Abstract
Background
Multiple sclerosis (MS) is an autoimmune disease leading to physical, emotional and cognitive disability. High body mass index (BMI) may impact cognitive function and brain volume in MS. Yet, there is paucity of evidence addressing the impact of BMI on cognitive function and brain volume in MS.
Objectives
The purpose of this study was to examine the effects of BMI on normal appearing brain volume and cognitive function in patients with relapsing–remitting MS.
Methods
A secondary data analysis of the NIH CombiRx study was conducted. Multivariate regression and mixed model analyses were executed to analyze the effect of BMI on brain volume and cognitive function.
Results
The mean baseline age of the 768 participants was 38.2(SD = 9.4) years. 73% were female and 88.8% were Caucasian. The mean BMI was 28.8 kg/m2(SD = 6.7). The multivariate regression and mixed model analyses failed to show a clinical effect of BMI on brain volume and cognitive function.
Conclusion
BMI did not show an effect on cognitive function and brain volume among MS patients. Although there is increased interest in the effects of modifiable factors on the course of MS, the effects of BMI on brain volume and cognitive function are debatable and warrant further research.
ClinicalTrials.gov NCT00211887
Introduction
Multiple sclerosis (MS) is a neurological autoimmune, inflammatory degenerative disease leading to physical, emotional, and cognitive disability among young adults.1,2 Aside from race and genetics, which are non-modifiable risk factors for MS, recent studies have shown an association between risk of MS in adolescents and young adults and modifiable factors, such as obesity. 3 In fact, obesity has been recognized recently as a modifiable emerging risk factor for MS by the American Academy of Neurology (AAN), 4 with 70% of MS patients obese or overweight.5,6
Obesity has been linked with cognitive dysfunction and brain volume loss in healthy adults,7,8 and brain volume loss has been recognized as one of the best predictors for cognitive impairment in MS.9,10 Although multiple studies have shown a link between cognitive impairment and brain volume loss in MS, it is unclear if this association is triggered by modifiable risk factors such as body mass index (BMI) and/or non-modifiable factors, such as genetics, or a combination of both. Cross-sectional studies,11-18 provided preliminary and partial support for the relationship between high BMI and brain volume loss and cognitive dysfunction in MS, but there are also conflicting results. For example, Bove and colleagues (2019) and Galioto and colleagues (2019) did not show an association between BMI and cognition but Owji and colleagues (2019) demonstrated a negative correlation between BMI and cognitive function as measured by the Paced Auditory Serial Addition Test (PASAT) and Symbol Digit Modalities Test (SDMT). The role of BMI in MS continues to be controversial; therefore, there is a critical need for gaining a better understanding of the effect of BMI on cognitive function and brain volume in this population.
This study addressed a major methodological limitation of prior studies. In particular, most of the prior studies have been cross-sectional studies as compared to this study, which was based on a longitudinal randomized control trial (RCT) for 3 years. The purpose of this study was to examine the effects of BMI on normal appearing brain volume and cognitive function in adult patients with relapsing–remitting MS (RRMS) treated with interferon-β or glatiramer acetate while controlling for potential confounders of age, sex, ethnicity, duration of illness from diagnosis and from first symptom, relapses, disability, MS medications, and smoking. The revised Scaffolding Theory of Aging and Cognition (STAC-R) guided this study.19,20 The STAC-R consists of a model linking lifestyle activities, biological factors, cognition, and brain volume, depicting life course experiences that may enrich or deplete neuronal functions. The scaffolding model suggests that individuals with MS who accrue multiple neural insults throughout the course of their illness will exhibit loss of brain volume and poor cognitive function.19,20 Hence, we hypothesized that high BMI may accelerate brain volume loss and cognitive dysfunction.
