Abstract
We present a case of paraneoplastic tumefactive demyelination in a 55-year-old female with an underlying anaplastic thyroid carcinoma (ATC), alongside a review of the literature on all cases of tumefactive demyelination associated with non-CNS neoplasia. In the presented case the patient developed a right-sided subacute sensorimotor hemiparesis. The initial cerebral MRI revealed a bilateral frontoparietal tumefactive mass lesion with marked gadolinium uptake and mass effect. Cerebrospinal fluid revealed CSF-specific oligoclonal bands type III, with negative cell count, protein and pathogen testing. Brain biopsy indicated demyelination and T-cell infiltrates and foamy macrophages. A body CT revealed an anaplastic thyroid carcinoma. Despite steroids, plasma exchange, rituximab, and cancer treatment, the patient died due to clinical fluctuation and cancer progression. In addition to our case 9 cases of tumefactive demyelinating have been reported in patients with newly diagnosed extracranial neoplasia, most commonly seminoma germ cell tumour (7/10). 8/10 (80%) of patients were male, with mean age at diagnosis was 52.9 years 95% C.I. [43.8, 62.0]. 5/10 patients presented with sensorimotor hemiparesis and/or confusion/neurocognitive deficits. 4/10 with visual deficits and 2/10 with aphasia. In all cases neoplasia was diagnosed simultaneously or after neurological manifestations. All cases presented initially as solitary lesions. A malignancy specific-treatment as well as steroid treatment in different regiments were applied. In addition in 2/10 plasmapheresis was implemented and 1/10 patients received intravenous immunoglobulins. In the majority of cases including the presented case partial neurological improvement was documented whereas malignancy usually progressed. To our knowledge, this is the first report of paraneoplastic tumefactive demyelination associated with an ATC highlighting the importance of a thorough workup in these patients. This is the first reported case of paraneoplastic tumefactive demyelination associated with ATC, underscoring the necessity of a comprehensive diagnostic approach in similar patients.
Plain Language Summary
Why was this study done?
We reported a case of a rare brain condition called tumefactive demyelination in a patient with an aggressive type of thyroid cancer (anaplastic thyroid cancer, or ATC). Tumefactive demyelination is when the immune system attacks the protective covering of nerve fibers in the brain called myelin, creating large, tumor-like lesions. This condition is typically not seen in patients with cancer, so we wanted to explore this rare connection and review similar cases.
What did the researchers do?
We examined the case of a 55-year-old woman who developed weakness on one side of her body. Brain scans showed a large lesion in her brain, and further tests indicated myelin damage. A body scan later revealed she also had thyroid cancer. Despite treatment for both the brain lesion and the cancer, the patient’s condition worsened, and she passed away due to cancer progression. We also reviewed 9 similar cases of this brain condition occurring alongside cancer.
What did the researchers find?
In our case and the other cases reviewed, patients developed brain lesions around the time they were diagnosed with cancer. Most patients showed improvement in their neurological symptoms after treatment, but the cancer itself usually progressed. This highlights the difficulty in treating both conditions simultaneously.
What do the findings mean?
This is the first report of a patient with thyroid cancer developing this rare brain condition, and it suggests that thorough testing should be done for cancer patients who develop unusual neurological symptoms. Early detection and treatment of both conditions are important, but the prognosis remains challenging due to the aggressive nature of both cancer and brain lesions with inflamation.
Introduction
Tumefactive demyelination of the central nervous system (CNS) is an orphan neurological disease usually occurring in the context of multiple sclerosis (MS). It affects 1 to 3 per 1000 cases with an annual incidence of 0.3/100 000(1). These lesions can mimic brain tumors, which implies a significant diagnostic challenge, especially in the absence of other demyelinating lesions. It can be the initial presentation in various pathological entities with overlapping clinical and radiographic features. The majority of patients with TDLs have or later develop multiple sclerosis (MS) but a smaller number of cases experience a monophasic course or will be diagnosed with rarer disorders such as neuromyelitis optica spectrum disorders (NMOSD), myelinoligodendrocyte glycoprotein antibody-associated demyelination (MOGAD) or acute disseminated encephalomyelitis (ADEM).
