (IFNα) synthesis (following blockade of IL-23 and Th17 activity). Activation of T cells by IFNα goes on to produce TNF-α and IL-17, which in turn sustain inflammatory cascades for the development of psoriatic plaques and pustules. Besides, serum in PPP patients demonstrates increased levels of IL-17 and IL-22-related cytokines without significant elevation of IL-23 and IL-8 (thereby accounting for why ustekinumab is not effective in every patient).37, 38 Currently, there are only case reports and case series exemplifying the utility of ustekinumab in PPP (Table 1),39–43 highlighting the need for RCTs to further strengthen these observations.
Palmoplantar Psoriasis (Non-pustular)
This type of psoriasis affects the skin of the palms and soles, featuring hyperkeratotic plaques and causing significant functional disability. Often, the response to this variant of psoriasis to treatment is delayed, accounted for by the presence of subclinical trauma and chronic pressure amounting to systematic koebnerization. 44 By antagonizing IL-23, ustekinumab indirectly blocks IL-22 and prevents the occurrence of epidermal hyperplasia and acanthosis. 45 The utility of ustekinumab in non-pustular palmoplantar psoriasis has been elaborated in two case reports (Table 1).44, 45
Nail Psoriasis
Nail psoriasis (NP) commonly manifests in patients with plaque psoriasis and PsA, causing cosmetic disfigurement that profoundly distresses the patient. Treatment for NP can be quite challenging owing to the intricate nail anatomy compared to the usual skin. Besides, if NP is the sole manifestation of the disease, prior to starting any systemic therapy, both pros and cons need to be adequately weighed. A practical suggestion would be to initiate topical therapy, and if unresponsive, intramatriceal drug injections could be an avenue to contemplate, owing to the localized approach, which has higher levels of safety and cost-effectiveness when compared to systemic therapy. A literature search revealed one open-label, uncontrolled, non-randomized study, one retrospective evaluation of NP from the PHOENIX trial, and one case report depicting the beneficial role of ustekinumab in NP (Table 1).46–48 However, Iqarashi et al. 49 in their report failed to elucidate a significant improvement in the Nail Psoriasis Severity Index (NAPSI) with ustekinumab for NP.
Hidradenitis Suppurativa
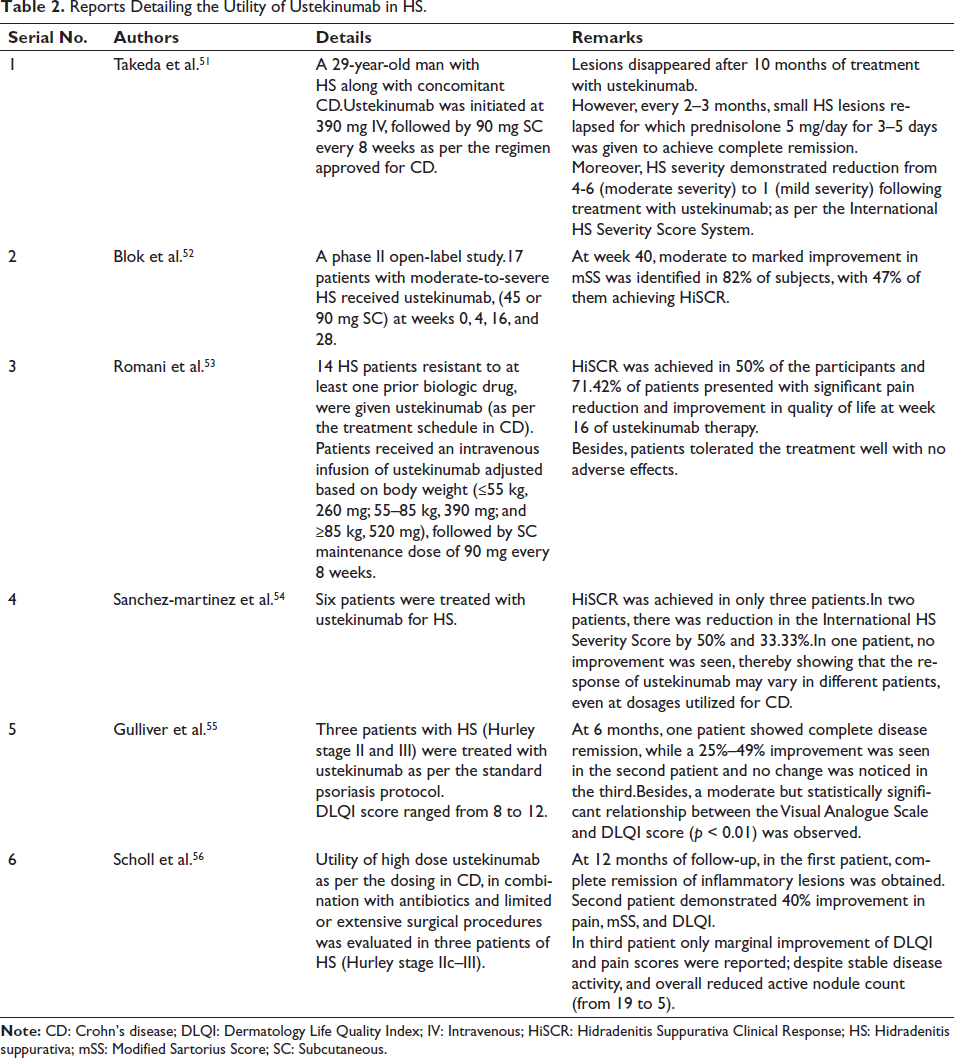
Hidradenitis suppurativa (HS) is a chronic, recurrent inflammatory skin disease arising from hair follicles, particularly affecting intertriginous zones, often associated with systemic comorbidity. 