Methodology
Parent Trial-CombiRx and ethics statement
This study is a secondary data analysis of the CombiRx trial, a phase III, multicenter RCT sponsored by the National Institutes of Neurological Disorders and Stroke (US NIH Grant/Contract U01NS045719, R21NS41986; NIH identifier number NCT00211887). This trial randomized individuals to one of three disease-modifying therapies (DMTs): interferon (25%), glatiramer acetate (25%) or both interferon and glatiramer acetate (50%). Participants were naïve to treatment at entry. The CombiRx trial was approved by the applicable central or institutional review boards and the Data and Safety Monitoring Committee (DSMC) appointed by National Institutes of Health (NIH)/ National Institute of Neurological Disorders and Stroke (NINDS) before site initiation and recruitment of participants. Written, informed consent was obtained prior to any screening procedures or enrollment. The trial was registered at www.clinicaltrials.gov/ct2/show/NCT00211887; for more information, please see www.CombiRx.org).21,22 The present study utilized deidentified data for the secondary analysis and was considered exempt by Mount Sinai Icahn School of Medicine institutional review board (IRB-16-1247) and New York University institutional review board (IRB-FY2019-2463).
Secondary Analysis Study
A total of 768/1008 patients completed the 3 years and were included in this secondary analysis, as they had sequential brain MRI films and brain volume calculations, BMI, and cognitive function measurements for 3 years. White and gray matter volume at 36th month was missing in 219 patients with RRMS in the CombiRx study (21.7%). Intention to treat (ITT) analysis of the full sample (n = 1008) was performed with 5 replicates of imputation based on BMI and brain volume using IBM SPSS version 23. 23 There were no exclusion criteria in this secondary data analysis.
Study Measures
BMI was calculated based on weight in kilograms divided by the square of height in meters, which were measured during the study every 3 months by the research team. BMI was evaluated both as a continuous variable and as a categorical variable divided into normal (<25 kg/m2), overweight (25-29.9 kg/m2), and obese (≥30 kg/m2). Brain volume was acquired using a standardized protocol that included 7 separate scan series.21,22,24,25 The brain volume analyzed in this study was based on the normal appearing white matter (NAWM) and normal appearing gray matter (NAGM). MRI abnormalities are seen in NAWM and NAGM in early RRMS and the lack of correlation between NAWM or NAGM and lesion abnormalities suggests that they are developed by partly independent mechanisms.26-29 Cognitive function was assessed by the PASAT. The PASAT tests memory, speed of information processing, concentration, and attention, with scores from 0–60 while higher scores indicating better cognitive performance.30-32 In MS studies, the PASAT Cronbach’s alpha was .90 and the test–retest coefficients ranged between .90 and .97.30,31,33 Assessment of construct validity of the PASAT showed good correlations with other cognitive tests on attention, working memory, processing ability, and speed.32,33 The CombiRx study included 2 PASAT screening visits before the baseline visit in an effort to diminish the well-known learning curve for the test. Substantial improvement of the PASAT between the first and second screening visits was observed, with a smaller change between the second screening visit and baseline.21,24 Although the assumption of homoscedasticity of errors of the PASAT was met, it had multiple outliers and its residuals did not follow a normal distribution. Therefore, in this study, the PASAT was dichotomized with a cutoff of 53 based on the PASAT median of the sample.
Continuous independent variables included age, duration of illness from first symptom in years, and number of relapses in the last 3 years. Categorical variables included sex, race (Caucasian, African American, other), disease modifying therapies (DMTs), systolic and diastolic blood pressure, and smoking (ex-smoker, never smoked and current smoker). Lastly, disability was measured by the Expanded Disability Status Scale (EDSS) with entry criteria of EDSS <6.0. The upper limit of the EDSS was determined by the CombiRx researchers. Their goal was to include only naïve relapsing–remitting patients and not those that transition into secondary progressive MS. The EDSS score was determined based on a neurological exam every 3 months by the neurologist. The EDSS quantifies disability in MS with scores from 0–10 in .5 point increments with higher scores indicating higher disability and monitors changes in the level of disability over time.
