Abstract
Hemorrhagic stroke (HS) in childhood accounts for almost 50% of childhood strokes, is among the top ten causes of deaths, or determines lifelong disability. These facts form significant socio-economic and demographic problems. The purpose of this review is to analyze current knowledge about HS in children. The data on HS terminology are presented, taking into account the International Classification of Diseases 11 edition. Attention is paid to the epidemiology of HS in children, including the results of individual local studies. The risk factors of HS in children were studied with an analysis of the causal, pathophysiological mechanisms of HS of various etiologies. The ideas about the clinical manifestations of HS in children are described. The analysis of HS treatment in children was carried out with an emphasis on achievements in neurointensive therapy of the acute period of HS. This review also includes information on the outcomes of HS in children.
Introduction
Hemorrhagic stroke (HS) in childhood requires careful study, as it is not uncommon, determines high mortality and disability of children. The importance of the problem is emphasized by the high social significance, as well as the lack of awareness of HS among doctors of various pediatric specialties. There are few studies of HS in childhood. There is limited information on risk factors, clinical and radiological correlation, and HS outcomes in children. There are no clear recommendations for working with such patients, proven, generally accepted diagnostic and treatment strategies. The purpose of this review is to analyze current knowledge about HS in children. We conducted a literature review in publications without language restriction by keywords: hemorrhagic stroke in children, definition, terminology, classification, epidemiology, risk factors, clinic, treatment, outcome of HS in children.
Terminology of Hemorrhagic Stroke in Children
HS occurs as a result of bleeding from the cerebral vessels.1,2 In 1995 the World Health Organization has proposed the definition of stroke as “rapidly developing clinical signs of focal (or global) brain dysfunction, symptoms of which last 24 h or longer or lead to death for no apparent reason other than vascular origin”. 3
Intracranial haemorrhage non-traumatic etiology.

Thus, the ICD 11 edition reflects, first of all, the anatomical site of bleeding. Thus, subarachnoid (SAH) hemorrhage is characterized by extravasation of blood between the inner, “spider” membrane and the substance of the brain. Intracerebral (ICH) bleeding is characterized by hemorrhage into the parenchyma (IPH) or ventricles of the brain (IVH). Subdural hemorrhage is bleeding between the soft and dura mater. There are also indications of hemorrhages lobar, hemispheric, in the brainstem and cerebellum of the brain.
In childhood, such characteristics of HS are also relevant, such as pediatric or pediatric stroke (occurs in children aged 29 days to 18 years), as well as neonatal or perinatal stroke: develops from the 20th week of pregnancy on the 28th day of the child’s life.4–6
Epidemiology of Hemorrhagic Stroke in Children
Indicators of HS frequency in children.
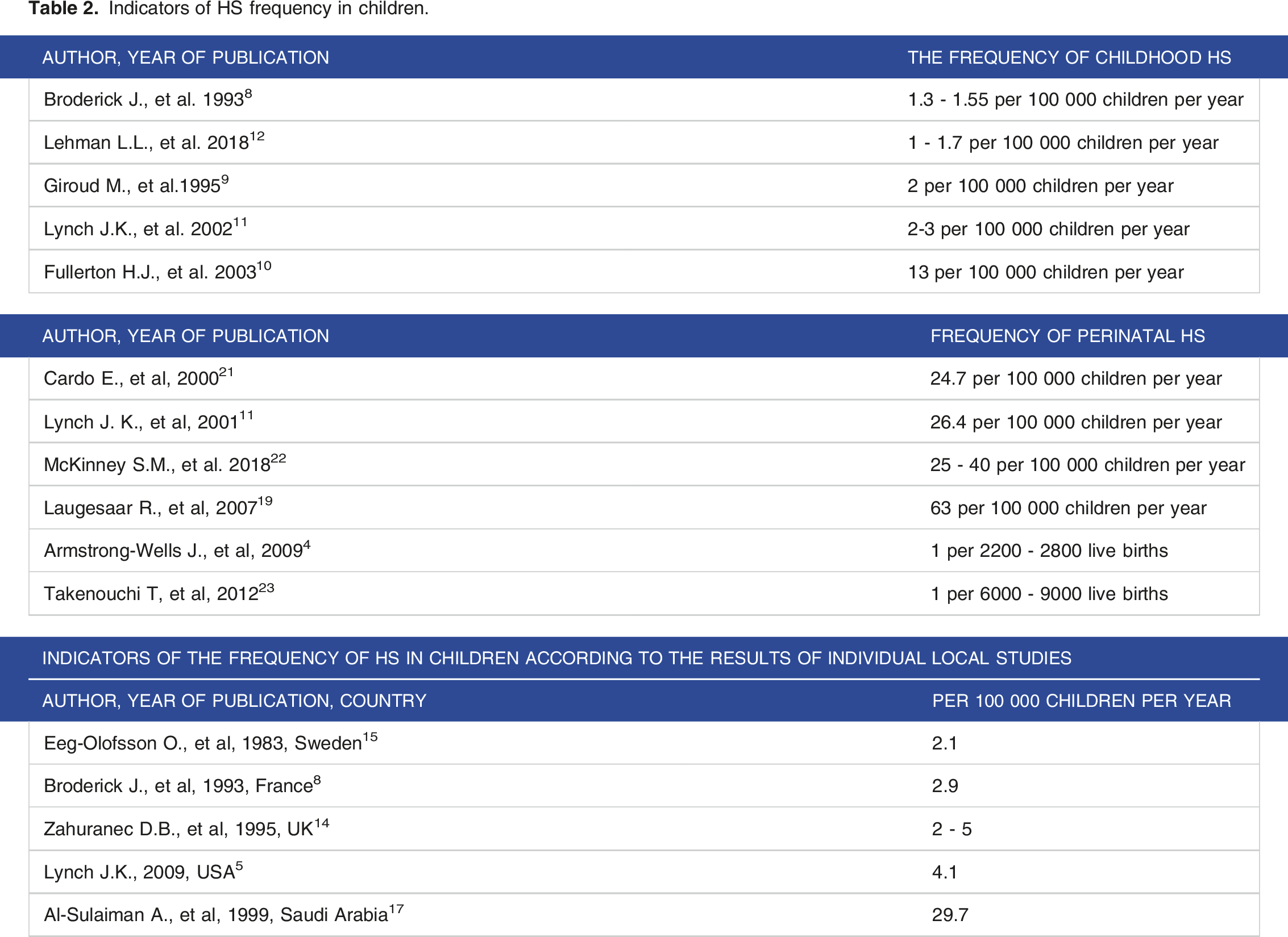
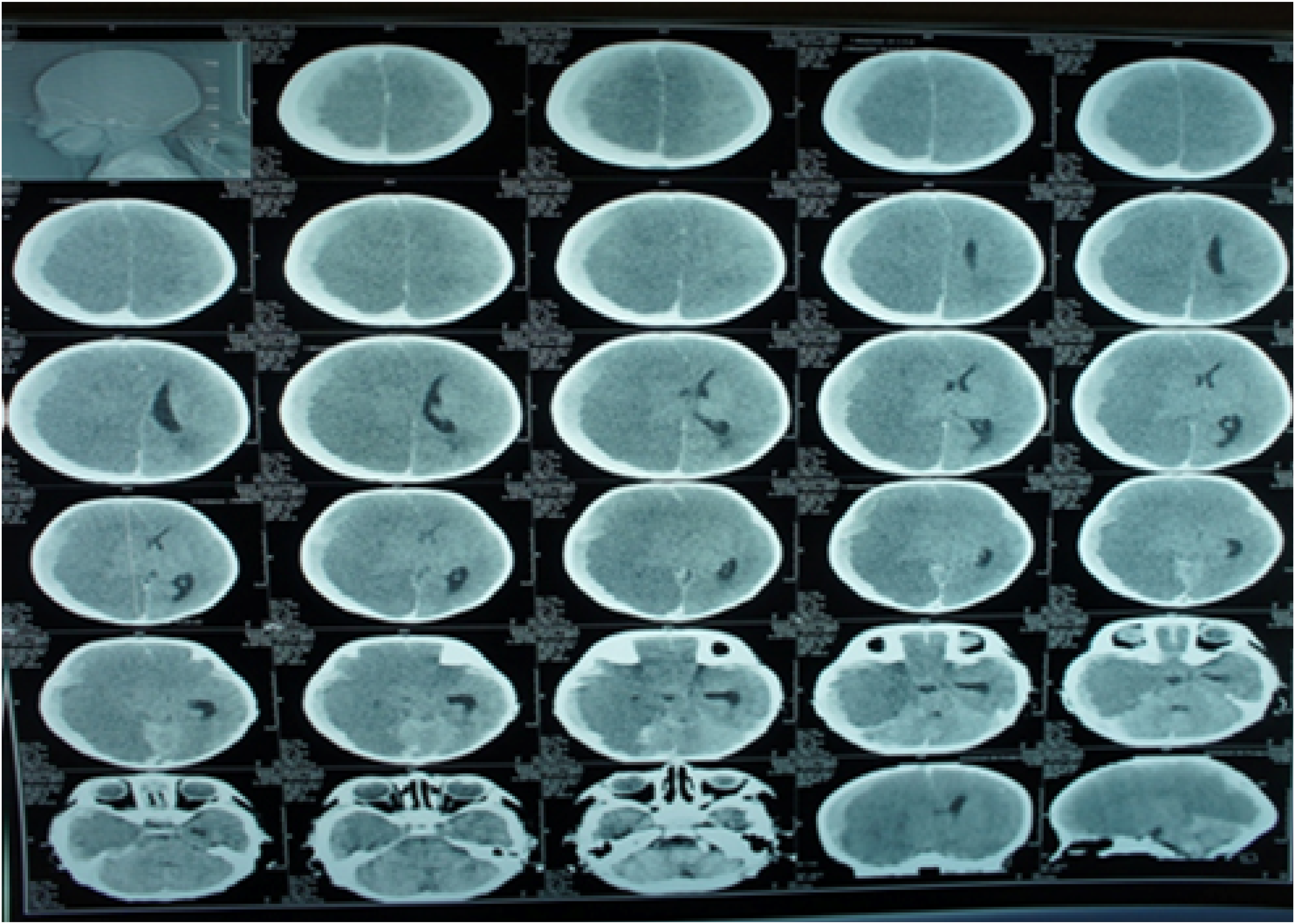
There are studies indicating the frequency of ICH and SAH. ICH (Figure 1) occurs with a frequency of 0.71 - 1.4 per 100 000 children.8,25 SAH is 18-22% (Figure 2). Computed tomogram of intraventricular hemorrhage. Computed tomogram of subarachnoid hemorrhage.
The combination of ICH and SAH occurs in 32% of cases.16,26,27
The distribution of HS by gender in most studies indicates that boys have a higher risk of stroke than girls by about 28%. The ratio of boys and girls with HS is 1.2 - 1.5:1.8,10,16,28
Risk Factors for Hemorrhagic Stroke in Children
Cerebrovascular Pathology and Hemorrhagic Stroke in Children
The most common cause of HS in children is cerebrovascular pathology. Thus, Beslow L.A., et al, found that vascular system abnormalities caused 91% of HS cases in children. 29 Other authors claim that cerebral vascular abnormalities are detected in children with HS in 32.2% - 67% cases.28,30–34 Abnormalities of the vascular system include arteriovenous malformations (AVM), intracranial aneurysms (IA) and cavernous malformations (CM). In studies by Jordan L.C., et al, pediatric HS, 31% of patients had cerebral AVM, IA - 13%, CM - 15%. 26 Giroud M., et al, among 23 children with HS, 9 had AVM, 2 had IA, and 5 had CM. 35