50 The utility of ustekinumab in HS has been described in one phase II open-label trial, one multicenter retrospective study, and a few case series (Table 2).51–56 The role of ustekinumab in HS is associated with inhibition of IL-23 and IL-12. It has been hypothesized that inflammation in HS is caused by abnormal Notch signaling. These signals induce differentiation of outer hair root sheath cells, which are destroyed easily and release keratins that trigger macrophage activation via Toll-like receptors. This consequently activates dendritic cells which release IL-23 to activate Th17 cells. Furthermore, IL-12 and IL-23 are also produced by CD68+ and CD32+ macrophages in lesions of HS. This subsequently deteriorates the balance between Th17 and regulatory T cells. By suppressing both IL-23 and IL-12, ustekinumab enables the restoration of this imbalance.57, 58
Reports Detailing the Utility of Ustekinumab in HS.
Despite the above postulation, Gkine and Bewley 59 displayed the development of HS with ustekinumab in a 19-year-old Caucasian woman treated for psoriasis. The reason for this remains unclear, but it has been suggested to be a probable paradox reaction to anti-IL12/2. Physicians should therefore be aware of this potent complication, with ustekinumab, in the event of which other treatment options for HS need consideration.
Pyoderma Gangrenosum
Pyoderma gangrenosum (PG) is an ulceronecrotic neutrophilic dermatosis characterized by painful ulcers commonly involving the lower extremities. 60 Currently, ustekinumab’s efficacy in PG has only been highlighted in case reports.
Gonzalez et al. demonstrated ustekinumab to be effective in treating PG in a 29-year-old woman with CD who had failed to respond to adalimumab. Ustekinumab was initiated at a dose of 260 mg intravenously (IV), following which the patient improved tremendously. The patient was then maintained on ustekinumab at 90 mg every 8 weeks. With this treatment, the patient remained in remission. 60
A French report further established the profitability of ustekinumab in a 56-year-old woman with plaque psoriasis/PsA, wherein PG had been precipitated following adalimumab therapy. Following three doses of ustekinumab (45 mg subcutaneously (SC), as per the standard protocol for plaque psoriasis), PG had completely healed along with clearance of psoriasis lesions. 61
Nunes and colleagues likewise outlined the utility of ustekinumab in treating PG in a patient with CD resistant to treatment with azathioprine, cyclosporine, and infliximab. Complete healing of PG was witnessed after administration of the first dose of ustekinumab (520 mg IV). The patient was then maintained on ustekinumab 90 mg SC every 8 weeks. 62
Ustekinumab’s role in PG was further highlighted in a report from North Carolina, wherein PG induced by secukinumab in a patient with psoriasis was successfully treated with ustekinumab (90 mg SC on weeks 0, 4, and then every 8 weeks). Significant improvement was noticed after two doses of ustekinumab, with almost complete re-epithelialization of the ulcer. 63
