Abstract
Background
The paddle lead (PL) and cylindrical lead (CL) remain the main implant categories in spinal cord stimulation (SCS) for treating neuropathic pain. Surgeons often complain about the greater trauma associated with PL implantation, while percutaneous endoscopic technique offers a promising approach for minimizing the trauma associated to PL implantation. However, there remains a dearth of real-world case study on endoscopy-assisted CL implantation.
Purpose
This study aimed to demonstrate the endoscopic-assisted approach and outcomes of CL implantation in SCS for managing neuropathic pain.
Research Design
A retrospective case series.
Study Sample
Patients aged 18 years and above with chronic neuropathic pain persisting for at least three months, refractory to standardized conservative treatment, were enrolled between January 2021 and March 2023.
Data Collection and Analysis
The surgical key steps including puncture, working cannula placement, endoscopic laminotomy and endoscopic CL introduction were demonstrated. Characteristics as demographics, follow-up time, visual analog scale (VAS) score, pain disability index (PDI) score and patient-reported outcomes measurement information system (PROMIS) scale were assessed.
Results
Successful CL implantation under endoscopy was achieved in all patients, including 3 with failed back surgery syndrome, 2 with complex regional pain syndrome and 2 with chronic pelvic pain. No spinal cord injuries, dural tears, lead migration, lead fractures, or postoperative infections were observed. VAS score of regional pain, PDI score as well as PROMIS of patient’s quality of life were all significantly improved after surgery.
Conclusion
Percutaneous endoscope-assisted CL implantation offered a new alternative technique for SCS in managing neuropathic pain.
Keywords
Introduction
Neuropathic pain remains a debilitating condition causing suffering and disability for millions of people worldwide. Spinal cord stimulation (SCS) has been widely employed for treating various pain conditions affecting trunk and limbs. 1 It works by delivering electrical pulses to leads placed on the dorsal column of the spinal cord, which modulate neural activity to reduce the sensation of pain reaching the brain. 2
Currently, paddle leads (PL) and cylindrical leads (CL) are the 2 dominant types of leads used for SCS. The advantages of implanting the paddle lead (PL) include better paresthesia coverage and a lower lead migration rate. 3 However, notable drawbacks include the requirement for a larger skin incision, paraspinal muscle dissection, and laminectomy/laminotomy, resulting in greater invasiveness. 4 Specifically, laminectomy or laminotomy requires excising a portion of the vertebral lamina, a process associated with significant risks, including iatrogenic spinal instability. This instability has been identified as a potential contributor in the manifestation of persistent postoperative back pain and the development of adjacent segment disease. 5
In contrast, percutaneous implantation of cylindrical lead (CL) has been identified by majority pain physicians as the first-line option when performing SCS. 6 As a minimally invasive technique, percutaneous CL implantation demonstrates effective outcomes while also maintaining a lower incidence of lead fractures. 3 However, because CL is made of flexible plastic, it is susceptible to deflection when encountering obstructions along the insertion path. 7 Consequently, successful CL implantation may sometimes be impeded by inherent anatomical constraints such as a narrowed interlaminar foramen, impinged osteophytes, hypertrophic ligamentum flavum, or scoliosis.7,8 The open access of PL implantation thus becomes necessary as a salvage modality. Nevertheless, to the best of our knowledge, there is scarce research in the literature regarding strategies to facilitate the CL implantation.
The evolution of percutaneous endoscopic techniques and their integration into minimal invasive surgery offer a promising tool for CL implantation. Endoscope-assisted CL implantation presented as an innovative approach for mitigating the trauma associated with open access while allowing for flexible lead placement using a rigid working-channel endoscope under direct visualization. 9 However, after searching PubMed, Web of Science, and Cochrane, no relevant technical reports on real-world case studies were identified. In this vein, this paper aimed to provide a detailed surgical description, shedding light on the practical aspects and outcomes of endoscope-assisted CL implantation for SCS in managing neuropathic pain.
