Abstract
Migraine is a complex and heterogenous disorder whose disease mechanisms remain disputed. This narrative review summarizes functional MRI (fMRI) and diffusion tensor imaging (DTI) findings and interprets their association with migraine symptoms and subtype to support and expand our current understanding of migraine pathophysiology. Our PubMed search evaluated and included fMRI and DTI studies involving comparisons between migraineurs vs healthy controls, migraineurs with vs without aura, and episodic vs chronic migraineurs. Migraineurs demonstrate changes in functional connectivity (FC) and regional activation in numerous pain-related networks depending on migraine phase, presence of aura, and chronicity. Changes to diffusion indices are observed in major cortical white matter tracts extending to the brainstem and cerebellum, more prominent in chronic migraine and associated with FC changes. Reported changes in FC and regional activation likely relate to pain processing and sensory hypersensitivities. Diffuse white matter microstructural changes in dysfunctional cortical pain and sensory pathways complement these functional differences. Interpretations of reported fMRI and DTI measure trends have not achieved a clear consensus due to inconsistencies in the migraine neuroimaging literature. Future fMRI and DTI studies should establish and implement a uniform methodology that reproduces existing results and directly compares migraineurs with different subtypes. Combined fMRI and DTI imaging may provide better pathophysiological explanations for nonspecific FC and white matter microstructural differences.
Introduction
Migraine is a highly prevalent, multifactorial primary headache disorder that affects 1 in 10 people worldwide.
1
Headache attacks are recurring and debilitating, with associated neurological symptoms.
2
The migraine cycle is characterized by distinct phases including: interictal, preictal, prodrome, aura, headache or ictal, and postictal.2,3 The aura phase preceding attacks commonly involves sensory and speech disturbances with possible brainstem or motor symptoms.3,4 Migraine is further divided across a spectrum of subtypes that include migraine with aura (MA), migraine without aura (MwoA), episodic migraine (EM), and chronic migraine (CM), defined by the
The pathogenesis for migraine remains disputed due to the complexity of the disorder. Large bodies of evidence suggest migraine pathophysiology involves altered excitability of trigeminovascular nociceptive pathways, while the migraine aura has been widely attributed to the phenomenon of cortical spreading depression.5,8 Persistent interictal alterations in neuronal networks likely explain the sensory, cognitive, emotional, and motor disturbances that migraineurs frequently experience during and outside of headaches, which manifest as visual and auditory hypersensitivity, cognitive impairment, and psychiatric comorbidities.2,5,8-10
Although not applied in routine clinical practice towards migraine, neuroimaging has studied these underlying neurological phenomena. 11 Functional MRI (fMRI) enables noninvasive investigation into migraine-related reorganized connectivity of specific brain regions and networks functionally responsible for sensory processing, as well as the measurement of abnormal brain responses to sensory stimulation observed with migraine sensory hypersensitivities. Diffusion tensor imaging (DTI) allows for noninvasive examination of migraine-related structural alterations to cell and axon integrity to highlight underlying neuronal pathology and plastic changes in migraine. Together, functional and white matter microstructural differences have elucidated disruptions to pain-related networks that likely underlie migraine pathophysiology and may serve as effective imaging biomarkers for predicting disease course and therapy. 12
In this comprehensive narrative review, we separately detail fMRI and DTI findings, first discussing their respective associations with migraine symptoms and phase. Next, we examine comparison studies between migraine subtypes based on presence of aura and chronicity. Finally, we aim to clarify discrepancies within the migraine literature and propose future research directions for each imaging modality, with an emphasis on combining fMRI and DTI to study the interplay between functional and white matter microstructural changes.
Methods
