Abstract
This review summarizes the off-label applications of topical sirolimus (rapamycin) for dermatological conditions beyond its approved use for angiofibromas associated with tuberous sclerosis, focusing on efficacy, safety, and reported adverse effects. Off-label use of mammalian target of rapamycin inhibitors like sirolimus has shown promising potential in dermatology. We analyzed the literature on the efficacy and safety of topical sirolimus for a range of dermatological conditions, including inflammatory eruptions, genodermatoses, bullous disorders, neoplasms, and others. Publications on topical sirolimus for other vascular anomalies, such as lymphatic malformations, were excluded to maintain focus on other lesser-known dermatological uses, except for studies involving post-laser combination therapies for vascular lesions. Topical sirolimus demonstrated notable efficacy in several genetic and benign proliferative skin disorders, including pachyonychia congenita, trichilemmomas, trichoepitheliomas, and Kaposi sarcoma, with a generally favorable safety profile and minimal systemic absorption. However, variability in formulations, dosing regimens, and the predominance of small-scale studies limit definitive conclusions. Further research is warranted to standardize treatment protocols and validate these findings.
Introduction
Sirolimus, or rapamycin, is an mammalian target of rapamycin (mTOR) inhibitor originally FDA-approved as an oral immunosuppressant for preventing organ rejection and treating lymphangioleiomyomatosis. 1 More recently, topical sirolimus – HYFTOR® (Nobelpharma America, LLC, Bethesda, MD, USA) – was approved for the treatment of facial angiofibromas associated with tuberous sclerosis. 2 Beyond its immunosuppressive properties, sirolimus demonstrates antineoplastic effects by inhibiting cell proliferation and angiogenesis.3,4
Mechanistically, sirolimus binds FKBP12 within target cells to form an immunosuppressive complex, which inhibits mTOR, a serine/threonine protein kinase central to pathways regulating cell growth, proliferation, survival, and angiogenesis, including the PI3K/Akt signaling cascade.3,4 By disrupting this pathway, sirolimus prevents cytokine-driven T-cell proliferation and halts cell cycle progression from G1 to S phase. 3 Additionally, sirolimus may reduce the production of nonfunctional keratin by downregulating genes such as KRT6 and KRT16, potentially alleviating symptoms in conditions like pachyonychia congenita (PC). 5
While the registered topical formulation is the only commercially available topical sirolimus product, various compounded formulations (solutions, mouthwashes, creams, and ointments) ranging from 0.001% to 8% concentration are used in clinical practice.6-10 Despite its expanding off-label use in dermatology, current literature lacks a comprehensive synthesis of efficacy, safety, and practical considerations across different dermatological indications.
This review aims to systematically summarize the off-label applications of topical sirolimus for dermatologic conditions beyond tuberous sclerosis-associated angiofibromas, with a focus on reported clinical outcomes, safety profiles, and limitations of current evidence.
Materials and Methods
A comprehensive literature search was conducted using PubMed, Scopus, and Embase to identify studies from the past 50 years on the off-label topical application of sirolimus for dermatological conditions beyond angiofibromas in tuberous sclerosis. Search terms included “topical sirolimus,” “topical rapamycin,” and “off-label application.” The search was limited to articles published in English.
Studies were included if they reported on the clinical use of topical sirolimus in human subjects for dermatologic conditions other than tuberous sclerosis-associated angiofibromas. Both monotherapy and combination therapy studies (eg, topical sirolimus combined with laser or other treatments) were considered. In contrast, studies focusing primarily on vascular or lymphatic malformations (eg, lymphangiomas) were excluded, to maintain the review’s focus on lesser-known dermatologic uses of topical sirolimus.
The initial search results were screened by title and abstract to exclude irrelevant studies. Full-text reviews were then performed to assess eligibility based on inclusion and exclusion criteria. Case reports, case series, retrospective and prospective studies, randomized controlled trials (RCTs), and literature reviews were included. Preclinical animal studies were excluded, except when used to support mechanistic insights in conditions with limited human data.
Study quality was assessed descriptively based on study design, sample size, and the level of evidence, following a simplified hierarchy adapted from the Oxford Centre for Evidence-Based Medicine. Level 1 evidence was assigned to RCTs, Level 2 to non-randomized prospective studies, Level 3 to retrospective studies, and Level 4 to case reports and case series. This assessment was intended to contextualize the strength of the available data rather than as a formal systematic review grading.
In total, 47 studies met the inclusion criteria and were included in this review.
Results
The off-label topical application of sirolimus has been reported for a broad range of dermatologic conditions. For clarity, results are organized by disease categories, including genodermatoses, skin tumors, bullous diseases, inflammatory dermatoses, vascular proliferative lesions, and other dermatologic conditions.
An overview of included conditions is presented in Table 1, and details of study interventions and clinical outcomes are summarized in Table 2.
Search Results: Off-Label Topical Sirolimus Applications.