Data Analysis and Management
Excel data sheets with de-identified data provided by the principal investigators of the CombiRx trial were imported into IBM SPSS version 23 23 for data analyses. The statistical significance for this secondary data analysis study with non-directional hypotheses was set at a 2-tailed alpha level of .05. Descriptive statistics were performed to portray the sample characteristics. In addition, Chi-square and t-tests were performed to analyze the association between categorical and continuous variables. Multivariate linear regression, multivariate logistic regression, and mixed model of interaction with time analyses were executed to analyze the effect of BMI on brain volume or cognitive function while controlling for age, sex, ethnicity, smoking, blood pressure, disease duration, relapses, and disability.
Results
Demographics and Disease Related Variables
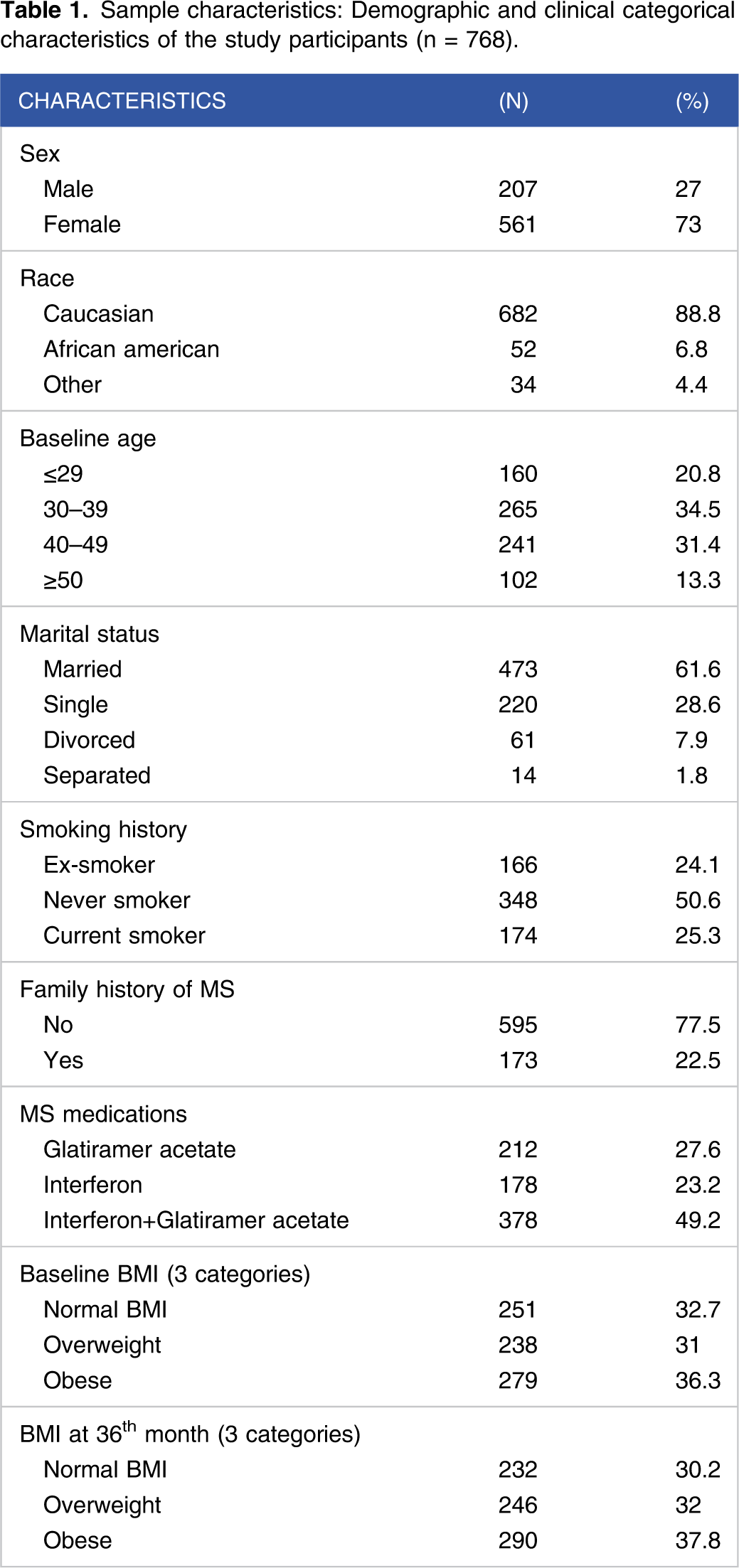
Sample characteristics: Demographic and clinical categorical characteristics of the study participants (n = 768).
