Abstract
Trends in health care delivery systems have shifted as a result of the modern uses of biomaterials in medicine. Contrary to traditional medicine, modern healthcare are now useful in solving problems that were considered impossible some years back. One of the most significant factors to the most recent advancements in implant development has been the use of calcium based materials in the creation of necessary implants in the form of soft and hard tissues. With the advent of naturally sourced materials in the manufacturing of biomaterials, lots of attention are now focused on the different sources of agro-based resources that can be used for the product developments. These agro-based materials are now been considered for sustainable and ecological purposes in several areas of applications globally in the recent times. Hence, the review was carried out with focus on the sources, relevance, processing techniques and applications of tricalcium phosphate based biomaterials in modern day healthcare delivery. This review provides a historical and prospective picture of the crucial functions that materials based on tricalcium phosphate will play in fulfilling human requirements for medication.
Keywords
Introduction
The emergence of biomaterials has significantly transformed healthcare delivery, particularly, in tissue engineering, regenerative medicine, and personalized therapies. However, early biomaterials were inert and served as passive supports. But recently, the advancements in biomaterials have focused on materials that can actively modulate biological responses, such as immunomodulatory biomaterials.1-3 Precision biomaterials are now designed for individual patients using digital technologies and big data.4,5 Additive manufacturing, like 3D printing, allows for the creation of complex structures that mimic natural tissues, enhancing applications in bone regeneration and wound healing.6-8 Biomaterials are synthetic materials being used to create devices that can replace biological parts and work well with living tissue. The development of biomaterials has reversed the conventional medical practice of amputating damaged parts. 9 Within the biomedical field, many biomaterials, including biomedical implants, are being created for various applications. Consequently, a fast-developing class of biomaterials known as biomedical implants is being utilized to replace damaged or sick soft or hard tissues in the human body. Since these materials were created to satisfy structural and biocompatibility requirements, it is anticipated that they would be secure and well-tolerated by the body as the patient grows.9,10
Mineralized tissue called bone is essential for the body’s support and defense. On the other hand, several conditions, including trauma, age, inflammation, infections, and malignancies, can result in bone abnormalities and have an impact on everyday life and health. 11 Significant bone defects greater than 2 cm or more than 50% of the defect’s circumference often cannot mend spontaneously and require using biomaterials for repair. Still, certain faults can heal up rather rapidly. 12
Every year, millions of people have bone grafting issues that needs medical attention. Treatment of tissue defects represents a major challenge in clinics due to issues involving shortage of donors, inappropriate sizes, abnormal shapes, and immunological rejection. 13 Hence, to address these problems which have huge financial impact, it is imperative from a scientific and clinical standpoint to create improved biomaterials for tissue healing. 14 In recent times, bone tissue engineering, membrane-guided regeneration, Ilizarov technique, and bone transplantation are typical therapies for bone defects. 15 However, these approaches frequently fail to satisfy clinical requirements. The gold standard for autologous bone grafting is limited by the following factors: risks of infection, blood loss, reoperation after surgery, and limited tissue availability. 14
Tricalcium phosphate (TCP) possesses a crystalline structure and chemical makeup similar to bone, making it a biocompatible and bioabsorbable material. Interestingly, when compared to other bone replacements, its rate of biodegradation is higher than that of hydroxyapatite (HA). 14 TCP is distinguished by its unique crystalline and physical characteristics, as well as by its very pure and homogeneous chemical makeup. There are 3 different forms of TCP: β-TCP, which is the low-temperature version, and α- and α′-TCP, which are the high-temperature forms. Nevertheless, as α′-TCP can only be found above ~1430°C and quickly turns back to α-TCP when the temperature drops below the transition point, it has little practical use. On the other hand, β-TCP exhibits stability at room temperature, but at around ~1125°C, it reconstructs itself to become α-TCP, a state that lasts until the temperature drops to normal temperature.
Currently, due to environmental concern, biomaterials are being developed from readily available agricultural by-products due to their comparable bio-mineral contents. 16 While hydroxyapatite (HA) has been widely developed and used as an alternate inorganic source for bone and tooth replacements and grafting, 17 less attention has been given to tricalcium phosphate (TCP) based biomaterials which is one of the reasons for this study. Therefore, this review focused on tricalcium phosphates (TCP), which have shown significant potential in bone healing. Studies, such as those by Vdoviaková et al, 18 demonstrated that, TCP ceramics can effectively fill bone defects and promote new bone formation characterized by immature bone with a high density of osteoblasts and disorganized collagen. Hence, the review highlights the importance of TCP as bioceramics in enhancing osteoconductivity and calls for more TCP based advanced biomaterials with bioactive properties to stimulate the body’s regenerative processes for improved bone healing and regeneration. It also underscored the methods for the synthesis of TCP from natural materials for sustainable development.