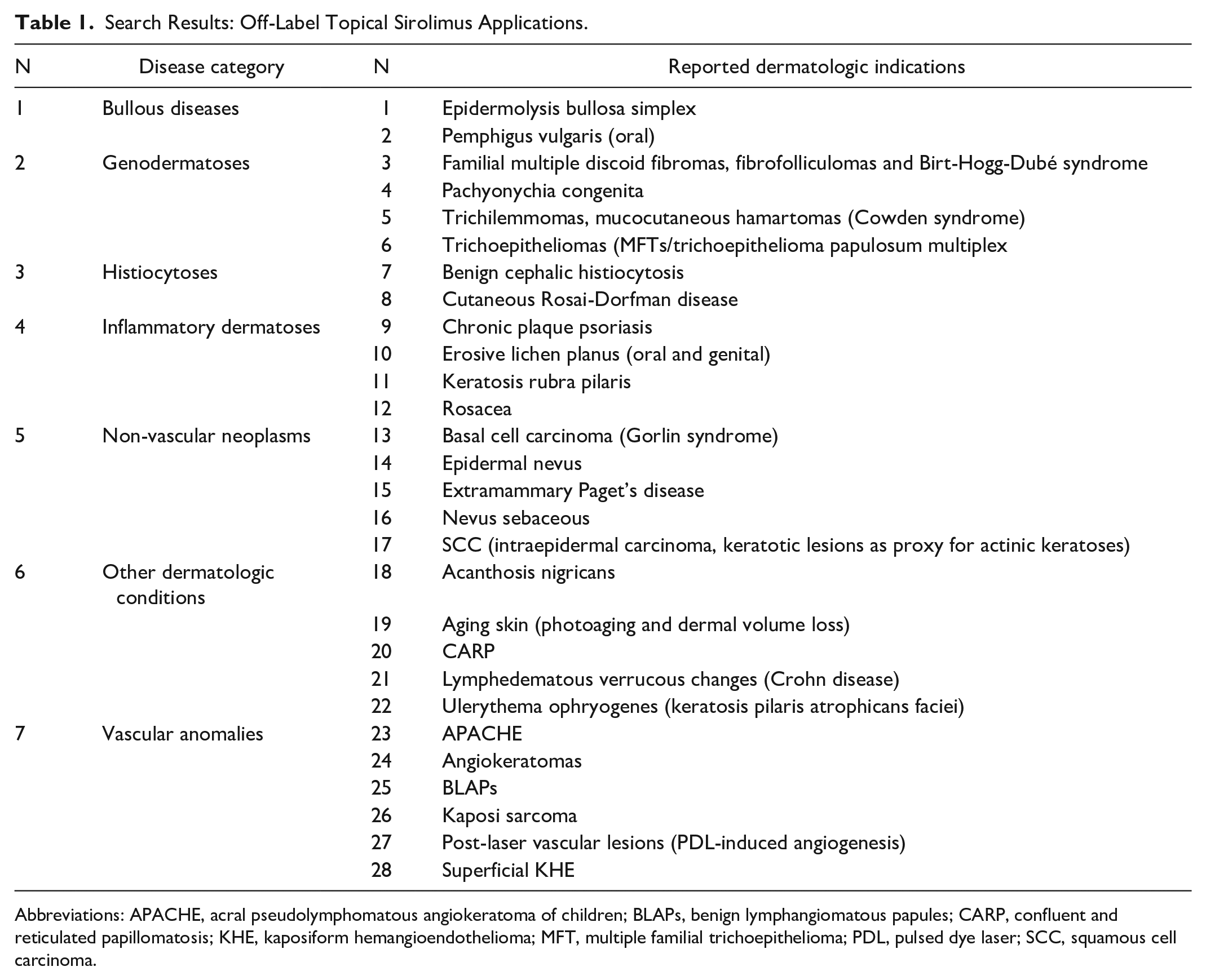
Abbreviations: APACHE, acral pseudolymphomatous angiokeratoma of children; BLAPs, benign lymphangiomatous papules; CARP, confluent and reticulated papillomatosis; KHE, kaposiform hemangioendothelioma; MFT, multiple familial trichoepithelioma; PDL, pulsed dye laser; SCC, squamous cell carcinoma.
Reported Cases of Off-Label Topical Sirolimus Application for Dermatological Conditions.