Sample characteristics: Demographic and clinical continuous characteristics of the study participants (n = 768).

Baseline and month 36th summary of changes between baseline and month 36 with effect size (n = 768).

Note. The brain volume was evaluated based on paired t-tests using SPSS IBM version 23. The BMI and categorical cognitive function (PASAT) were evaluated based on paired t-test (continuous) and McNemar test (categorical). Cohen’s d effect size for 2-tail t-test was calculated based on the absolute value of the mean difference between the baseline and 36th month groups divided by .5 times the pooled standard deviation (Cohen’s d = |m2 – m1|/[.5(sd1 + sd2)], n1 = n2).
BMI, Cognitive Function, NAWM, and NAGM Brain Volume
The multivariable logistic regression analyses and the mixed model analysis failed to show an effect of BMI on cognitive function in patients with RRMS. BMI was evaluated for effects on cognitive function as a continuous variable and as a categorical variable due to its meaningful clinical implications. Least squares means adjusted for age, sex, race, and treatment group showed mean values of 2.7 for BMI < 25 kg/m2; 3.2 for those ≥25 kg/m2 and <30 kg/m2; and 2.0 for those with BMI ≥30 kg/m2 (P = .21).
BMI and Normal Appearing Brain Volume or Cognitive Function at Baseline and 36th Month (n = 768).

Hierarchical linear regression model for baseline BMI and the 36th month white matter (n = 768).

Note. Dependent variable = white matter brain volume at 36th month; Duration Dx = duration of illness from diagnosis; SBP = systolic blood pressure; DBP = diastolic blood pressure.
Mixed model for repeated measures analysis: Baseline BMI/continuous, time, and normal appearing gray matter brain volume longitudinally (n = 768).

Dependent Variable: Gray Matter Brain Volume. EDSS = Expanded Disability Status Scale; ref = reference group. The interaction of baseline BMI*Time = F(1/2684.13) = 9.435, P = .002.
Mixed model: Baseline BMI/categorical, time, and NAGM brain volume longitudinally (n = 768).

Note: The Categorical Baseline BMI*Time = F(2, 2683.19) = 5.963, P = .003.
Obese BMI as reference was chosen by the SPSS Mixed model analysis.

Predicted gray matter volume in 3 BMI groups over 3 years.
Discussion
This study found that being overweight predicted an increase in NAWM brain volume as compared with normal BMI, but surprisingly the obese group was not different from the normal BMI group. Thus, overweight patients with RRMS had higher white matter volume (12 mL) than those with normal BMI at 36 months, but it was not a clinically meaningful change. Interestingly, a few non-MS studies have supported the protective effect of high BMI. These studies reported larger regional white matter brain volumes in obese individuals compared to normal weight controls, possibly due to increased density of the lipid-based myelin sheath.34,35 Importantly, sex acted as a confounder in the association between BMI and NAWM brain volume likely attributed to the fact that most of the study participants were females who have different developmental phases.
