Abstract
Background
Blebs are small bulges on the surface of intracranial aneurysms (IAs) that increase rupture risk. Among Chinese individuals, the prevalence, distribution, and clinical, geometric, and local hemodynamic characteristics of small- and medium-sized (<15 mm) blebbed IAs remain unclear.
Objectives
To investigate the prevalence, distribution, and associated clinical, geometric, and hemodynamic features of blebs in small- and medium-sized IAs among Chinese patients, and to identify predictors of aneurysm rupture and bleb formation.
Design
A retrospective observational study.
Methods
CTA or DSA data from 214 patients with ruptured (RIAs) and unruptured IAs (UIAs) (<15 mm), with or without blebs, were analyzed. Three-dimensional reconstruction, geometric measurement, and computational fluid dynamics (CFD) analysis were conducted using Mimics and ANSYS Fluent. Hemodynamic parameters were assessed across the neck, body, and dome, and logistic regression was used to identify predictors of rupture and bleb formation.
Results
Aneurysms from 214 patients (93 men, 121 women; mean age 59.90 ± 11.76 years) were analyzed. Blebs were found in 107 aneurysms (56.7% of RIAs, 39.1% of UIAs). They were more frequent in ACoA, PCoA, and bifurcation aneurysms (all P < 0.05) and were associated with rupture. RIAs had larger blebs than UIAs (2.73 ± 1.28 mm vs 2.06 ± 1.07 mm, P = 0.009). Blebbed IAs exhibited larger size, more irregular shape, higher AR, SR, BN, HWR, and lower NWSS, TAWSS, OSI (all P < 0.05). SR was the strongest rupture predictor (AUC = 0.718, SR > 1.3144). Aneurysms at ACoA (OR = 8.812,CI:2.455-31.634), PCoA (OR = 6.376,CI:2.094-19.414), and high SR (OR = 2.738,CI:0.98-7.651) were significant rupture risk factors. PCoA (OR = 2.261,CI:0.759-6.739) and SR (OR = 4.683,CI:1.937-11.324) independently predicted bleb formation.
Conclusion
Blebs are common in small- and medium-sized IAs, especially at the ACoA, PCoA, and bifurcations, and are associated with an increased risk of rupture. Larger blebs are more often seen in ruptured IAs. A high SR is a key predictor of both rupture and bleb formation. Further studies on bleb-related hemodynamics in the neck, body, and dome are warranted.
Keywords
Introduction
Intracranial aneurysm (IA) is a common cerebrovascular lesion with potentially fatal risks, with a prevalence of approximately 3% in the general population. Most IAs remain asymptomatic; however, an estimated 0.2%-2.4% rupture annually, leading to subarachnoid hemorrhage (SAH).1,2 Aneurysmal SAH is a severe and life-threatening condition characterized by high mortality and significant disability rates, making it a major threat to patient health.1,2 Therefore, identifying high-risk aneurysms and implementing early clinical interventions are of great clinical significance.1,2
Previous studies have demonstrated that secondary saccular protrusions on the aneurysmal wall, known as “Blebs” or daughter sacs, are closely associated with aneurysm instability.3,4 Blebs are widely regarded as key indicators of irregular aneurysm morphology and vascular wall instability.3,5,6 Their presence is significantly correlated with an increased risk of rupture.1,2,5 However, the underlying mechanisms of bleb formation and their role in aneurysm rupture risk assessment remain incompletely understood.3,7,8 In particular, there is no consensus regarding their geometric characteristics and hemodynamic mechanisms.3,7,8 Furthermore, most existing aneurysm risk models and research data are derived from clinical samples of Western or Japanese populations, raising concerns about their applicability to other populations, especially the Chinese Han population.1,9
Notably, epidemiological data suggest significant regional differences in aneurysm rupture risk. In particular, ruptured aneurysms in the Chinese Han population exhibit distinct clinical characteristics compared to Western and Japanese patients.1,2,9 For instance, Chinese patients often present with aneurysm rupture at a smaller diameter (≤5 mm).3,9 This observation underscores the necessity for population-specific research to enhance the accuracy of rupture risk prediction and improve clinical prevention and treatment strategies.1,4,9 However, there is currently a lack of comprehensive studies on the geometric features and hemodynamics of aneurysmal blebs in the Han population. Most existing studies have been limited by small sample sizes, resulting in insufficient evidence.7,9
This study aims to address this research gap by analyzing the morphological and hemodynamic characteristics of intracranial aneurysms in the Chinese Han population and elucidating the relationship between aneurysmal blebs and rupture risk.4,8-10 Specifically, this study will compare the geometric parameters (eg, morphological complexity, size, and bleb distribution)3,5,6,11,12 and hemodynamic features (eg, wall shear stress and flow patterns)4,7,8,13,14 between aneurysms with and without blebs. We hypothesize that aneurysms with blebs exhibit significant differences in geometric structure and hemodynamic characteristics, which may be closely related to their susceptibility to rupture. The findings of this study are expected to provide valuable insights for optimizing rupture risk assessment and individualized clinical decision-making for Chinese Han patients with intracranial aneurysms.