Impact of Agro-Based Resources on Sustainability of Biomaterials
Application of agro-based materials in polymer composites for various applications has form part of the new trends in advanced materials development. This class of materials based on source of origin, belongs to natural materials. Natural materials have been reported to be biodegradable and contribute to the development of green economy due to their several advantages such as environmental friendliness, energy efficiency, ease of manufacturing, low cost, and sustainability. 19 Natural materials have recently become more attractive to technology and innovation as substitute for synthetic materials due to these merits in several areas of applications including biomedical. 20 Several works have been carried out to reveal the potential applications of natural materials sourced from plants, animals and minerals. Among the commonly used materials are; snail shells, eggshells, cow bone, fish bones, and scales.21-23
The use of agricultural by-products for the production of biomaterials helps reduce environmental pollutions and supports the creation of eco-friendly biomaterials that can replace petroleum-based products. Recently, agro-wastes, like plant matter and food residues, are valuable for producing bioplastics which lowers fossil fuel dependence and reduce the amount of plastic pollutions in our environments. 24 Bioplastics made from renewable resources such as starch and cellulose from agricultural by-products is sustainable and efficient alternatives for various applications in modern days. 25 Thus, these developments are being advanced based on current and future concerns for sustainable human and environmental needs.
Nowadays, valorizing agro-industrial food wastes is emerging as an effective method for producing high-value biomaterials. Lignocellulosic biomass, a major component of agricultural waste, can be converted into nanocellulose and other valuable products using sustainable biorefinery techniques. 26 This process not only tackles waste management issues but also supports the circular economy by turning waste into useful resources.26,27 Also, the use of agro-based resources in biomaterials development reduces greenhouse gas emissions compared to traditional manufacturing process with synthetic materials, thereby, lowering the carbon footprint significantly.28,29 The development of biocompatible materials from agro-wastes, such as egg and snail shells, demonstrates the potential for transforming waste into high-value medical products.22,30,31
Sources and production methods for tricalcium phosphate
With the chemical formula Ca3(PO4)2, tricalcium phosphate (TCP) is the calcium salt of phosphoric acid. It is one of the 3 calcium phosphate polymorphs that make up the CaO–P2O5 system, along with β-TCP, α-TCP, and α′-TCP. 32 It is frequently utilized in many different contexts, such as calcium supplements, dental products, and food additives. The sources of tricalcium phosphate can be categorized into natural and synthetic as shown in Table 1.
Sources of tricalcium phosphate.

Synthesis of tricalcium phosphate
The synthesis of tricalcium phosphate (TCP) can be achieved through various methods, each influencing the material’s properties and applications. TCP exists in different polymorphic forms, primarily α-tricalcium phosphate (α-TCP) and β-tricalcium phosphate (β-TCP), which can be synthesized using distinct approaches.
One common method for synthesizing α-TCP involves the thermal treatment of calcium phosphate precursors. Typically, calcium and phosphate sources, such as calcium carbonate and phosphoric acid (calcium phosphate precursors), are mixed and heated at high temperatures (around 1300°C) to facilitate the formation of α-TCP. The temperature and calcination duration impact crystallinity and phase purity, with higher temperatures enhancing strength but risking unwanted phase formation. 41 Optimizing these conditions is essential to ensure α-TCP’s quality for clinical applications. This process often results in a coarse powder that requires milling to achieve the desired particle size for biomedical applications. High-temperature synthesis is crucial because it ensures the stability and crystallinity of the α-TCP phase, which is essential for its bioactivity and mechanical strength. 34
Likewise, β-TCP can be synthesized using several methods, including wet chemical precipitation, mechanochemical synthesis, and sol-gel processes. One common approach for producing β-TCP is the wet chemical precipitation method, which allows for precise control over variables like temperature, pH, and reactant concentrations. For instance, adjusting the concentrations of diammonium hydrogen phosphate and calcium nitrate can affect the particle size and bioactivity of β-TCP. 42 The wet chemical precipitation method involves mixing calcium nitrate and diammonium hydrogen phosphate in an aqueous solution, where the pH is carefully controlled (typically between 8 and 10.8) using sodium hydroxide. This method allows for the formation of nano-sized β-TCP particles, which can be advantageous for enhancing the material’s surface area and reactivity. 43
Another approach for synthesizing β-TCP is through the calcination of biological or synthetic apatite at temperatures above 700°C. This method can yield biphasic calcium phosphate (BCP) materials, which consist of both hydroxyapatite and β-TCP, allowing for tailored solubility and mechanical properties suitable for various clinical applications. 44
Synthesis of TCP can be optimized by adjusting the ratios of the constituent phases to achieve specific biological responses. Also, the incorporation of additives during the process can modify the properties of TCP. For instance, the addition of iron to tricalcium phosphate has been explored to enhance its toughness, making it more suitable for load-bearing applications. 45 Furthermore, the use of biopolymers or other organic compounds during synthesis can improve the bioactivity and integration of TCP with biological tissues. 46 Thus, the influence of production process on the properties of TCP cannot be over emphasized.