Our literature search was conducted by 3 authors who independently assessed the eligibility criteria for our included studies. This search was conducted on the PubMed database in January 2023. The breakdown and results of our search strategy are presented in Figure 1. Flow chart of literature search performed for functional MRI and diffusion tensor imaging studies in migraine.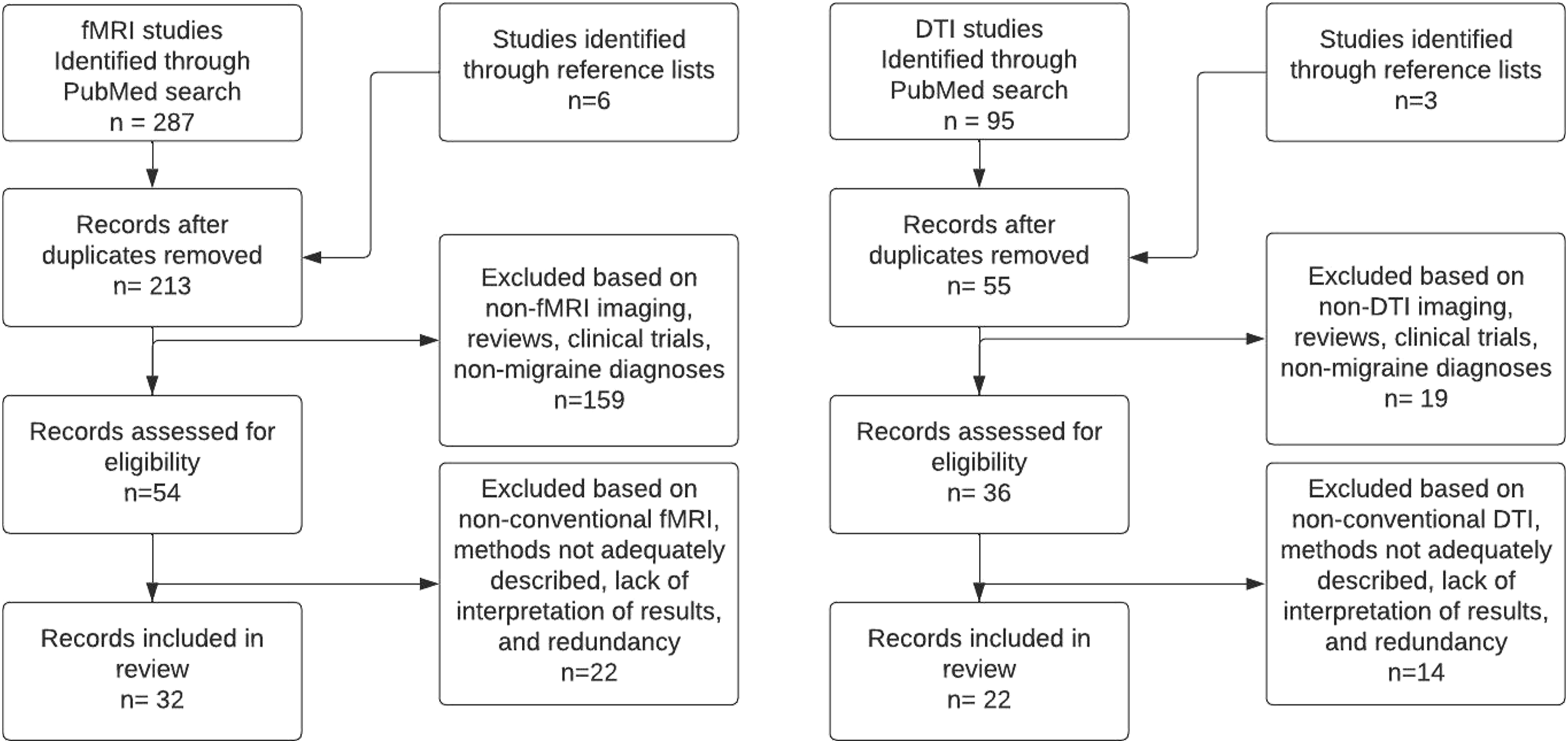
In our fMRI search, we identified studies that used conventional resting state fMRI (rs-fMRI) analysis and task-based fMRI using sensory stimulation to compare functional connectivity (FC) and blood oxygen level-dependent (BOLD) activation between migraineurs against healthy controls and across migraine subgroups. The search terms we used included “resting state fMRI and migraine,” “functional connectivity and migraine,” and “sensory stimulation and fMRI and migraine.” We filtered articles with dates ranging from January 2010 to January 2023. Our inclusion criteria evaluated studies based on relevance, original results, findings supporting or contradicting previous studies, and direct comparisons based on presence of aura and chronicity. The reference lists of our included studies and 6 previous review articles were also evaluated for additional relevant studies. We excluded reviews, clinical trials, in vitro or ex vivo studies, animal studies, non-migraine diagnoses, nonconventional rs-fMRI analysis, and studies with inadequate methods or interpretation of results.
In our DTI search, we identified studies that used tract-based or region of interest analysis to compare diffusion indices between migraineurs and healthy controls and across migraine subgroups. The search terms we used included “diffusion tensor imaging and migraine” and “white matter microstructure and migraine.” Due to fewer search results available compared to fMRI, we did not filter articles based on date. Akin to our fMRI search, we evaluated studies based on relevance, original results, findings supporting or contradicting previous studies, and direct comparisons based on presence of aura and chronicity. The reference lists of our included studies and 2 previous review articles were also evaluated for additional relevant studies. We excluded reviews, clinical trials, in vitro or ex vivo studies, animal studies, non-migraine diagnoses, nonconventional DTI analysis, and studies with inadequate methods or interpretation of results.
Literature Search Results
Our fMRI search selected 32 studies, all of which included age and sex-matched healthy controls. Of these studies, 22 performed rs-fMRI, and 10 performed task-based fMRI with sensory stimulation. 6 studies directly compared MA to MwoA, and 4 directly compared CM to EM. Among our listed studies, 1 case report and 2 case series were included due to the usage of longitudinal imaging to track migraine phase-specific findings.
Our DTI search selected 22 studies, all of which included age and sex-matched healthy controls. Of these studies, 6 directly compared MA to MwoA, and 4 directly compared CM to EM. 7 studies combined imaging with fMRI.
Functional Connectivity and BOLD Response Changes in Migraine
fMRI is an imaging method that indirectly measures neural activity in specific areas of the brain based on changes in regional blood flow. rs-fMRI measures the spontaneous fluctuations in BOLD signals between different brain regions when the subject is lying still, with no task being performed.5,13 The recorded BOLD signals detect the degree of synchronization in signal frequencies between different voxels on brain images.5,13 A high degree of synchronization reflects a strong connectivity in neural activity between the two correlating brain regions.5,13 Many functional imaging studies have also investigated sensory processing in migraineurs by exposing participants to noxious or visual stimuli to compare differences in BOLD signals and changes in regional activation against healthy controls. 14 Both rs-fMRI and task-based fMRI over the past decade have demonstrated many changes in FC and BOLD responses in migraineurs.
Functional Connectivity Changes in Migraine Compared to Healthy Controls
Functional Connectivity Changes Detected With Resting State Functional MRI in Migraineurs Compared to Healthy Controls.
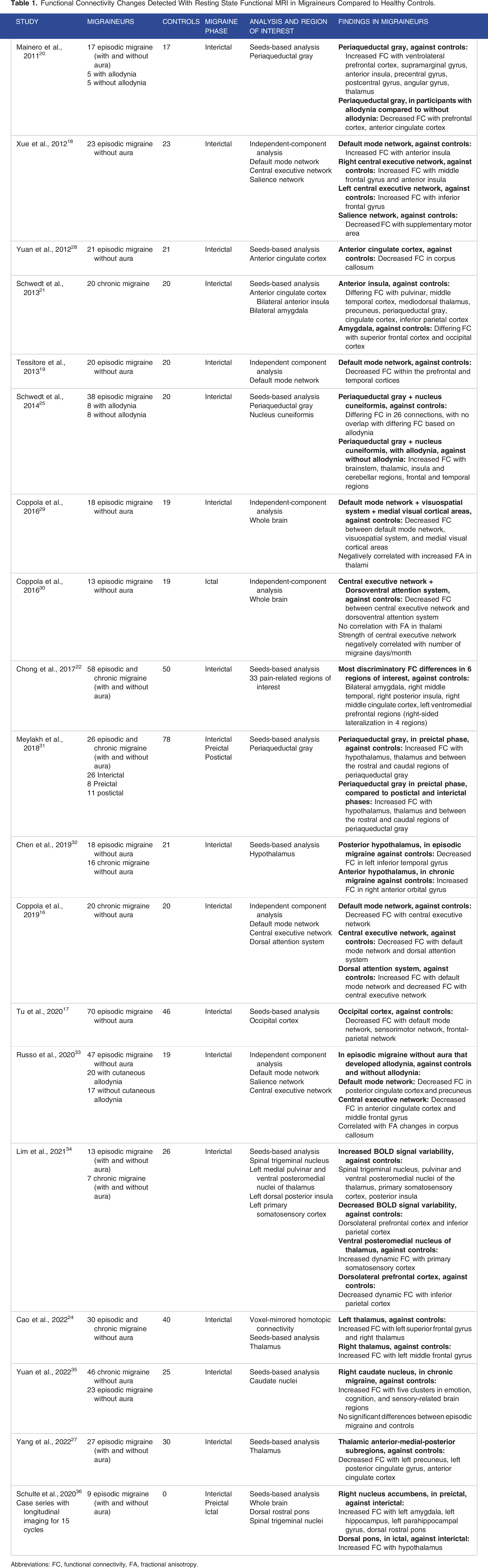
Abbreviations: FC, functional connectivity, FA, fractional anisotropy.