High BMI was found to have a statistically significant effect on NAGM volume. The continuous and categorical baseline BMI models estimated contradictory effects on the NAGM brain volume changes throughout the 3 years of the study, possibly indicating different cutpoints matter or some lack of fit of the linear model to the data with the continuous BMI. Although these results were statistically significant, their clinical meaningfulness is questionable in view of the lack of consistency, the large sample size and the resulting high power for statistical significance in the analyses. Nonetheless, in prior studies, higher BMI appeared to be associated with similar reductions in gray matter volume and brain parenchymal volume.19-21
This study also found that BMI had no effect on cognitive function as assessed by the PASAT. The majority of the patients in the study were newly diagnosed patients with short duration of illness and high cognitive performance and all were treated with one or more DMTs, which might have been the reason for the absent relationship between BMI and cognitive function. The PASAT scores improved from baseline to month 36th, which could be explained by the data analysis showing that the missing 36th month data were of those with the most impaired cognitive performance who either withdrew from the study or refused to take this difficult test. Similarly to our secondary analysis results, a recent large study (n = 8713) of patients with MS revealed no association between the Processing Speed Test (PST) and BMI as continuous or categorical variable. 36 Contrastingly, other cross-sectional evidence has found that obesity is linked to reduced cognitive functions, particularly in executive, attention, and memory domains, which are highly prevalent in MS. 17 In addition, although an effect between BMI and cognitive function has been seen mostly in the aging healthy population and partially in a few MS studies,17,18 the interaction between BMI and cognitive function in patients with MS, and the role of BMI as a risk factor for cognitive dysfunction are complex and highly debated. However, if an association exists between cognition and BMI, the mechanisms are unclear, and it might be through complex mechanisms that involved depression, exercise, or other factors associated with both obesity and cognitive function.
Limitations of the Study
This study had several limitations that warrant discussion. The CombiRx primary study did not include a placebo control arm; therefore the comparisons and findings are related to patients with RRMS on DMTs (glatiramer acetate or interferon beta-1a or combination of both). The lack of age-sex matched control group influenced the ability to ascertain the effects of the disease itself, BMI and/or other covariates on the outcomes. Additionally, the CombiRx study included patients with MS who were newly diagnosed, and therefore had minimal functional (mean EDSS < 2) or cognitive disability. Patients with longer disease duration might have had greater disability that may have affected their BMI, brain volume and cognitive function.
In addition, the follow-up time of this study was 3 years, and while among the longest RRMS trial, still may not have been enough time to detect meaningful changes in BMI, cognitive function, and brain volume that could be found in longer follow-up periods. Furthermore, BMI is often considered to be an inaccurate measure of body fat content and does not take into account muscle mass, bone density, overall body composition, and racial and sex differences. Other measures such as waist circumference, body fat percent, and other reliable methods to measure normal, overweight and obesity conditions may provide a better assessment of their impact on the course of MS. Additionally, as noted above there are some limitations related to the PASAT. Nonetheless, recent evidence supports a correlation of the PASAT with a highly sensitive test like the SDMT that is now often used in MS research and clinical practice. Finally, the exclusion of patients with major comorbidities in the CombiRx study is both a strength and a limitation. While it somewhat limits generalizability to a wider group of individuals with MS, it also helped isolate the effects of obesity separate from other comorbid diseases and their treatments. In addition, other variables, such as physical activity and mood disorders, were not included in the CombiRx trial. These factors have an impact on brain volume and cognitive function in people with MS, which can affect the results of the study.
Conclusion
This study showed questionable effects of BMI as a continuous or categorical variable on cognitive function and normal appearing brain volume. Consistently, there was no predictable effect of the BMI on cognitive function as measured by the PASAT. Furthermore, the likelihood of meaningful impact of BMI on cognition or brain volume in early RRMS patients over 3 years seems remote due to the rigor and sample size of this trial, the consistency between the 3 year completers and the ITT results with imputation. To address some of the issues raised given the mixed nature of the results, future longitudinal prospective research studies should include a few anthropometric measurements and other cognitive tests assessing their associations and the impact of these measurements on the course of MS.
Footnotes
Acknowledgments
The completion of this secondary analysis could not have been accomplished without the support of Dr. Deborah Chyun, and Drs. Stephen Krieger and Linda Hartmann. In addition, thanks to Dr. Shiela Strauss for her support and assistance. Finally, my deepest gratitude to my family.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