Methods
General information
This study was performed accoding to the principles of the Declaration of Helsinki, and approved by the Ethics Committee of the local hospital (No.2021073205). All subjects provided their informed consent to participate in this study and all participants consented to publication if any identifying information is included in the manuscript. Between January 2021 and March 2023, enrolled patients in our department were older than 18 years old and experienced chronic neuropathic pain lasting for at least 3 months, despite undergoing standardized conservative treatment.
Illustration cases
The schematic diagram of this technique was illustrated as Figure 1. The 2 patients presented below were shown for elaborating surgical procedures. The first case was a 72-year-old male diagnosed with failed back surgery syndrome (FBSS) secondary to a lumbar internal fixation surgery for L3-L5 disc herniation. The second case was a 54-year-old male also diagnosed with FBSS secondary to a lumbar internal fixation surgery for L2 compression fracture. The chronic pain of lower limb and foot was refractory to conservative therapy for more than 3 months. The schematic illustration of percutaneous endoscopic cylindrical lead implantation for spinal cord stimulation. The yellow pillar indicated as the endoscopy; the red line indicated as the implanted cylindrical lead; the purple tube indicated as the puncture needle.
Operative procedure
Case 1
This patient has been experiencing consistent pain and a burning sensation in his lower limbs for the past 2 years, with a daily average visual analog scale (VAS) score of 7 out of 10. The preoperative CT scans revealed a complete obstruction of interlaminar foramina and ossification of the ligamentum flavum at T11-L2 levels (Figure 2(A) and (B)). He was positioned in a prone posture on a fluoroscopic operating table. A mobile C-arm X-Ray generator (Ziehm Solo FD, Ziehm Imaging GmbH, Germany) was used to conduct the spine anteroposterior (AP) and lateral fluoroscopy to identify the target spinal segment. Under fluoroscopic guidance, the target level (L1/2 in this illustration case) and the puncture point were determined. After that, 10 mL of 0.5% lidocaine was injected from the skin at the puncture point deep into muscle for local anesthesia. An 18-gauge (18G) percutaneous puncture needle was introduced at the bony prominence on the right side of L1/2 interlaminar foramen. After