Resting state functional MRI data comparing migraineurs without aura (MwoA) and healthy controls (HC). (A) Spatial group maps of 3 pain-related intrinsic connectivity networks (ICN), including the default mode network (DMN), central executive network (CEN) split into right (rCEN) and left (lCEN), and salience network (SN). (B) Group comparison maps of functional connectivity (FC) in the DMN, CEN, and SN (
rs-fMRI studies emphasize that abnormal changes to FC in migraineurs contribute to increased regional hyperexcitability and decreased pain inhibition, which may explain the pathophysiology behind hypersensitivities to sensory stimuli in migraine.14,20,26 For instance, abnormal FC is visible in the ascending trigeminal spinal-thalamo-cortical pathways and top-down modulatory pathways.8,34 In particular, interictal FC changes in the brainstem descending pain modulatory system between the periaqueductal gray and related pain processing and modulatory regions likely contribute to central sensitization, dysfunctional inhibitory pathways, and allodynia during migraine attacks.20,25
Furthermore, FC alterations in hypothalamic-brainstem interactions have been found across different phases of the migraine cycle. By longitudinally monitoring EM in a 2016 case study and 2020 case series, Schulte et al demonstrated that the hypothalamus shows increased preictal FC with the spinal trigeminal nuclei and increased ictal FC with the dorsal rostral pons compared to the interictal phase.36,37 Meylakh et al in 2018 expanded upon these findings with a case-control study demonstrating increased preictal FC across the periaqueductal gray, thalamus, and hypothalamus. 31 Large-scale network phase-specific changes have also been reported, such as decreased FC strength in the central executive network during the ictal phase. 30 Together, these differences suggest cyclical regulation of complex regional interactions involving periodic changes in pain sensitivity and higher cortical function.30,31
Changes in BOLD Activation in Migraine Compared to Healthy Controls
Changes in BOLD Responses in Task-Based Functional MRI Using Sensory Stimulation in Migraineurs Compared to Healthy Controls.

Abbreviations: FC, functional connectivity, DTI, diffusion tensor imaging, FA, fractional anisotropy.
Light stimuli at low intensity capable of inducing discomfort in both MA and MwoA reveal a widening photoreactive area in the primary visual cortex, where increased interictal activation has been recognized especially in MA.23,38,40 Noxious thermal stimulation on EM reveals greater activation of the temporal pole and a reduced threshold for painful heat during the ictal phase. 26 Nociceptive stimulation with ammonia gas does not result in normal habituation in migraineurs, but rather sensitization to pain, accompanied by increased activity in the anterior insula, thalamus, and middle cingulate cortex. 41 Together, these studies support a strong association between regional hyperexcitability and hyperresponsiveness to visual and painful stimuli innocuous to healthy controls. 14 Such alterations likely relate to sensitivities to environmental stimuli during and between migraine attacks and perceptual changes that precipitate attacks, including aura, photophobia, phonophobia, and cutaneous allodynia.14,23,26,38,40
Of further note, additional phase-specific regional activation has been found in hypothalamic-brainstem interactions that complement phase-specific FC changes. 44 Prior to an attack in the prodrome phase, the spinal trigeminal nuclei, visual cortex, and anterior hypothalamus exhibit increased activity.37,39,44 In contrast, during the ictal phase, the spinal trigeminal nuclei are more weakly activated whereas the rostral pons and posterior hypothalamus are strongly activated, suggesting their roles in acute pain.37,42 Given increased FC between the rostral pons and the hypothalamus also recorded during the ictal phase, the rostral pons has been termed in some studies as the “migraine generator.”36,37,39 These varying states of excitability and activation in hypothalamic-brainstem regions help explain the functional changes that occur as migraineurs transition from a pain-free state to a painful state, as well as other disease factors such as perceptual changes and attack frequency.31,39,42
fMRI Comparisons Between Migraine with Aura and Migraine without Aura
Functional MRI Studies Comparing Migraine With Aura Against Migraine Without Aura.