Methods
Data
The Clinical and Geometric Characteristics of Ruptured and Unruptured Aneurysms With and Without Blebs

Study Flow Diagram and Graphical Abstract of the Study (A) Study Flow Diagram Explaining the Steps of Our Analysis. AVF: Arteriovenous Fistula; AVM: Arteriovenous Malformation; MMD: Moyamoya Disease; IA: Intracranial Aneurysm; RIA: Ruptured Intracranial Aneurysm; UIA: Unruptured Intracranial Aneurysm; CTA: Computed Tomography Angiography; DSA: Digital Subtraction Angiography; CFD: Computational Fluid Dynamics (B) Graphical Abstract of the Study
Bleb Identification and Geometric Parameters
Aneurysm blebs were defined as secondary outpouchings clearly distinct from the original aneurysm wall. They were identified by a noticeable change in surface curvature at the aneurysm neck.. We referred to Salimi’s study and divided IAs into three regions (nonsoftware partitioning method): the neck, body, and dome.
8
Two experienced neuroradiologists independently evaluated the presence and size of blebs on 3DRA or CTA images using Mimics Research 21.0 (Materialise, Belgium) (Figure 2). Although formal inter-rater agreement statistics (eg, Cohen’s kappa) were not calculated due to the retrospective nature of the dataset, all discrepancies were resolved through joint review. This approach has been supported by prior studies reporting substantial agreement for similar visual assessment protocols (κ > 0.70). Geometric parameters were also measured using Mimics and are defined in Table S2. 3D Reconstruction, Geometric Measurement, and IA Region Partitioning (A-D) Reconstruction of an IA in 3D With Mimics Research 21.0 (E) IA Geometric Measurement after 3D Reconstruction (F) Aneurysm Geometric Parameter Diagram. H: Height; L: Length; Wneck: Aneurysm Neck; α: Angle of Incidence; β: Aneurysm Angle. The Diameter of the Aneurysmal Vessel (Parent Vessel Average Diameter, Dv) Included the Vessel Diameter for Sidewall-type IAs (Diameter of the Aneurysmal Artery Near the Neck of the Aneurysm (D1) and the Proximal End of the Aneurysmal Artery From the Neck of the Aneurysm (1.5D2) of the Average Diameter of the Vessels) and Bifurcation-Type IAs (Average Diameter of the Aneurysmal Vessels and Their Branches) (G) Diagram Illustrating the Division of the IA Into the Neck, Body, and Dome, Showing the Neck, Body, and Distance Lines
Hemodynamic Modeling
The 3D aneurysm models generated using 3-matic Research 13.0 were imported into ICEM CFD 2021 R2 (ANSYS, USA). We used an unstructured tetrahedral mesh with a diameter of 0.4 mm, resulting in approximately 3 to 6 million grid elements. In the present study, it was assumed that blood is a Newtonian fluid, and energy transfer and the influence of gravity were not considered. A rigid IA wall was assumed to have no sliding boundaries, disregarding the effects of IA wall thickness and elasticity on hemodynamic parameters. The blood flow conditions at the inlet of the vessel were set on the basis of the pulse velocity measured by TCD (the average flow velocity at the entrance of the ICA for individuals aged 40 to 60 years measured by TCD was 0.92 ± 0.10 m/s) and the outlet pressure condition (no stress, ie, static pressure value 0). Hemodynamic analysis of the MESH vascular model was performed with ANSYS Fluent 2021 R2 software, in which the Navier Stokes equation governed the simulation process. This software was used to simulate three cardiac cycles and export the data of the last cardiac cycle after 800 stages (0.001 s per stage). The exported calculation results were saved in CDAT format for analysis, and the obtained image data were postprocessed via CFD Post 2021 R2 software (ANSYS 2021 R2).
Data Analysis
A contingency table and Pearson’s chi-square test were used to analyze associations between categorical variables and the presence of blebs unless the assumptions of the chi-square test could not be met; in those cases, Fisher’s exact test was used. In addition, the geometry of the aneurysms was characterized by computing variables that describe various aspects of their morphology, including the size of the aneurysm sac—length (L), height (H), width (W), and size ratio (SR)—and the width of the aneurysm neck—neck width (Wneck). The degree of elongation and shape irregularity were quantified using the aspect ratio (AR), bottleneck ratio (BN), and height-to-width ratio (HWR). The presence of an irregular shape was also noted (Table 1). For between-group comparisons, the t test was used for normally distributed data, and the Wilcoxon rank-sum test was used for nonnormally distributed data. On the basis of the maximum Youden index of the ROC curve, the optimal threshold was determined, and multiple logistic regression was performed on parameters with significant differences (P ≤ 0.001) in clinical and geometric aspects (Model a: risk of rupture; Model b: presence of bleb). Flow variables were calculated to assess the hemodynamic environment of the aneurysms, including normalized wall shear stress (NWSS), time-averaged wall shear stress (TAWSS), normalized TAWSS (TANWSS), and TAWSS gradient (TAWSSG). Other parameters included the oscillatory shear index (OSI), relative residence time (RRT), flow velocity (VE), and vorticity (VOR) (Table S2). The hemodynamic characteristics of the neck, body, and dome regions of aneurysms with and without blebs were compared with the nonparametric Wilcoxon (Mann‒Whitney U) test. As shown in Figure 2G, local area parameters were measured by randomly selecting multiple points near the neckline, body line, and dome point and then taking the mean of these points to represent the hemodynamic parameters of the corresponding area. The hemodynamic variables were then normalized by the average values over the parent artery. A post-hoc power analysis was conducted for subgroup comparisons to confirm statistical adequacy (Tables S4–S6, Figure S3). Logistic regression models adjusted for key clinical covariates were tested to validate robustness (Models 1-6; Table S7 and Figure S4). Multiple comparisons were corrected using Bonferroni and Benjamini-Hochberg methods (Tables S8–S10, Figure S5). All statistical analyses were carried out in SPSS l9.0 (SPSS, Inc., Chicago, Illinois) and R. P < 0.05 was considered to indicate statistical significance.