confirming the puncture position through several AP X-rays (Figure 2(C)), a scalpel was used to make a skin incision of around 0.8 cm (Figure 2(D)). Dilators of varying sizes were systematically introduced over the puncture needle until a working cannula was smoothly established (Figure 2(E)–(H)). Once fluoroscopic inspection confirmed the successful insertion of the working cannula into the target area, a spinal endoscope (joimax® GmbH, Karlsruhe, Germany) was inserted into the cannula (Figure 2(I)). The endoscope was connected to a light source and camera (Karl Storz Endoscopy Stack System, Tuttlingen, Germany), as well as a pump that for continuous saline flushing. Key surgical procedures of percutaneous endoscopic cylindrical lead implantation for spinal cord stimulation. Preoperative anterior view of lumbar CT 3D reconstruction with the red arrow indicating the narrowed targeted L1/2 interlaminar foramina (A) and CT axial view indicated ossification of the ligamentum flavum (B); intraoperative position for needle puncture (C) and skin incision for further cannulation (D); determine the cannula position (E) and insert the cannula (F); locate the inferior edge of L1/2 interlaminar foramen in AP (G) and lateral (H) view of radiographs; the endoscopic laminotomy procedure (I and J); the endoscopic lead introduction (K and L demonstrated needle puncture and visualization; M and N shown the first lead introduction while O and P shown the second lead introduction); the status when completing the leads insertion under endoscopic guidance (Q, R, S) and anchoring the leads to the skin (T). (CT=computed tomography; AP = anteroposterior).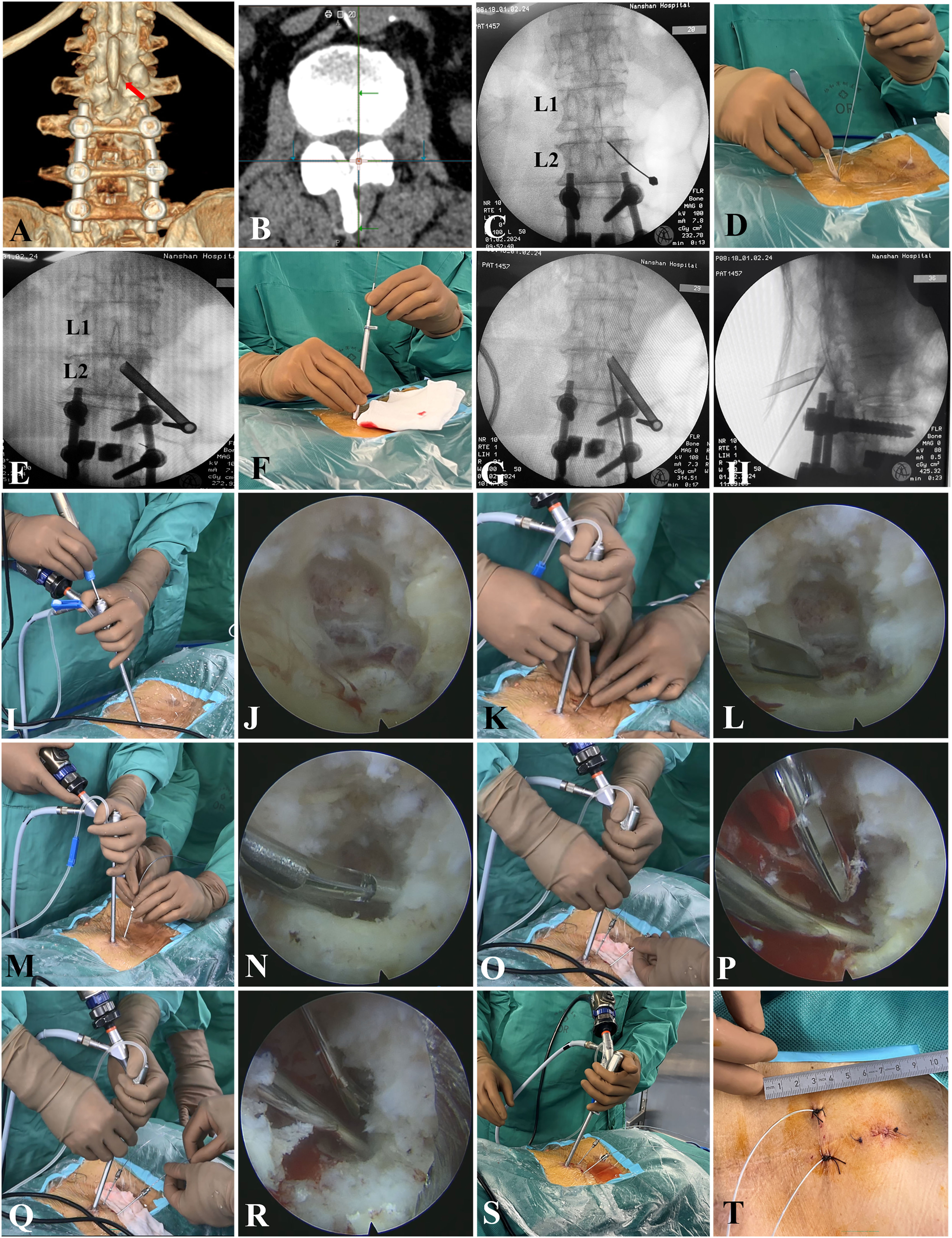
Soft tissue was meticulously excised using punch forceps and Kerrison rongeurs under precise endoscopic guidance so that the target bony prominence which located at the medial junction of the inferior facet joint and the superior facet joint were fully exposed. A 3.5-mm-diameter abrasive head together with a Kerrison punch as well as an ultrasonic bone curette were employed to carefully grind down the bony prominence and enlarge the interlaminar foramen (laminotomy). The patient presented with severe ossification of the ligamentum flavum, making inserting CL challenging. As a result, a Kerrison punch and a radiofrequency ablation tip were used to subsequently remove the ligamentum flavum, facilitating further lead insertion (Figure 2(J)).