Vascular abnormalities in most cases are represented by AVM. 36 AVM is a vascular pathology in which veins and arteries intertwine with each other to form a “vascular tangle”. The peculiarity of this vascular formation is that there are no capillaries in it. In this regard, blood from the arteries directly enters the veins, tissues do not receive nutrients and oxygen from the blood, there is no metabolism.37,38 Lin C., et al, consider AVM as the leading cause of hemorrhage in children with spontaneous ICH. 39 A study of spontaneous ICH in children showed that vascular malformations cause bleeding in 91% of subjects, in studies by Meyer-Heim A.D., et al, in 47% of children. 29 In 53% of patients, hemorrhage occurred as an acute event, and in 47% of cases, a prolonged course of AVM was observed. 32 Al-Jarallah A., et al, revealed a congenital vascular anomaly in 42.6% of children with non-traumatic intra-parenchymal hemorrhage. In 33.8% of cases, there was AVM or fistula. 31 Krings T., et al, state that the probability of the first cerebral hemorrhage in AVM is 2-4% per year. 40 Studies by Carlin T.M., et al, in 96% of cases revealed AVM in children under 5 years of age. 41 Earley C. J. et al, showed that AVM was the cause of ICH in 29% of cases. 25 Gabriel R.A., et al, Plummer N.W., et al, found that AVM determines 8.6% of non-traumatic SAH and 1% of cerebral strokes.42,43 One bleeding in AVM is accompanied by a mortality rate of 25%. 44
The next common abnormality of the vascular wall, accompanied by HS, is IA - deformation of the cerebral arteries in the form of bulging of their walls at the site of thinning. Hitchon P., et al, Gabriel R.A., et al, believe that IA are recorded with a fairly high frequency in the general population: 10.3 per 100 000 per year.43,45 Huang J., et al, identified 1377 cases of IA. The ratio of boys and girls was 2.2:1. 11% of aneurysms were formed in the area of bifurcation of the internal carotid artery (ICA), 42% - in the posterior circulation. 37% of the aneurysms were gigantic. In 58% of cases, aneurysms were accompanied by SAH. 46 Sanai N., et al, identified 43 aneurysms in 32 pediatric patients. In 22% of cases, aneurysms were accompanied by SAH. The locations of the aneurysm included the ICA (13 lesions), the middle cerebral artery (11 lesions) and the basilar artery (6 lesions). Of the 43 lesions, 17 (40%) were giant aneurysms, and 22 (51%) had fusiform/dolichoectatic morphological features. 47 Jordan L.C., et al, in the HS study (IPH, SAH and IVH) indicate that aneurysms mainly determine SAH (57%), while among children with pure ICH, an aneurysm was detected in only 2%, and among children with mixed hemorrhage (IPH and SAH) - at the level of 5%. Krings T., et al, confirm that cerebral vascular aneurysms account for 13% of HS, which are mainly manifested by SAH. Aneurysm may be associated with arterial dissection (50%), bacterial or mycotic infections (15%), trauma (5-10%) or idiopathic (30%). 48 Pappachan J., et al, are of the opinion that aneurysms in children with HS are relatively rare, among which 10-15% are post-traumatic and about the same number are fungal. 49
Another vascular anomaly accompanying HS in children is CA. CA represent intra or extracranial vascular lesions with calcifications. CA occurs sporadically in 13%, and as family cases in 50%. CA develops as a result of cerebral vascular aneurysms, as well as symptomatic hypertension due to kidney diseases, aldosteroma, pheochromocytoma, hypercorticism, and aortic coarctation. 49 CA is the cause of HS in children in 5-15% of cases.26,50
Violations of Coagulating and Anticoagulating Properties of Blood and Hemorrhagic Stroke in Children
Al-Jarallah A., et al, among 68 children with intra-parenchymal cerebral hemorrhage, blood clotting disorders were detected in 22 (32.4%) patients. 31
Prothrombotic or coagulation disorders that lead to the development of HS may be hereditary or acquired.
The most common hereditary prothrombotic abnormalities include deficiency of antithrombin III, antiphospholipid antibodies, plasminogen, prothrombin gene, 20210A VII, VIII coagulation factors (hemophilia).51–56 Some authors cite data that 10-30% of childhood HS occur as a result of both hereditary hemophilia and von Willebrand disease, thrombocytopenia, liver dysfunction, vitamin K deficiency.57–59 In a study by Sharma S., et al, Yilmaz C., et al, the most common risk factors for HS were blood clotting disorders associated with vitamin K deficiency.60,61
Lo W.D., et al, Al-Jarallah A., et al, revealed genetic or acquired hemostasis defects in HS in children in 9% and thrombocytopenia in 6% of cases.31,62
Acquired coagulation disorders in children provoke liver and kidney diseases, including nephrotic syndrome with loss of coagulation factors. 63
The direction of hemostatic disorders in HS was represented by hypocoagulation in about 10% of cases. 28
Cerebral Venous Vessel Thrombosis and Hemorrhagic Stroke in Children
The incidence of venous vascular thrombosis (CSVT) of the brain in children averages 0.29 - 0.67 per 100 000 children per year.64,65 In the neonatal period, CSVT is 2.6 per 100 000 children per year.65,66 The mortality rate for CSVT in children is 6 - 30%.67,68
Risk factors for CSVT in newborns are acute conditions associated with the perinatal period: asphyxia during childbirth, generalized infection, dehydration, and the tendency of newborns to thrombosis.64,69
In older children, significant CSVT factors are injury to the dural sinuses during intracranial surgery or traumatic brain injury, taking oral contraceptives, glucocorticosteroids, protein S deficiency, anemia, connective tissue pathology, liver failure, nephropathy, malignant neoplasms, septic conditions or local infection in the head and neck,64,70–76 prothrombotic disorders.64,77,78
