Abstract
Migraine is a complex headache characterized by changes in functional connectivity and cerebral perfusion. The perfusion changes represent a valuable domain for targeted drug therapy. Arterial spin labeling is a noncontrast imaging technique of quantifying cerebral perfusion changes in the migraine setting. In this narrative review, we will discuss the pathophysiology of the different categories of migraine, as defined by the International Classification of Headache Disorders-3 and describe a category-based approach to delineating perfusion changes in migraine on arterial spin labeling images. We will also discuss the use of arterial spin labeling to differentiate migraine from stroke and/or seizures in the adult and pediatric populations. Our systematic approach will help improve the understanding of the complicated vascular changes that occur during migraines and identify potential areas of future research.
Introduction
According to the 2016 Global Burden of Disease Study, more than 1 billion individuals suffer from migraines worldwide. 1 In that study, the growing impact of migraine was illustrated by a staggering 51.8% increase in the number of years lived with disability in 2016, compared with that in 1990. 1 The economic burden of migraine in 2016 was approximately $36 billion in the United States alone. 2
Arterial spin labeling (ASL) is a noncontrast imaging modality that holds great promise in the evaluation of abnormalities in cerebral blood perfusion during different types of migraine. A literature review of the applications of ASL in migraine over the past decade revealed several case reports and case series consisting of fewer than 50 migraineurs (ie, patients who suffer migraines). In this review, we will detail the dynamic perfusion changes in migraine, with or without typical, hemiplegic, brainstem, and retinal types of aura, as defined by the International Classification of Headache Disorders-3 (ICHD-3). 3 We will also discuss the use of ASL to differentiate migraine from other neurologic disorders in the adult and pediatric populations. Finally, we will identify areas of potential research in uncommon subtypes of migraine that have not been studied with ASL.
Pathophysiology of Migraine
Historically, the pathophysiology of migraine was explained by 2 theories, the nerve-storm theory proposed by Edward Liveing in 1873 4 and the vasomotor theory of cerebral arterial vasodilation proposed by Peter Wallwork Latham, also in 1873. 4 The vasomotor theory was further explored by Wolff and colleagues in a series of experiments that demonstrated the headache-inducing effects of vasodilatory drugs, such as nitroglycerin, and the therapeutic effects of vasoconstrictor agents, such as ergotamine. They described 2 components of the attack: the aura phase, which is caused by intracranial cerebral vasospasm, and the headache phase, which is caused by extracranial vasodilation; these occur with intramural vasogenic edema and sterile inflammation. 5 This theory was later challenged by Olesen and colleagues, who demonstrated that the headache phase begins well after the resolution of drug-induced vasodilation. 6 Furthermore, with the development of sumatriptan and its effect on both vascular and neural physiology, there was a resurgence of Liveing’s nerve-storm hypothesis. 7 The current understanding of migraine is that neither theory alone completely accounts for this complex process.
In genetically susceptible individuals, physiological changes caused by stress and hypoglycemia can trigger the depolarization of small, pseudo-unipolar neurons in the trigeminal ganglion. In turn, these cells activate second-order neurons in the trigeminal nucleus caudalis in the medulla and in the dorsal horn of the upper cervical spine. These second-order neurons project to other brainstem nuclei, and the contralateral dorsomedial and ventroposteromedial nuclei of the thalamus. Resultant alterations in functional connectivity among these structures and several cortical regions leads to a complex pattern of cortical spreading depolarization (CSD) throughout the cortices, causing the aura symptoms that accompany migraine.8,9 The vascular changes are associated with the wave of cortical depolarization and are characterized by an initial transient hyperemia, followed by decreased blood flow, which persists after the initial wave passes. 9 The initial hyperemia is most likely related to an increase in extracellular concentration of potassium, which in turn increases the concentration of calcitonin gene–related peptide (CGRP), a potent vasodilator. 10 The subsequent oligemia may be related to an increased influx of calcium into the pericytes, with resultant capillary vasoconstriction. 11 Although most of these mechanisms are common in different categories of migraine headaches, their location and clinical manifestations may differ. We will discuss the underlying mechanisms of each category of migraine and highlight the differences in vascular changes that are detected by ASL.