At 3 cm caudally from the working channel, 10 mL of 0.5% lidocaine was administered 1 cm laterally to the right of the spine midline, for local anesthesia. An 18 G percutaneous puncture needle was deployed in a slightly cephalad direction (Figure 2(K)). The needle under the guidance of fluoroscopy was slowly advanced. At this point, carefuladjustment of the depth and direction of the puncture needle were made under direct endoscopic visualization (Figure 2(L)).
An 8-contact CL (Intellis™, Medtronic Inc, Minnesota, USA) was inserted through the puncture needle after removing the stylet (Figure 2(M)). The implant depth was confirmed by fluoroscopy as the lead was advanced (Figure 2(N)). This process continued until the lead was slightly shifted off the midline to the right, with its tip at the T7-T10 epidural space for achieving better lower limb stimulation 10 (placed at T9 for this case). An external pulse generator was connected and the stimulation mode, including frequency, amplitude as well as pulse width were programmed to ensure the adequate stimulation based on the patient’s self-reported paresthesia. This patient was finally configured to receive electrical pulses at a frequency of 60 Hz, with an amplitude of 1.7 mA and a pulse width of 350 µs. The puncture needle was then withdrawn, and the lead was secured to the nearby skin.
To provide concordant paresthesia coverage and avoid as much as possible the risk of lead migration, an additional CL (identical to the previous 1) was placed 3 cm caudal to the working cannula and 1 cm lateral to the left of the spine midline in the same fashion (Figure 2(O) and (P)). After repeating the above programing procedure successfully (Figure 2(Q)–(S)), the puncture needle was also withdrawn, and the lead was secured to the nearby skin.
Bleeding sites were carefully detected and addressed using a radiofrequency ablation tip under the endoscopic field. The endoscope and working cannula were finally extracted and the incision was sutured (Figure 2(T)). Intraoperative and postoperative X-rays and CT scans were performed for evaluating leads position, as depicted in Figure 3. Evaluation of the cylindrical lead position following percutaneous endoscopic implantation. Intraoperative fluoroscopy positioning of implanted lead (A); Postoperative CT 3D reconstruction image (B), AP X-ray image (C), coronal (D) and lateral CT views (E) for evaluating lead position. (CT=computed tomography; AP = anteroposterior).
Case 2
This patient had a burst fracture of the L2 vertebra accompanied by a cauda equina injury. He underwent open reduction and internal fixation 1 year after the initial injury, followed by removal of the internal fixation 2 years later. The primary symptoms reported by the patient including needle-like and cutting pain, as well as cold sensation in the left calf, sole of the foot, and dorsum of the foot. Pain intensity, assessed by VAS, reached 9-10 in the sole and remained at a VAS score of 7 in the dorsum of the foot. The preoperative CT scan of the case shown a bony obstruction of interlaminar foramina and at the target T12-L1 puncture level (Figure 4(A) and (B)). Illustration of endoscope-assisted cylindrical lead implantation with preservation of the ligamentum flavum. The preoperative CT images shown bony obstruction of the target interlaminar foramina (A) without ligamentum flavum ossification (B); intraoperative fluoroscopy assisted needle puncture and working channel placement (C); needle puncture under endoscopic visualization (D); introducing the cylindrical lead under endoscopic guidance (E); inserting of the lead until planned location was reached (F); the postoperative CT confirmed that the lead placement was satisfactory (G-I).
The needle entry-point determination (Figure 4(C)) and working channel establishment were similar to the procedures described in the above case. The ligamentum flavum was meticulously preserved during the laminotomy. The lead was introduced under endoscopic visualization and penetrated through the ligamentum flavum until the target site of stimulation contact was reached (placed at T8 for this case), as presented in Figure 4(D)–(F). The remaining procedures were also identical to those described in the case mentioned above (except the lead used was ProclaimTM, Abbott Medical, TX, USA). Postoperative CT scan was performed for evaluating leads position, as depicted in Figure 4(G)–(I).
Outcome measures
Primary outcome measures assessed including pre- and postoperative VAS score for back, leg and pelvic pain,11,12 pain disability index (PDI) score 13 and patient-reported outcomes measurement information system (PROMIS) scale 14 were collected. Patients were followed, postoperative X-ray or CT were performed as appropriate.