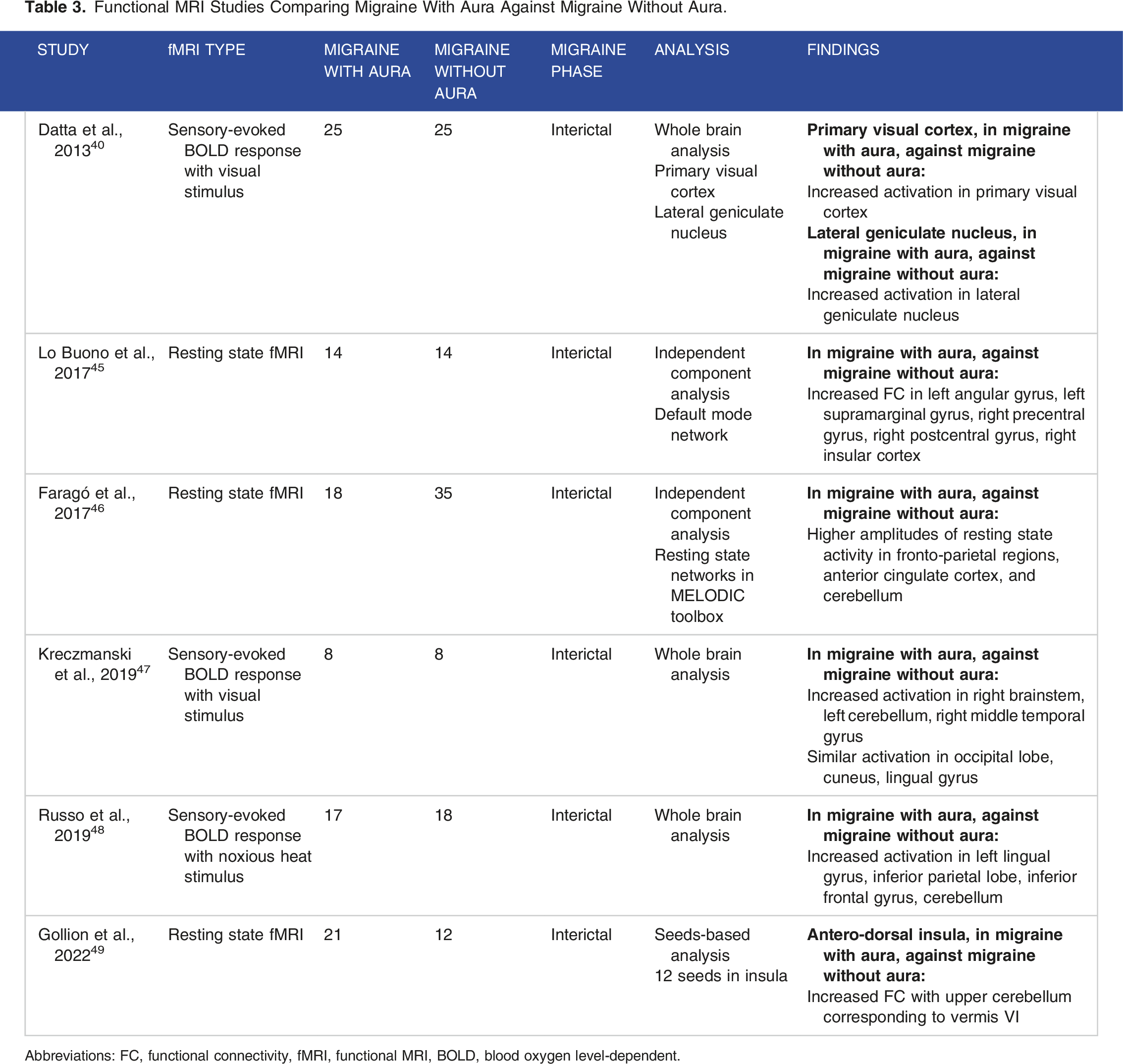
Abbreviations: FC, functional connectivity, fMRI, functional MRI, BOLD, blood oxygen level-dependent.
Compared to MwoA, MA demonstrates broad differential FC and BOLD responses in large-scale networks. rs-fMRI studies have uniquely reported increased FC in the anterior cingulate cortex, precentral gyrus, postcentral gyrus, angular gyrus, supramarginal gyrus, insula, and cerebellum.45,46,49 Task-based fMRI studies have reported increased activation in the primary visual cortex, lateral geniculate nucleus, middle temporal gyrus, lingual gyrus, frontal gyrus, inferior parietal, brainstem, and cerebellum.40,47,48
Areas where both FC and BOLD response changes have been recorded include the frontoparietal regions, which may relate to changes in executive function and pain processing and modulation, potentially explaining hypersensitivity symptoms and sensory changes experienced during the aura phase.45,46,48 Notable overlap with increased FC and BOLD responses in the brainstem and cerebellum also appear to be unique to MA.47-49 Greater activation of cortical areas involving advanced visual processing, in conjunction with nociceptive trigeminal activation and associated cerebellar changes, may suggest an integrated pathophysiological model specific to MA involving dysfunction in pain-modulating, limbic, and advanced visual regions (Figure 3).
48
Kincses et al in 2019 proposed that more pronounced cortical hyperexcitability due to cortical spreading depression drives greater maladaptive plasticity and neuroinflammatory degeneration, ultimately resulting in more white matter alterations and a more noxious state in MA.
50
Group comparison of blood oxygen level-dependent (BOLD) response in advanced visual network (including lingual gyrus, inferior parietal lobe, inferior frontal gyrus, and medial frontal gyrus) between migraineurs with aura (MwA), migraineurs without aura (MwoA), and healthy controls during trigeminal heat stimulation. Increased BOLD activation observed in MwA. Reprinted from “Advanced visual network and cerebellar hyperresponsiveness to trigeminal nociception in migraine with aura,” by Russo A, Tessitore A, Silvestro M, et al, 2019, J Headache Pain, 20(1), 46. DOI: 10.1186/s10194-019-1002-3. Copyright (2019) by BioMed Central. Permission under the Creative Commons CC BY license. http://creativecommons.org/licenses/by/4.0/.
Yet inconsistences have also been found in the brain regions reported to be uniquely affected in MA, such as marked hyperexcitability and interictal activation of the visual cortex and lingual gyrus that have been similarly observed in MwoA.17,37,40,47 These differing results are likely due to a limited number of direct comparisons between MA and MwoA, with many regional FC and BOLD response changes whose results have not been reproduced. Additional factors of disease course or chronicity are also overlooked.
fMRI Comparisons Between Episodic Migraine and Chronic Migraine
Functional MRI Studies Comparing Chronic Migraine Against Episodic Migraine.

Abbreviations: FC, functional connectivity, fMRI, functional MRI, BOLD, blood oxygen level-dependent.
There is considerable overlap in regional BOLD signal abnormalities related to the sensory-discriminative, cognitive, and integrative domains of the pain experience in both subtypes.21-23 Another notable similarity finds greater activation of the posterior hypothalamus during headaches in both EM and CM which is not observed during headache-free phases, implying that the posterior hypothalamus plays a major role in acute pain. 42 A crucial difference in regional activation, as reported by Schulte et al in 2017, is increased activation of the anterior hypothalamus in CM. 42
But differences in functional changes are also apparent between EM and CM on rs-fMRI.21,22,32,42 For instance, when compared to healthy controls and EM, CM has shown differential FC with the anterior hypothalamus, caudate nucleus, anterior cingulate cortex, middle cingulate cortex, and lingual gyrus.32,35,42 These regional differences may be suggestive of disruptions to cognitive, emotional, and sensory brain networks unique to migraine chronification. Examples of correlations with clinical factors such as increased body mass index values may partially explain comorbidities found with CM, including obesity, sleep disturbances, and autonomic disorders.32,35,42
Also of note, Schwedt et al in 2013 observed that the time since onset of CM is correlated with FC strength involving the anterior insula, mediodorsal thalamus, and periaqueductal gray. 21 In a similar respect, Chong et al in 2017 found FC differences compared with healthy controls to be more accurately determined in migraineurs with a long migraine history of over 14 years compared to migraineurs with a shorter disease history. 22 These changes in CM imply that disease burden and course may drive functional reorganization in the brain, but the number of studies that have examined these changes is limited. 22 More direct comparisons between the migraine states are necessary to track the functional changes that occur during the transition from EM to CM.