CSVT determines thrombosis in cerebral veins, superficial or deep venous sinuses, obstruction of blood flow, venous stasis, impaired transport of cerebrospinal fluid.64,66,73,79
The frequency of HS in CSVT has maximum values: 6.5 - 25.0 per 100 000 children per year.80–82
In newborns, CSVT is a common cause of intraventricular hemorrhages.65,83 To a lesser extent, CSVT causes primary subdural and subarachnoid hemorrhage.73,84
Infections and Hemorrhagic Stroke in Children
Up to a third of HS cases in children occur in the context of infection. 85
Of 104 Saudi children with HS, infectious and inflammatory diseases were identified as a risk factor in 18 (17.3%) children. 86 Observations by Lo W.D, et al, showed that infection of the central nervous system, including as a component of systemic disease, was the cause of the development of HS in 9% of cases. 62 Cases of childhood HS have been reported with microbial associations such as chlamydia, enterovirus, parvovirus 19, influenza A virus, coxsackie, mycoplasma.87–89
The spectrum of infection in HS also includes bacterial meningitis, Haemophilus influenzae, Streptococcus pneumoniae, Staphylococcus aureus, systemic lupus erythematosus, neurobrucellosis, as well as congenital toxoplasmosis and congenital rubella syndrome.90,91 There are studies that have shown that the persistence of herpes viruses, Cytomegalovirus, Epstein-Barr virus, as well as bacterial, tuberculous meningitis and HIV infection can serve as risk factors for HS.64,92–95 In HIV infection, hemorrhage was accompanied by secondary HIV - induced vasculitis, vasculopathy, thrombocytopenia or coagulopathy. 96
Hemorrhagic complications in the presence of an infectious agent are triggered by a systemic inflammatory process, endothelial dysfunction, which, when combined with prothrombotic activity, leads to damage to the vascular wall, vasculopathy, thrombosis, occlusion of the vessel lumen.86,97–99
Vasculitis and Hemorrhagic Stroke in Children
Cerebral vasculitis (CV) is more common in children over the age of 14 and can be of primary or secondary, as well as infectious and non-infectious etiology.100,101
In primary vasculitis there are no diseases of organs and systems with concomitant inflammation of blood vessels.102,103
Secondary CNS vasculitis occurs in systemic lupus erythematosus, Schenlein-Genoch purpura, polyarteritis nodosa, Takayasu arteritis, Wegener’s granulomatosis, Kawasaki disease, malignant neoplasms.104–109
In all cases of HS (average 4.6%) vasculitis, it provokes inflammation, damage and dysfunction of the vascular wall endothelium, lymphocytic, transmural infiltration from T - and other inflammatory cells, thrombosis.102,103
Cardiovascular Diseases and Hemorrhagic Stroke in Children
Cardiovascular diseases increase the risk of developing HS (2.6 - 6.8%) in children. Among cardiovascular diseases, congenital and acquired heart defects can lead to the development of HS in children. The development of HS in heart defects is mainly associated with intracardiac bypass surgery from right to left in most defects.110–112 HS in children can develop in the early stages after surgical, invasive heart interventions, in pediatric cardiac surgery patients on extracorporeal membrane oxygenation.113–120 Acquired heart diseases (endo and myocarditis, cardiac arrhythmias, cardiomyopathy, rheumocarditis) are also classified as a risk factor for HS in children. 118
In childhood, HS occurs less frequently against the background of arterial hypertension (0.3 - 2.2% of cases).121–124
However, in the observations of Al-Jarallah A., et al, there was no association of HS with systemic hypertension in children. 31
Oncological Diseases and Hemorrhagic Stroke in Children
HS is a known complication in patients with malignant oncological diseases.62,123
Lo W.D., et al, in a 7-year retrospective analysis of 85 children with non-traumatic intracranial hemorrhage, brain tumors were found in 15% of cases, Al-Jarallah A., et al - in 13.2%.31,62
However, other studies show a lower probability of developing HS in children with cancer (0.55 - 4%).125–129
Bowers D.C., et al, calculated the time interval for the development of HS in brain tumor survivors, which averaged 9.8 years after diagnosis of leukemia, and an average of 13.9 years since diagnosis of cancer. 130
Zadeh C., et al, revealed HS in 40% of children with leukemia, Kyrnetskiy E.E., et al, in 30 children with brain tumors, in 19 children with acute leukemia and 2 children with lymphoma.131,132 In studies by Noje C., et al, the prevalence of HS in children with acute lymphoblastic leukemia (ALL) was 2.6%, with acute myeloblastic leukemia (AML) - 3.3%, with various types of lymphoma - 1.6%. 129 Di Mario F.J., et al, identified HS with the same distribution between ALL, AML and lymphoma. 133 In the observations of Packer R.J., et al, HS was found in 4% of children with ALL, 13% with AML, 1% with lymphoma, 6% with neuroblastoma, 5% with bone tumor and 1% with other tumors. 125