Arterial Spin Labeling in Migraine
During ASL, radiofrequency inversion pulses are applied to protons in the carotid and vertebral arterial blood at the skull base, before those protons enter the intracranial compartment, a technique known as labeling. 12 These pulses will invert the longitudinal magnetization of protons in arterial blood water. 13 A brief post-label delay is then implemented to allow the labeled protons to enter the imaging slice of interest and equilibrate between the intravascular and extravascular compartments. 14 The negatively magnetized protons in arterial water produce a 1%-2% signal loss from the positively magnetized static protons. 15 Control images of the same slice of interest without radiolabeled water are then acquired, and those images are subtracted from the images with radiolabeled water. This subtraction generates a perfusion image, which enables the quantification of CBF which is measured in mL/100 g/min. 16 There are currently several techniques of radiofrequency inversion pulse application or proton labeling, including the pulsed technique which employs a single pulse or short pulse train, the continuous technique which employs the constant application of a pulse train and the pseudocontinuous technique (PCASL) which employs a train of closely spaced, low flip-angle, slice-selective pulses in conjunction with gradients oriented in the direction of blood flow. 17 PCASL is currently the most commonly used method in clinical practice and is advocated by the International Society for Magnetic Resonance in Medicine and the European Consortium for ASL. 18 All the case reports and series discussed in this review article employed PCASL in interrogating perfusion changes in migraineurs.
Migraine Without Aura
According to the ICHD-3, the diagnosis of migraine without aura requires at least 5 attacks of headaches lasting 4-72 hours, with at least 2 of the following traits: unilateral location, pulsating quality, moderate or severe intensity, aggravated by or causing avoidance of routine physical activity, and at least 1 of the following events during the headache: nausea and/or vomiting, photophobia and phonophobia, and not accounted for by any other diagnosis. 3
The pathophysiology of migraine in patients without accompanying aura has been linked to several theories. One theory is that migraineurs without aura experience a clinically silent cortical spreading depolarization. This theory was supported by 1 study that evaluated the efficacy of tonaberstat, an inhibitor of cortical spreading depolarization in animal models, in 39 migraineurs; the investigators showed a significant decrease in the frequency in patients who experienced migraines with aura but no effect on those who experienced migraine without aura. 19 Other proposed mechanisms for migraine without aura include shear-induced platelet aggregation, which leads to local serotonin and CGRP release and nitric oxide formation. 20 Further studies of the electrophysiological changes in migraineurs without aura will help us better understand the pathophysiology of this entity.
Although nonspecific T2/FLAIR (fluid-attenuated inversion recovery) images of white matter hyperintensities occur in migraineurs without aura, no associated diffusion-weighted imaging findings have been reported in this migraine category.21,22 Advanced imaging studies evaluating structural changes in migraineurs without aura have demonstrated decreased gray matter volume in several supratentorial and infratentorial locations, including the medial frontal cortex, the dorsal anterior cingulate gyrus, the occipital lobe, and the trigeminal nerve nucleus in the brainstem, which are known areas of activation during the attack.23,24 One study by Qin et al also demonstrated an increase in axial, mean, and radial diffusivity in the spinal trigeminal nerve nucleus and the right inferior cerebellar peduncle. 24 Whether these changes are a cause or a consequence of repeated migraine attacks is unknown. More longitudinal case-control studies will establish a more definitive association between structural abnormalities and migraine in these patients without aura. Similar to studies on structural alterations, those on resting-state functional magnetic resonance imaging (MRI) in migraineurs without aura have shown decreased functional connectivity in several networks, including the anterior cingulate–prefrontal cortex, the insula–prefrontal cortex, and in components spanning the visual cortex, posterior insula, and primary somatosensory cortex.25,26 These studies support the theory that clinically silent cortical spreading depolarization is a component of the pathophysiology of migraine without aura.