Limitations in Existing fMRI Studies and Future Directions
Although migraine literature has found numerous FC and BOLD response changes related to brain region, migraine phase, and migraine subtype, there is a lack of clear consensus on reported functional changes in migraine. 15 The reported findings are diverse with some overlap, but differences across studies are cited across affected regions, trends in FC strength, and lateralized activity.22,23,37,39 Many results have not been reproduced and may not be specific to migraine and migraine subtype. 15 Other limitations include limited sample sizes, few direct comparisons between migraine types, technical limitations of 1.5T or 3T MRI scanners with limited resolution of BOLD images, and unaccounted migraine comorbidities.
Future studies with a stricter and uniform methodology should focus on finding reproducible migraine-specific FC patterns and consider longitudinal follow-up to track imaging findings in conjunction with clinical progression. While migraine neuroimaging has previously failed to produce a robust and reliable fMRI biomarker, Tu et al in 2020 recently found a fMRI-based neural marker for MwoA that supports the existing data, using discriminatory FC patterns with good accuracy, sensitivity, specificity, and retest reliability.12,17 Continued investigation into phase- and course-specific biomarkers with similar criteria are recommended, and other imaging modalities such as DTI would identify associated structural differences for a more coherent understanding about reported functional changes.
White Matter Microstructural Changes in Migraine
Diffusion imaging studies of migraine have primarily focused on DTI, which is widely available in clinical MRI settings and suited for analyzing white matter microstructural abnormalities. By utilizing the sensitivity of the diffusion sequences to the anisotropic diffusion of water molecules, DTI measures the directional alignment of white matter tract fibers where displacement of the molecules is greatest.5,51 With the standard analysis approaches of tract-based spatial statistics, region of interest analysis, or whole brain voxel-wise analysis, the measures obtained from DTI include fractional anisotropy (FA), mean diffusivity (MD) or apparent diffusion coefficient (ADC), axial diffusivity (AD), and radial diffusivity (RD).51-53
Differences in these measures between migraineurs and healthy controls reflect white matter microstructural differences associated with migraine. FA is highly sensitive to changes to axonal integrity, fiber density, and myelin but often nonspecific, suggesting cell death, cell shrinkage or swelling, axonal loss, demyelination, and more.29,53 MD is often measured in conjunction with FA, indicating changes in the extracellular space due to cell shrinkage or loss of axonal or dendritic connections.29,53 AD is reflective of axonal integrity and affected by axonal injury and brain maturation, while RD is more reflective of axonal myelination and susceptible to demyelination and axonal degeneration.53,54 It is debated whether such changes contribute to a predisposition to migraine or result from repeated attacks, which may be explained as maladaptive plastic modifications, accumulated white matter damage, or secondary functional alterations after repeated stimulation.55,56
Microstructural Changes to White Matter Tracts in Migraine Compared to Healthy Controls
DTI Studies of Migraineurs vs Healthy Controls, Including Comparisons Based on Presence of Aura and Chronicity.

Abbreviations: FA, fractional anisotropy, MD, mean diffusivity, ADC, apparent diffusion coefficient, RD, radial diffusivity, AD, axial diffusivity, FC, functional connectivity.
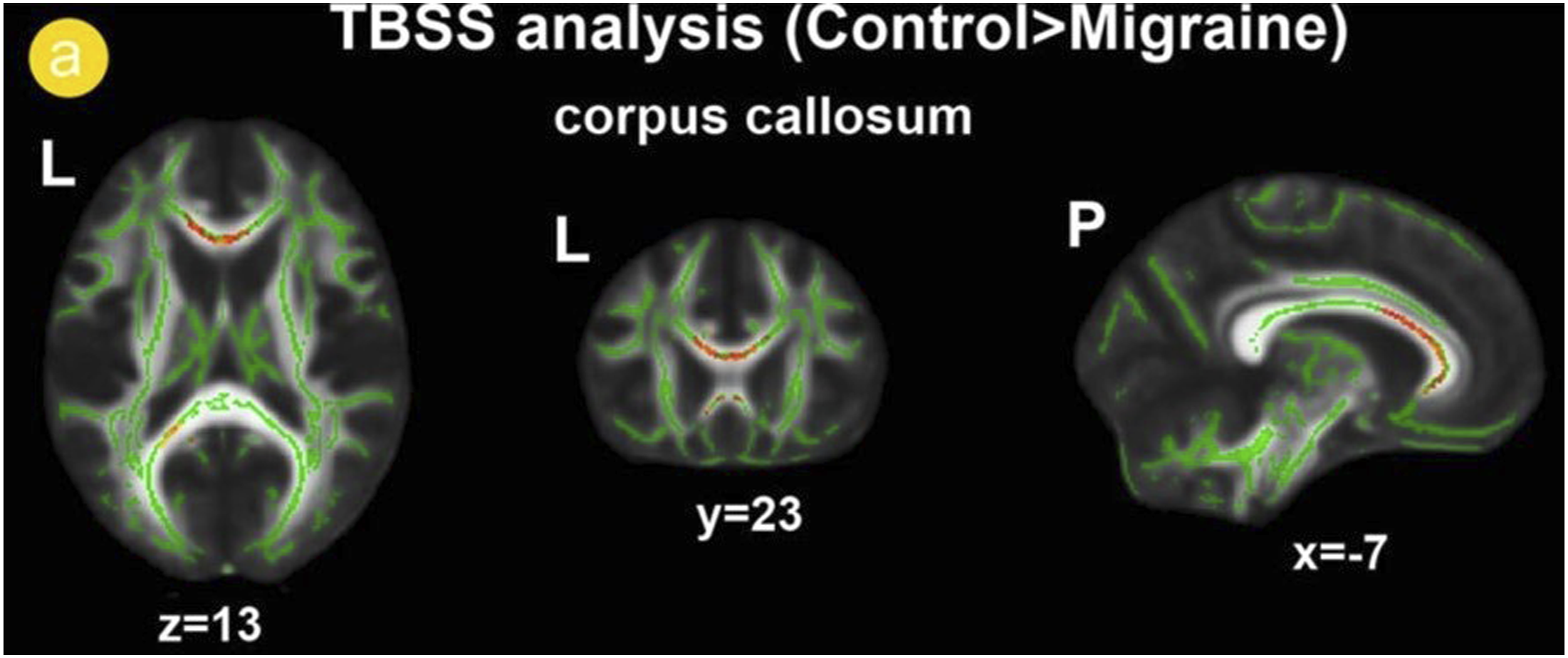
Reduced fractional anisotropy is found in the genu and splenium of the corpus callosum in migraineurs without aura compared to healthy controls, using tract-based spatial statistics (TBSS) analysis. The normal white matter skeleton is shown in green, and voxels with significant differences are shown as red-yellow. Reprinted from “Reduced fractional anisotropy of corpus callosum modulates inter-hemispheric resting state functional connectivity in migraine patients without aura,” by Yuan K, Qin W, Liu P, et al, 2012, PLoS One, 7(9), e45476. DOI: 10.1371/journal.pone.0045476. Copyright (2012) by PLoS One. Original image cropped to include top half only. Permission under the Creative Commons CC BY license. http://creativecommons.org/licenses/by/4.0/.