Most HS occurred in the early stages of cancer treatment. It was found that a dose of radiation to the skull of more than 50 Gray increased the risk of death from cerebrovascular disease by 17.8 times.134,135 HS was caused by a combination in the treatment of L-asparaginase, prednisone, radiation and chemotherapy.104,131,136–139
Modifiable risk factors for the development of HS in patients with leukemia and lymphoma include hypercoagulation and hyperviscosity.130,140 Many authors claim that vasculopathy, which occurs after several months or years with various tumors, as well as after radiation therapy, can lead to the development of HS.87,141–143
Genetic Polymorphisms, Hereditary Diseases in the Development of Hemorrhagic Stroke in Children
Sickle Cell Anemia
Sickle cell anemia (SCD) is a hereditary hemoglobinopathy, which is manifested by the production of abnormal hemoglobin S, a change in the shape and properties of red blood cells. HS in SCD occurs with a frequency of 17 - 44 per 100 000 people.144–147 Mortality in the first 2 weeks after HS in SCD is 26%, recurrence of HS is 6.4 cases per 100 000 with an average time to recurrence of 7.2 months. 148
Factors that can lead to cerebrovascular damage in SCD include low hemoglobin concentration, chronic anemia, low oxygen content in the blood, which leads to impaired cerebral blood flow. Other factors include a high white blood cell count, as well as SCD treatment measures: blood transfusion within the last 14 days, treatment with corticosteroids and nonsteroidal anti-inflammatory drugs. 148 In patients with SCD and stroke, blood clots, accumulation of sickle-shaped erythrocytes and other cells, damage to the endothelium, hyperplasia of the internal elastic membrane, scarring of the media, abnormal blood flow rate, formation of aneurysms, more often in the vertebrobasilar system, were also observed in the affected large arteries.149–152
Moyamoya Disease or Syndrome
Moyamoya disease or syndrome is a non-atherosclerotic, non-inflammatory, non-amyloid vasculopathy characterized by progressive stenosis of the distal intracranial sections of the internal carotid artery, less often the proximal sections of the anterior, middle, posterior and basilar arteries with the development of basal collateral vessels. 153 Genetic polymorphism of HLA class II genes is associated with Moyamoya disease. Moyamoya syndrome is also known, which occurs against the background of associated pathology: SCD, Down syndrome, neurofibromatosis, previous irradiation of the skull.154,155 MMD occurs with a frequency of 0.086 per 100 000 children, in the first decade of life, more often in girls.38,156,157 Most patients with MMD have subarachnoid hemorrhages. The clinical course in 20% of cases of the disease may manifest HS with progressive neurological disorders, hemiplegia, seizures, mental retardation and motor deficits.38,158–161
Hyperhomocysteinemia
Hyperhomocysteinemia (Hyper-Hcy) is a genetically determined, hereditary metabolic defect, the primary link of which is a violation of the metabolism of sulfur-containing amino acids. The cause of Hyper-Hcy may be a deficiency of cystathionine β - synthetase, homocysteine methyltransferase, as well as a mutation of the methylenetetrahydrofolate reductase (MTHFR) gene involved in homocysteine metabolism (polymorphism A1298C of the MTHFR gene). MTHFR is also a key enzyme in folic acid metabolism in vivo.162,163
Hyper-Hcy may cause a deficiency of folic acid or vitamin B2, B12. 2 The mechanism of cerebrovascular disease in Hiper-Hpu is hypercoagulation, hyperaggregation of platelets, which leads to thrombosis, especially of the carotid arteries, arterial dissection, vascular endothelium damage, neurotoxic effect.21,164–166 A number of studies have shown an association of HS with elevated cholesterol levels in the blood. At the same time, the risk of ICH increases.167,168
Antiphospholipid Syndrome
Antiphospholipid syndrome (APS) is a systemic autoimmune disease with a high titer to phospholipids (PL) of antibodies such as antibodies to cardiolipin, antibodies to lupus anticoagulant and antibodies to cofactor proteins (prothrombin, protein C, protein S, annexin V, prostacyclin and beta2-glycoprotein-I (beta2-GP-I). 169 The main consequence of APS, which can provoke HS, are arterial or venous thrombosis of various localization.68,170,171 Ferro J.M., et al, found antiphospholipid antibodies in 22.6% of patients with CSVT, Martinelli I, et al, in 9 patients out of 121 examined patients with CSVT.68,172 The most common forms of APS are primary and secondary APS. In childhood, secondary APS is more often observed, which develops when taking medications (psychotropic, hormonal, contraceptive, novocainamide, interferon alpha in high doses), viral and bacterial infections, autoimmune diseases, as well as malignant neoplasms.170,173
HS with neurological symptoms is observed in about 20% of patients. 174
The risk of developing HS is associated with such a genetic pathology as a mutation of collagen formation, which determines defects in the structure of blood vessels. 28
HS may also develop against the background of arteriopathy associated with heterozygous mutations in the CTSA gene. 175
Metabolic Hemorrhagic Stroke in Children