In contrast, perfusion studies using ASL in migraine without aura have reported conflicting results. In 2017, Gil-Gouveia et al used 3T ASL to study the perfusion changes in migraine without aura in 13 female patients. They analyzed the average CBF in 10 regions of interest during headache and headache-free periods. The time interval between the headache and headache-free periods was a minimum of 48 hours with no pharmacologic intervention. The investigators did not identify any significant differences in average CBF between the 2 periods. 27 However, a study by Younis et al in 2021 challenged those results. They used ASL to compare perfusion changes in 26 migraineurs without aura during headache and headache-free periods in a double-blinded, randomized, crossover study on 2 separate days. On Day 1, patients were randomized to receive either a CGRP infusion or placebo tablet, and on Day 2, they were randomized to receive either a sildenafil tablet or an infusion of isotonic saline. Perfusion significantly increased within the dorsolateral pons, ipsilateral to the side of pain during headaches and relative to headache-free periods. 28 Both case series comparing perfusion changes during the headache and headache-free periods excluded chronic migraineurs. However, the study by Gil-Gouveia et al included spontaneous migraine attacks without aura over a wide period, from onset of symptoms (4-69 hours) and segregated patients during the early phase of attack (less than 5 hours) from the later phase (more than 5 hours). In contrast, the study by Younis et al employed pharmacologic methods of migraine induction with CGRP and sildenafil and only evaluated changes during the first 6 hours.27,28 Therefore, the increased CBF in the dorsolateral pons, ipsilateral to the side of the attack observed in this study, may be related to pharmacologic intervention and may be transient. Further studies evaluating perfusion changes at different time points, with a focus on the dorsolateral pons, may help clarify the current controversy.
ASL studies on perfusion changes in migraine without aura in between attacks have also yielded mixed results. A study by Chen et al in 2018 used ASL to compare perfusion in 15 migraineurs without aura who were scanned between attacks and in 15 controls. Their results suggested increased CBF in the left temporal pole (Broadman area 38A) in migraineurs, relative to controls (Figure 1).
29
A larger case-control study by Giani and colleagues in 2019 compared 21 female migraineurs without aura who were scanned between attacks with 15 sex-matched controls. The investigators calculated the average CBF from 114 cortical and subcortical areas and compared the perfusion values among migraineurs and between migraineurs and controls. No significant difference in the average CBF was found between migraineurs and controls or among migraineurs.
30
These 2 studies evaluating perfusion changes between attacks of migraine without aura differed in subject composition, with the study by Chen et al including only episodic migraineurs, and the study by Giani et al including episodic and chronic migraineurs. Furthermore, Chen et al clearly defined the timing of image acquisition as 6-14 days after the last migraine attack; Giani et al did not define image timing.29,30 These differences in study design suggest that Brodmann area 38 may have a definitive role in the pathogenesis of episodic migraine, owing to its complex integrative role in the auditory, somatosensory, and paralimbic pathways.
30
A summary of ASL findings in migraineurs without aura is given in Table 1. The brain region with hyperperfusion located in the left superior temporal lobe (Broadman area 38) in patients with episodic migraine compared with normal controls. Reprinted from Chen Z et al, Evaluation of gray matter perfusion in episodic migraine using voxel-wise comparison of 3D pseudo-continuous arterial spin labeling. J Headache Pain. 2018; 19 (1):36. https://doi.org/10.1186/s10194-018-0866-y. http://creativecommons.org/licenses/by/4.0/. Perfusion Changes Detected by Arterial Spin Labeling in Migraineurs without Aura. aControls completed ASL imaging during a headache-free period.

Migraine With Typical Aura
According to the ICHD-3, migraine with typical aura consists of visual and/or sensory symptoms and/or speech/language symptoms, with gradual development, duration of each symptom no longer than 1 hour, a mix of positive and negative features, and complete reversibility. 3