These alterations extend to the cerebellum and brainstem, involving vermis VI extended to lobules V and VI, the inferior cerebellum peduncle tract, spinal trigeminal nuclei, red nuclei, and periaqueductal gray.64,66,67 The affected white matter tracts indicate a diffuse and concomitant involvement of visual, trigeminal, and thalamic pathways, which support the proposal of dysfunctional trigeminovascular-related visual and pain processing pathways in migraine, with interference in transmission, modulation, and multimodal conduction and integration.55,66
White matter microstructural changes in the visual pathway provide several interesting insights into migraine. Granziera et al in 2006 found increased cortical thickness in the middle temporal visual area and visual area 3 accessory motion processing pathways to be correlated with reduced FA in the underlying white matter, superior colliculus, and lateral geniculate nucleus. 68 These findings suggest that structural changes to white matter are accompanied by changes to gray matter.57,68 Additionally, visual area 3 accessory has been previously suggested as the source of cortical spreading depression manifested during the aura phase, but changes to visual area 3 accessory in both MA and MwoA imply that cortical spreading depression may be an underlying phenomenon even if aura is absent.8,68
White Matter Microstructural Differences Based on Presence of Aura, Migraine Phase, and Age
However, it remains important to distinguish between these two migraine subtypes as white matter microstructural differences have been found in MA and MwoA. 61 Differences in FA depending on aura presence have been reported in the corpus callosum, optic radiations, parieto-occipital regions, trigeminothalamic tract, and periaqueductal gray.11,58,61-63 Yet, existing DTI studies involving direct comparisons between MA and MwoA have been limited in number and do not appear to demonstrate a replicable pattern of white matter microstructural changes, with Granziera et al even finding no significant FA differences between these subgroups. 68
Studies on phase-specific white matter microstructural changes have been limited, but Coppola et al in 2014 found increased interictal FA values in the thalamus that were positively correlated with the number of days since the previous migraine attack, but were normalized during the ictal phase. 65 Coppola et al followed up with 2016 studies that used both fMRI and DTI, observing that increased FA in the thalamus is negatively correlated with interictal FC changes in the default mode network and visuospatial system, compared to non-significant FA values and the absence of such a correlation during the ictal phase.29,30 Together, these findings may highlight a decrease in neuronal connections in the thalamus that may underlie functional network changes during attack-free periods and onset of the ictal phase.29,30,65 Additionally, these transient FA values suggest dynamic changes in water mobility that shift with migraine phase, which may help explain the heterogeneous DTI results within the migraine literature. 11
Microstructural contributions to migraine are further complicated when comparing white matter changes in pediatric and adult migraineurs. Most notable is increased FA in the optic radiations of pediatric subjects reported by Messina et al in 2015, compared to reduced FA in adults reported by Rocca et al in 2008.55,62 Together, these structural findings help demonstrate that the pathophysiological changes in migraine appear scattered and diffuse, alter visual perception, and may differ depending on aura presence, migraine phase, and patient age.55,57,62,68
White Matter Microstructural Differences Between Episodic Migraine and Chronic Migraine
DTI analysis also provides further insight into the structural differences between EM and CM. CM appears to involve global white matter changes when compared to EM, differing in numerous white matter tracts that include the corpus callosum, cerebral peduncles, thalamic radiations, superior and inferior longitudinal fasciculi, corona radiata, external capsule, anterior and posterior limbs of the internal capsule, cerebellar peduncles, and corticospinal tract (Figure 5).56,71 More specifically, reduced AD in these regions suggest decreased fiber density and axonal loss.