Only nine cases of paraneoplastic tumefactive demyelination are described until now.2-10 Here we present a case of a 55-year-old woman with a tumefactive demyelinating lesion (TDL) without concomitant MS diagnosis, representing a paraneoplastic disorder with an underlying anaplastic thyroid cancer. In addition, we perform a review of the literature including all nine PubMed listed cases of tumefactive demyelinating lesions associated with non-CNS malignancies published.
Methods/Literature Search
The literature search was conducted in PubMed until February 08, 2024. The following keywords were used “paraneoplastic tumefactive demyelinating lesions” OR “paraneoplastic tumefactive demyelination” and a reference screening of the identified publications was performed (see Figure 1). Cases with paraneoplastic demyelination not representing tumefactive demyelinating lesions or cases with demyelination in the context or coexisting with classical autoimmune/paraneoplastic neurological syndromes such as autoimmune encephalitis or autoimmune cerebellar degeneration were beyond the scope of this work and where not included in the literature review. Structured Literature Search Presented in a Flow Chart.
Several cases and case series are described in the literature with tumor-like demyelinating lesions in connection to CNS-lymphoma.11-17 These lesions represent a separate entity known as “sentinel lesions of primary central nervous system lymphoma (PCNSL)” and therefore where not included in the present literature review but are discussed separately in the section “discussion”.
Case Report
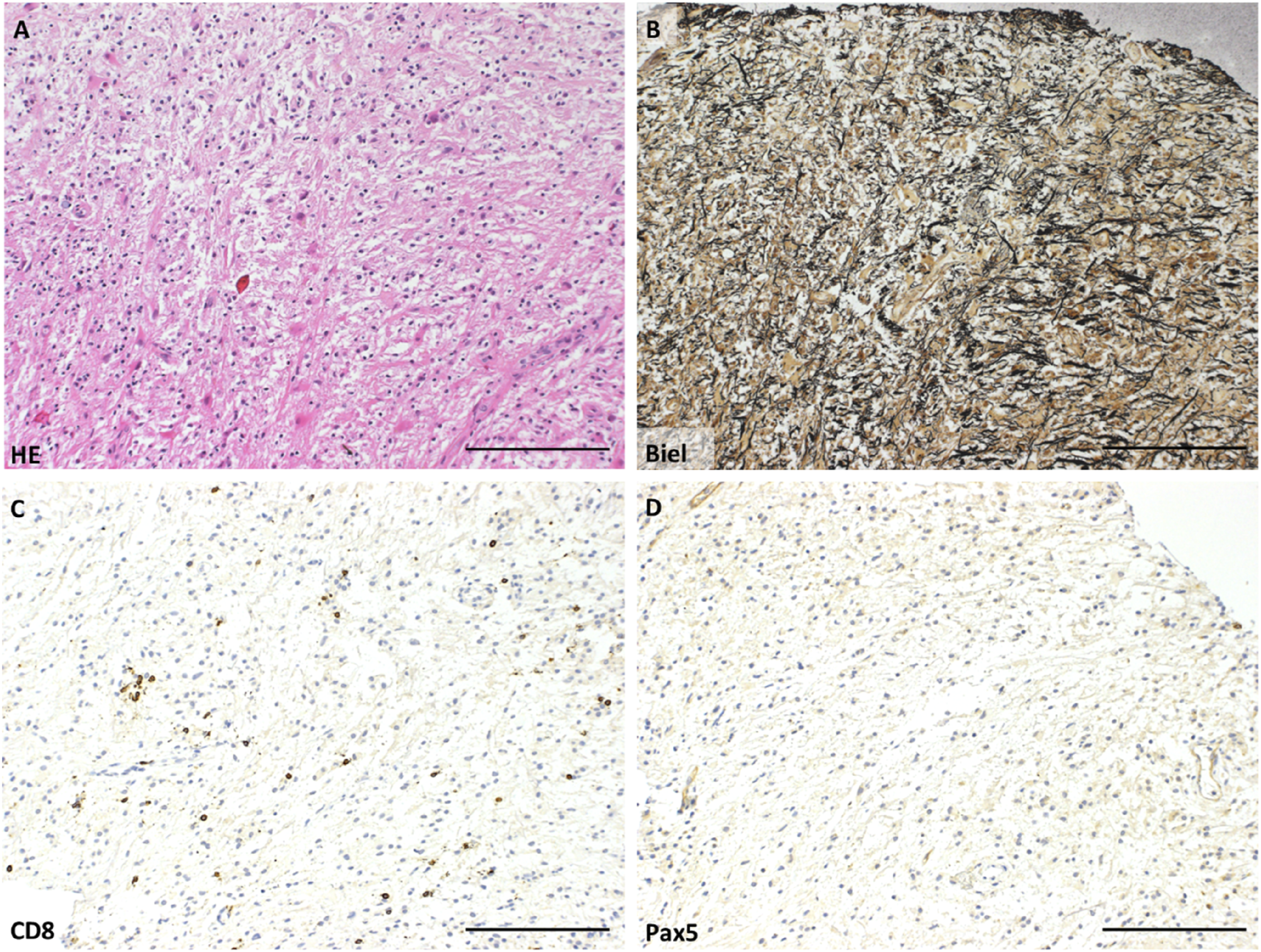
A previously healthy 55-year-old female patient developed a right-sided subacute progressive sensorimotor hemiparesis. The initial cerebral MRI (cMRI) revealed a mass lesion extending from the left frontoparietal periventricular white matter over the corpus callosum to the right frontal periventricular