This study adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines. The completed STROBE checklist is provided as supplementary material.
Results
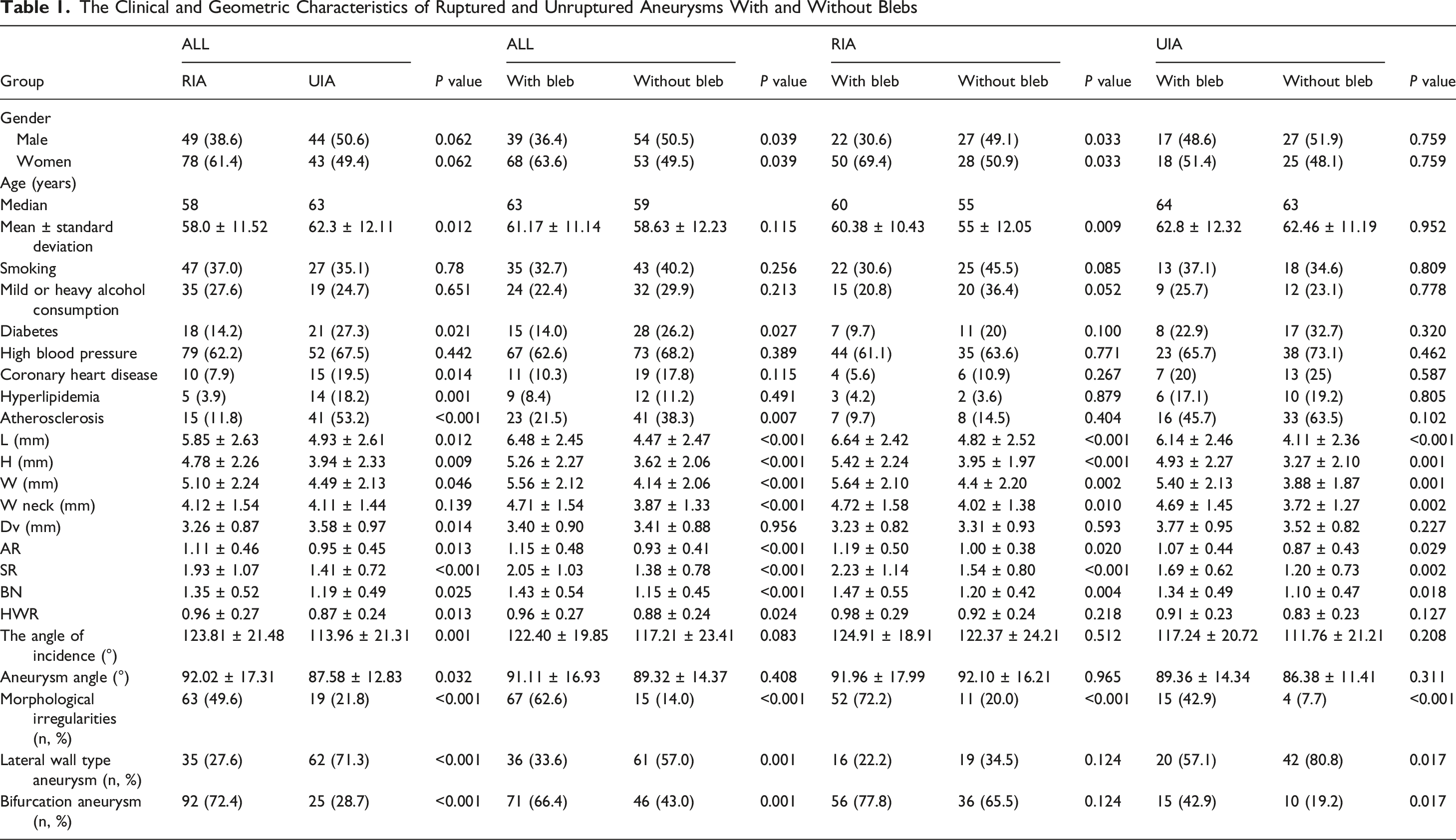
Bleb Distribution, Frequency, and Size
The Size and Positional Characteristics of Ruptured and Unruptured Aneurysms With and Without Blebs

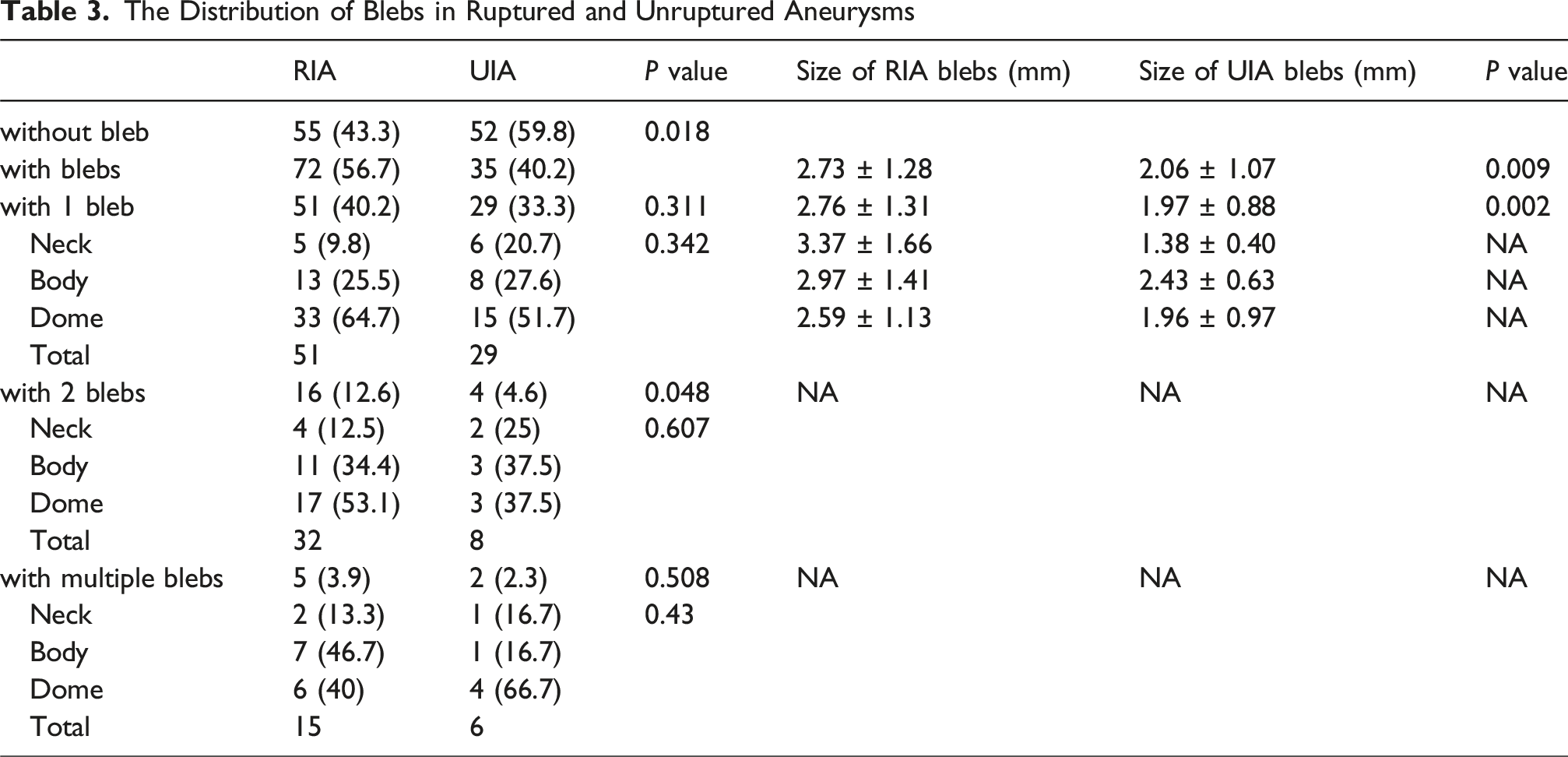
The Distribution of Blebs in Ruptured and Unruptured Aneurysms
Associations with the Presence and Geometry of Blebs
We compared the geometric differences between IAs with and without blebs and determined the risk factors among the geometric parameters for the presence of blebs and IA rupture with logistic regression. As shown in Table 1, there were significant differences between aneurysms with and without blebs. Specifically, age (P = 0.115), coronary heart disease history (P = 0.115), hypertension history (P = 0.389), drinking history (P = 0.651), and smoking history (P = 0.780) were not associated with the presence of blebs. Aneurysms with blebs were detected in a greater percentage of female patients (63.6%) than male patients (36.4%). Patients with IAs without blebs (38.3%) were more likely to have a history of atherosclerosis than those with IAs with blebs (21.5%) (P = 0.007). In addition, aneurysms located in certain anatomical sites tended to be more likely to have blebs. Aneurysms with blebs were most prevalent in the ACoA and PCoA (29.9%), followed by the MCA (15.0%), ICA (15.0%), and V-BA (6.5%), while the smallest proportion (3.7%) were found in the ACA. Aneurysms with blebs were more likely to occur at artery bifurcations than on the sidewall. In terms of geometry, aneurysms harboring blebs had greater L (P < 0.001), H (P < 0.001), W (P < 0.001), Wneck (P < 0.001), AR (P < 0.001), SR (P < 0.001), BN (P < 0.001), and HWR (P = 0.024) values and had more irregular shapes (P < 0.001). The numbers of RIAs and UIAs with single or multiple blebs are presented in Table 3. A more detailed distribution of blebs is presented according to aneurysm anatomy (neck, body, dome), location, and rupture status (RIA vs UIA) (Table S11).