Influence of production methods on the quality of tricalcium phosphate
The synthesis methods for tricalcium phosphate (TCP) play a critical role in determining its physicochemical properties, crystallinity, morphology, and biological performance in medical applications. Also, pH control during synthesis is vital as it influences phase formation and precipitation kinetics. Likewise, the additives incorporated during synthesis affect TCP’s properties. For example, adding polymers like alginate or collagen can improve TCP scaffold porosity and interconnectivity, facilitating better cell infiltration and tissue integration. 46 The developed composite materials often demonstrate improved biological responses, such as increased cell attachment and proliferation, compared to pure TCP. 46
Solid state method
Solid-state synthesis is characterized by several key steps, including the selection of appropriate raw materials, mixing, and subsequent high-temperature treatment. 36 In the solid-state method, the most common precursors used are calcium carbonate (CaCO₃) and calcium phosphate compounds such as dicalcium phosphate (DCP) or calcium oxide (CaO). The reaction typically requires a high temperature, often exceeding 800°C, to facilitate the solid-state reaction between these precursors, resulting in the formation of TCP.36,47 For instance, Kariya et al demonstrated that by modifying the heating process in a dental casting mold, β-TCP could be synthesized effectively, highlighting the importance of temperature control in the solid-state method. 47 However, it is important to note that this method often requires long reaction times and high sintering temperatures, which can lead to challenges in achieving uniform particle size and morphology. 37 Moreover, the solid-state method can also be influenced by the presence of additives or modifiers, such as magnesium oxide (MgO), which can alter the phase composition and enhance the properties of the final product. This was illustrated by Altomare et al, who explored the synthesis of new β-TCP variants with different metal substitutions, demonstrating how solid-state reactions can be tailored to produce materials with specific characteristics. 36 The resulting β-TCP produced via solid-state methods is known for its bioactivity and osteoconductivity, making it suitable for various biomedical applications, including bone grafts and scaffolds for tissue engineering.48,49
Solid State Reaction is a traditional method for synthesizing TCP, typically requiring high temperatures (around 1300°C) and extended reaction times. This method often results in coarse powders that necessitate milling to achieve the desired particle size and homogeneity. 34 While this technique can produce materials with good crystallinity, high temperatures can lead to the formation of unwanted phases if the precursors are not pure. 39 Moreover, the long synthesis time can be a significant drawback for large-scale production. 37
Flame spray pyrolysis
It is a method that allows for the production of amorphous tricalcium phosphate nanoparticles. In this technique, a liquid precursor solution is introduced into a flame, where it undergoes rapid thermal decomposition. The resulting nanoparticles exhibit a Ca:P ratio of 1.5, which is essential for the formation of TCP. 50 This method is advantageous due to its ability to produce nanoparticles with controlled morphology and size, which are critical for enhancing the bioactivity of the material in biological environments. 37 This technique allows for the production of nanoparticles with controlled morphology and size, which can enhance the material’s bioactivity. 51 However, the purity of the final product can be compromised due to the presence of residual combustion by-products, which may affect the material properties adversely. 52 Additionally, the complexity of the equipment and the need for precise control over the reaction conditions can limit its widespread application.
Sol-gel method
It’s another prevalent synthesis route for TCP, known for its simplicity and ability to produce nanosized particles at relatively low temperatures. This method involves the conversion of precursor solutions into a gel, which subsequently undergoes drying and heat treatment to form TCP. The sol-gel process has been shown to yield high-purity materials with uniform composition, making it suitable for biomedical applications. 35 Specifically, the sol-gel-derived TCP can be tailored to achieve desired properties such as porosity and mechanical strength, which are vital for bone grafting materials. 53
The Sol-gel Method is advantageous for its ability to produce highly homogeneous materials at relatively low temperatures compared to solid-state methods. This technique allows for the incorporation of various dopants and can yield materials with tailored properties. 54 However, the sol-gel process can be time-consuming, involving multiple steps such as gelation, drying, and calcination, which may extend the overall synthesis time. 38 Furthermore, achieving complete conversion to TCP can be challenging, and the final product may require further processing to enhance crystallinity.55,56,111
Hydrothermal treatment
It’s a synthesis method that involves the use of high-pressure and high-temperature water to facilitate the crystallization of TCP from precursor materials. This technique allows for the production of highly crystalline TCP, which is beneficial for its mechanical properties and bioactivity. 57 The hydrothermal process can also be combined with other methods to enhance the characteristics of the synthesized TCP, such as improving its solubility and biological response. 38 Hydrothermal Treatment is characterized by its ability to produce high-purity materials due to the controlled environment in which the reaction occurs. This method can yield TCP with desirable microstructures and enhanced bioactivity. 58 However, the necessity for high pressure and temperature can complicate the synthesis process and increase operational costs. 37 Additionally, the scalability of this method can be limited due to the specialized equipment required.