Statistical analysis
Data were collected from baseline and follow-ups. The numerical data was presented as mean ± standard diviation (SD). The SPSS 27.0 (SPSS, Inc, Chicago, IL, USA) was used to performed the statistical analysis. If the data follow a normal distribution, t test was used to compare the VAS and PDI scores at 3 months, 6 months, and 12 months with the baseline. Conversely, if the data do not meet the normality assumption, the Wilcoxon signed-rank test was conducted. P < 0.05 was considered statistically significant.
Results
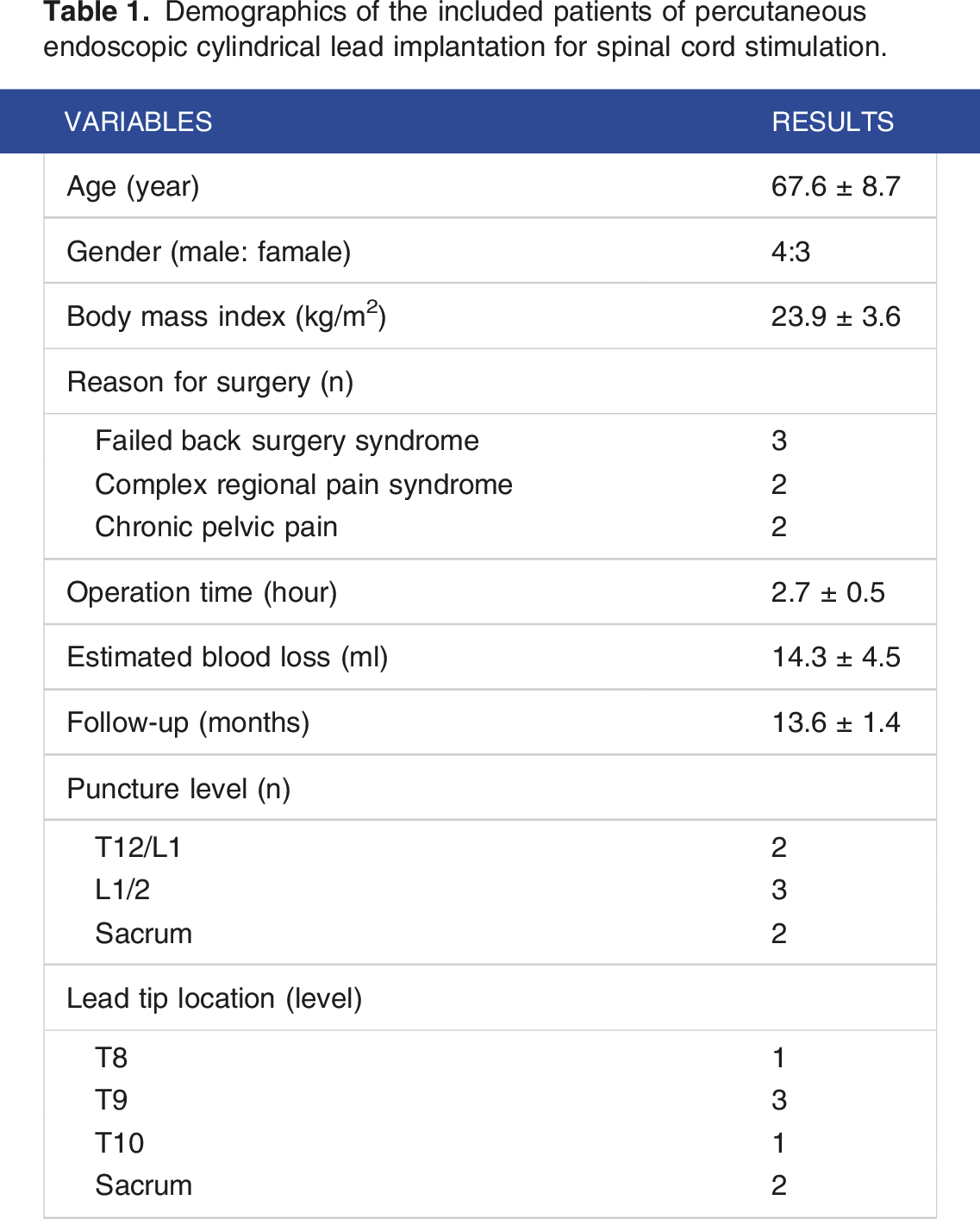
Demographics of the included patients of percutaneous endoscopic cylindrical lead implantation for spinal cord stimulation.