area with peripheral gadolinium uptake, perifocal flair hyperintensities and some space occupying effect with compression of the posterior horn of the left lateral ventricle (Figure 2). A stereotactic biopsy of the cerebral lesion revealed a demyelinating inflammatory pattern with lymphocytic infiltrates and foamy macrophages in a perivascular and parenchymal location. A CNS neoplasia was ruled out (Figures 3 and 4). Due to clinical severity without sufficient treatment response and to rule out a false negative result concerning neoplasia biopsy was performed twice with comparable findings. A broad laboratory work up demonstrated an inflammatory CSF profile with CSF-specific oligoclonal bands (OCBs) type III but no other relevant findings (Table 1). Initial MRI. Axial T2 (A), T1 Post Contrast (B), DWI (C) and ADC Map (D) Show a Left Frontoparietal Well Defined Lesion With Peripheral Spiculated and Linear Enhancement With Peripheral Diffusion Restriction Representing Active Demyelination (Advancing Front). The Lesion on the Left Side was Also Extending to the Corpus Callosum (Not Shown). No Significant Space Occupying Character. There is Also Punctate Enhancement on the Right. (A) H/E -Staining Showing a Macrophage-Rich Lesion, With Reactive Astrocytes. No Evidence of Malignant cells. (B) Bielschowsky Silver Impregnation Shows Preserved Axons. No Evidence of Necrotic lesions. (C) CD8 Staining Shows Single CD8 + Cytotoxic lymphocytes. (D) Pax-5-Staining Shows No Evidence of Pax5 + B Cells, Arguing Against Lymphoma. A, B, C, D Scale bar 100 µm. (A, B) Proteolipid Protein Staining (PLP) Shows Demyelination (A) and Macrophages Phagocyting PLP (B). (C) Myelin Oligodendrocyte Glycoprotein Staining (MOG) Shows Macrophages Phagocyting MOG (Evidence of Early Active Demyelinating). (A) Scale bar = 100 µm, B and C Scale bar = 50 µm. Presentation of the Relevant Laboratory Diagnostics Performed in the Presented Case.