Some authors distinguish “metabolic stroke” (MS) as a consequence of a metabolic disorder of an innate or acquired nature. 176
Congenital metabolic disorders determine Handy - Schueller - Christchen disease, deficiency of biotin, nicotinic acid, vitamins B, C, Fabry disease and others. MS can complicate the course of certain mitochondrial diseases and syndromes: Mytochondrial Encephalopathy, Myepathy, Lactatacidosis, Stroke like episodes (MELAS), Pearson-Murrow syndrome and others. In these conditions, there are diseases of internal organs and concomitant metabolic disorders. The main biochemical sign of the entire mitochondrial pathology is lactate acidosis, an increase in the level of lactic and pyruvic acids in the blood and liquor, aciduria. Violations of local fibrinolysis, vasculopathy, angiopathy are observed, which determines both transient and persistent disorders of cerebral circulation.177–179
MS is observed as a consequence of acquired somatic diseases in children with severe diabetes mellitus, chronic liver and pancreatic diseases, after undergoing severe intestinal surgery, anesthesia, uncompensated alkalosis or acidosis.64,92,180 Pappachan J., et al, describe the clinical signs of MS: persistent vomiting, hypo or hyperglycemia, organic acidemia, urea cycle disorders, mitochondrial disorders. 49
Burdened Perinatal History as a Risk Factor for Hemorrhagic Stroke in Children
Shirokostyuk L. A. points out that 63% of children with cerebrovascular pathology have equal proportions of maternal diseases, unfavorable course of pregnancy and childbirth in the mother, difficulties of adaptation in the neonatal period. 181 An important role in the occurrence of HS was played by intranatal factors: signs of uteroplacental insufficiency, fetal distress, rupture of the membranes, early discharge of amniotic fluid, contaminated water, severe infections, childbirth using instruments (forceps or vacuum aid), emergency cesarean section, prematurity and postponement, an Apgar score of less than 7 points, intensive care at birth, hypoglycemia, congenital heart disease.4,91,182–187
Drug Use
Mainly in the adolescent population, it is not uncommon to develop hemorrhages due to taking drugs such as amphetamines, ecstasy, cocaine, phencyclidine, sniffing glue.188,189
Unknown Risk Factors for Hemorrhagic Stroke in Children
Unknown HS risk factors in children after a thorough diagnostic search, according to Malik A.A., et al, account for 30% of cases. 57
Cohort studies by Beslow L.A., et al, Jordan L.C., et al, showed an unknown etiology of childhood strokes in 9-23% of cases, Lanthier S., et al, DeVeber G., et al in 10-15%, Chung B., et al - 12%.16,28,29,64,123
Median frequency of HS risk factors in children are shown in Diagram 1. Median frequency of HS risk factors in children. Row 1 - cerebrovascular pathology (32,2 - 91%), row 2 - hemostasis disorders (10 - 3.4%), row 3 - infections (9 - 33%), row 4 - vasculitis (0 - 4.6%), row 5 - cardiovascular diseases (2.6- 6.8%), row 6 - oncological diseases (0 - 15%), row 7 - genetic polymorphisms, hereditary diseases (0 - 20%), row 8 - unknown (9 - 30%).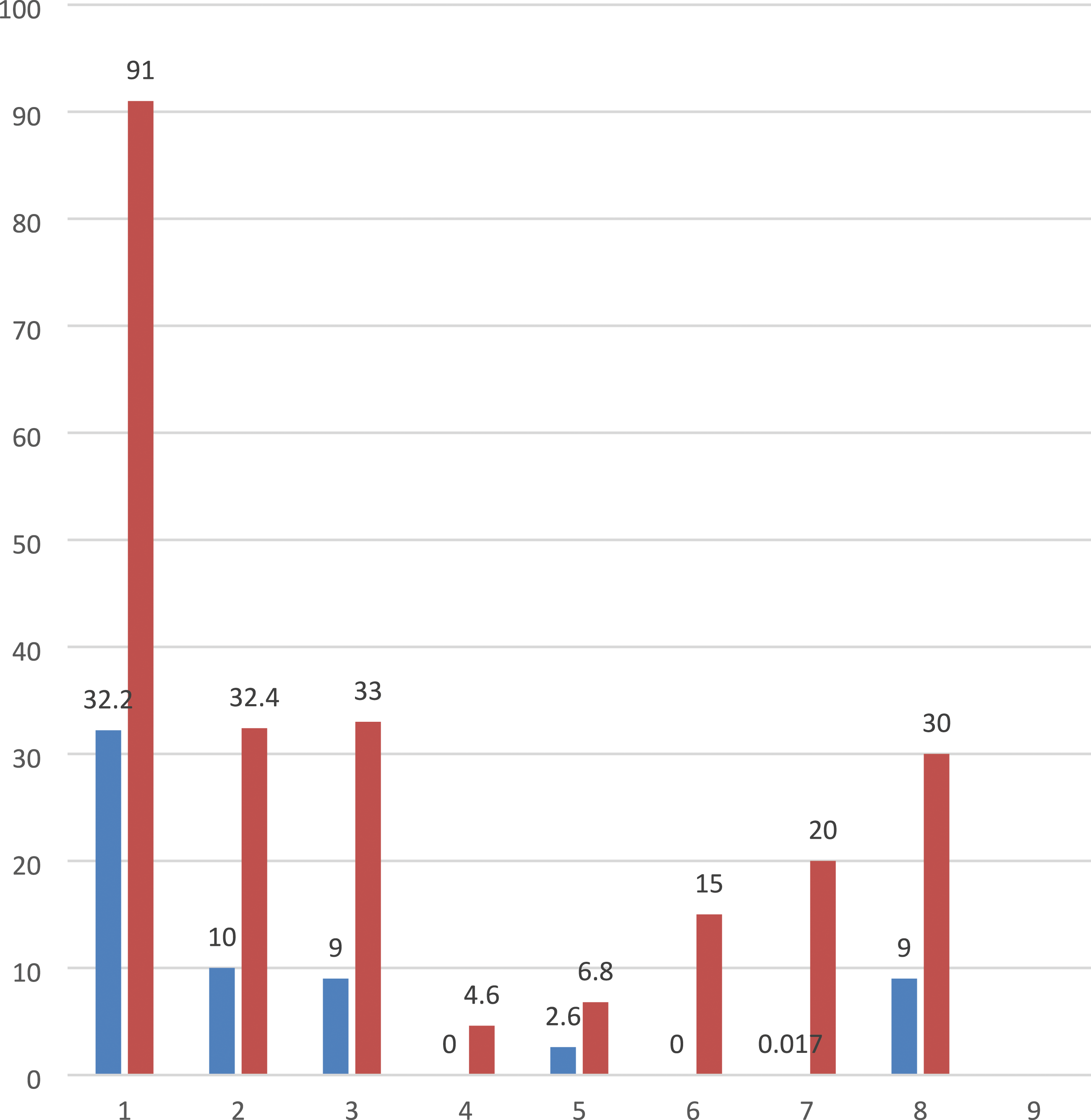
Neurological Manifestations of Hemorrhagic Stroke in Children
In older children, the main signs of HS are: headache, vomiting, difficulty speaking, visual deficits, coordination disorders, changes in consciousness, hemi and tetraplegia, seizures.13,15,16,25,31,33,34,190,191 Beslow L.A., et al, observed acute symptomatic seizures in intracranial hemorrhage in 36% of patients. 192 Fox S., et al, revealed that intracranial hypertension and cerebral edema rapidly progress after HS. 191
The younger the child, the more nonspecific the initial clinical features of HS may be. 13 Thus, seizures are considered to be common initial manifestations of HS in young children.64,192,193 Lo W.D. believes that in children under 6 years of age, the most common signs of HS include both seizures and changes in mental status, vomiting, respiratory distress, lethargy, weakness. Young children do not report headaches. 13 Meyer-Heim A., et al, Calder K., et al, pointed to more diverse manifestations of the acute period of HS, when along with irritability or drowsiness there were problems associated with feeding the baby, vomiting and symptoms of peripheral hemodynamic disorders, a bulging large fontanel.32,194
Certain types of stroke are characterized by different clinical manifestations. Thus, SAH can manifest itself by sudden onset, headache, irritability, photophobia, loss of vision, inability to move limbs, difficulty speaking, loss of consciousness, periodic seizures and meningeal syndrome.
With IVH, unconsciousness, respiratory rhythm disturbances with hypoventilation, apnea, generalized tonic seizures or decerebral rigidity, bradycardia, arterial hypotension, fontanel bulging, tetraparesis, and schizophrenia are more common. 195