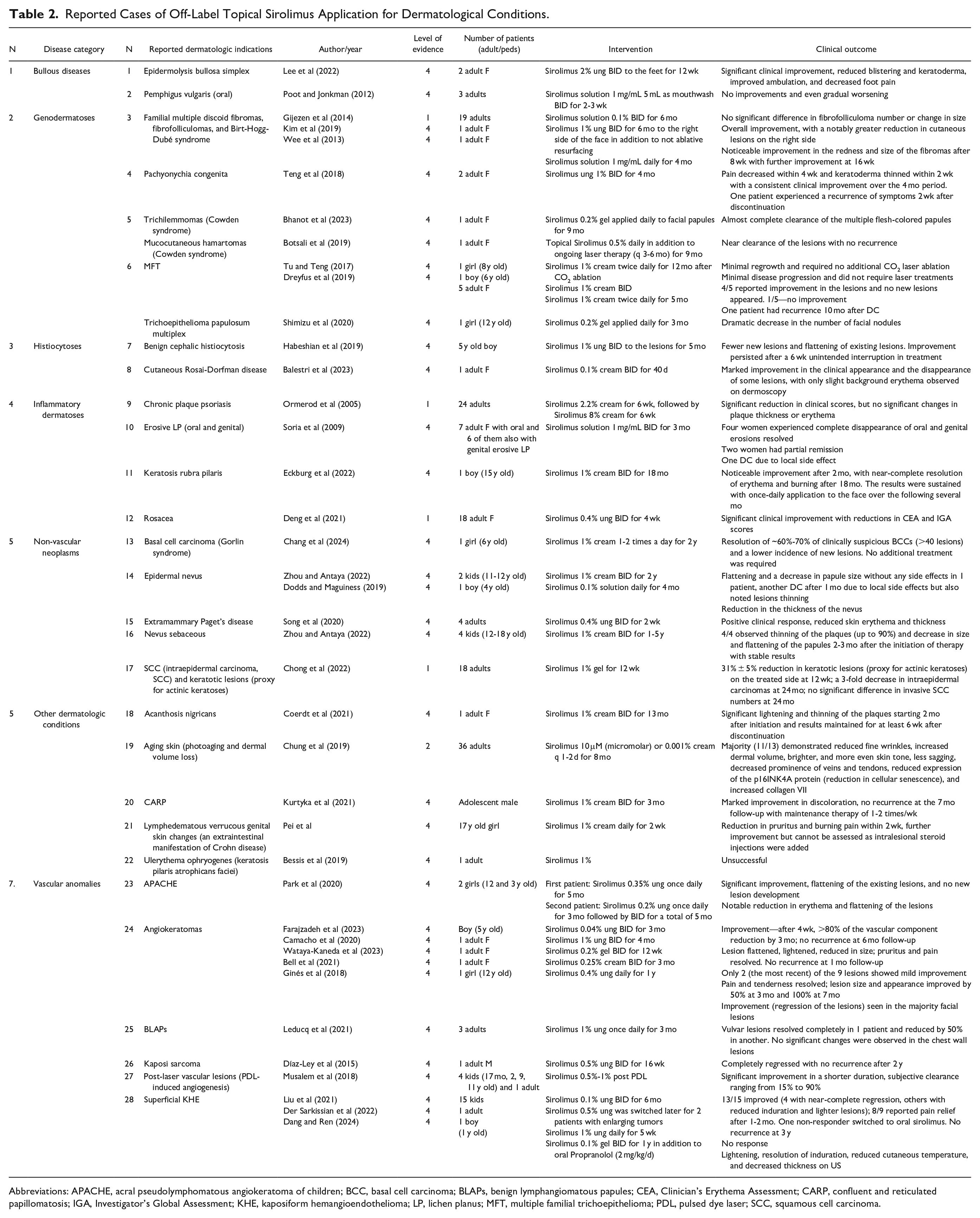
Abbreviations: APACHE, acral pseudolymphomatous angiokeratoma of children; BCC, basal cell carcinoma; BLAPs, benign lymphangiomatous papules; CEA, Clinician’s Erythema Assessment; CARP, confluent and reticulated papillomatosis; IGA, Investigator’s Global Assessment; KHE, kaposiform hemangioendothelioma; LP, lichen planus; MFT, multiple familial trichoepithelioma; PDL, pulsed dye laser; SCC, squamous cell carcinoma.
Bullous Diseases
Epidermolysis bullosa simplex
Epidermolysis bullosa simplex (EBS) is a hereditary blistering disorder caused by mutations in keratin 5 or 14, leading to skin fragility and intraepidermal blistering in response to minor trauma. 11 Lee et al reported 2 adult female patients with plantar EBS and painful plantar keratoderma who were treated with sirolimus 2% ointment applied twice daily for 12 weeks. 11 Both patients experienced significant clinical improvement, including reduced blistering, thinning of keratoderma, improved ambulation, and decreased foot pain. No adverse events were reported. mTOR inhibition may reduce keratinocyte hyperproliferation and inflammatory signaling, thereby enhancing epidermal stability and reducing mechanical stress-induced injury. 11
Pemphigus vulgaris
Pemphigus vulgaris (PV) is an autoimmune blistering disease of the skin and mucous membranes caused by autoantibodies against desmoglein 1 and 3, with desmoglein 3 being predominantly targeted in mucosal-dominant PV. 12 Poot and Jonkman treated 3 PV patients with painful oral erosions using sirolimus 1 mg/mL solution – Rapamune® (Pfizer Inc., New York, NY, USA) – twice daily as a mouthwash. No improvements were observed, with gradual worsening noted, and treatment was discontinued in favor of high-dose steroids with azathioprine. 13 No specific adverse events were observed. Pretel et al demonstrated that sirolimus protected keratinocytes from acantholysis by inhibiting apoptosis, suggesting a potential disease-specific mechanism of action. 14
Genodermatoses
Familial multiple discoid fibromas, fibrofolliculomas, and Birt-Hogg-Dubé syndrome
Birt-Hogg-Dubé (BHD) syndrome is a rare autosomal dominant genodermatosis characterized by multiple fibrofolliculomas, trichodiscomas, and acrochordons. It is caused by mutations in the folliculin gene, impacting mTOR signaling. 15 Gijezen et al conducted a RCT of 19 patients with BHD using 0.1% sirolimus solution twice daily for 6 months, which showed no significant reduction in fibrofolliculoma size or number. 15 However, Kim et al reported improved cosmetic outcomes using 1% sirolimus ointment combined with 1540 nm nonablative laser. 16 Wee et al also described a case of familial multiple discoid fibromas in a 27 year-old male who experienced improved size and redness of the fibromas after applying sirolimus 1 mg/mL solution daily for 4 months. 17 No adverse events were reported.