High-temperature combustion synthesis
High-temperature combustion synthesis is characterized by exothermic reaction of precursors that leads to the formation of TCP at elevated temperatures. This method is particularly effective for producing TCP with specific stoichiometries and phases, such as α-TCP and β-TCP, which have different biological behaviors. 39 The combustion synthesis route can also be optimized to control the particle size and morphology, which are critical for their application in bone regeneration. 59 High-Temperature Combustion Synthesis is a rapid method that can produce TCP at lower temperatures than traditional solid-state methods. This technique can lead to the formation of TCP with good crystallinity and specific morphologies. 60 However, the combustion process can introduce impurities from the fuels used, which may affect the material properties negatively. 52 Moreover, controlling the combustion conditions is crucial to avoid the formation of undesired phases.
Precipitation method
Chemical Precipitation methods are widely used for their simplicity and ability to produce materials with high purity and uniformity. 42 This method allows for the control of particle size and morphology through careful manipulation of reaction conditions such as pH and temperature. 61 However, the precipitation process can be sensitive to environmental factors, and achieving consistent results may require extensive optimization. 42 Additionally, the drying and calcination steps can add to the overall synthesis time.
These methods have been used to synthesized tricalcium phosphate by many researchers using difference by-products from animal sources.
Synthesis of tricalcium phosphate from eggshells
(a). Beta tricalcium phosphate (β-TCP) was synthesized from eggshells using precipitation method at temperature variations of 600°C-1000°C and sintering durations of 1-5 hours by Sani et al. 62 The results revealed a notable amount of β-TCP composition and Ca/P ratio in the final product where highest β-TCP composition of 81% was achieved at 1000°C for 5 hours of sintering. However, the Ca/P ratio of 1.74 did not meet the desired value of 1.5 which implies that further optimization is needed. Conversely, materials with a higher Ca/P ratio may not provide the same level of biological response, as indicated by the work of Hossain, which discusses the implications of different calcium phosphate compositions in drug delivery and scaffold applications. 63 Achieving the optimal ratio of 1.5 is essential for ensuring the desired physical and biological properties, which are critical for their effectiveness in medical and dental applications. Further optimization of synthesis methods and conditions is necessary to meet this target, thereby enhancing the performance of calcium phosphate-based biomaterials. 64
(b). In another research, collected eggshells were washed and immersed in boiling water for 30 minutes to remove any surface contaminants. The washed eggshells were dried in the oven for 3 hours before crushed into smaller flakes using alumina mortar. The flakes were calcined at a temperature of 900°C for 4 hours in the furnace for a complete transformation of CaCO3 into CaO powders to ensure complete carbon dioxide removal (CO2) as reported by previous researchers.65,66 CaP powders were prepared by using the wet chemical precipitation method followed by calcination at different temperatures. X-ray diffraction (XRD) analysis shows that the 2 types of CaP patterns present are hydroxyapatite (HA) and β-Tricalcium phosphate (β-TCP). Fourier transform infrared (FTIR) shows phosphate ion band in every sample while scanning electron microscopy (SEM) shows the transformation of structure from needle-like to more fluffy and rounded-edge structure from uncalcined to 1000°C. From the results obtained, CaP extracted from eggshell waste was successfully synthesized from the precipitation method. This method contributes to the materials processing cost reduction and increases the application of natural materials instead of synthetic ones.
(c). Synthesis and characterization of calcium phosphate from eggshells of different poultry with and without the eggshell membrane have been carried out. Previous researches have used quail, hen, duck, and pigeon eggshells as a calcium source to obtain calcium phosphate materials via the environmentally friendly wet synthesis. Using the eggshells with the organic membrane, the biphasic calcium phosphate materials composed mainly of HA were obtained. The second mineral phase was β-TCP in the case of using quail, hen, and pigeon eggshells, and octacalcium phosphate (OCP) in the case of duck eggshells. The HA content in the obtained materials depended on the amount of membrane in the eggshells and decreased in the order of pigeon, duck, hen, and quail eggshells, respectively. The eggshell membrane removal from the eggshells caused the reduction in HA content and the presence of the more soluble β-TCP or OCP phase in the obtained materials. The calcium ions release profile in the PBS buffer indicates the potential biomedical application of these materials. 67
Synthesis of tricalcium phosphate from snail shell
(a). Ardiansyah et al 40 carried out the synthesis of calcium phosphate bioceramics from Achatina fulica snail shells. The snail shells were cleaned and dried under direct sunlight before grinding and filtering using mesh size of 100. Calcium oxide (CaO) powder from snail shells was obtained by calcination at 1000°C for 5 hours. Calcium hydroxide (Ca(OH)2) suspension was prepared by weighing 18.5195 g of CaO powder and adding 500 mL of deionized water. The H3PO4 solution was made by diluting 85% H3PO4 solution in deionized water to get a 0.6 M H3PO4 solution. The synthesis of calcium phosphate bioceramics was carried out based on a modified method. A volume of 500 mL of 0.6 M H3PO4 solution in a burette was dropped at a speed of 6 mL/min into a beaker containing 500 mL of 1M Ca(OH)2 suspension at a temperature of 60 ± 2°C with constant stirring. While XRF analysis revealed calcium content of 81.83% in the calcined CaO raw material XRD analysis confirmed the formation of tricalcium phosphates and β-calcium pyrophosphate. The synthesized particles exhibited irregular surface shapes with agglomeration, typical of the precipitation method, including flux, sphere, and fracture shapes.