With intracerebellar hemorrhages revealed symptoms of compression of the brain stem: bradycardia, apnea, increased intracranial pressure with swelling of the fontanel, divergence of the sutures of the skull, moderate dilation of the ventricles, as well as paresis of facial muscles, tetraparesis, opisthotonus. Subdural hemorrhage shows signs of compression of the brain stem: anisocoria with no photoreaction of the pupils, sopor or coma, as well as rigidity of the occipital muscles, opisthotonus and bradycardia.33,196
Bulbar dysfunction and dysarthria indicate involvement of the brain stem, while aphasia suggests involvement of the basal ganglia, thalamus, or hemispheres of the brain. 197
In children with spontaneous ICH, headache, loss of consciousness and vomiting were common symptoms upon admission. 39
Therapeutic Measures for Hemorrhagic Stroke in Children
HS in childhood in most cases requires urgent neurosurgical care. In this regard, at the beginning of HS therapy, a neurosurgeon’s conclusion on the need for neurosurgical intervention is necessary. The presence of indications for surgical intervention dictates the interaction of the neurosurgeon with the anesthesiological teams. Currently, the possibilities of an augmented reality (AR) system are being considered - projecting three-dimensional images of the anatomy of the brain or spinal cord onto realistic models of patients, which helps in neuronavigation and surgical planning. 198
The polyethologicity of HS in childhood often requires a multidisciplinary approach to identify the root cause of HS, which determines further HS therapy in children. HS treatment methods determine the need for patients to stay in the intensive care unit, in conditions of monitoring vital signs (pulse, blood pressure, respiratory rate, saturation, etc.). Emergency therapy is: maintenance of respiratory, cardiac activity, blood pressure, oxygenation, water-salt metabolism, treatment of cerebral edema, glucocorticosteroid therapy, optimization of glycemia, prevention and correction of hyperthermia, infections.6,38,199
Restoration of Respiration and Gas Exchange in Children with Hemorrhagic Stroke
Restoration of respiration, patency of the respiratory tract, gas exchange is required by patients with impaired consciousness (sopor, coma), respiratory rhythm (for example, apnea), loss of protective reflexes of the respiratory tract (cough and vomiting), as well as in the presence of known clinical signs of respiratory insufficiency: cyanosis, indicators of partial oxygen tension in arterial blood (PaO2): 50 - 79 mmHg, carbon dioxide (PaСО2): 45 - > 70 mmHg, arterial oxygen saturation (SaO2): 94 - 75%. In the limit values of these indicators, as well as in case of cardiorespiratory destabilization, patients should be intubated and transferred to artificial lung ventilation (ALV).200,201
Stabilization of Hemodynamics in Hemorrhagic Stroke in Children
Stabilization of hemodynamics in children with HS primarily involves the management of blood pressure (BP), since an increase in blood pressure in children with HS can lead to continued or repeated bleeding, impaired cerebral perfusion.202–204 However, there are no recommendations for specific target ranges of blood pressure in children.38,205 Fujimura M., et al, suggest reducing high blood pressure by 15-25% during the first 24 hours, with a gradual decrease in the future. 206 Rivkin M.J., et al, recommend the treatment of blood pressure in children when it exceeds normal values by 15% for more than an hour or at any time when blood pressure exceeds 20%. 207 Vasospasm is treated with hemodilution to improve perfusion, calcium channel antagonists, in particular nimodipine, as well as antihypertensive drugs, beta blockers, possibly ACE inhibitors.49,204,208 However, the question of the effectiveness of these drugs remains open. 209
Infusion Therapy for Hemorrhagic Stroke in Children
Infusion therapy for HS in children is necessary to maintain euvolemia, perfusion pressure, and regulation of intracranial pressure (ICP). Intravenous access is necessary for this purpose. However, the permissible amounts of infusion therapy for HS in children require clarification. Thyagarajan B., in the absence of contraindications, some infusions are recommended to be administered in the volume of enteral nutrition. In infancy, the optimal nutritional substrate is breast milk. In case of impaired swallowing function, as well as in patients who are in a state of somnolence or coma, due to the high risk of aspiration, it is necessary to install a gastric probe. 210
There is insufficient research on the qualitative composition of infusion therapy for HS in children. Lo W., in children with HS, recommends the use of saline solutions of liquids, since hypotonic solutions contribute to the development of cerebral edema. 13
The use of hypertensive solutions for infusions in patients with HS effectively reduced intracranial pressure. 211 A combination of crystalloid solutions with hydroxyethyl starch-based colloids is acceptable for HS. 212
When the hemoglobin (Hb) level is < 5 g/dl, hemotransfusion is required at the rate of 10 mL / kg. The hemoglobin level should be maintained at 100 g/l. This provision is consistent with the recommendations of the World Health Organization (WHO). 38
Treatment of Cerebral Edema, High Intracranial Pressure in Hemorrhagic Stroke in Children