In BHD syndrome, sirolimus may act by reducing abnormal follicular proliferation.15,16
Pachyonychia congenita
PC is a rare autosomal dominant disorder of cornification caused by heterozygous mutations in keratin genes, most commonly KRT6A, and is characterized by painful palmoplantar keratoderma, blistering and maceration, nail dystrophy, oral leukokeratosis, and follicular keratosis. 18 Teng et al described 2 adult female patients with PC who applied sirolimus 1% ointment twice daily for 4 months. 5 Both reported reduced plantar pain within 4 weeks and thinning of plantar keratoderma. Clinical improvement was sustained during treatment, although 1 patient experienced symptom recurrence 2 weeks after discontinuation. No systemic adverse events were reported. Sirolimus is thought to reduce nonfunctional keratin production by downregulating keratin gene expression such as KRT6 and KRT16. 5
Trichilemmomas, mucocutaneous hamartomas (Cowden syndrome)
Cowden syndrome is a rare autosomal dominant genodermatosis associated with PTEN mutations, characterized by mucocutaneous hamartomas including facial trichilemmomas, oral papillomas, and acral keratoses. 19 In a 67 year-old woman with Cowden syndrome, sirolimus 0.2% gel was applied once daily for 9 months, resulting in near-complete clearance of facial trichilemmomas. 19
Botsali et al reported sustained improvement over 9 months in mucocutaneous hamartomas with sirolimus 0.5% ointment in combination with periodic erbium:YAG laser treatment in a 57 year-old female with Cowden syndrome. 20 No adverse events were reported. Sirolimus may be effective in Cowden syndrome through inhibition of the overactive PI3K/AKT/mTOR pathway driven by PTEN deficiency. 19
Trichoepitheliomas (multiple familial trichoepitheliomas)/trichoepithelioma papulosum multiplex
Trichoepitheliomas are benign follicular neoplasms that may occur sporadically or in inherited patterns such as multiple familial trichoepitheliomas (MFT) or Brooke-Spiegler syndrome, typically presenting as skin-colored papules on the central face. 6 Tu and Teng described 2 siblings with MFT, one of whom was treated with sirolimus 1% cream twice daily following CO2 laser ablation, with minimal regrowth after 12 months, while the untreated sibling showed slower progression after 7 months of sirolimus without laser treatment. 6
Dreyfus et al reported 5 adult females with MFT treated with sirolimus 1% cream twice daily for 5 months; 4 showed lesion improvement and no new lesion development. 21
Shimizu et al described a 12 year-old girl with MFT who experienced dramatic reduction in facial nodules after using sirolimus 0.2% gel once daily for 3 months. 22 No adverse events were reported. The suppression of abnormal follicular proliferation is thought to be how sirolimus may aid patients with trichoepitheliomas. 6
Histiocytoses
Benign cephalic histiocytosis
Benign cephalic histiocytosis (BCH) is a self-limiting, non-Langerhans cell histiocytosis that typically affects infants and young children, presenting as asymptomatic red-yellow papules on the head and neck. 23 Habeshian et al treated a 5 year-old boy with BCH using sirolimus 1% ointment twice daily for 5 months, resulting in flattening and reduction of lesions. 23 Improvement persisted despite a 6 week treatment interruption. No adverse events were reported.
Cutaneous Rosai-Dorfman disease
Rosai-Dorfman disease (RDD) is a rare non-Langerhans cell histiocytosis characterized by dermal or subcutaneous nodules composed of S100-positive histiocytes. 24 Balestri et al reported a 47 year-old woman with cutaneous RDD treated with sirolimus 0.1% cream applied twice daily for 40 days. 24 Clinical improvement included flattening of nodules and partial resolution of lesions. No adverse effects were observed.
The proposed mechanism involves inhibition of histiocytic proliferation through modulation of the mTOR signaling pathway. 24
Inflammatory Dermatoses
Chronic plaque psoriasis
Chronic plaque psoriasis is a common inflammatory skin disorder characterized by erythematous plaques with silvery scale, resulting from immune-mediated keratinocyte hyperproliferation.