56
Interestingly, these same studies demonstrate no significant differences in diffusion indices between EM and controls, which suggest that migraine chronification involves more diffuse and pronounced white matter microstructural changes detectable through diffusion imaging.56,71 Reduced axial diffusivity (AD) is found in 44 regions in the Johns Hopkins University ICBM-DTI-81 White Matter Atlas and White-Matter Tractography Atlas in chronic migraineurs (CM) compared to episodic migraineurs (EM). The normal white matter skeleton is shown in green, and voxels with significant differences are shown as red-yellow. Reprinted from “White matter changes in chronic and episodic migraine: a diffusion tensor imaging study,” by Planchuelo-Gómez Á, García-Azorín D, Guerrero ÁL, et al, 2020, J Headache Pain, 21(1), 7. DOI: 10.1186/s10194-019-1071-3. Copyright (2020) by BioMed Central. Original image cropped to include top half only. Permission under the Creative Commons CC BY license. http://creativecommons.org/licenses/by/4.0/.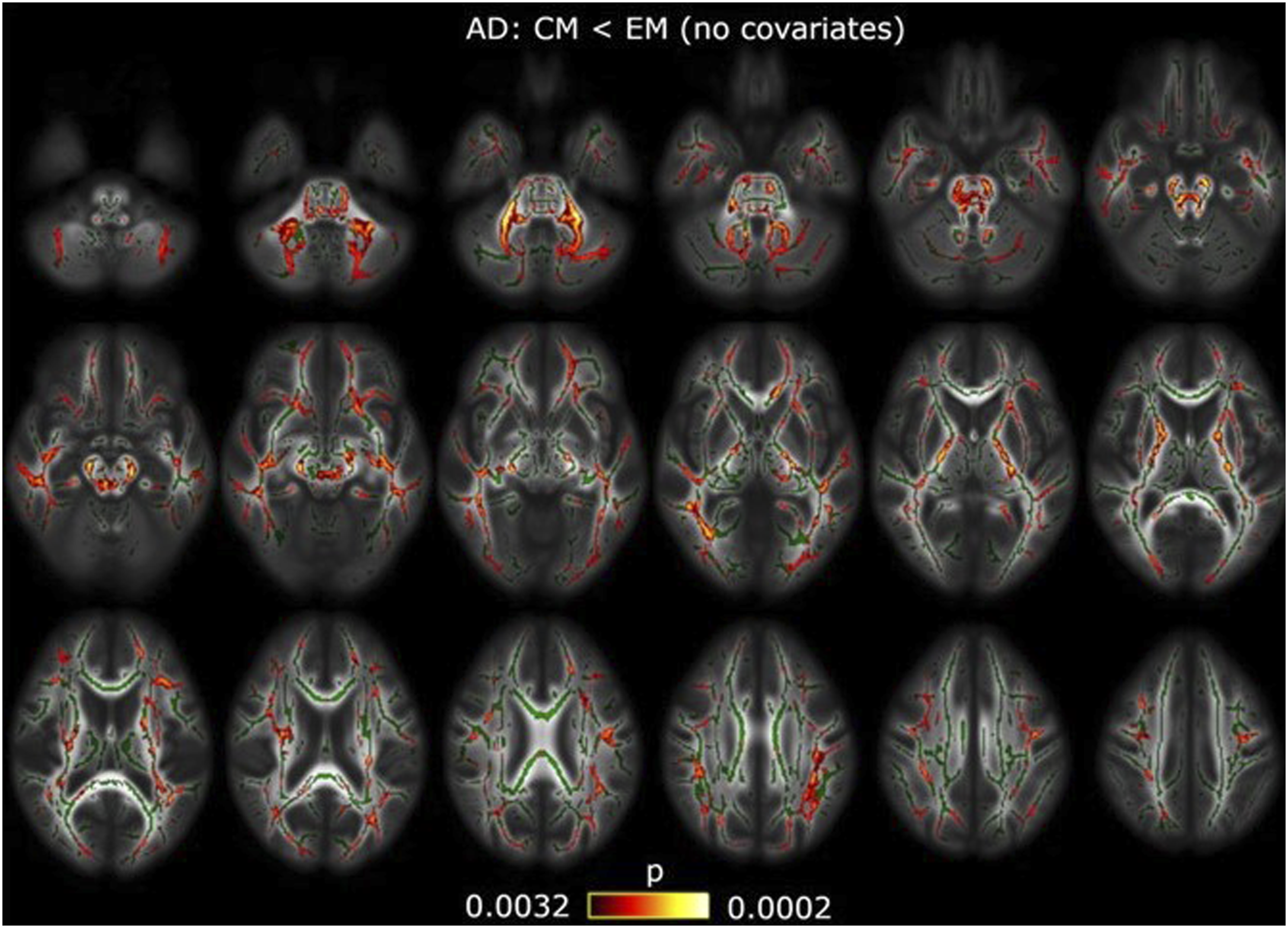
This proposed explanation is supported with more white matter differences that involve reduced FA in the anterior insula, anterior cingulate gyrus, and uncinate fasciculus associated with components of pain processing, including modulation, resilience, emotion, and cognition. 70 Given that higher FA in these regions is correlated with individuals with a higher cognitive reserve, Gomez-Beldarrain et al in 2016 proposed that individuals without CM and with intact cognition likely possess stronger white matter tracts with higher pain resilience. 70 Therefore, while EM may involve white matter alterations, the transition to CM may involve a much greater loss of axonal integrity that contributes to pain hypersensitivity, decreased cognition, and dysfunctional inhibition.56,70 Moreover, Planchuelo-Gómez et al in 2020 also found increased FA and reduced RD in the external capsule in correlation with time since the onset of CM, suggesting neural plasticity and reorganization for adapting to a more continuous painful state in CM.56,57
Limitations in Existing DTI Studies and Future Directions
On the other hand, conflicting trends in FA and other DTI measures have been reported across different studies.27,55,56,61,62 These apparently contradictory results may suggest a coexistence of both debilitated and enhanced white matter networks within the brain, involving both axonal damage and plastic adaptation. 57 However, a study in 2015 by Neeb et al has even found no significant differences in DTI measures across CM, EM, and healthy controls. 69 Consequently, existing studies using DTI face many limitations in methodology that may explain these different results within the literature. These flaws include small sample sizes, few replication studies, few comparisons between migraine types, participant usage of medication and frequency of usage, and unaccounted comorbidities including anxiety and depression.57,69
There is no migraine-specific biomarker tracking white matter microstructural changes, necessitating future studies to distinguish migraine subtypes and minimize confounding variables. Longitudinal studies are also needed to evaluate phase-specific structural changes and long-term alterations that occur beginning at youth and during the transition from EM to CM.
Furthermore, more advanced diffusion modalities may be implemented that overcome DTI limitations. In 2020, Planchuelo-Gómez et al used apparent measures using reduced acquisitions (AMURA), utilizing the ensemble average diffusion propagator to provide better modeling of the white matter fiber architecture, yielding initial results that complement DTI analysis of migraine. 57 Other more advanced diffusion imaging techniques such as high angular resolution diffusion imaging (HARDI) may also provide more accurate characterization of white matter changes in future studies. Moreover, multi-shell diffusion MRI can also be used to decompose the neurite signal from the rest of the voxels to indirectly measure the neurite density index. 72 These techniques would provide information about the microscopic damages to white matter that are hidden to structural imaging techniques. Doing so would calculate several metrics including neurite density and the degree of axonal fanning, estimate microglial and myelin contents, and determine white matter tract integrity. These proposed research directions with an improved methodology may elucidate an identifiable biomarker of white matter changes, as well as any interactions with changes in gray matter and functional connectivity.