Another publication revealed ustekinumab in combination with vacuum-assisted closure (VAC) therapy to be highly effective in treating PG in a 62-year-old man who had been diagnosed with myelodysplastic syndrome. Ustekinumab (90 mg SC, standard psoriasis regimen) was commenced owing to treatment failure with corticosteroids, thalidomide, infliximab, adalimumab, and immunoglobins. Within 2 weeks of the first dose of ustekinumab, dramatic improvement was observed, following which VAC therapy was applied every 5 days to the ulcer. This treatment was continued for 8 weeks, with complete healing occurring at 20 weeks. Ustekinumab was then continued every 8 weeks as maintenance for 18 months. This revealed surgical treatments to be advantageous along with biological therapy once the correct immunosuppressive environment was achieved in treating PG. 64
The role of ustekinumab in PG can be attributed to the heightened expression of IL-23 in tissue samples from PG lesions at both transcriptional and protein levels. 65
Atopic Dermatitis
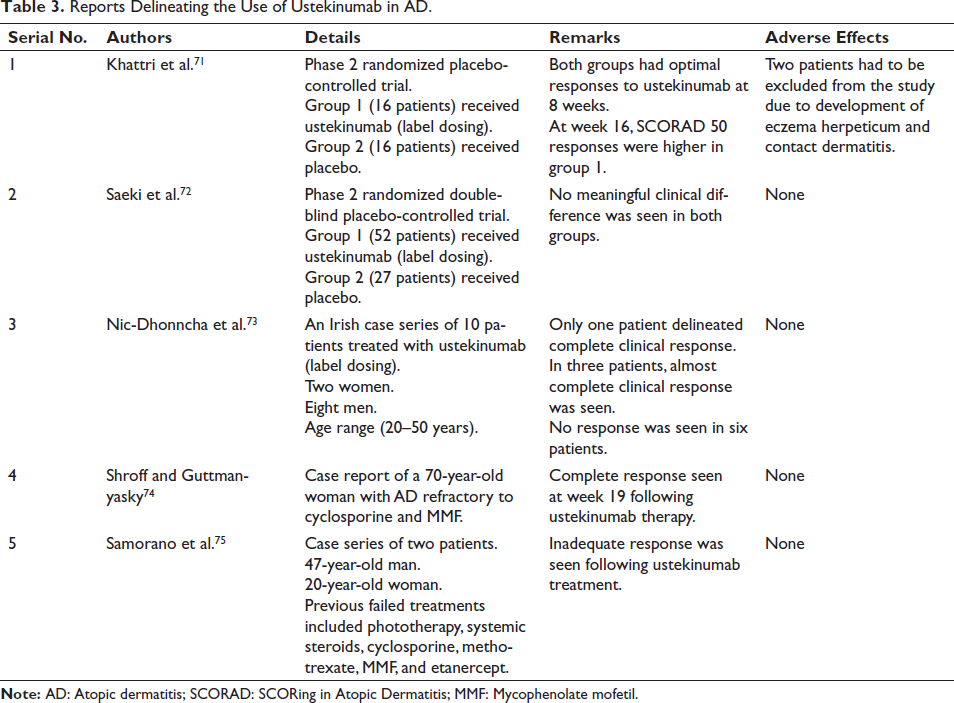
Atopic dermatitis (AD) is a chronic inflammatory disorder of the skin characterized by intense pruritus and xerosis. The pathogenesis of AD is believed to be driven by a type 2 helper T-cell (Th2)-mediated immune response. However, during the chronic phase of the disease, there is a switch towards the Th1-mediated immune response, wherein IL-12 comes into play and brings about this change.66–68 Besides, based on Th1/Th2 cell responses, evidence suggests the involvement of Th17 cells, in the immunopathology of AD. The exact mechanism of action of ustekinumab in AD is still not fully understood. Based on the above postulation, ustekinumab’s role in AD occurs by downregulating Th1 and Th17 inflammatory pathways once it binds to IL-12 and IL-23.69, 70 However, despite these conclusions, the therapeutic effects of ustekinumab for AD have demonstrated disparity, ranging from complete responsiveness to no response at all, as demonstrated in RCTs, case series, and case reports (Table 3).71–75
Reports Delineating the Use of Ustekinumab in AD.
Interestingly, AD, following ustekinumab therapy, has also been observed. Ishuyi and coworkers reported the emergence of AD in a 59-year-old man with psoriasis following ustekinumab treatment, with a SCORAD of 10 being reached after 172 weeks of ustekinumab therapy, and rapid improvement of AD after cessation of ustekinumab. 76 In another patient with concurrent psoriasis and AD, ustekinumab resulted in complete remission of psoriasis but worsening of AD with SCORAD values increasing from 9 to 15; and improvement of AD with subsequent halting of ustekinumab treatment. 77 The underlying mechanism for this can be ascribed to IL-12 blockade as well as indirect IL-17 inhibition, resulting in an immunological imbalance favoring increased activity of the Th2 pathway. The resultant Th2 dominance, in turn, accounts for AD-like symptoms.77, 78 This variation in clinical responses illustrates that ustekinumab may not be a very suitable agent for AD. Nonetheless, it can be tried as an unconventional drug in patients when treatment with approved biological drugs have failed.