10
Ormerod et al conducted a randomized, double-blind study in 24 adult patients, applying 2.2% sirolimus for 6 weeks followed by 8% sirolimus for another 6 weeks.
10
Significant improvements were observed in clinical scores (
Erosive lichen planus (oral)
Erosive lichen planus (LP) is a chronic inflammatory mucocutaneous condition marked by painful erosions of the oral mucosa, often co-occurring with genital involvement. 25 Soria et al reported on 7 steroid-refractory female patients treated with sirolimus 1 mg/mL oral solution used twice daily for 3 months. 25 Oral lesions resolved completely in 4 patients, and genital lesions in all 6 with genital involvement. Mild local discomfort was noted initially and resolved in most cases, though 1 patient discontinued treatment. One patient had detectable systemic sirolimus levels, but no systemic adverse effects were observed.
Keratosis pilaris rubra
Keratosis pilaris rubra (KPR) is a rare inflammatory keratinization disorder characterized by facial erythema, follicular hyperkeratosis, and burning discomfort. 26 Eckburg et al described a 15 year-old boy with KPR treated with sirolimus 1% cream applied twice daily for 18 months. 26 The patient experienced near-complete resolution of erythema and burning. Sustained improvement was maintained with once-daily application for several additional months. No adverse events were reported.
Rosacea
Rosacea is a chronic inflammatory skin condition characterized by facial erythema, flushing, and inflammatory papules. 27 Deng et al identified hyperactivation of mTORC1 signaling in affected epidermis, contributing to disease exacerbation. 27 In a RCT of 18 female patients, 10 patients applied sirolimus 0.4% ointment twice daily for 4 weeks, resulting in significant improvement in Clinician’s Erythema Assessment (CEA) and Investigator’s Global Assessment scores. No adverse events were reported.
Although the included studies in inflammatory dermatoses did not directly investigate the mechanism of action, the observed therapeutic effects of sirolimus may be attributed to its ability to regulate T-cell activation, keratinocyte proliferation, and inflammatory cytokine signaling. These immunomodulatory and antiproliferative effects may contribute to clinical improvement in conditions such as psoriasis, LP, and rosacea.10,25,27
Non-Vascular Neoplasms
Basal cell carcinoma
Basal cell carcinoma (BCC) is the most frequent skin cancer, often associated with dysregulation of the Hedgehog and mTOR signaling pathways. 28 Chang et al reported a 6 year-old girl with Gorlin syndrome who applied sirolimus 1% cream once or twice daily for 1 year. 28 Approximately 60% to 70% of over 40 clinically suspicious BCCs on the eyelids and shoulders resolved, and no new lesions developed over 2 years of continuous treatment. No adverse effects were reported. The therapeutic effect is thought to result from inhibition of basal cell proliferation. 28
Epidermal nevus
Epidermal nevus (EN) is a congenital hamartomatous proliferation of the epidermis, typically following Blaschko’s lines. 7 Zhou and Antaya reported 2 adolescent patients with EN on the posterior neck treated with sirolimus 1% cream applied twice daily. 7 One patient discontinued treatment after 1 month due to dryness, flakiness, and redness, though some lesion thinning was observed. The second patient used sirolimus for 2 years and experienced lesion flattening without adverse effects. Dodds and Maguiness described a 4 year-old boy with extensive EN on the torso, neck, and upper arms in a Blaschkoid distribution, associated with an FGFR3 mutation, who was treated with sirolimus 0.1% solution applied daily to the neck and axilla for 4 months. 8 Treatment resulted in reduced nevus thickness. The therapeutic effect of sirolimus may stem from its inhibition of the FGFR3 pathway, contributing to decreased keratinocyte proliferation. 8
Extramammary Paget’s disease
Extramammary Paget’s disease (EMPD) is a rare intraepithelial adenocarcinoma typically affecting apocrine gland-bearing areas. 29 Song et al reported 4 patients with EMPD treated with sirolimus 0.4% ointment applied twice daily for 2 weeks. 29 Histological evaluation revealed a reduction in PAS+, CEA+, and CAM5.2+ Paget cells and decreased mTOR activity, along with improved epidermal structure. No adverse events were reported. These findings suggest that sirolimus may reduce tumor cell proliferation and improve epidermal architecture in EMPD. 29
Nevus sebaceous
Nevus sebaceous (NS) is a congenital hamartomatous lesion, often located on the scalp, face, or neck, and associated with postzygotic mutations in the RAS/MAPK pathway. 7 Sirolimus, acting downstream via mTOR inhibition, may reduce cellular proliferation in these lesions. Zhou and Antaya described 4 adolescents (ages 12-18) with NS treated with sirolimus 1% cream twice daily for 1 to 5 years. 7 All patients experienced lesion thinning within 2 to 3 months, and 1 achieved 90% flattening of scalp lesions, maintaining results with reduced application frequency. Reported side effects included cracking, itching, bleeding, and folliculitis, which were managed with topical hydrocortisone.