A body CT demonstrated a hyperplastic left thyroid lobe without signs of lymphatic or hematogenous metastasis. In neck ultrasound scan the lesion measured 30 × 44 × 55 mm, exhibited an inhomogeneous structure, sharp borders, and colloid granulations (Figure 5), a fine needle biopsy confirmed a Bethesda category V carcinoma with squamous differentiation (Table 2). (A) Computer Tomography of the Neck Revealing a Large Heterogeneous Lesion Measuring 47 mm With Peripheral Enhancement and Small Calcifications in the Left Thyroid Lobe Resulting in a Mild Displacement of the Trachea to the Right. No Pathologically Enlarged Thoracic Lymph nodes. (B) Thyroid Ultrasonography Showing a Heterogeneous, Well-Defined, Solid, Predominantly Hypoechoic Mass With Macrocalcifications and Minimal Vascularization (Not Shown) of the Left Thyroid Lobe Measuring 51 × 39 × 41 mm. Epidemiological, Clinical, Radiological and Pathological Characteristics of all Reported Cases With Paraneoplastic Tumefactive Demyelination Lesions. Abbreviations: Abs, antibodies; ACE, angiotensin converting enzyme; ANA, Antinuclear Anbiody, anti-ds-DNA: anti-double stranded DNA; ANCA: Antineutrophil Cytoplasmic Antibodies; ANNA3, Antineuronal Nuclear Antibody Type 3; Anti-AQP4, anti-Aquaporin 4; Anti-MOG: anti- myelin oligodendrocyte glycoprotein (MOG); Anti-Ri, anti-neuronal nuclear antibody 2 (ANNA2); Anti-Yo, Purkinje cell antibody type 1; Anti-Hu, antineuronal nuclear antibody-type 1, ANNA-1; b.c.c: blood cell count; CASPR-2, contactin associated protein-2; CRMP-5, collapsin response mediator protein-5; CSF, cerebrospinal fluid; CT: computer tomography; ENA: extractable nuclear antigen; FDG-PET, fluorodeoxyglucose- Positron-Emission-Tomography; GAD,glutamatedecarboxylase; GABA, γ-aminobutyric acid; HIV, human immunodeficiency virus;; Lg-1, leucine-rich gliomainactivated 1; Ma-1/Ma-2, paraneoplastic neuronal antigen; NMDA, N-methyl-d-aspartate; NAA, N-acetylaspartate; PCA2, Purkinje Cell Cytoplasmic Antibody Type 2; RF, Rheumatoid factor; SOX-1, anti-glial nuclear antibody (AGNA) and sex-determining region Y-box; Tr (DNER), delta/notch-like epidermal growth factor related receptor; Zic4, zinc finger protein. IgG, IgM, immunoglobulin G, M; Gd, Gadolinium ; LDH, Lactate Dehydrogenase; OCB, oligoclonal bands; RNP, ribonucleoprotein; SS-A/SS-B, Sjögren’s syndrome A/B.

A total thyroidectomy was performed due to infiltration of the prelaryngeal musculature and adherence to surrounding tissues, the surgery included resection of the involved muscles and mobilization of the enlarged thyroid gland. Neck dissection at levels VI and VII was conducted because of the presence of enlarged adherent lymph nodes. Intraoperative frozen section analysis confirmed anaplastic thyroid carcinoma without differentiated components. Postoperatively, the surgical incision margins were tumor-free, confirming localized disease at the time of surgery. Histopathology confirmed the diagnosis of anaplastic thyroid carcinoma. The tumor was confined to the thyroid gland without evidence of extrathyroidal spread to distant lymph nodes. The tumor size, 30 × 44 × 55 mm, and infiltration of surrounding musculature were consistent with advanced disease. We diagnosed a paraneoplastic tumefactive demyelination of the central nervous system, which was the first clinical manifestation of the cancer occurring prior to metastasis or other cancer related local or wasting symptoms. Treatment was as follows: In addition to the dexamethasone (8 mg per day), which had led to a partial improvement of her neurological symptoms, high dose methylprednisolone iv treatment (1000 mg/d over 3 consecutive days) and subsequently 5 sessions of plasmapheresis every other day were started and some additional clinical improvement was observed.
Tumor-specific treatment in the localized disease stage consisted of postoperative radiotherapy (ED 2 Gy, GD 66 Gy) combined with weekly chemotherapy with doxorubicin.
Subsequently the patient rapidly deteriorated and progressed exhibiting a spastic tetraparesis, an apraxia and a left tactile extinction. The patient did not develop any clinical findings consistent with involvement of the peripheral nervous system. In particular, there were no findings of muscle atrophy, trophic disturbances, hyporeflexia, or sensory disturbances consistent with polyneuropathy, mononeuropathy, or radiculopathy.