We used ROC curve analysis and the maximum Youden index to determine the optimal threshold values for the predictor variables. As shown in Figure 3, the SR was the strongest predictor of blebs (SR > 1.3144, AUC = 0.718, 95% CI, 0.649-0.788; P = 0.001). According to Figure 3G, significant risk factors for rupture included location in the ACoA (OR = 8.812; 95% CI = 2.455-31.634) and PCoA (OR = 6.376; 95% CI = 2.094-19.414) and SR (OR = 2.738; 95% CI = 0.98-7.651). As shown in Figure 3H, logistic regression analysis revealed two independent risk factors for the presence of blebs: location in the PCoA (OR = 2.261; 95% CI = 0.759-6.739) and SR (OR = 4.683; 95% CI = 1.937-11.324). In Chinese individuals, blebs were common in small-, medium-, and high-SR IAs (especially RIAs) and in IAs located in the ACoA, PCoA, and bifurcations. ROC Curves and Logistic Regression Analysis (A-F) Comparison of the AUC Values of Independent Risk Factors for Rupture and the Presence of Blebs; (A-C) Risk Factors for Rupture; (D-F) Risk Factors for the Presence of Blebs. AR: Aspect Ratio; SR: Size Ratio; BN: Bottleneck Ratio; RIA: Ruptured Intracranial Aneurysm (G-H) Logistic Regression Forest Map (G) Association Between Risk Factors and Aneurysm Rupture (H) Risk Factors for the Presence of Blebs. ACoA: Anterior Communicating Artery; PCoA: Posterior Communicating Artery; ACA: Anterior Cerebral Artery; MCA: Middle Cerebral Artery; PcirA: Posterior Circulation Aneurysm; AR: Aspect Ratio; SR: Size Ratio; BN: Bottleneck Ratio; HWR: Height Width Ratio
Multivariate logistic regression sensitivity analyses confirmed that the size ratio (SR) remained an independent predictor for both aneurysm rupture and bleb formation, even after adjusting for key clinical covariates such as age, sex, hypertension, smoking, and diabetes. Higher SR values consistently showed strong associations with rupture risk (Models 1-3; adjusted OR range: 3.05-3.81, all P < 0.05) and bleb formation (Models 4-6; adjusted OR range: 5.59-7.26, all P < 0.001). Aneurysm location at ACoA and PCoA and bifurcation morphology were also significant independent predictors of aneurysm rupture across multiple adjusted models (Table S7).
Hemodynamics of Blebs in Different Regions
Hemodynamic Comparison of Aneurysms With and Without Bleb in the Neck, Body, and Dome Regions


(A-H) A Ruptured MCA Aneurysm With Two Blebs and a Length of 6.88 mm (A) Volume Rendering of a Three-Dimensional Rotational Angiography Image (B-C) Aneurysm Bleb1 and Bleb2 are Visible During Surgery (D) A Rupture Point is Identified Near Aneurysm Bleb2 During Surgery (E) Wall Shear Stress (WSS) Distribution (F) Mean Gradient Oscillatory Number (GON) Distribution (G) Mean WSS Gradient (TAWSSG) Distribution (H) Streamline Distribution (I∼N) Analysis of an Unruptured MCA Aneurysm With No Blebs and a Length of 5.56 mm at an Arterial Bifurcation (I) Volume Rendering of a Three-Dimensional Rotational Angiography Image (J-K) Blebs and the Neck, Body, and Dome of the Aneurysm are Not Visible During Surgery (L) WSS Distribution (M) Time-Averaged Wall Shear Stress (TAWSS) (N) Mean GON Distribution
Sample Size and Power Analysis
To further validate the adequacy of our sample size, we performed a comprehensive post-hoc power analysis for various subgroup comparisons. These included ruptured vs unruptured aneurysms and the presence vs absence of blebs (see Supplemental Tables S4–S6). The analysis demonstrated satisfactory statistical power (Power > 0.8) for most critical comparisons; for example, comparisons of aneurysm length (L), height (H), and width (W) between aneurysms with and without blebs approached or reached a power of 1.0. However, some subgroup analyses—particularly involving aneurysm locations within small subgroups and certain hemodynamic parameters—did not achieve adequate statistical power (Power < 0.8), suggesting these specific comparisons may be limited by insufficient sample size and warrant cautious interpretation (refer to Supplemental Tables S4-S6 and Supplemental Figure S3 for detailed results).
To account for multiple comparisons and control for Type I error, we applied Bonferroni and Benjamini-Hochberg (BH) corrections across all statistical tests performed (Tables S8–S10). After Bonferroni correction, most associations became non-significant due to its conservative nature, while BH correction revealed several consistently significant associations. Specifically, aneurysm morphology (length, height, AR, SR, morphological irregularities, lateral wall and bifurcation aneurysm), and hemodynamic parameters (especially NWSS, TANWSS, TAWSS, TAWSSG, and SSR at the aneurysm dome) remained significantly associated after correction (Tables S8, S10). Heatmaps illustrating adjusted P-values visually highlighted these significant findings, enhancing clarity (Figure S5).
Discussion
The aim of this study was to investigate the prevalence, geometric characteristics, and hemodynamic features of bleb aneurysms in the Chinese Han population to supplement existing studies on aneurysmal blebs. Previous clinical evidence suggests significant pathophysiological differences between small and large intracranial aneurysms (IAs). 9 An intraoperative study demonstrated that smaller aneurysms (7-10 mm) typically have thin and translucent walls, whereas larger aneurysms (12-20 mm) often exhibit thick, irregular, and opaque walls.9,15 Meng et al further proposed that different aneurysm sizes might exhibit distinct hemodynamic profiles and rupture mechanisms; specifically, smaller aneurysms could be associated with high WSS and positive WSS gradients, while larger aneurysms might correlate with low WSS and high OSI.9,16 Consequently, we hypothesized that aneurysm size might reflect differences in morphological, hemodynamic, and clinical characteristics, necessitating the exclusion of aneurysms larger than 15 mm from our analysis to ensure a homogenous study population focusing specifically on small- to medium-sized aneurysms.