(b). In the study, carbonated apatite powder (CAp) and β-tricalcium phosphate (β-TCP) was made from leftover snail shells. They are produced via thermal decomposition followed by chemical precipitation method with phosphoric acid in a one step process. A calculated amount of the CaO powders produced from the snail shells was exothermically reacted with ammonium diphosphate solution in a drop wise manner. The powders were mixed and stirred for a day, then filtered and dried in an oven. The final powder was made by crushing the dried material. They then heated the powder at a very high temperature and then characterized using different analytical techniques. 23
(c). Sèmiyou et al 68 studied snail shell powders from 2 different types of snail shells, Lanistes varicus and Achatina achatina snail shell to see if there powder could be used to make calcium phosphate bioceramics. They used co-precipitation and microwave irradiation methods to mix the powders and found that they contained mostly calcium carbonate. The results revealed that the synthesized calcium phosphate bioceramics contain a mixture of Hydroxyapatite (HA: Ca10(PO4)6(OH)2) and Apatitic Tricalcium Phosphate (TPa: Ca9(HPO4) (PO4)5(OH). The study of the antibacterial activity of the synthesized bioceramics showed that only those obtained with Lanistes varicus powder significantly inhibit the growth of Staphylococcus aureus with a lasting effect. On the other hand, inhibition of growth of Klebsiella oxytoca is partial and resistance to antimicrobial activity of bioceramics was noticed. Thus, bioceramics synthesized from Lanistes varicus snail shell powder has antibacterial property and should be used against the growth of pathogens that cause dental cavities.
Preferable methods for the synthesis of TCP
Among various methods for synthesizing tricalcium phosphate (TCP), the microwave-assisted co-precipitation method stands out as one of the best. This method is advantageous due to its ability to produce high-purity β-TCP with controlled particle sizes, reduced synthesis time, and enhanced homogeneity and crystallinity compared to conventional methods like solid-state reactions or wet chemical synthesis. 69 Microwave-assisted synthesis utilizes rapid heating, which can significantly enhance reaction rates and improve the quality of the resulting TCP, making it particularly suitable for biomedical applications where material consistency is crucial as reported by Cestari. 69 Additionally, this method often results in better phase control and reduced impurities, which are critical for biocompatibility and performance in medical implants.
Application of Tricalcium Phosphate
Tricalcium phosphate materials primarily function as osteoconductive materials, allowing bone growth either on their surface or within their pores, channels, or tubes. Calcium phosphate is biocompatible and effective in promoting the formation of hard tissue. It has been utilized in various applications such as a capping agent, in cleft palate procedures for creating apical barriers, in apexification, treating vertical bone defects, and as coatings for implants. Tricalcium phosphate is a resorbable phase of calcium phosphate with favorable properties, including its ability to support bone growth. 70 Table 2 presents some areas of applications for tricalcium phosphate in biomedical industries.
Application and function of tricalcium phosphate.

Tricalcium phosphate (TCP) is a calcium salt of phosphoric acid, commonly used in a variety of applications due to its biocompatibility, osteoconductivity, and similarity to the mineral component of bones. Its uses span across medical, dental, and nutritional fields. The beta form of TCP (Beta TCP or β-TCP) is particularly noteworthy and preferred over other forms of TCP for its superior properties in these applications. 77 Some of its superior properties include:
Bioactivity and Biocompatibility: β-TCP interacts positively with biological tissues, promoting cell attachment, proliferation, and differentiation and it is non-toxic and does not elicit an immune response when implanted in the body. 76
Osteoconductivity: β-TCP provides an ideal scaffold for bone growth, aiding in the repair and regeneration of bone tissues. Its structure allows for the infiltration of bone cells and blood vessels, facilitating the integration with natural bone. 71
Resorbability: β-TCP gradually dissolves and is replaced by new bone tissue over time. This property is crucial for bone healing, as it allows for the gradual transfer of mechanical load to the newly formed bone. 71
Mechanical Strength: Although not as strong as some other bone graft materials like hydroxyapatite, β-TCP has sufficient mechanical strength to support bone tissue during the healing process, making it suitable for load-bearing applications. 77
Major roles of tricalcium phosphate-based biomaterials
Tricalcium phosphate (TCP)-based biomaterials have become crucial in addressing medical challenges, especially in orthopedics and dentistry. They excel in bone regeneration and defect repair by mimicking natural bone’s mineral composition, which supports new bone tissue growth. TCP scaffolds foster osteoblastic differentiation and bone healing by providing a favorable environment for cell attachment and proliferation.78,79 Also, TCP can be combined with polymers like polycaprolactone or collagen to improve strength and elasticity for load-bearing applications. 80 TCP’s antibacterial properties are enhanced through modifications with natural substances such as propolis, reducing infection risks during healing. 81 Beyond bone regeneration, TCP materials are used in vertical bone augmentation and maxillary reconstruction, offering advantages over autogenous grafts due to lower morbidity and complications.82,83 Generally, TCP-based biomaterials represent a significant advancement in regenerative medicine, addressing many limitations of traditional bone grafting methods and promising improved patient outcomes in orthopedic and dental surgeries.