Squamous cell carcinoma
Squamous cell carcinoma is a common skin cancer derived from epidermal keratinocytes, particularly in immunosuppressed individuals. 9 In a 12 week RCT involving 18 adult solid organ transplant recipients, each patient applied 1% sirolimus gel to the dorsum of 1 hand and forearm, while vehicle alone was applied to the contralateral side. 9 The sirolimus-treated side showed a 31% reduction in keratotic lesions at 12 weeks and an 81% decrease in intraepithelial carcinomas over 24 months. One participant developed contact dermatitis. Sirolimus may exert antineoplastic effects by downregulating keratinocyte proliferation and may suppress premalignant transformation. 9
Other Dermatologic Conditions
Acanthosis nigricans
Acanthosis nigricans (AN) is a velvety hyperpigmented thickening of the skin, often associated with insulin resistance or seen in familial cases caused by FGFR3 mutations. 30 Coerdt et al described an 18 year-old female with Fitzpatrick type IV/V skin and an FGFR3 mutation, who applied sirolimus 1% cream twice daily for 13 months. 30 She experienced significant lightening and thinning of plaques, with results maintained for at least 6 weeks after discontinuation. No adverse effects were reported.
Aging skin
Aging skin is characterized by dermal atrophy, fine wrinkles, pigment irregularities, and loss of elasticity, partly due to cellular senescence and reduced collagen integrity.31,32 Chung et al conducted a placebo-controlled trial involving 36 participants over age 40 with photoaged dorsal hands, who applied sirolimus 0.001% cream every 24 to 48 hours for 8 months. 31 Nineteen participants discontinued or were lost to follow-up, but among the 13 who completed the study, 11 showed clinical improvements by 4 months. These included reduction in fine wrinkles, increased dermal volume, more even skin tone, less sagging, and decreased visibility of veins and tendons. Histologic analysis demonstrated reduced expression of p16INK4A (a marker of cellular senescence) and increased collagen VII. Blagosklonny proposed the broader use of topical sirolimus for skin aging, based on its ability to potentially slow the aging process and enhance skin structure. 32
Confluent and reticulated papillomatosis
Confluent and reticulated papillomatosis (CARP) is a rare dermatosis characterized by hyperpigmented, reticulated plaques on the trunk. 33 Kurtyka et al described an adolescent male with CARP and AN who was treated with sirolimus 1% cream twice daily for 3 months. 33 The patient showed marked improvement in discoloration, and results were maintained with 1 to 2 times weekly application for 7 months. No adverse events were noted.
Lymphedematous verrucous changes of the genital skin
Lymphedematous verrucous changes of the genital skin are chronic, thickened, wart-like lesions often associated with inflammation or secondary lymphedema. 34 Pei et al described a 17 year-old female with Crohn’s disease and persistent erythema, swelling, and verrucous papules on the mons pubis and labia. 34 She was treated with sirolimus 1% cream for 2 weeks, resulting in a reduction in pruritus and burning. However, intralesional triamcinolone was added, limiting attribution of long-term improvement to sirolimus alone. No adverse events were reported.
Ulerythema ophryogenes
Ulerythema ophryogenes (UO), or keratosis pilaris atrophicans faciei, is a rare condition causing eyebrow and facial follicular atrophy and erythema and may occur as part of genetic syndromes such as cardiofaciocutaneous (CFC) syndrome. 35 Bessis et al reported a case of UO in CFC syndrome treated with sirolimus 1% cream without clinical improvement. 35
Vascular Anomalies
Acral pseudolymphomatous angiokeratoma of children
Acral pseudolymphomatous angiokeratoma of children (APACHE) is a rare, benign dermatosis characterized by multiple, asymptomatic, erythematous papules in acral regions. 36 Park et al described 2 pediatric patients with APACHE treated with sirolimus 0.2% and 0.35% ointment twice a day for 5 months, leading to significant flattening of the lesions and reduction in erythema. 36 No adverse effects were reported.