Utility of Combined fMRI and DTI Imaging in Migraine
Pairing DTI and fMRI in the same studies to examine coexisting functional and white matter microstructural differences should also be considered in future investigation. While these imaging modalities have traditionally been studied separately, existing studies of healthy volunteers have observed that applying such multimodal imaging may have useful research and clinical application in relation to migraine. Lanyon et al in 2009 used fMRI and DTI to identify visual motion perception pathways between the middle temporal area and superior temporal cortex with the pulvinar nucleus of the posterior thalamus, active in the setting of a dysfunctional primary visual cortex. 73 Zou et al in 2022 combined imaging to identify prominent prefrontal-parietal and insula-cingulate connectivity that more accurately predicts individualized pain threshold compared to using fMRI and DTI alone, highlighting key roles of the insula, precuneus, and calcarine in pain sensitivity. 74 These regional connectivity features related to visual and pain modulatory pathways support reported changes to FC and diffusion indices in the migraine literature.
In combined imaging studies on migraine patients, Moulton et al in 2011 investigated the role of the temporal pole in the transition between interictal and ictal phases, observing extensive white matter connectivity between the temporal pole and pulvinar nucleus that may serve as an afferent pathway for transmitting nociceptive information and the basis for observed phase-specific functional changes. 26 Coppola et al in 2014 and 2016 supported these results with correlations between increased FA in the thalamus and FC changes in the default mode network, visuospatial system, and medial visual cortical areas, proposing that deficient thalamic microstructural connections contribute to interictal changes to functional networks.29,30,65 More recently, Faragó et al in 2019 and Russo et al in 2020 also demonstrate how FA and RD differences correlated with FC changes in large-scale brain networks, suggesting a joint role of neuroanatomic and brain activity-related alterations in migraine pathophysiology.33,58 More specifically, functional and white matter microstructural disruptions co-occurring in central pain modulatory circuits may ultimately contribute to presence of aura and development of allodynia.33,58
Consequently, multimodal imaging using both fMRI and DTI carries exciting potential in migraine research by providing better pathophysiological explanations for nonspecific FC and white matter microstructural differences currently being reported. Examining the interplay between brain structure and function can help elucidate existing knowledge gaps and reconcile discrepancies between studies.
Discussion
Our fMRI and DTI literature search resulted in many diverse findings—some reproduced, many others not. By performing a comprehensive review, we deduced several noteworthy functional and white matter microstructural trends and related them to migraine clinical presentation and subtype. However, while most studies we included demonstrated some degree of significant result, many findings did not overlap. Several studies demonstrated no significant differences across comparisons against controls or between migraine subgroups.27,35,56,68,69,71 In turn, our ability to gather these findings into a coherent pathological pattern was limited.
Some of these discrepancies can be explained by the differences in study designs. We ensured that all our included studies defined migraine based on the
Additionally, we ensured most of our included studies took similar approaches in analysis, but the type of analysis performed inevitably can affect results. While both seeds-based and independent component analyses in fMRI yield similar results in primary resting state networks, results can vary in secondary sub-networks depending on factors such as regional seed placement, scan time, and FC metrics within networks vs between networks.75,76 Functional changes observed with rs-fMRI vs task-based fMRI can be difficult to connect as well, with differences focusing on temporal statistical relationships between brain regions vs triggered regional activation. Similarly, tract-based spatial statistics and region of interest labeling in DTI share many of the same advantages in detecting FA changes but differ based on automated or manual region of interest selection, whole brain vs regional analysis, and registration of white matter microstructural differences unique to individual participants. 77 Furthermore, we omitted studies utilizing less conventional methods, such as Granger causality analysis of fMRI, that may help explain some of these assorted findings. 78
Ultimately, current limitations of fMRI and DTI studies may be overcome with a standardized uniform protocol in migraineur recruitment, analysis of important demographic and clinical factors related to migraine, and collaboration between different investigating parties on methodology and data. Longitudinal studies and utility of combined fMRI and DTI imaging, as well as more advanced functional and diffusion imaging modalities, should also be considered to bridge existing gaps in results and interpretation.
Conclusion
rs-fMRI and task-based fMRI migraine studies have observed a wide array of alterations in functional connectivity and regional activation in pain-related networks. These functional changes can be phase-specific and likely disrupt multimodal sensory-integrative pathways in pain processing and modulation. MA appears to entail unique pathogenesis mechanisms involving the frontoparietal, brainstem, and cerebellar regions associated with pain modulation and executive function. CM can be distinguished from EM based on diencephalic, insular, limbic, and occipitotemporal changes which may reflect comorbidities associated with chronicity.
DTI studies on brain imaging in migraineurs have observed white matter microstructural changes in major cortical white matter tracts extending to the brainstem and cerebellum. These alterations are associated with functional and structural gray matter changes, supporting proposed dysfunction of multimodal visual and pain processing pathways. White matter differences are more diffuse and extensive in CM, suggestive of pathogenic or adaptive differences unique to chronicity.
However, interpretations of the reported fMRI and DTI measure trends have not achieved a clear consensus. Future studies should establish a uniform methodology to reproduce existing results and conduct more direct comparisons between migraineurs with different subtypes of the disease. Alternative fMRI and advanced diffusion imaging techniques may overcome some of these limitations. Combining fMRI and DTI imaging has related the co-occurrence of functional and white matter microstructural changes to migraine phase and pain sensitivity; increased utilization of combined imaging will improve understanding of the underlying structural and functional interplay in migraine pathophysiology.
Footnotes
Author Contributions
Alexander Lerner: Conceptualization, Data curation, Supervision, Writing-original draft, Writing-review & editing
Giuseppe Barisano: Conceptualization, Data curation, Writing-original draft
Daniel Phung: Conceptualization, Data curation, Investigation, Methodology, Writing-original draft, Writing-review & editing
Wilson Xu: Conceptualization, Data curation, Investigation, Methodology, Writing-original draft, Writing-review & editing
Soniya N Pinto: Conceptualization, Data curation, Writing-original draft
Nasim Sheikh-Bahaei: Conceptualization, Data curation, Supervision, Writing-original draft, Writing-review & editing
Validation Error Authors: Brendon Chou, Alexander Lerner, Giuseppe Barisano, Daniel Phung, Wilson Xu, Soniya N Pinto, Nasim Sheikh-Bahaei.
Brendon Chou: Conceptualization, Data curation, Investigation, Methodology, Writing-original draft, Writing-review & editing
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author’s Note:
Presentation at meeting: Presented at American Society of Neuroradiology 2022.