Role of Beta-tricalcium phosphate in bone repair and its mechanism to regulate osteogenesis
Significant bone loss due to trauma, tumor removal, infection, and congenital disorders often results in delayed healing, posing a challenge for orthopedic surgeons, thus, bone autografts have traditionally been used. 84 However, their limitations have prompted the search for alternative bone substitutes. 85 The main challenges in repairing large bone defects include inadequate mesenchymal stem cell (MSC) recruitment, poor vascularization, and insufficient growth factor stimulation. To achieve successful bone regeneration, enhancing MSC adhesion, growth factor release, and the angiogenic potential of biomaterial scaffolds is crucial.
Beta-tricalcium phosphate (β-TCP) is highly regarded by orthopedic surgeons for its biocompatibility and bioactivity in bone repair. 86 It has been shown to significantly enhance bone regeneration compared to nanostructured carbon implants, porous titanium, and even bone autografts. β-TCP scaffolds have been optimized by adjusting their physical properties, adding ionic components, and incorporating growth factors to improve bone healing. The release of Ca2+ from β-TCP is essential for MSC and osteoblast proliferation and differentiation, and it may influence blood clot structure at fracture sites, affecting bone regeneration. 87 The porous surface of bone materials improves mechanical interlocking and stability with surrounding bone tissues. 88 Porosity allows for the absorption of growth factors and enhances interactions with pro-osteogenic factors, 89 which positively impacts bone formation. 90 Porous β-TCP also enhances dissolution, MSC infiltration, and new bone formation. 91 Figure 1 illustrates the effect of β-TCP on bone healing.

A diagram shows how β-TCP scaffolds aid bone healing. Macropores (>100 μm) allow entry of growth factors, MSCs, blood vessels, oxygen, and nutrients better than micropores (<5 μm). High porosity increases the scaffolds’ surface area, enhancing effectiveness. Rough surfaces boost MSC adhesion and proliferation, aiding bone regeneration. 91
High surface roughness in materials enhances bone-forming cell attachment and osteogenic activity, aiding osteointegration, whereas low roughness may impede it. For instance, a calcium phosphate-coated titanium alloy with 8 to 10 Ra showed better collagen adhesion than one with 2 to 3 Ra. β-TCP materials can be optimized for roughness and pore sizes to improve bone formation, protein adsorption, cell infiltration, and neovascularization. However, β-TCP’s rapid degradation rate can limit osteoblast migration and colonization, hindering bone healing. 92 To address this, β-TCP is often combined with other materials to form composite scaffolds with improved biomechanical properties. β-TCP influences bone repair by regulating growth factors, cytokines, and ions, promoting osteoblast differentiation, vascularization, and growth factor release, and affecting blood clot formation to enhance bone healing. The release of Ca2+ from β-TCP is essential for MSC and osteoblast proliferation and differentiation, as shown in Figure 2.