In the present study, our post-hoc power analysis highlighted the limitations posed by sample size on certain subgroup comparisons. Although the overall sample size was deemed reasonable, insufficient statistical power (Power < 0.8) was identified in a few subgroup analyses. These findings indicate that future research should consider larger sample sizes to robustly confirm or refute the differences observed in these specific subgroups.
The sensitivity analyses demonstrated that the association between SR and aneurysm rupture and bleb formation remained significant after controlling for potential clinical confounders such as age, sex, hypertension, smoking, and diabetes. These findings underscore the robustness of SR as a crucial morphological indicator of rupture risk and bleb development, independent of clinical risk factors. Additionally, the adjusted models highlighted the role of aneurysm location (especially at ACoA and PCoA), bifurcation morphology, and smoking status, further emphasizing the multifactorial nature of aneurysm rupture pathogenesis.
Our rigorous application of multiple comparisons corrections strengthened the robustness of the study findings. While Bonferroni correction identified fewer significant results due to its stringent threshold, Benjamini-Hochberg correction provided a balanced approach, confirming key morphological and hemodynamic parameters as genuinely associated with aneurysm rupture and bleb formation. These findings underscore the importance of adjusting for multiple testing, thus reducing false-positive outcomes and enhancing the validity of our conclusions.
Bleb Prevalence and Clinical Factors
This study compared the location of blebs in IAs, the frequency of blebs, the distribution of blebs in the RIA and UIA groups, and the clinical baseline conditions of the IAs with blebs group and the IAs without blebs group. In the present study, 50% of patients had aneurysms with blebs (usually single blebs and rarely multiple blebs), and 14.2% of the blebs occurred in the neck region, while the majority occurred in the aneurysm dome or body. The findings of this study are consistent with previous work by Salimi et al. 17 Additionally, we made interesting observations regarding RIAs and UIAs with blebs. Approximately 43.3% of the RIAs in our database did not have blebs; 40.2% had one bleb, 12.6% had two blebs, and 3.9% had multiple blebs. Among all aneurysms with blebs, the prevalence of those with multiple blebs was 6.5%. Additionally, many UIAs were characterized by the presence of blebs, as 40.2% of the aneurysms with blebs did not rupture in our sample.
Smoking increases the risk of aneurysm growth and rupture.18-20 In a large-scale prospective population study, smoking status and female sex were closely associated with the risk of developing UIAs and SAH. 21 In this study, no significant differences in smoking history were observed between the two groups, likely due to selection bias or small sample sizes. Although smoking status and hormonal factors (indirectly assessed by gender) were included in the analysis, neither emerged as significant independent predictors, potentially reflecting demographic or ethnic differences. Future studies with larger samples and detailed hormonal assessments are warranted.
In the present study, aneurysms with blebs were more likely to occur in patients without a history of atherosclerosis and in females than in patients with atherosclerosis and males, respectively. In their fifth decade, women may be subject to hormonal changes during menopause, which may explain the greater incidence of cerebral aneurysms in female patients. 19 Salimi et al reported that the presence of blebs in IAs was associated with the presence of dental infections and the use of hormone replacement therapy. 17 However, in the present study, those variables were not analyzed because of a lack of relevant medical history data. Importantly, although the data on small- and medium-sized IAs (<15 mm) were obtained from the Chinese Han population (<15 mm), the clinical baseline values varied among the patients. We also confirmed the findings of Salimi et al, who reported that there was no significant difference in the prevalence of blebs between the Chinese Han population and European and American populations.
Bleb Distribution and Size
Studies have shown that the IAs in the ACoA and PCoA have greater risks of rupture than those at other sites do. 22 Size, location, the presence of blebs, and SAH history are independent predictors of aneurysm rupture. 23 Therefore, a better understanding of ruptured aneurysms and blebs would be beneficial. 17 Blebs have been reported to develop more frequently in patients with previously ruptured aneurysms and in large or elongated aneurysms, as well as in aneurysms located at certain locations, such as the ACoA and PCoA, although this observational design does not allow us to confirm direct causality. 8 According to our research, the ACoA, PCoA, and bifurcated arteries had more aneurysms with blebs than did the ICA. Additionally, we stratified all aneurysms on the basis of size (<5 mm, ≥ 5 mm, or <15 mm), and RIAs and blebs were more likely to be observed in the ACoA and PCoA regardless of aneurysm size. Therefore, we speculate that ACoA and PCoA aneurysms may rupture more frequently due to their relatively high tendency toward blebbing.
Although a similar prevalence of blebs was observed between the ACoA and PCoA, the risk of rupture may be lower for MCA IAs than for ACoA or PCoA IAs. Salimi et al reported that the flow conditions differ across IA locations and that IAs may develop into different forms if they are exposed to different flow conditions. This may result in different rupture pathways, regardless of the development of blebs, which could explain the differences in rupture rates and blebs in IAs at different locations. 10 Blebs were predominantly located at the dome across both ruptured (RIA) and unruptured (UIA) aneurysms, with the dome accounting for the highest proportion in all locations. Specifically, blebs on RIA frequently involved the dome in ACoA, PCoA, and MCA aneurysms, whereas in UIA, dome blebs were predominantly observed in ICA and PCoA aneurysms.