A cerebral MRI indicated several new FLAIR/T2 hyperintense, gadolinium enhancing lesions sub-/juxtacortikal and on the right cerebellar peduncle. Again, a steroid pulse treatment (Methylprednisolon 1000 mg/d for 3 days with subsequent oral tapering) and rituximab (MabThera®, Roche, Switzerland; 1000 mg twice) was combined with the above-described radio-chemotherapy.
Eight weeks later, a local relapse of thyroid cancer and new lung metastases were diagnosed. The patient was diagnosed with community-acquired pneumonia and was treated with Co-Amoxicillin. Later that month the patient was admitted to the oncology ward due to exacerbation of thoracic pain and exhaustion with impaired mobility, in the context of a pre-existing right-sided hemiparesis, Clinical examination revealed bilateral basal crackles. Laboratory findings demonstrated markedly elevated inflammatory markers. A chest X-ray showed progression of perihilar opacities, increasing signs of congestion, and bilateral compressive atelectasis. Given the suspicion of nosocomial pneumonia, antibiotic therapy with Cefepime was initiated. Tests for urinary Legionella and pneumococcal antigens were negative, as was a COVID-19 swab. Under the prescribed therapy, inflammatory markers regressed, and the antibiotic treatment was discontinued after seven days.
During the subsequent course, the patient experienced progressive clinical deterioration and worsening dyspnea. Despite decreasing inflammatory markers, these symptoms were interpreted as multifactorial, likely due to pleural effusions and pulmonary metastases. On November 21, 2021, thoracentesis was performed, draining 900 mL of pleural fluid. This provided temporary improvement in respiratory symptoms. However, a rapid decline in the patient’s general condition ensued, with worsening dyspnea.
Prior to this, the patient and her partner had decided against intensive care measures, including mechanical ventilation. Therefore, comfort care was initiated. The Patient passed away a few day later. (see Figure 6 for detailed timeline of clinical events). Timeline of the Relevant Clinical Events, Diagnostic Examinations (in Light Blue) and Treatment (in Green).
Review of the Literature
We report the first case of a paraneoplastic tumefactive demyelination (TDL) associated with an anaplastic thyroid carcinoma. In the literature 9 cases of TDL connected to non-CNS malignancies have been described (2-10, Figure 7). Together with our case 7/10 patients had seminomas or germ cell carcinomas as underlying malignancies, 1/10 had a renal cell carcinoma,
6
a chronic myelomoncytic leukemia,
2
and an anaplastic thyroid carcinoma. Pie Chart Representation of all the Published Cases of Paraneoplastic Tumefactive Demyelination.
8/10 (80%) of the patients with paraneoplastic tumefactive demyelinating lesion were male leading to a female to male ratio of 1:4, which is remarkable for autoimmune diseases. Mean age at diagnosis was 52.9 years (95% Confidence Interval [43.8, 62.0], min 32, max 71).
In the majority of published cases including our case the malignancy was discovered either simultaneously4,5,9 or shortly after the diagnosis of the TDL3,8 usually as a result of the work up for the Tumefactive brain lesion. In one case 6 the malignancy was discovered 21 months later and in one case 10 a testicular mass later diagnosed as seminoma was known to the patient for 3 years before the diagnosis of TDL but didn’t seek medical attention. In 2 cases2,7 the frame of the diagnosis of the 2 entities was not clearly defined.
Clinical presentation was as follows: Sensorimotor hemiparesis and confusion/neurocognitive deficits were most common and occurred in 5/10 each, followed by visual deficits (4/10) and aphasia (2/10, Table 2). Information about imaging findings were available in 9/10 cases. All cases presented initially as solitary lesions. At least some mass effect was seen in 6/9 lesions, Gadolinium enhancement was evident in 5/9 lesions.
Biopsy was performed in all cases but one 5 revealing demyelination with inflammatory infiltrations with a predominance of CD68-immunoreactive macrophages and perivascular lymphocytic infiltration.