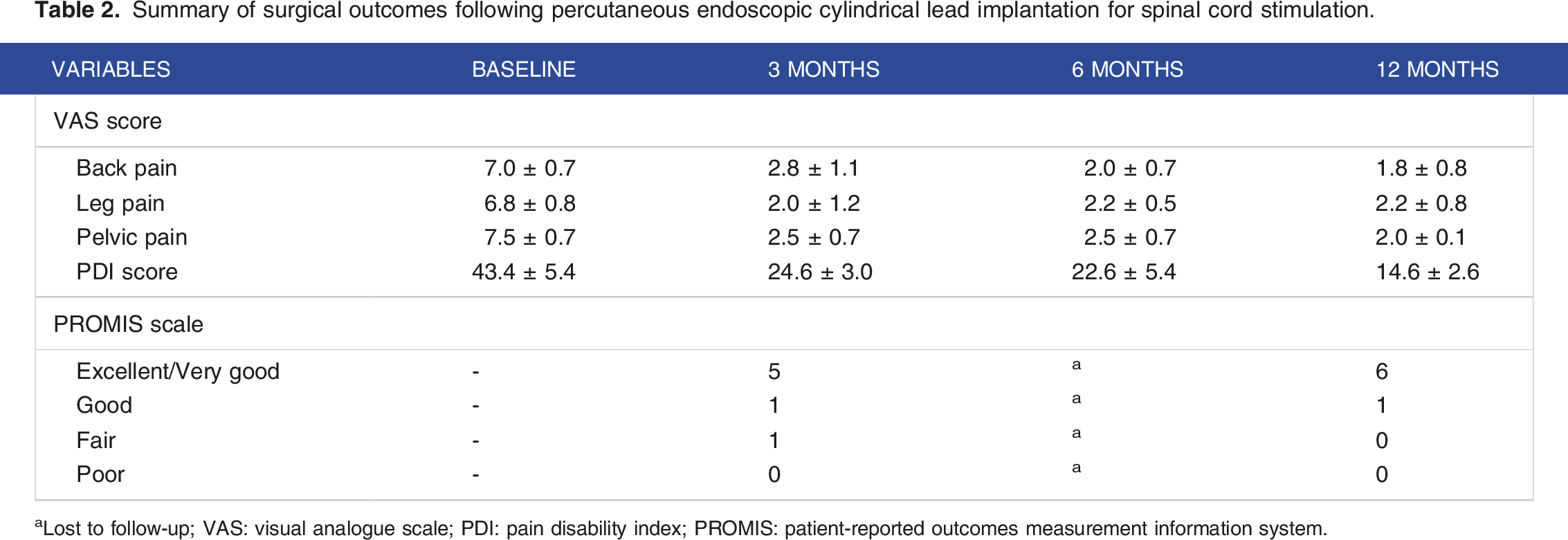
Patients were subsequently followed up for an average duration of 13.6 ± 1.4 months. Complications such as spinal cord injuries, dural tears, lead migration, lead fractures, and spinal cord infections were not observed. One patient experienced postoperative incisional pain, which was effectively alleviated with intramuscular tramadol (100 mg twice daily for 2 days). As presented in Figure 5 and Table 2, VAS score of leg, back and pelvic pain, as well as PDI scores were all significantly improved at all follow-up stagescompared to baseline (P < 0.05). The PROMIS scale yielded 71.4% of patients reporting excellent or very good quality of life at the 3-month follow-up, increasing to 85.7% at the 12-month follow-up. Outcome assessment of pain, functional capacity, and quality of life after endoscopic cylindrical lead implantation. Mean VAS (A) and PDI (B) scores were recorded from baseline to 12 months follow-up; The PROMIS scale was assessed at the 3 (C) and 12-month (D) follow-up. Summary of surgical outcomes following percutaneous endoscopic cylindrical lead implantation for spinal cord stimulation. aLost to follow-up; VAS: visual analogue scale; PDI: pain disability index; PROMIS: patient-reported outcomes measurement information system.