According to Yogesh et al, 20 various pathways to rupture have been identified, such as wall degeneration and weakening under abnormally high blood flow conditions (more common in ACoA aneurysms), as well as progressive wall remodeling and degradation under abnormally low blood flow conditions possibly mediated by inflammation (most common in MCA aneurysms). 20 In the present study, we did not investigate the rupture pathways of IAs at different locations and with different characteristics of the IA wall in depth. However, the results revealed that the bleb size in the RIA group was greater than that in the UIA group (P < 0.05), suggesting that larger blebs may correlate with a greater likelihood that the IA will rupture. In summary, in Chinese individuals, blebs were common in small and medium-sized IAs, and in IAs located at the ACoA, PCoA, and bifurcations, while RIAs were more likely to have larger blebs. These conclusions should be validated with a larger sample.
Geometry and Hemodynamics
In a study by Bor et al, aneurysm size, dome/neck ratio, and multilobulation were risk factors for aneurysm growth. 24 According to Lindgren et al, irregular or multilobed shapes are strongly associated with rupture for IAs of various sizes. 5 Bjorkman et al reported that only IA size and shape irregularity were independently associated with IA rupture, with the latter having the strongest association. 25 Salimi et al reported that the formation of blebs in small aneurysms is associated with unstable blood flow and shape irregularity. 8 The present study revealed that compared with those without blebs, aneurysms with blebs had larger L (P < 0.001), H (P < 0.001), W (P < 0.001), AR (P < 0.001), SR (P < 0.001), BN (P < 0.001) and HWR (P = 0.024) values and more irregular shapes (P < 0.001). These findings are consistent with Salimi et al, indicating that aneurysms with blebs often exhibit an irregular morphology and appear more likely to rupture. However, because this was an observational study, our results indicate association rather than direct causality. In addition, the multiple logistic regression results of this study indicate that the SR value was an important risk factor for the incidence of blebs (OR = 4.683; 95% CI = 1.937-11.324), revealing that the size of the parent artery should also be considered when assessing IAs. In summary, in Chinese individuals, blebs were common in high-SR IAs. According to Xu et al, aneurysm width, AR, and anterior fornix projection are independent risk factors for IA rupture, 26 whereas another study suggested that aneurysm rupture may be caused by the velocity and pattern of blood flow within the aneurysm. 27 The greater the AR is, the greater the height or the smaller the neck of the IA; the smaller the neck is, the greater the obstruction to blood flow within the aneurysm. 26 The lower the blood flow velocity is, the greater the degree of hemodynamic changes within the aneurysm, leading to a degenerative process. 26 As the IA forms from the neck, it is more likely that larger-volume and smaller-neck IAs will have thinner walls, making them more susceptible to WSS if the IA wall is not reshaped. 26 The results of our study suggest that the SR values are meaningful, as higher SR values were associated with IA rupture and the presence of blebs. Higher SR values may reflect exacerbation of the hemodynamic state described above, but further validation is still needed.
Salimi reported that bifurcation aneurysms are exposed to higher flow conditions than sidewall aneurysms. Compared with sidewall aneurysms, bifurcation aneurysms have a greater maximum WSS and a more concentrated WSS distribution and are larger, longer, and more twisted in shape. 10 Similar to Salimi et al, our study revealed that bifurcation aneurysms were more likely to rupture and contain blebs. Salimi et al reported that strong and concentrated inflow jets; complex and unstable flow patterns; and concentrated, oscillatory, and nonuniform WSS flow patterns lead to blebs in IAs. 8 Our research method differs from that of Salimi et al, as we did not apply software to remove blebs; instead, IAs with blebs were divided into three regions, namely, the neck, dome, and body, for hemodynamic parameter analysis and comparison between aneurysms with and without blebs. Hemodynamic comparisons between aneurysms with and without blebs revealed that IAs with blebs had smaller NWSS (Pdome < 0.001), TAWSS (Pneck = 0.004; Pdome < 0.001), TANWSS (Pneck = 0.028; Pdome = 0.001), and TAWSSG values (Pneck = 0.028; Pdome = 0.001). Interestingly, the OSI (P = 0.011) and RRT (P = 0.021) of the necks of aneurysms with blebs were lower than those of the necks of aneurysms without blebs, but there were no differences between those values at the domes. Compared with IAs without blebs, IAs with blebs had a smaller SSR value (P = 0.003) and a greater VOR value (P = 0.007) in the dome region. According to the results of this study, IAs with blebs can exhibit hemodynamic characteristics that differ from those of IAs without blebs, particularly concerning the OSI, RRT, SSR, and VOR parameters. Interestingly, after we divided IAs with blebs into RIA and UIA groups to compare their hemodynamic characteristics, we found that the RIA group had lower OSI (Pbody < 0.001) and RRT (Pbody < 0.001) values than did the UIA group. Accordingly, IAs containing blebs with lower OSI and RRT values in the body region may be more likely to rupture. These observations may help explore the mechanisms linking blebs to aneurysm rupture. However, prospective studies are needed to establish any direct causality. Further validation with large samples is still needed because of the limited number of cases and associated selection bias. Due to potentially low flow conditions present in the bleb, the wall may be weakened or degraded, and the geometric shape of the bleb could concentrate internal stresses within the wall, supporting the hypothesis that blebs form in regions of slow circulation and may be linked to rupture events. 11 However, these data should be interpreted as indicative of association rather than causation. Salimi et al reported that not all blebs are the same; some blebs have thin, translucent walls, whereas others have thick, atherosclerotic walls, and aneurysms can rupture at the sites of thin blebs, those of atherosclerotic blebs, or far from the blebs. 7 Moreover, the blood flow characteristics of neck fixed-type IAs are thought to be more stable than those of neck growth-type IAs. 28 In contrast, neck growth type IAs may have less favorable flow conditions. 28 According to our research, blebs may alter the hemodynamic characteristics of different regions of aneurysms, and more attention should be given to the neck and dome regions of the aneurysms. Interestingly, when blebbed RIAs and UIAs were compared, the body region was still considered important. In this study, we divided the IAs into three regions for local hemodynamic comparison to provide reference values for further analysis of the aneurysm walls, assessment of the risk of UIA rupture, evaluation of secondary rupture risks in RIAs, and treatment during surgery. Further research is necessary to verify the wall conditions of different blebbed aneurysms.