Angiokeratomas
Angiokeratomas are benign vascular neoplasms characterized by dilated capillaries in the superficial dermis with an overlying hyperkeratotic surface. They may present as solitary lesions or as multiple lesions in the context of genetic disorders such as Fabry disease. 37 Farajzadeh et al described a 5 year-old boy with a 5 × 6 cm angiokeratoma on the proximal thigh treated with sirolimus 0.04% ointment twice daily for 3 months. 38 Over 80% of the vascular component resolved, with sustained improvement and no complications at 6 months. Camacho et al reported an 18 year-old woman with a painful and pruritic 1.4 × 1.4 cm angiokeratoma on the right buttock, who achieved complete resolution of symptoms and lesion flattening after 4 months of sirolimus 1% ointment twice daily. 39 Wataya-Kaneda et al treated a 47 year-old woman with Fabry disease and 9 angiokeratomas on the trunk, head, and upper extremities using sirolimus 0.2% gel twice daily for 12 weeks; only 2 lesions showed mild improvement. 37 Bell et al treated a 61 year-old man with Fordyce-type angiokeratomas using sirolimus 0.25% cream twice daily, reporting a 50% subjective improvement after 3 months and complete resolution of pain with 100% improvement in lesion appearance after 7 months. 40 Ginés et al also reported a 12 year-old boy with facial angiofibromas treated with 0.4% sirolimus ointment once daily for 1 year, with improvement noted in the malar and infraorbital areas, but no change in the nasal region. 41 These cases suggest sirolimus may have a role in reducing vascular proliferation and associated inflammation.37-40
Benign lymphangiomatous papules
Benign lymphangiomatous papules (BLAPs) are benign lymphatic proliferative lesions that typically arise due to lymphatic obstruction, often following surgery or radiotherapy for malignancy. 42 Leducq et al reported 3 adult patients with BLAPs after radiotherapy for endometrial or breast cancer. 42 Two patients had vulvar lesions and 1 had lesions on the anterior chest wall, associated with oozing, pruritus, and lymphorrhea. All were treated with sirolimus 1% ointment once daily for 3 months. The vulvar lesions resolved completely or partially, whereas chest wall lesions showed minimal improvement. The authors suggested this difference may reflect regional variations in epidermal thickness, occlusion, and local humidity. No adverse effects were reported. The therapeutic effect of sirolimus in BLAPs may be attributed to its ability to suppress abnormal lymphatic endothelial proliferation and local inflammatory responses, as observed in other lymphatic disorders. 42
Kaposi sarcoma
Kaposi sarcoma (KS) is a vascular neoplasm linked to human herpesvirus 8 and characterized by endothelial proliferation and inflammation. 43 Díaz-Ley et al described a 73 year-old man with classic (Mediterranean) KS localized to the right foot and ankle who was treated with sirolimus 0.5% ointment twice daily for 16 weeks. 43 Complete regression was achieved, with no recurrence noted after 2 years of follow-up. Mild pruritus occurred during early treatment but resolved spontaneously. This case suggests sirolimus may offer a well-tolerated topical option for localized KS via inhibition of endothelial proliferation. 43
Post-laser vascular lesions (pulsed dye laser-induced angiogenesis)
Port-wine stains (PWS) are congenital capillary malformations that are often treated with pulsed dye laser (PDL). Despite PDL’s effectiveness, treatment outcomes are limited by post-laser angiogenesis and vascular regeneration, which may reduce blanching.44-46 Musalem et al retrospectively reviewed 5 patients with PWS who were treated with PDL followed by sirolimus 0.5% to 1% cream. 45 Subjective clearance ranged from 15% to 90%, with minimal adverse effects—1 case of transient burning. Several patients showed marked improvement when sirolimus was introduced after a plateau in PDL response, suggesting a potential role for sirolimus in enhancing laser outcomes. Although direct mechanistic data in humans are limited, preclinical studies have shown that sirolimus may inhibit angiogenic pathways, including VEGF and HIF-1α expression, supporting its proposed effect in this setting. 44
Superficial kaposiform hemangioendothelioma
Superficial kaposiform hemangioendothelioma (KHE) is a rare, locally aggressive vascular tumor that may present in infancy or adulthood. 47 In a retrospective study of 15 children with KHE treated with sirolimus 0.1% ointment twice daily for 6 months, 13 patients improved, and 4 achieved near-complete regression. 48 Two additional patients responded to a higher concentration (0.5% ointment), and no relapses occurred after 3 years. In contrast, Der Sarkissian et al described a 65 year-old man with neck KHE who failed to respond to 1% sirolimus ointment after 5 weeks. 47 Dang and Ren presented a 1 year-old boy with a 10 month history of KHE on the left mandible and prothorax, treated with sirolimus 0.1% gel twice daily plus oral propranolol (2 mg/kg/day) for 1 year. 49 The child showed noticeable improvement in induration, thickness, and lesion color. Across all cases, topical sirolimus was well tolerated, with no adverse effects reported. These reports suggest sirolimus may be most effective in pediatric superficial KHE, particularly when initiated early or combined with adjunctive therapies.47-49
Topical formulation and adverse effects
Formulation concentrations of sirolimus ranged from 0.001% to 8% and were compounded into creams, ointments, and gels,6-10 in addition to the commercially available HYFTOR® 0.2% gel. 2 Dosing was typically once or twice daily. Notably, no comparative studies have evaluated the relative efficacy or adverse effects of different concentrations or dosing frequencies.7-9,43
Topical sirolimus was typically well tolerated, with skin irritation as the most common side effect, which often resolved with continued use, temporary discontinuation, or adjunctive use of topical hydrocortisone.7,9,31 Less frequent side effects included superficial mucosal erosions, mild facial acne (with facial application), folliculitis, and allergic contact dermatitis.9,16,34 Two patients reported headaches that resolved with simple analgesia, 9 but no significant systemic adverse events were noted.7,9,31 Blood levels, when measured, remained below the immunosuppressive threshold (4 ng/mL), minimizing concerns about systemic immunosuppression.9,41 In vascular and lymphatic lesions, topical sirolimus was also well tolerated, including in pediatric populations with long-term use.20,27,41
Discussion
The application of topical sirolimus for various dermatologic conditions demonstrates a wide spectrum of efficacy, with outcomes largely dependent on the underlying pathology and treatment parameters. Sirolimus, an mTOR inhibitor, has shown therapeutic potential through modulation of the cell growth, proliferation, angiogenesis, lymphangiogenesis, and inflammation.3,4
Topical sirolimus appears particularly effective for genetic and benign proliferative skin disorders such as PC,5,18 trichilemmomas, 19 trichoepitheliomas,6,21,22 and epidermal nevi.7,8 In these conditions, it reduced lesion size, discomfort, and recurrence, with enhanced outcomes reported when combined with adjunctive laser therapy.16,20 For example, patients with trichilemmomas and trichoepitheliomas showed minimal regrowth following laser-assisted sirolimus delivery, and those with PC experienced reduced keratoderma thickness and pain, improving quality of life.