Discussion
This technical report represents the first demonstration of the surgical procedure and the clinical significance of percutaneous endoscope-assisted CL implantation for SCS in managing neuropathic pain. All enrolled patients achieved satisfactory outcomes in terms of pain relief, pain-related disability, and health-related quality of life over an 1-year follow-up period.
In the existing literature, there have been extensive discussions regarding the selection between PL and CL for SCS.3,6,9,15,16 While there is no consensus indicating 1 method to be superior over the other in treating neuropathic pain, a growing body of research suggests that percutaneous CL offers reduced invasiveness compared to PL, facilitating also easier implantation and removal of leads.3,15,17,18 However, the challenge lies in the fact that the percutaneous approach often constrained by inherent anatomical structures, such as narrow interlaminar spaces and bony obstructions. 7 The employ of endoscopic-assisted percutaneous CL implantation allows for easier laminotomy in the targeted interlaminar space. Furthermore, under direct endoscopic visualization, partial scar tissue or adhesions within the spinal canal can be excised, 19 thereby facilitating lead manipulation within the spinal canal. For case 1 demonstrated in this study, we removed the ossified ligamentum flavum under direct endoscope visualization and successfully placed the lead.
Despite these advancements, there is currently a scarcity of literature concerning achieving smoother and less invasive implantation of CL. Li et al 9 performed an anatomical study using 4 fresh adult cadavers and demonstrated the feasibility of implanting surgical PL in the thoracic and cervical segments using a unilateral biportal endoscopic technique, offering a minimally invasive alternative for SCS. However, the operation of cadavers has rendered its efficacy in live patients unknown. MacDonald et al 7 demonstrated a novel surgical technique using a disposable malleable suture retrieval snare to precisely position PL during initial placement or revision, aiming to optimize stimulation coverage for chronic pain management. The authors noted that employing an absorbable suture could prevent potential fixation points that might impede electrode revision or removal. However, they also highlighted the ongoing risk of neurologic injury associated with the bulk of paddle-style leads.
This study has several limitations. Firstly, it included a small sample size and a short follow-up period, necessitating larger prospective controlled studies for more conclusive evidence. However, the primary objective of this study was to offer a comprehensive surgical procedural guide for endoscopic-assisted CL implantation and to demonstrate the feasibility of this technique. The preliminary findings of this study might pave the way for future clinical uses. Secondly, this technique relied on the surgical skill level of the operating physician, particularly when performing laminotomy under endoscopic guidance. Therefore, this challenge might pose a significant limitation to junior practitioners with little experience (Supplemental Material).
Conclusions
In this study, we elaborated on a surgical procedure using a single endoscope that achieved laminotomy and facilitated smooth CL introduction, and demonstrated minimal invasiveness as well as favorable patient outcomes. Laminotomy can be performed at interlaminar foramen of targeted segment under the assistance of endoscopy, which significantly expanded the adaptability of percutaneous CL implantation.
Supplemental Material
Footnotes
Author contributions
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded by the Science, Technology, and Innovation Commission of Shenzhen Municipality (Grant No. JCYJ20230807115918039) from Zhouyang Hu; the Nanshan District Health Science and Technology Major Project (Grant No. NSZD2023026; NSZD2023023) from Zhouyang Hu and Guoxin Fan; Nanshan District Health Science and Technology Project (Grant No. NS2023002; NS2023044) from Xiang Liao and Guoxin Fan; the National Natural Science Foundation of China (Grant No. 82102640) from Guoxin Fan, the National Key Research and Development Program of China (Grant No. 2022YFC3602203) from Xiang Liao, the Medical Scientific Research Foundation of Guangdong Province of China (Grant No. A2023195) from Guoxin Fan.
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