Alopecia Areata
Alopecia areata (AA) is an autoimmune disorder resulting in non-scarring alopecia with a considerable impact on a patient’s quality of life. Although there are a number of therapeutic agents for AA, that though effective, cannot be used for prolonged periods owing to safety concerns, and often cessation of treatment is heralded again by hair loss.79, 80 Currently, ustekinumab’s utility in AA has been outlined in case reports and case series alone. The role of ustekinumab in AA is contributed by its antagonistic action on IL-12, which enables the development of naive T cells to Th1 cells. 81
Alesia and colleagues demonstrated hair regrowth in three pediatric patients following ustekinumab treatment. Two patients received a stat dose of 90 mg of ustekinumab, and the third patient was given 90 mg of ustekinumab at months 0, 3, and 6. In all three patients, the SALT score improved by 71.43%–100%. 81 In another study, similarly, ustekinumab (90 mg SC) was found to be effective in AA, given at weeks 0, 4, and 16. Besides, in one patient with alopecia universalis, improvement was substantial, with regrowth of all body hair, eyebrows, and all of the scalp at week 49. 82 Again, ustekinumab’s favorable effect on AA was iterated in a 39-year-old South Asian woman. Dosed at 90 mg (0, 4, and every 8 weeks), by 20 weeks, hair density had visibly increased. 83
Lupus Erythematosus
Lupus erythematosus (LE) is associated with dysregulation of the IL-17/IL-23 axis along with a reduction in the total number of T-regulatory FoxP3+ cells. Increased levels of IL-23 produced predominantly by dendritic cells and macrophages promote the expansion of Th17 cells, which in turn elaborates IL-17 production, a cytokine significantly elevated in the skin of patients with systemic LE and discoid LE. IL-17 further stimulates the production of autoantibodies and inflammatory cytokines (TNF-α, IL-1β, IL-6, IL-8, IL-22, etc.) and chemokines (CCL2, CCL7, CCL29, CXCL1, and CXCL5). There is thus an imbalance between proinflammatory and anti-inflammatory cytokines with a deranged homeostasis. Ustekinumab, in LE, ameliorates this deranged homeostasis by preventing differentiation of T cells to Th17 via IL-12/23 inhibition.84–87
Though there are a number of case reports where ustekinumab has been beneficial in LE, RCTs are currently lacking in this context. Further details have been propounded in Table 4.88–93 However, despite the propitious effect of ustekinumab in LE, it has also been reported to induce subacute cutaneous LE. 94 The cause of this reaction occurs secondary to the diversion of T cell differentiation to the alternative pathway of T helper cell IL-22 production via IL-6, leading to excess TNF-α production and stimulation of inflammatory responses. 95
Reports Highlighting the Efficacy of Ustekinumab in LE.

Bechet’s Disease
The role of ustekinumab in Bechet’s disease (BD) occurs via its antagonist activity against IL-23 and the associated Th17 pathway that plays a major role in BD. 96 Ustekinumab has demonstrated efficacy in two patients with multirefractory BD (unresponsive to colchicine, cyclosporine, methotrexate, azathioprine, anakinra, and anti-TNF-α agents).97, 98 In order to strengthen these findings, RCTs are warranted.