Migraineurs with typical aura have a genetic and hormonal predisposition, with a 4-fold increased relative risk in first-degree relatives. 31 The slowly propagating wave of cortical spreading depolarization (∼3-5 mm/min) has been demonstrated in visual aura in animal studies and in the human visual cortex on functional MRI studies.32,33 The link between cortical spreading depolarization and the subsequent headache has been suggested by an inflammatory cascade, through the release of CGRP and vasodilation, with direct depolarization of nociceptive afferents via the release of potassium. 34 Cortical spreading depolarization may also directly stimulate the trigeminal nucleus caudalis via direct connections from layer 5 of the insula and primary somatosensory cortex. 35
MRI findings during the aura attack include reversible cortical restricted diffusion spanning multiple vascular territories and reversible cortical venous engorgement in the symptomatic hemisphere.36,37 Magnetic resonance angiogram (MRA) findings were more variable; in a case-control study comparing 33 patients with acute stroke with 33 migraineurs with typical aura, vasoconstriction of the middle and posterior cerebral arteries was seen in 15.2% of patients, and vasodilation of the same arteries was seen in 6.1% of patients. 38
Case-control studies on volumetric changes in fewer than 100 migraineurs with typical aura have demonstrated decreased gray matter volume in the frontal and temporal lobes in migraineurs with aura. However, these gray matter volumetric changes did not correlate symptomatically to the structures involved in producing the aura (ie, the occipital cortex for visual aura and the somatosensory cortex for sensory aura).39,40A more recent volumetric analysis by Burke et al using a novel mapping technique demonstrated significant gray matter volume loss localized to a common brain network, with connectivity to the extrastriate visual cortex, a region previously implicated in cortical spreading depolarization. 41 Similarly, studies of resting-state functional MRI have demonstrated alterations in functional connectivity in the extrastriate visual cortex, lingual gyrus, and occipital cortex.42,43
ASL studies on perfusion changes in migraine with typical aura consist of case reports and case series of fewer than 20 patients. A case report by Law-ye et al in 2017 demonstrated the utility of ASL in a previously healthy 17-year-old male patient presenting with aphasia. After diffusion-weighted and FLAIR imaging ruled out acute stroke, ASL imaging demonstrated hypoperfusion involving multiple vascular territories in the left cerebral and right cerebellar hemispheres. 44 Another case report published in 2017 by Burns et al illustrated the biphasic changes on ASL in a 25-year-old migraineur presenting with right upper limb paresthesia and aphasia. The ASL map during the aura phase demonstrated a mean relative decrease of 49% in CBF in the left cerebral hemisphere, compared to the contralateral side. The ASL map acquired 25 hours later, during the headache phase of the attack, demonstrated hyperperfusion in the left parieto-occipital region, with a mean relative increase of 44% in CBF compared to the contralateral side. 45
Case series evaluating ASL findings in migraineurs with typical aura involve a combination of patients with typical, motor, brainstem, and retinal aura. In 2018, Wolf and colleagues used ASL to evaluate 4 migraineurs with aura. Only 1 of their patients demonstrated a typical aura with left-sided vision changes and hemihypesthesia. ASL imaging of this patient demonstrated hypoperfusion in the right temporo-occipital region and hyperperfusion in the adjacent right frontotemporal region. The regional hyperperfusion in the right frontotemporal region was thought to represent the early headache phase of the attack, given that the patient began experiencing headache symptoms towards the end of the scan. ASL was also performed during the follow-up exam, when the patient was asymptomatic, and demonstrated resolution of hypoperfusion and hyperperfusion. Dynamic susceptibility–weighted perfusion imaging performed concurrently confirmed these findings. 46 Another case series published in 2018 by Cadiot et al used ASL to evaluate 17 patients (aged 11-16 years) presenting with migraine with aura. ASL imaging was done during the aura phase of the attack. Ten patients had typical aura: 1 had only sensory disturbances, and 9 had a combination of sensory symptoms, visual disturbances, and aphasia. All 10 patients demonstrated parieto-occipital hypoperfusion, in the hemisphere contralateral to the deficit. The patterns of hypoperfusion did not conform to any vascular territory. A unique feature of this study was that the findings of hypoperfusion correlated with concomitant vasospasm in the affected cerebral hemisphere on time-of-flight MRA. 47
Changes in Perfusion Detected by Arterial Spin Labeling in Migraineurs with Typical Aura.