CSF analysis was available in 7/10 cases. In 4/7 cases CSF results were normal.5,7,8,10
CSF pleocytosis is described in 1/7 case 6 and elevated protein in 2/7 cases.3,6 CSF-specific oligoclonal bands were only present in our case (1/7).3,8-10
In all cases, where clinical data were documented, a malignancy specific-treatment (chemotherapy and/or radiation and/or tumor excision) as well as steroid treatment in different regiments were applied. In 4 cases i.v. methylprednisolone was applied with or without oral tapering, in 2 cases in a dosing of 1000 mg/d for 3 days, in 1 case in a dosage of 160 mg/d. In one case the dosage was not defined. Dexamethasone was given in 6 cases (8 mg/d in two cases, 12 mg/d in one case, 16 mg/d in one case, dosage not defined in two cases). In one the exact regimen of oral steroids was not documented. In 2/10 cases , including our case, plasmapheresis was implemented 7 and 1/10 patients received iv immunoglobulins (dosage unknown). 10
In the majority of the cases including the presented case partial neurological improvement was documented at least at some point in the disease course,6-10 whereas malignancy usually progresses despite anti-cancer treatment.
Discussion
Here we present a patient with paraneoplastic tumefactive demyelination with underlying anaplastic thyroid cancer and demonstrate the difficulties in diagnosing and managing this emerge disease.
In general, tumefactive demyelination (TDL) associated with an anaplastic thyroid carcinoma has not been described thus far. Anaplastic thyroid carcinoma (ATC) is rare, where disease management is closely tied to prognosis resulting in a median survival of 6.1 months and an 1-year overall survival of 20 %. 18 Therapy management depends on disease stage, but is mostly combined by surgery, chemotherapy and radiation. 19 Location of distant metastasis are with decreasing frequency lung, bone, liver, brain, adrenal and abdominal. 19 Although brain metastasis are rarely seen, tumefactive demyelination was never reported.
TMD was historically regarded as an unusual presentation of multiple sclerosis. Nowadays recent advances in neuropathology and neuroradiology have shown that TDL represent an umbrella term for many different diagnostic entities. 20
Disease-modifying drugs (e.g. fingolimod, natalizumab) used in MS have been associated with the occurrence of TDLs, especially after drug initiation or cessation. 21 Moreover, various pathological entities such as atypical MS variants (Marburg’s type, Balo’s concentric sclerosis, Schilder’s disease, acute disseminated encephalomyelitis) as well as Neuromyelitis optica spectrum disorders (NMOSD) and Myelin Oligodendrocyte Glycoprotein Antibody associated disease (MOGAD) have been shown to present with tumefactive lesions.1,21,22 TDLs can however be caused by a variety of disorders (infectious diseases, other systemic or neurological autoimmune diseases, medications, paraneoplastic aetiology) as well as an idiopathic monophasic or relapsing disease with recurrent TDL.1,20 Recently two studies tried to address the clinical, radiological as well as immunopathological differences of TDLs in different contexts, especially in context of MS, NMOSD and MOGAD(20,23), whereas a recent review discusses the pathophysiology and clinical findings in these diseases as well as the current, emerging and experimental treatments. 24 According to these studies close clinical follow-up, radiological features and eventually immunopathology can help us to stratify these lesions in disease entities and better organize treatment strategies. The radiological findings of the TDL in our case not typical for a MS (Central vein sign, hypointense rim), NMOSD or MOGAD as described by Cacciaguerra et al. 23
Interestingly in our case the pattern of involvement of the perivascular spaces and subependymal region could represents spreading of the inflammation via the glymphatic pathway (Figure 8). Longitudinal Follow-Up With Coronal T1 Post Contrast Showing the Lesion Dynamic. On the Initial Presentation (A) the Lesion Presented on the Left Side With Peripheral Spiculated and Linear Enhancement With Infiltration of the Corpus Callosum Reaching the Subependymal Region. Linear Enhancement on the Right Side was Also Seen Representing Enhancement of the Perivascular Spaces, which Increased on the Follow-Up Exams (B) Later on (C) Further Extension of the Enhancement in the Subependymal Region was Seen on the Left Side, as Well as New Lesions in the White Matter. This Pattern of Involvement of the Perivascular Spaces Represents Spreading of the Inflammation via the Glymphatic Pathway. The Subependymal Region Also Plays an Important Role in Brain Fluid Dynamics and Clearance. No Central Vein Sing or Hypointence Rim Lesions where Seen.