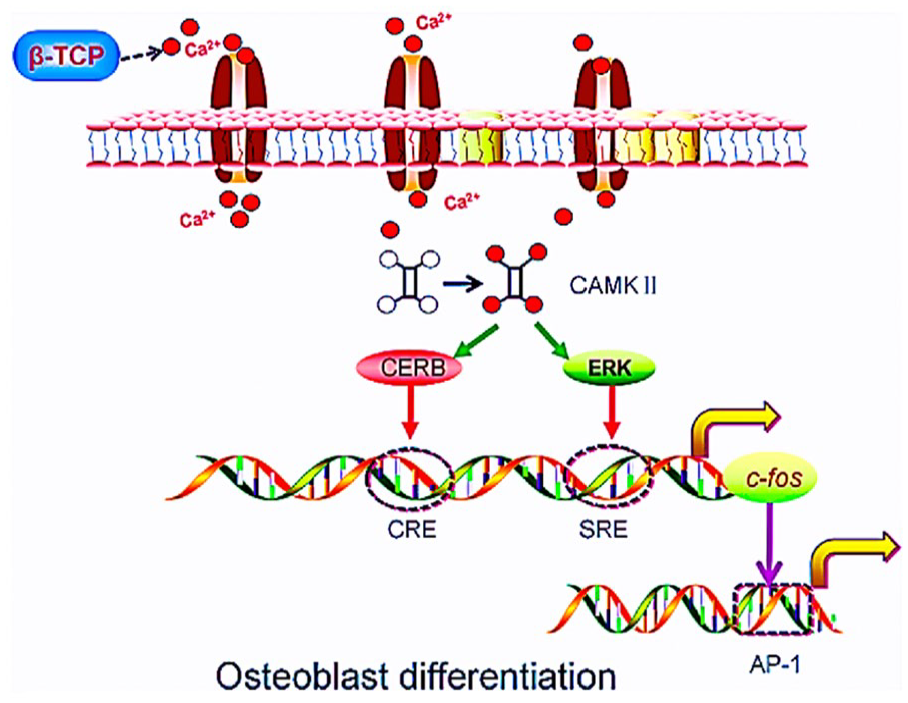
Ca2+ released from β-TCP activates the α-CaMKII pathway, modulating CREB and extracellular ERK activity. This activation increases the transactivation of SRE and CRE, which regulate the c-fos promoter, leading to higher expression of AP-1 and promoting osteoblast differentiation. 92
Studies indicate that incorporating growth factors like the BMP family into scaffolds enhances their biological performance. 94 β-TCP is believed to improve bone regeneration by interacting with growth factors such as BMPs, which are crucial for cell migration, proliferation, tissue angiogenesis, and new bone formation at defect sites. Growth factors play a significant role in regulating angiogenesis during bone formation (Figure 3). Vascular endothelial growth factor (VEGF) regulates osteogenesis through the PI3K/AKT, Raf-MEK-ERK, and PLCγ-IP3 signaling pathways after binding to VEGFR2. Platelet-derived growth factor (PDGF) promotes angiogenesis by activating PDGFR-αα, PDGFR-αβ, and PDGFR-ββ receptors. Ca2+ released from β-TCP binds to calmodulin (CaM), enhancing endothelial cell proliferation and facilitating neovascularization and bone healing. Transforming growth factor beta (TGF-β) induces osteogenic gene expression through the SMAD signaling pathway after binding to TGFR2. Bone morphogenetic proteins (BMPs) promote osteogenic gene expression via BMPR and the activation of SMAD and p38-MAPK signaling pathways. 93

Role of growth factors in regulating angiogenesis during bone formation. 93
After implantation at a fracture site, substitute materials interact with peripheral blood, forming a blood clot around the graft. 95 This clot acts as a “natural scaffold,” activated by the extrinsic coagulation pathway during bone fractures. 96 Coarse, loose fibrin clots have increased roughness and porosity, making them more susceptible to fibrinolysis, which aids bone tissue repair and regeneration. 41 In contrast, dense clots with thin fibrin are resistant to fibrinolysis, delaying bone regeneration. 97 Fibrin structure impacts bone healing, as fibrinolysis releases growth factors essential for cell growth.97,98,108 Tight fibrin networks slow bone healing due to low porosity and delayed degradation, whereas β-TCP enhances bone regeneration by interacting with growth factors.15,55,56 Figure 4 illustrates fibrin network formation and β-TCP regulatory sites. Thrombin catalyzes the binding of monomeric fibrinogen to polymeric fibrin by releasing fibrinopeptides (Fp) A (light red arrows) and FpB (blue arrows) from fibrinogen. FpA-derived fibrin monomers aggregate to form fibrin oligomers, which then polymerize into protofibrils. These protofibrils undergo lateral polymerization via intermolecular interactions at αC regions, forming αC polymers (black dashed circles) and resulting in thick fibers. The cross-linked fibrin clot, a gel-like meshwork, is crucial for hemostasis. β-TCP may regulate fibrin polymerization at FpB cleavage sites and the primary binding sites (black arrowheads) of the fibrinogen γ chain (orange).