A 14-year-old boy presenting with acute dysarthria and right sensory deficit. Initial emergency MRI shows no ischemia on DWI (a), left temporo-occipital hypoperfusion (arrow heads) on the ASL perfusion map (b), and no vascular abnormalities on axial (c) and coronal (d) 3D TOF-MRA. Note that the low position of the TOF-MRA box does not allow visualization of the distal intracranial arteries, meaning that distal vasospasm may go underdiagnosed in this case. Reprinted from Cadiot D et al. Magnetic resonance imaging in children presenting migraine with aura: Association of hypoperfusion detected by arterial spin labelling and vasospasm on MR angiography findings. Cephalalgia. 2018 Apr; 38 (5):949-958. https://doi.org/10.1177/0333102417723570
Hemiplegic Migraine
According to ICHD-3, a hemiplegic migraine is marked by fully reversible, unilateral motor weakness, in addition to visual, sensory, and/or speech/language symptoms. 3 Hemiplegic migraine has a genetic basis, with both familial and sporadic forms identified. The familial form has an autosomal-dominant mode of inheritance. 48 Mutations in the ion transportation genes CACNA1A, APT1A2, and SCN1A have been identified in both forms of hemiplegic migraine, suggesting the genetic heterozygosity of this condition. CACNA1A gene mutations impair the calcium channels in presynaptic neurons, with a consequent increased influx of calcium and release of glutamate and potassium into the synaptic cleft. APTA1A2 mutations cause abnormalities of the sodium–potassium ATP pump, which impair the glial reuptake of glutamate, resulting in slow recovery from neuronal excitation. SCN1A mutations affect voltage-gated sodium channels and impair the inhibitory effects of GABA (gamma-amino butyric acid)-ergic neurons. Together, these mutations increase the concentrations of glutamate and potassium in the synaptic cleft, making the brain more susceptible to cortical spreading depolarization.49,50
Case reports on hemiplegic migraineurs have demonstrated cortical edema and gyriform enhancement of the hemisphere contralateral to the deficit.51,52 In addition, several cases of hemicerebral atrophy have been reported, particularly in patients with the familial form.53-55 A few case reports and case series have illustrated perfusion changes in the hemisphere contralateral to the deficit on ASL images (Figure 3). In 2016, Kim et al reported a case of a 16-year-old male patient with sporadic hemiplegic migraine, who presented with right-sided weakness. The investigators noted decreased CBF in the left cerebral hemisphere, which was more severe in the parieto-occipital region. These findings were further confirmed with dynamic susceptibility–weighted perfusion MRI and accompanied by vasoconstriction of the peripheral branches of the left, middle, and posterior cerebral arteries on MRA.