Differentiating tumefactive demyelinating lesions (TDLs) from gliomas or CNS lymphomas based on radiological features remains a significant challenge. Although no pathognomonic imaging signs exist, certain characteristics suggest demyelination. These include relatively mild edema and mass effect compared to lesion size, open-ring enhancement, diffusion restriction, and low perfusion on brain MRI.1,25,26 Interestingly, a small retrospective study reported that areas of enhancement in TDLs on MRI tend to appear hypodense on unenhanced CT scans. 27
In magnetic resonance spectroscopy (MRS), TDLs may show an increased choline-to-N-acetylaspartate (Cho/NAA) ratio, a finding also observed in neoplasms. Additionally, increased glutamate/glutamine peaks may support a diagnosis of TDL. Serial MRS is more informative than single-time-point imaging, as TDLs often demonstrate variable findings over time, while neoplasms typically show consistent progression. 1
In our case, the involvement of the frontal and parietal lobes, the lesion’s location within the white matter, peripheral diffusion restriction, and low relative cerebral blood volume (rCBV) were indicative of a tumefactive lesion. MRI spectroscopy findings were inconclusive: the absence of myelin breakdown products argued against glioma; the lack of glutamate and glutamine was inconsistent with a tumefactive lesion; and the absence of lipids and contrast leakage argued against lymphoma. Ultimately, pathological analysis revealed activated complement components within myelin-phagocytosing macrophages. This finding, along with nearly complete myelin loss, mild perivascularly accentuated inflammation, and the presence of an active macrophage border, made a diagnosis of steroid-pretreated lymphoma unlikely. Furthermore, Pax-5 staining showed no evidence of Pax-5+ B cells, further ruling out lymphoma.
In very rare cases, primary central nervous system lymphomas (PCNSL) initially present with steroid-responsive, multifocal demyelinating “sentinel” lesions, often representing TDLs.12,13,13,14,16,20 The exact pathomechanism of this phenomenon is not clear and different factors such as randomly coincidental findings, missed first biopsy, disruption of anti-tumor immune response by steroids, malignant transformation of TDL lesions to lymphoma during latent period and masking of diagnosis by corticosteroids have been proposed. 14
In cases where tumefactive demyelination occurs after brain radiation, post-radiation brain injury should be considered a plausible differential diagnosis. Radiation-induced neurotoxicity, a known side effect in cancer patients, primarily affects oligodendrocyte-lineage neural precursor cells, disrupting axonal myelination. Pathologic demyelination after therapeutic radiation depends on factors such as tumor volume, radiation dose, treatment field, and patient age.28,29
Post-treatment imaging of radiation-induced changes can be confusing and is categorized into acute, early delayed, and late delayed phases based on the time of onset. Acute and early delayed injuries are usually transient and reversible, while late delayed injuries tend to be irreversible. Katsura et al. reviewed the timeline and imaging features of radiation-induced changes, offering a framework for interpreting post-treatment scans. 28
Although clinical and imaging features are not always reliable, certain characteristics may help differentiate radiation necrosis from tumor progression. These include the time elapsed since radiation therapy, 30 combined MRI patterns,31,32 and findings from MR perfusion and spectroscopy. For instance, recurrent brain tumors often exhibit high Cho/Cr and Cho/NAA ratios, while radiation necrosis is associated with increased lactate and lipid peaks.33,34 Hypoperfusion with reduced rCBV is typical in radiation necrosis, whereas high-grade tumors show elevated rCBV. 35
To date, no studies have systematically compared the clinical and radiological features of tumefactive demyelination and post-radiation injury. In cases where imaging findings worsen over time or symptoms progress, biopsy remains the definitive method for distinguishing between these entities.