From a clinical perspective, these findings highlight the potential value of incorporating bleb presence and geometric factors (e.g., SR, shape irregularity) into risk stratification for small- and medium-sized IAs. Identifying aneurysms with high SR and blebs might help clinicians prioritize imaging follow-up or consider earlier intervention, although prospective studies are required to confirm any incremental benefit in management strategies.
Generalizability Considerations
This study was conducted in two hospitals in Tianjin, China, and most participants were of Chinese Han ethnicity. Therefore, the generalizability of the findings to other populations or regions may be limited. Differences in genetic backgrounds, environmental factors, and comorbid conditions could influence the epidemiology of IA formation and rupture. Larger, multicenter, and multi-ethnic studies are warranted to validate the applicability of our results and strengthen the evidence base for clinical decision-making.
Limitations
The small sample size of this study is one of its key limitations. However, this is one of the few dedicated studies on the blebs of small- and medium-sized IAs in the Chinese Han population with comprehensive information. (2) The imaging methods used may have affected morphological parameters. In addition, uncertainty exists regarding whether aneurysm rupture affects aneurysm morphology and influences morphological analysis. The morphological parameters were calculated from 3D angiograms, which are essentially monograms that do not consider thrombi. However, we used high-clarity CTA/DSA data to minimize systematic errors. (3) The findings require verification with histological data to demonstrate the direct relationship between hemodynamics and aneurysm wall properties. (4) Our CFD model is based on conventional assumptions, including rigid vessel walls and Newtonian blood rheology. These simplifications are widely accepted and have been shown to introduce only minor deviations in most cases. 29 To facilitate comparison with previous studies, we also avoided complex turbulence modeling. Nonetheless, we acknowledge that these assumptions may introduce certain systematic biases. For example, modeling blood as a Newtonian fluid may underestimate wall shear stress (WSS) in low-shear regions such as aneurysm domes, while assuming rigid walls may slightly overestimate WSS and flow velocities compared to physiological conditions.30,31 Future studies incorporating fluid-structure interaction (FSI) models could overcome these limitations and yield more physiologically accurate hemodynamic assessments.30,31 Finally, as most of our participants were of Chinese Han ethnicity, caution should be exercised when generalizing our findings to other ethnic or demographic groups. Population-based differences in vascular morphology and rupture risk warrant further validation in diverse cohorts.
Conclusions
Among Chinese individuals, blebs are common in small- and medium-sized IAs (especially RIAs), IAs with high SR values, and IAs located at the ACoA, PCoA, and artery bifurcations. IA rupture is more likely to occur in aneurysms with larger blebs. These findings complement the current research on blebs. Further investigations of the hemodynamics of the neck, body, and dome regions of IAs with blebs should be performed.
Supplemental Material
Supplemental Material - Prevalence, Geometry, and Hemodynamics of Small and Medium-Sized Intracranial Aneurysms With and Without Blebs in the Chinese Han Population
Supplemental Material for Prevalence, Geometry, and Hemodynamics of Small and Medium-Sized Intracranial Aneurysms With and Without Blebs in the Chinese Han Population by Xiaopeng Cui, Yan Zhao, Liang Wang, Yujing Jin, Zhenglong Yang, Yaohua Li, Zilin Zhao, Hengrui Zhang, Kai Wei, Zhennan Sun, Peng Huai, Lei Chen, and Xinyu Yang in Journal of Central Nervous System Disease
Footnotes
Ethics Approval
This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Tianjin Medical University General Hospital (Approval No. IRB2022-YX-175-01) and Tianjin Fifth Central Hospital (Approval No. WZX-EC-KY2024039).
Consent to Participate
The requirement for informed consent was formally waived by both ethics committees due to the retrospective design and de-identified nature of the data. However, all patients had previously signed written informed consent for the use of their medical records in research. All data were anonymized before analysis.
Consent for Publication
All patients provided written informed consent for publication of their anonymized data and any relevant medical images.
Author contributions
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by the Tianjin Health Research Project (Grant No. TJWJ2023QN102), the Peking University Binhai Hospital Research Innovation and Transformation Fund (Grant No. 2024-CX-01), and the projects of the Natural Science Foundation of Tianjin, China (No. 21JCQNJC00350).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The supporting data of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