Pityriasis Rubra Pilaris
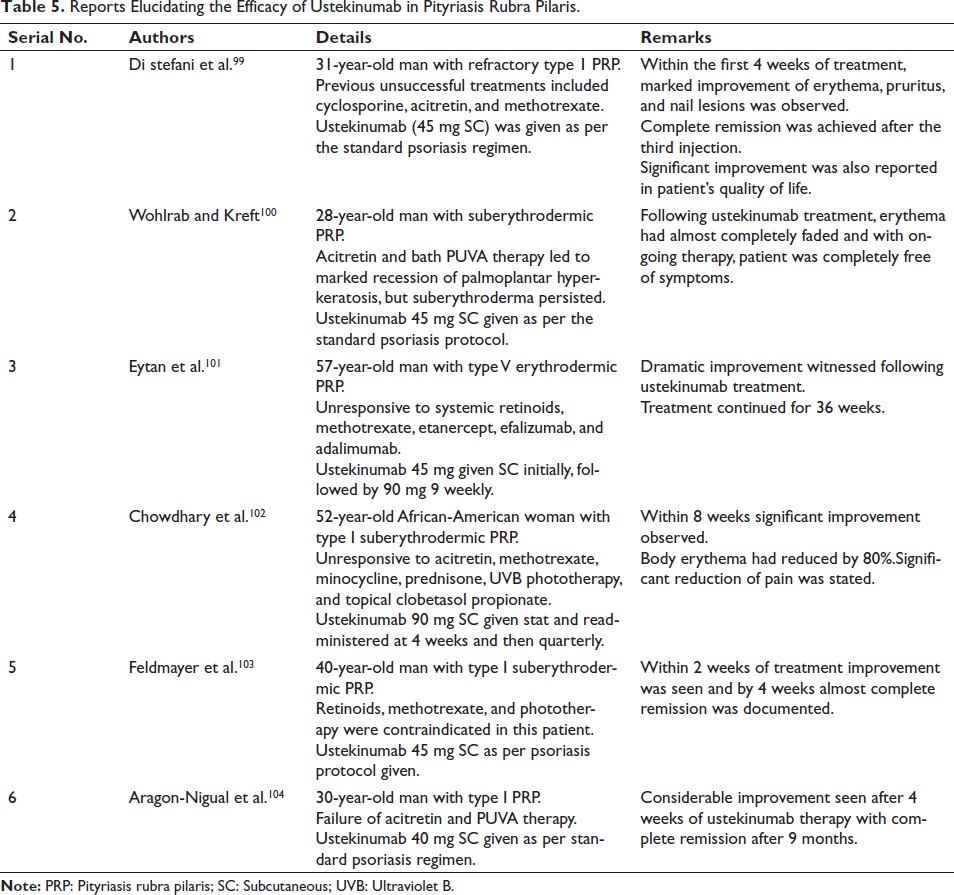
The exact mechanism of ustekinumab’s action in pityriasis rubra pilaris (PRP) is unclear. Because of its IL-12 and IL-23 blocking properties and subsequent suppression of cytokine release and immune activation, ustekinumab has been used in patients with unremitting PRP. However, information on the effects of ustekinumab on PRP does not meet the requirements of evidence-based medicine. 99 Currently, there are only case reports describing the appositeness of ustekinumab in PRP, which are summarized in Table 5.99–104
Reports Elucidating the Efficacy of Ustekinumab in Pityriasis Rubra Pilaris.
Miscellaneous
In an anecdotal report, ustekinumab (45 mg SC/in the standard psoriasis dosage) was associated with dramatic clinical improvement of recalcitrant oral lichen planus (LP) within 12 weeks in a 72-year-old woman unresponsive to systemic or topical steroids, ciclosporin, acitretin, and azathioprine. 105 It has been suggested that IL-23 is markedly elevated in LP lesions, which is suppressed by the antagonizing effects of ustekinumab on IL-23 and Th17/Tc17 cells. 106
Similarly, isolated reports have highlighted the utility of ustekinumab in synovitis acne pustulosis hyperostosis osteitis syndrome, bullous pemphigoid (BP), LP pemphigoid, and anti-laminin γ1 pemphigoid.107–110 Interestingly, the occurrence of BP has been associated with ustekinumab therapy, suggesting other cytokine pathways responsible for the development of iatrogenic pemphigoid.111–113
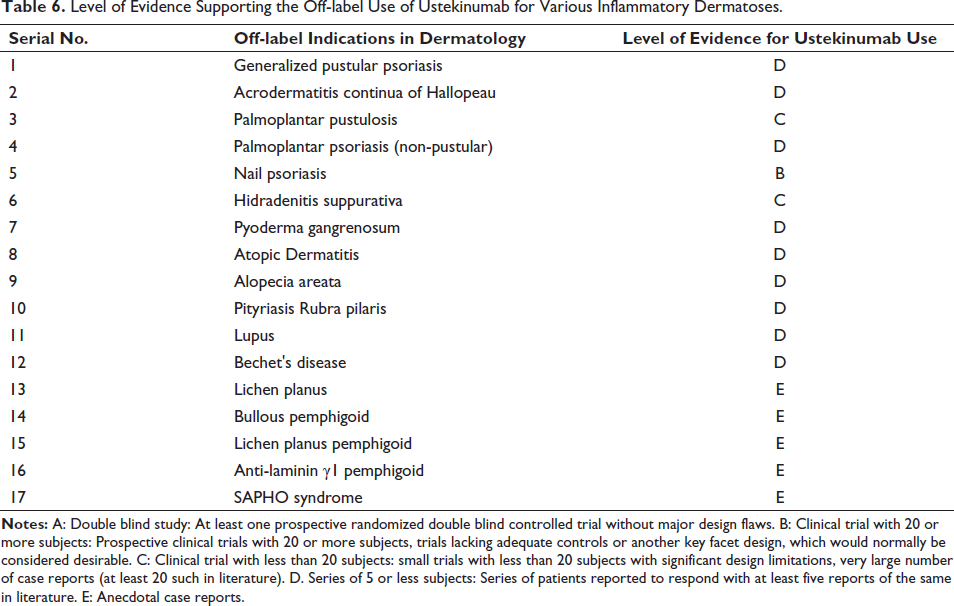
Table 6 delineates the level of evidence supporting the off-label use of ustekinumab for various inflammatory dermatoses.