56
In 2016, a case-control series reported by Boulouis et al included 4 patients with motor deficits among 11 migraineurs, and 3 of these 4 who were scanned within the first 6 hours of their aura onset demonstrated 25% or more decrease in CBF in the hemisphere contralateral to the deficit, as compared to controls. The remaining patient was scanned 17 hours after the onset of aura and demonstrated an increase in CBF that was greater than 25% in the hemisphere contralateral to the side of the deficit, as compared to controls.
57
In the work of Wolf et al, 2 of 4 patients presented with symptoms of a hemiplegic aura; 1 presented with left-sided hemiparesis and numbness and was scanned within 30 minutes of the aura. ASL findings in this patient revealed adjacent areas of hypoperfusion in the right parietal lobe and hyperperfusion in the temporal lobe, which resolved on follow-up. The other patient presented with right-sided hemiparesis and aphasia and was scanned 6 hours after symptom onset, that patient demonstrated only hyperperfusion in the contralateral parieto-occipital region, which resolved on follow-up.
46
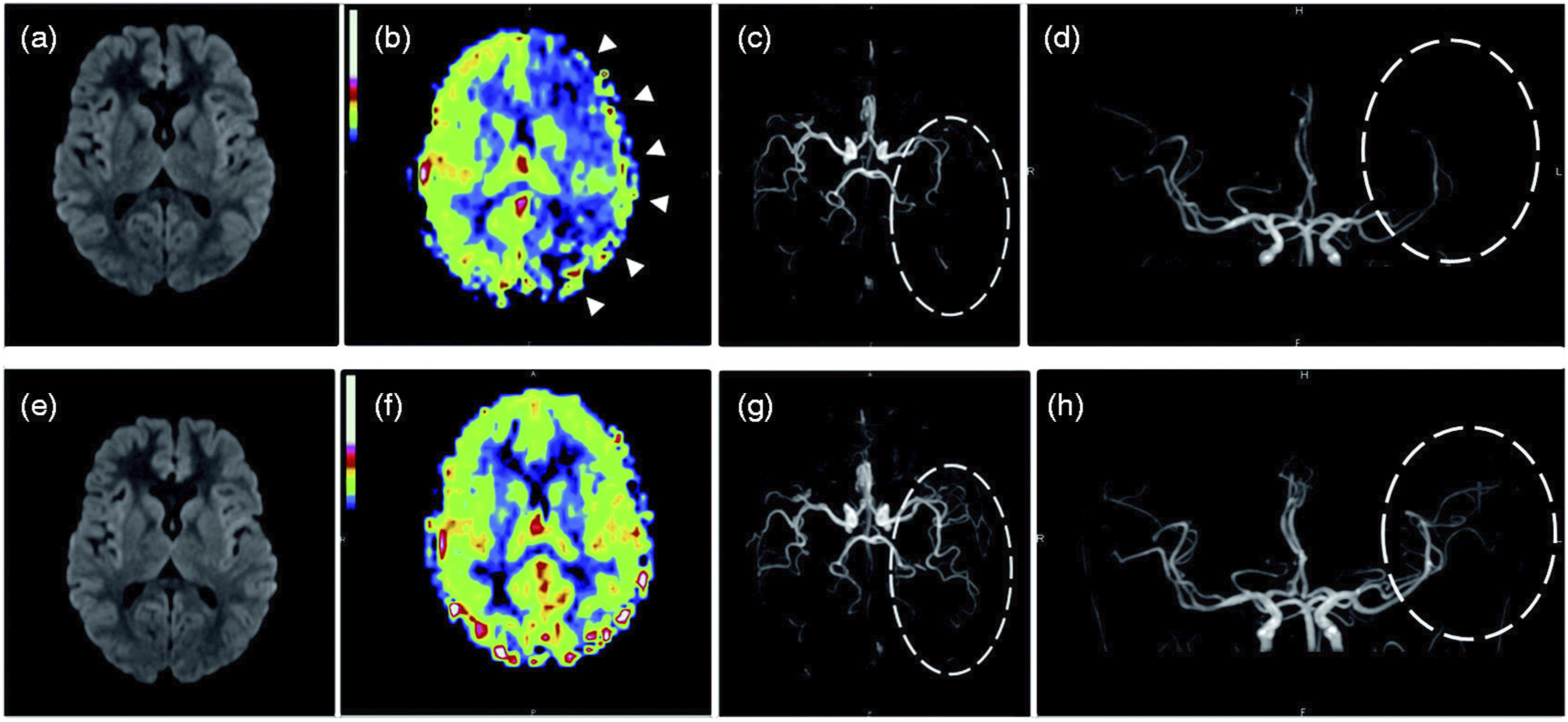
A case series of 17 patients published by Cadiot et al included 5 hemiplegic migraineurs, 4 of whom were scanned 2-10 hours after the onset of the aura. ASL images showed hypoperfusion in the contralateral frontal, temporal, parietal, and/or occipital regions, with corresponding areas of vasoconstriction on time-of-flight MRAs. One patient was scanned 2 days after the onset of the aura; ASL images and MRAs showed normal findings.
47
A summary of ASL changes in hemiplegic migraine is given in Table 3, with perfusion changes during the aura phase of the attack depicted in Figure 4. ASL demonstrates left hemispheric hypoperfusion (A). Note the hypo-oxygenated cortical veins on SWI along the left cerebral hemisphere (B). MRA notes thinning of the distal branches of the left middle and posterior cerebral arteries. Reprinted from Cobb-Pitstick KM et al. Time Course of Cerebral Perfusion Changes in Children with Migraine with Aura Mimicking Stroke. AJNR Am J Neuroradiol. 