After HS, neurological deterioration may progress for several days. One of the main causes of this is cerebral edema, increased ICP and secondary brain damage.213,214 So, in a study by Gebel J.M., et al, with IPH, the absolute volume of edema doubled in the first 24 h after treatment. 215
In childhood, intensive therapy of elevated ICP in HS is required, since children do not have cerebral atrophy of aging, which gives space to accommodate the mass effect. 191 In order to reduce sharply increased intracranial pressure, intracerebral hematomas must be removed, but it is not clear whether this leads to improved outcomes. 38 Beslow L.A., et al, propose to install an intraventricular catheter (IVC) for therapeutic as well as diagnostic purposes in some cases of pediatric HS. IVC will allow both drainage of cerebrospinal fluid and measurement of ICP. 29 Non–surgical methods for reducing elevated ICP include holding the headboard of the patient’s bed at an angle of 30°, which promotes venous drainage of the brain, as well as hyperventilation to pCO2 - 25-30 mmHg and reducing blood volume by stimulating osmotic diuresis. Hyperosmolar therapy with mannitol or hypertonic solution, as well as sedation, is recommended for this.6,215 Mannitol is not suitable for use when the osmotic plasma pressure is >320 mOsm, since high osmotic plasma pressure leads to dehydration, but mannitol can increase the osmotic pressure of the hematoma and change the osmotic pressure gradient.216–218
Alternatives to mannitol may be a hypertonic solution, which has a longer effect and does not violate the BBB, as well as furosemide. 219
Furosemide is allowed to be combined with mannitol or hypertonic solution.220,221
Glucocorticosteroid Therapy for Hemorrhagic Stroke in Children
Some authors believe that in conditions of severe stress effects in HS in children, including those caused by surgery under general anesthesia, replacement glucocorticosteroid therapy (GCS) is necessary.222–224
Lo W. it recommends the use of GCS for vasogenic edema of the brain. 13
However, hyperglycemia, suppression of the function of the hypothalamic-pituitary-adrenal system, infectious complications and diabetes are harmful as a result of the use of GCS.225,226
Treatment of Hemostasis in Hemorrhagic Stroke in Children
Recommendations for the management of hemostasis in HS in children require compensation for deficient blood clotting factors. 227 Thus, patients who have serious thrombocytopenia of non-immunological origin should be transfused platelet mass. Routine platelet transfusion is performed with a platelet count of less than 50 × 109 / L.228,229 Vitamin K-dependent HS should be prescribed vitamin K. To increase the level of coagulation factors, a prothrombin complex concentrate or freshly frozen plasma can also be used, which simultaneously reduces the risk of hematoma enlargement. However, the authors remind that these hemostatic measures pose a risk of thromboembolism. 13 To reduce the volume of bleeding and stabilize hematomas in children during the first 4 hours after HS, the use of recombinant factor VIIa is recommended.230,231
There is also a benefit from hematoma lysis with thrombolytics in cases of early treatment of HS.232,233 At the same time, some authors have identified the conversion of ischemic stroke to hemorrhagic stroke both naturally and with the introduction of a tissue plasminogen activator. 234 Thus, the effect of anticoagulants, thrombolytics on acute HS in children requires systematic study. 2
Treatment of Seizures in Hemorrhagic Stroke in Children
Al-Jarallah A., et al, found that seizures complicate childhood stroke in 10.3% of cases, Jordan L.C., et al, in 20%.31,193 Recurrent seizures increase the risk of recurrent hemorrhages, exacerbate brain damage and lead to negative long-term results. 235 In this regard, in clinical seizures, as well as convulsive activity, including those detected during EEG, treatment of this condition is required.38,236 Ferriero D.M., et al, recommend prescribing anticonvulsants for a short period of time after HS. 6 The first-line drug for the treatment of seizures is Phenobarbital, Phenytoin, the second line is Lidocaine, the third line is Midazolam, Levetiracetam, Clonazepam, Lorazepam.237–247
However, many authors note that there are no studies on the safety, efficacy, and specificity of the use of anticonvulsants in HS in various periods of childhood.38,215,248
Neuroprotection in Hemorrhagic Stroke in Children
Neuroprotection involves protecting the brain from secondary damage and degeneration in HS. Neuroprotective therapy includes medications, most of which are currently in clinical trials. 249
Relative neuroprotective properties are citicoline derivative, Edaravone, Erythropoietin, NeuroAid (MCLC601), Actovegin, magnesium sulfate (MgSO4), hypothermia. However, for the treatment of HS in children, these drugs need additional multicenter, randomized clinical trials, since the effectiveness of these neuroprotective agents was analyzed in ischemic stroke, in experimental animal studies, or the neuroprotective properties of these drugs had contradictory results.