Level of Evidence Supporting the Off-label Use of Ustekinumab for Various Inflammatory Dermatoses.
Adverse Effects
Reported cutaneous adverse effects associated with ustekinumab are pruritus, exfoliative dermatitis, LE tumidus, Jessner–Kanof’s lymphocytic infiltrate, and injection site erythema.114, 115 Common extracutaneous side effects observed are nasopharyngitis, headache, arthralgia, fatigue, bronchitis/upper respiratory tract infection, sinusitis, back pain, abdominal pain, and fever (in higher dosing schedules). 116
Serious side effects that need consideration include the risk of active tubercular infection, major adverse cardiovascular events (MACEs), and potential oncogenic complications. However, analysis of published register-based data has not demonstrated higher rates of severe tubercular infections when compared to either anti-TNF-α or conventional antipsoriatic drugs.117, 118 Similarly, though a meta-analysis of RCTs reported an increase in MACEs during the first month of ustekinumab therapy, no significant increase in frequency of MACEs was observed when compared to placebo. 119 Regarding the oncogenic potential, despite scarce reports of malignant tumors, cancer incidence itself was low in clinical trials and register-based data. 118 Even with the reassuring data, before starting ustekinumab, appropriate monitoring of the drug is essential, details of which have been elucidated in Table 7.120–123
Drug Monitoring for Ustekinumab.

Use in Specific Populations
Pregnancy
Although there are several cases of pregnant patients treated safely with ustekinumab, mostly in gastroenterological indications, the German S3 guideline and also the summary of product characteristic (SmPC) do not currently recommend using this medication for psoriasis during gestation. 124
Lactation
Only a few reports exist on the use of ustekinumab during lactation. The substance was found in breast milk in four out of six breastfeeding patients with chronic inflammatory bowel disease (IBD). The highest concentration (0.72–1.57 mg/mL) was measured during 12–72 hours after drug administration.
The breastfed infants did not demonstrate any negative effects from it. Despite this, owing to limited evidence, ustekinumab is not currently recommended for lactating mothers. 124
Pediatric
Ustekinumab can be used in pediatric patients with psoriasis. Besides, it is a safe and effective treatment option for pediatric and adolescent CD and Crohn’s-like IBD. 125
Future Direction
As of now, in highly recalcitrant dermatoses, the need for combination therapy is an avenue for further exploration with ustekinumab. Although the combination of ustekinumab with methotrexate 126 and acitretin 34 has been reported, simultaneous use with apremilast, fumaric acid esters, cyclosporine, and azathioprine are areas needing evaluation. Interestingly, the combination of ustekinumab and adalimumab for CD has been reported to have favorable results. 126 The need now arises to assess the utility of other biological drugs with ustekinumab in the form of dual biological therapy in those dermatoses showing partial responsiveness to ustekinumab.
Conclusion
Ustekinumab is a good biological drug for the treatment of psoriasis and PsA. Besides, it is the biological choice for pediatric patients with psoriasis owing to its weight-based dosing schedule. Moreover, currently, ustekinumab has been found to be effective in a number of off-label dermatological indications. Furthermore, it is well tolerated by patients with minimal side effects. Nevertheless, there are reports of serious adverse effects following ustekinumab usage. All these points need to be carefully evaluated prior to prescribing ustekinumab.
Data Availability Statement
The data that support the findings of this study are available in the supplementary material of this article.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and publication of this article.