2018 Sep; 39 (9):1751-1755. https://doi.org/10.3174/ajnr.A5693. http://creativecommons.org/licenses/by/4.0/. http://creativecommons.org/licenses/by/4.0/. Changes in Perfusion Detected by Arterial Spin Labeling in Patients with Hemiplegic Migraine. A 13-year-old girl presenting with acute right sensory deficit, dysarthria and decreased level of consciousness. Initial MRI (a–d) performed 9 h 16 min after the onset of aura shows no ischemia on DWI (a), left hypoperfusion in the frontal, temporal, parietal and occipital lobes (arrow heads) on the ASL perfusion map (b), and vasospasm of the distal branches of the left middle cerebral arteries and posterior cerebral arteries (ellipse) on axial (c) and coronal (d) 3D TOF-MRA. Follow-up MRI (e–h) performed one day later shows no ischemia on DWI (e), normalization of the ASL perfusion map (f), and resolution of vasospasm (ellipse) on axial (g) and coronal (h) 3D TOF-MRA. Reprinted from Cadiot D et al. Magnetic resonance imaging in children presenting migraine with aura: Association of hypoperfusion detected by arterial spin labelling and vasospasm on MR angiography findings. Cephalalgia. 2018 Apr; 38 (5):949-958. https://doi.org/10.1177/0333102417723570. http://creativecommons.org/licenses/by/4.0/.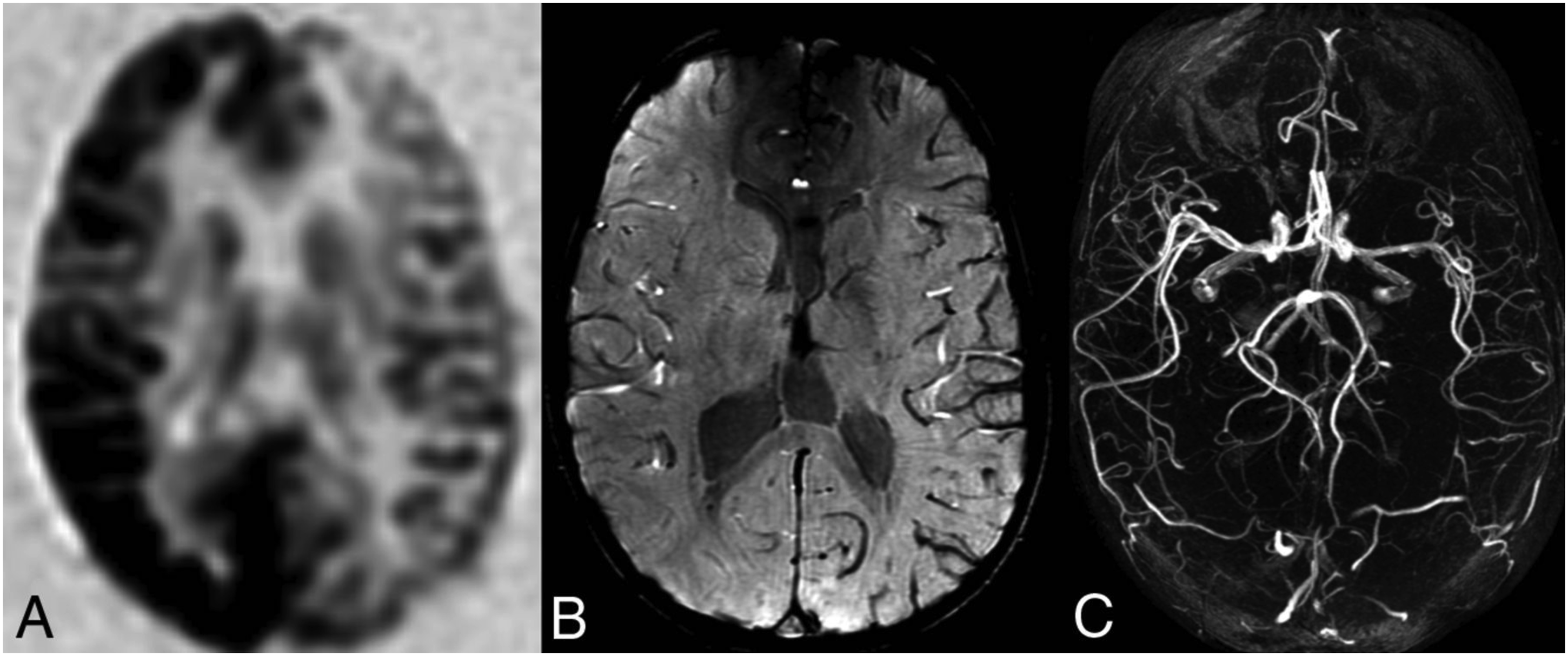

Migraine With Brainstem Aura
Migraine with brainstem aura is characterized by at least 2 of the following fully reversible symptoms: dysarthria, vertigo, tinnitus, hyperacusis, diplopia, ataxia, and decreased level of consciousness (Glasgow Coma Scale score, <13). 3 Migraine with brainstem aura was initially believed to be caused by vasospasm of the basilar artery. 58 However, that theory was never validated with imaging studies, and a more recent review by Demarquay and colleagues suggests a cortical origin of this aura. The investigators analyzed stereotactic electroencephalogram and functional mapping data from patients with symptoms of brainstem aura; they found that the symptoms could have originated from several cortical regions in the vestibular, auditory, and visual cortices. This finding further supports the role of cortical spreading depolarization in the origination of aura. 59
The ASL perfusion study of 17 migraineurs by Cadiot et al further supports a cortical origin of the brainstem aura. Their cohort included 12 patients with dysarthria and 3 with decreased levels of consciousness, all of whom demonstrated decreased perfusion in the cerebral cortex, predominantly in the left frontotemporoparietal region (Figure 4). 47 Additional case reports and case series on ASL findings in patients with brainstem aura are needed to validate this hypothesis.