A rare but important differential diagnosis for TDLs includes leukodystrophies.36,37 Thakkar et al. resently presented a detailed review of MRI findings for various leukodystrophies, including associated genetic and laboratory findings, which can aid in differentiation. 38
Concerning the management of patients with paraneoplastic neurological syndromes, it is critical to emphasize the importance of a careful weighing of risks and benefits when using both immunotherapy and tumor treatment. A nuanced approach ensures that treatment strategies are tailored to each patient’s unique clinical scenario, optimizing outcomes while minimizing potential risks. Furthermore In the context of neurological paraneoplastic syndromes (PNS), immunosuppressive agents used to treat the PNS may be a double edged sword — negatively impacting tumor progression via reduction of immune mediated cancer control while damping PNS symptoms. However, the effect of chemotherapy on the immune-mediated aspects of PNS is complex and may depend on several factors, including the type of malignancy, the specific chemotherapy used, and the nature of the paraneoplastic syndrome.
In our case, the patient received corticosteroids and rituximab to manage the TDL, alongside treatment for the anaplastic thyroid tumor, a malignancy known for its aggressive nature and poor prognosis. Corticosteroids, while critical for managing demyelination-related inflammation, are well-documented to have immunosuppressive effects, including dampening T-cell-mediated immune responses. This could theoretically reduce anti-tumor immune surveillance and potentially contribute to tumor progression. Rituximab, which primarily targets B-cells, has a distinct immunological profile compared to corticosteroids. While this does not rule out any indirect effects, it implies a potentially lower risk of promoting malignancy progression through the modulation of T-cell responses. It is generally challenging to definitively attribute the rapid progression of the malignancy in this case to immunosuppressive therapy. Additionally, the timing and duration of immunosuppressive therapy relative to tumor progression further complicate establishing a direct causal relationship. The aggressive biology of anaplastic thyroid carcinoma, characterized by rapid growth and resistance to conventional therapies, likely played a significant role.
The limited number of cases describing paraneoplastic tumefactive demyelination precludes systematic comparison of clinical, radiological, and histological features with other TDL etiologies. This represents a limitation of our study. However, findings from the 10 published cases of paraneoplastic TDLs generally align with those of tumefactive demyelination, typically manifesting as a monophasic disease or in the context of multiple sclerosis.
In all reported cases, the diagnosis of paraneoplastic syndrome was based on the coexistence of demyelination and malignancy. There is no evidence to support a definitive causal relationship between anaplastic thyroid cancer and TDL. As acknowledged in the literature, no specific antibodies have yet been linked to paraneoplastic TDLs, further supporting the idea that the mechanism underlying these cases remains cell and not antibody mediated, however this is still speculative. Nevertheless, while no direct causality has been established, one can consider the rarity of both conditions—anaplastic thyroid cancer and tumefactive demyelination- and the strict timely association as hinds for claiming an association between both diseases. Further investigations are needed.
Interestingly, the majority of published cases of paraneoplastic TDLs were associated with seminomas or germ cell tumors. This type of tumor is also associated with well-recognized paraneoplastic neurological syndromes and neuronal antibodies, such as brainstem/cerebellar syndromes with Kelch-like Protein 11 (KLHL11) antibodies, encephalitis, diencephalitis/brainstem encephalitis with Ma- or Ma2 antibodies, encephalitis with Anti-Ta-, NMDA-R, and anti-CRMP-5 antibodies.39,40 The known causal relationship of seminomas/germ cell tumors with these immune-mediated neurological syndromes further supports the possibility of a causal relationship also between malignancies, especially seminomas, and TDLs.
Conclusion
This study highlights the first reported case of paraneoplastic tumefactive demyelination associated with anaplastic thyroid carcinoma (ATC). Awareness of demyelinating lesions with paraneoplastic aetiology also in cancers such as ATC should be raised. Especially in single TDLs nor fulfilling the criteria/without typical clinical and radiological findings for a multiple sclerosis a broad paraclinical work up including whole body imaging in search of a malignancy is of importance. In paraneoplastic cases rapid treatment of the inflammation as well as the malignancy appears to be the key factor for prognosis. Future studies are necessary to elucidate the pathophysiological mechanisms underlying such rare associations.
Footnotes
Author Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
•
•
•
•
•
•
•
•
•
Data Availability Statement
Data supporting the findings of this study are available from the corresponding author upon request.