Polymeric fibrin network formation and β-TCP regulatory sites. 98
Limitations and Remedy for Tricalcium Phosphate
While TCP, especially β-TCP, offers significant benefits for biomedical applications due to its biocompatibility, osteoconductivity, and resorbability, it also has limitations such as mechanical weakness, fast resorption rate, brittleness, and handling difficulties as presented in Table 3. Advances in composite materials and formulation techniques can help mitigate these drawbacks and enhance the utility of TCP in both internal and external applications. 99
Limitations and Remedy for Tricalcium Phosphate. 99

Advancements in Implant Development With Calcium-Based Materials
The development of implants using calcium-based materials, especially calcium phosphate (CaP) biomaterials, has greatly advanced bone regeneration. Due to their high biocompatibility, bioactivity, and resemblance to the mineral structure of human bone, these materials are well-suited for orthopedic and dental uses.102,103 A key advancement in implant technology is the application of calcium phosphate (CaP) coatings on titanium implants. Since these coatings closely resemble the mineral makeup of bone, they promote better integration with surrounding tissue. Research indicates that calcium phosphate coatings improve osseointegration, enhancing the stability and durability of both dental and orthopedic implants. 100 The release of calcium ions from these coatings into the surrounding tissue encourages the formation of biological apatite, essential for bone regeneration and integration. 104 Additionally, incorporating ions like fluoride and zinc into calcium phosphate coatings has shown antibacterial properties, which are critical in preventing infections after surgery. 105
Another important advancement is the development of bioactive glass ceramics and hydroxyapatite (HA) coatings. These materials offer a scaffold for bone growth and actively contribute to the biological processes essential for osseointegration. HA coatings, in particular, have been found to enhance early bone healing by replicating the structure of natural bone minerals. 106 Recent innovations feature calcium-doped titanium surfaces, which have been shown to enhance the adsorption of fibrinogen—a protein vital to the early stages of wound healing and implant integration. This technique not only helps reduce bacterial colonization but also creates a conducive environment for bone regeneration. 107
Furthermore, the mechanical properties of calcium phosphate ceramics have been optimized for load-bearing applications. Studies suggest that biphasic calcium phosphate (BCP) ceramics possess ideal solubility and osteoinductivity, making them highly suitable for bone grafts.108-111 The inclusion of calcium oxide in BCP ceramics has further improved both their mechanical and biological properties, enhancing their clinical utility.109-111 These materials demonstrate excellent biocompatibility and show promising outcomes in tissue integration and mechanical performance.112,113 The incorporation of calcium silicate into composite materials is being investigated to enhance the bioactivity and mechanical strength of dental implants which further expand the potential applications of calcium-based materials in implantology. 113 Thus, advancements in calcium phosphate biomaterials have tackled challenges related to immune responses and infection risks. The creation of bioactive calcium phosphate coatings has significantly reduced the chances of post-implantation inflammation, as these materials do not produce excessive reactive oxygen species.114,115 This biocompatibility is essential for the successful implementation of implants in clinical settings.
Future Outlook
Tricalcium phosphate (TCP) is a prominent material in the biomedical field, particularly in bone repair and regeneration. Recent advancements have focused on the development of ecologically inclined TCP-based materials, emphasizing sustainable practices in their production and application. This review explores the relevance, processing techniques, and future applications of these materials. Tricalcium phosphate (TCP) is increasingly recognized for its pivotal role in biomedical applications, particularly in bone repair and regeneration, due to its biocompatibility, bioactivity, and resorbability. Current research focuses on enhancing these properties through the development of ecologically inclined TCP materials, which prioritize sustainability throughout their lifecycle. This includes sourcing raw materials from biogenic sources, employing eco-friendly synthesis methods, utilizing additive manufacturing techniques, and implementing surface modifications that enhance osteointegration and antimicrobial properties. Applications of TCP span bone tissue engineering, dental implants, drug delivery systems, and orthopedic implants, with advancements in hybrid composites, porous carriers, and bioactive coatings driving innovation. Future prospects for TCP-based materials include the development of multi-functional composites, an increased focus on sustainability through life cycle assessments and circular economy principles, regulatory and clinical advancements to streamline approval processes, and market growth driven by heightened awareness and adoption of sustainable medical solutions. These efforts aim to address the dual goals of improving patient outcomes and promoting ecological health, positioning ecologically inclined TCP materials as a promising frontier in medical material science.
Conclusion
In conclusion, the integration of ecologically inclined practices in the development and application of tricalcium phosphate (TCP) materials holds significant promise for the biomedical field. By emphasizing sustainability from raw material sourcing to advanced processing techniques, and by exploring innovative applications in bone tissue engineering, dental implants, drug delivery systems, and orthopedic implants, TCP-based materials are set to transform medical treatments while mitigating environmental impact. The future outlook is bright, with potential advancements in material properties, regulatory support, and market adoption driving the evolution of these materials. As researchers, industry leaders, and regulatory bodies continue to collaborate, ecologically inclined TCP materials are poised to become a cornerstone of sustainable and effective biomedical solutions, improving both patient outcomes and ecological health.
Footnotes
Author Contributions
Isiaka Oluwole Oladele - Conceptualization and Supervision, Samson Ademola Adekola - Research and Writing, Newton Itua Agbeboh – Review, Corrections and Correspondence, Baraka Abiodun Makinde-Isola - Research and Writing Benjamin Omotayo Adewuyi - Leadership Supervision and Management.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