Encouraging results were also observed in vascular anomalies—including angiokeratomas,9,27-29,30 superficial KHE,48,49 and port-wine stains44-46—especially when sirolimus was used in conjunction with PDL therapy. In such cases, sirolimus likely exerts its benefit through inhibition of angiogenic and endothelial proliferative pathways.44,46 Conditions characterized by vascular proliferation and inflammation, such as KS, 43 also responded well, with 1 report noting complete lesion regression sustained for 2 years.
While sirolimus has shown positive outcomes in several inflammatory conditions such as psoriasis, 10 oral LP, 25 and CARP, 33 results have been mixed in disorders like PV13,14 and BLAPs. 42 These differences may reflect the degree to which the mTOR pathway contributes to disease pathogenesis.
Variability in reported outcomes also stems from the lack of standardized formulations and dosing regimens. Concentrations ranged from 0.001% to 8%,6-10 delivered in various vehicles and applied at different frequencies,7-9,43 complicating cross-study comparisons. Although preclinical (murine) studies were not included for efficacy assessment, they were selectively referenced to support proposed mechanisms—such as the inhibition of post-laser angiogenesis 44 and lymphatic endothelial proliferation 42 —in settings where human mechanistic data were lacking. Overall, topical sirolimus maintains a favorable safety profile, with most adverse effects being mild and localized. Systemic absorption was minimal, with measured blood levels consistently below the immunosuppressive threshold.9,41 This favorable safety profile makes topical sirolimus an appealing option for localized dermatologic treatment, particularly in patients who are unsuitable for systemic immunosuppressive therapies.
Conclusion
Topical sirolimus demonstrates promising off-label therapeutic potential across a wide range of dermatologic conditions, particularly in genetic and benign proliferative disorders, vascular anomalies, and select inflammatory dermatoses. Its mechanism of action—via mTOR inhibition—enables targeted modulation of keratinocyte proliferation, angiogenesis, lymphangiogenesis, and immune responses. Reported outcomes include lesion size reduction, symptom control, and high local tolerability, with minimal systemic absorption and a low incidence of adverse effects. Although most of the current evidence is derived from case reports and small series, the consistency of reported clinical benefits supports its potential utility in dermatology, especially in patients who may not tolerate systemic immunosuppressive therapies. However, the heterogeneity of formulations, dosing regimens, and outcome measures hinders direct comparison and generalizability.
Further research is essential to establish standardized treatment protocols, assess long-term safety, and better understand disease-specific mechanisms of action. Larger, controlled studies are needed to define optimal concentrations, formulations, and dosing schedules. With a stronger evidence base, topical sirolimus may become an accepted and effective treatment option for a range of complex cutaneous conditions.
Limitations
There are several limitations in this literature review. A key limitation is that many included studies are case reports or case series without a comparator group, which increases the risk of publication bias and limits the strength of the conclusions. There was considerable heterogeneity in treatment protocols, including sirolimus concentrations, formulations, frequency, and duration of application, as well as variability in outcome reporting. These differences make direct comparisons between studies difficult and limit the generalizability of findings.
Furthermore, sirolimus topical therapy was often combined with surgical or laser modalities, making it challenging to isolate its independent therapeutic effect. Although preclinical (murine) studies were excluded from clinical efficacy analysis, they were referenced selectively to support proposed mechanisms of action, particularly in vascular and lymphatic proliferative conditions where human mechanistic data are limited.
Additionally, standardized clinical scoring tools were rarely used, and long-term follow-up was often lacking, particularly for assessing safety and sustained response. A detailed assessment of individual study quality and methodological limitations was beyond the scope of this review due to its broad coverage. Future research should aim to address these gaps through prospective, controlled studies with uniform endpoints and extended follow-up periods to better evaluate the long-term efficacy and safety of topical sirolimus.
Footnotes
Author Contributions
Tatiana Lapa: data curation, writing—original draft preparation. Matt Sandre: conceptualization, methodology, supervision, writing—review and editing.
Data Availability Statement
Data sharing not applicable—no new data generated.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
Not applicable.