If a child who has had HS has a treatable risk factor for stroke, the underlying disease should be treated. 38 There are recommendations for the treatment of some probable risk factors HS. For example, microsurgical and endovascular treatment methods are used in children with AVM, KA.47,250
It is useful to add folic acid, vitamin B6 and B12 to the treatment of hyperhomocysteinemia, along with weight control and diet changes. Children with SCD are recommended to receive blood transfusion for a long time for primary and secondary prevention of stroke, until a decrease in sickle cell hemoglobin <20-30% with hematocrit >30%.251,252 It is also recommended to administer hydroxyurea, hematopoietic stem cell transplantation, gene therapy, direct and indirect revascularization.253–255
Supportive treatment for CSVT should include adequate hydration, control of seizures, treatment of increased intracranial pressure, and thrombolytic therapy. 38
Calcium channel blockers, aspirin, and surgical revascularization methods are recommended for the treatment of MMD. 159
The Outcome of Hemorrhagic Stroke in Children
The average risk of recurrent HS in children is 20-35%. In children with one identified HS risk factor, the probability of recurrent HS is within 8%, with a combination of two or more factors, the recurrence of HS is 42%.28,256–258 Children with hematological disorders, vascular malformations, and tumors had a high risk of recurrence.28,32,259 In children with medical etiology (for example, thrombocytopenia, hypertension), the 5-year cumulative recurrence rate was 13%. However, there were no relapses in children with idiopathic HS. 37
Neurological Outcome in Hemorrhagic Stroke in Children
Long-term neurological deficit after HS was found in 45-50% of children.62,258 Two years after perinatal HS, moderate or severe deficiency was detected in 5% of children. 185
The nature of neurological complications after HS in older children was determined by hemiparesis in 25.0% of cases, aphasia (7.4%), epileptic seizures (10.3%), hydrocephalus (4.4%). 31 In the observations of Chang B., et al, of the 14 patients with HS, 41% of the survivors had long-term neurological disorders, including mental retardation (N = 11), epilepsy (N = 7) and hemiplegia (N = 10). Seizures at initial admission were a significant risk factor for long-term neurological deficits. 16
Blom I., et al, observed 36 patients after HS. Six children had epileptic seizures, 11 had hemiparesis of varying severity, 3 had symptoms of cerebellar ataxia, one child had persistent tetraparesis, and one had paraparesis. Signs of cognitive impairment were found in 15 patients. Most had low self-esteem, as well as emotional, behavioral, and health problems. 258 Some studies have revealed persistent neurological deficits in 65-66% of children who have had HS. Subsequently, problems with the development, education of children, as well as convulsive disorders developed.28,34,256,260,261
Other authors, in half of the children who suffered a HS, motor and cognitive impairments were subsequently observed.14,19,62,159 Beslow L.A., et al, after examining 26 patients with ICH, clarified that motor disorders were observed in 38% of survivors, and cognitive disorders in 50%. 29 During long-term follow-up of 42 children with spontaneous ICH, 3% of children retained visual deficits, speech delay within 25 - 36%.39,182 However, Bruno C.J., et al, argue that 1 year after perinatal HS, moderate or severe deficits are observed in few children. 262
There are studies that have studied the factors accompanying the adverse neurological outcome of HS in children. Thus, a more severe outcome of HS was observed in newborns and children <3 years old, as well as in the presence of aneurysms, hematological disorders, infratentorial hemorrhage location, a score on the Glasgow coma scale (SCG) at admission ≤ 7, a change in mental status within 6 hours after hospitalization.32,54 At the same time, Lo W.D. did not find a positive correlation between the initial Glasgow coma scale, the localization of bleeding and the outcome of HS. 13
The ratio of intracerebral hemorrhage volume to total brain volume was also important for HS outcomes: the volume of hemorrhage, which is 2% of the total brain volume, leads to moderate disability, 4% to severe disability.123,263 In a prospective, single-center, cohort study of children with ICH, with an average follow-up period of 3 to 7.5 months, neurological disorders were detected in 71% of survivors in patients with a hemorrhage volume of more than 2% of the total brain volume. At the same time, 55% of children had significant disabilities. 29 A poor result after HS was observed with a combination of several risk factors. 28
Mortality in Hemorrhagic Stroke in Children
Estimates of the annual mortality rate after HS in children vary in individual studies. Some authors claim that HS is among the top ten causes of death in childhood, which is 2-3 cases per 100 000 children. 11 Other authors give a mortality parameter of 0.6 cases per 100 000 children per year.28,264 Chung B., et al, determined that this indicator is 0.34 cases per 100 000 children. 16
Indicators of HS outcome in children.

Conclusion
Thus, an analysis of current knowledge about HS in childhood shows the variable nature of the results. This applies primarily to epidemiology, outcomes, as well as the cause of HS development in children. However, the information presented presents childhood HS as a common pathology that has a tendency to relapse, determines early disability and a high incidence of deaths. ICH is more often registered, then SAH. Information about other types of HS specified in the ICD 11 edition is limited in children. HS affects boys more often than girls. The causes of this phenomenon are unknown. HS is more common in children under 2 years of age.
Determination of the etiology, assessment of risk factors for HS in children is an area that requires systematic study with the formation of high-risk groups for the development of HS in children. This is explained by the fact that the nosologies that determine HS in children include numerous intra and extracerebral causes. Cerebrovascular pathology occupies a leading position in the spectrum of nosologies. Obvious risk factors for HS in children include impaired coagulating and anticoagulating properties of blood, cardiovascular, oncological diseases, and infections. The likely target group for HS development is children with CSVT, vasculitis. The state of health of the child’s parents, obstetric history, perinatal factors are important, especially in the implementation of perinatal HS. Rare genetic, hereditary diseases appear, in the structure of which HS can develop in children.
The multifactorial nature of HS in childhood determines the variety of clinical manifestations, which requires a timely differentiated, multidisciplinary approach to the diagnosis of HS.
It is necessary to search for the best strategies for providing neurointensive care to children in the acute period of HS, based on multicenter, randomized clinical trials and a convincing evidence base in accordance with the type of HS, age of children, predictors of the disease.
Instructions are required to optimize the respiratory function in HS in children with its violation, which include a list of indications for tracheal intubation, timing of connection to a ventilator, setting parameters and modes of ventilation, procedures for stopping ventilation. Infusion therapy is important in HS in children, which allows to maintain homeostasis, nutrition of the body, administration medicines, especially in conditions of restriction of oral administration of fluids, electrolytes, nutrition in seriously ill patients. However, there are no clear guidelines for determining the daily fluid requirement in children at different age periods, volume, quality of enteral nutrition, composition of infusion media in HS. The latter is especially important, given the cerebral edema, increased intracranial pressure accompanying HS.
Recommendations for the management of hemostasis in children with HS require clarification, in accordance with the direction of hemostatic disorders. There are few therapeutic possibilities for effective neuroprotection, symptomatic therapy of HS in children.
Footnotes
Author Contributions
Contribution of the author
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent to Publication:
All the authors have read the manuscript and agreed to it for publication.