Retinal Migraine
According to ICHD-3, the aura of a retinal migraine is characterized by fully reversible, monocular, positive and/or negative visual phenomena (eg, scintillations, scotomata, or blindness) confirmed by clinical visual field examination and/or the patient’s drawing of a monocular field defect and at least 2 of the following traits: aura spreading gradually over 5 minutes or longer and symptoms that last 5-60 minutes, accompanied by or followed within 60 minutes by headache. 3
Changes in Perfusion Detected by Arterial Spin Labeling in Migraineurs with Retinal Aura.

Hemiplegic Migraine vs Stroke vs Seizure
Clinically, the presentation of hemiplegic migraine may mimic an ischemic stroke, and evaluation of perfusion changes on ASL is often helpful in establishing the diagnosis. ASL, when used in conjunction with DWI and FLAIR sequences, demonstrates hypoperfusion in a vascular territorial distribution in ischemic stroke. While DWI and FLAIR sequences are helpful in estimating the infarct core, ASL can estimate the surrounding salvageable tissue ie, penumbra, thereby helping select patients for endovascular revascularization procedures.
61
Furthermore, ASL can often help localize the site of vascular occlusion, when used in conjunction with SWI sequences. An intraluminal thrombus manifests as a focus of high signal on ASL, due to the trapped, labeled arterial spins, with a corresponding focus of increased susceptibility on SWI.
62
Collateral vessels surrounding the infarct core are seen as serpiginous areas of increased signal intensity on ASL, due to delayed blood flow causing arterial transit artifact (Figure 5).
63
By contrast, these specific findings are conspicuously absent in hemiplegic migraineurs, who typically demonstrate no signal abnormalities on DWI images, with hypoperfusion in a non-vascular distribution during the aura phase of the attack (<12 hours from symptom onset) and hyperperfusion during the headache phase of the attack (>17 hours from symptom onset).
57
There are certain pitfalls to be aware of when using ASL to distinguish stroke from hemiplegic migraine. It is important to know that with a small infarct core, the area of hypoperfusion on ASL is often overestimated, and can appear non-territorial in distribution. The same is true in elderly patients with delayed arterial transit times, resulting in overestimation of the hypoperfused territory.
64
Furthermore, in patients with ischemic stroke and spontaneous reperfusion, there may be areas of increased perfusion on ASL, confounding the results. In these cases, careful correlation with areas of signal abnormality on structural imaging, as well as evaluation for vascular occlusion on MRA can help establish the diagnosis.
65
Mismatch DWI-ASL and collateral vessels. 60-year-old male presents to the ED with sudden onset acute aphasia and right facial droop. (a) MRI obtained approximately 1 h after the onset of symptoms demonstrates subtle restricted diffusion (white arrowheads) without corresponding hyperintense FLAIR signal in the left frontoparietal lobe and posterior aspect of the left temporal lobe, likely reflecting hyperacute to acute ischemic infarct in the left MCA territory. (b) MR perfusion images demonstrate the area of increased Tmax and MTT with decreased CBF without decreased CBV representing penumbra without core infarct. (c) Qualitative grayscale ASL maps demonstrate a wedge-shaped area of low signal in the left MCA territory (short white arrows) which is larger than the area of subtle restricted diffusion on diffusion-weighted images representing DWI-ASL mismatch or penumbra, also corresponding to the area of penumbra on MR perfusion images. Also, serpiginous high signal (long white arrows) surrounding the area of low signal on ASL images (short white arrows) due to Arterial Transit Time (ATA) compatible with the slow flow in collaterals and luxury perfusion. ASL ATA shows collateralization which portends a good prognosis in stroke. (d and e) Follow-up perfusion and ASL MR images after 24 h demonstrate interval resolution of previously seen wedge-shaped areas of low signal and resolution of perfusion deficits confirming penumbra on prior images. Findings are related to treatment with IV TPA. Reprinted from Daftari Besheli L et al. Arterial Spin Labeling technique and clinical applications of the intracranial compartment in stroke and stroke mimics - A case-based review. Neuroradiol J. 2022 Aug; 35 (4):437-453. https://doi.org/10.1177/19714009221098806. http://creativecommons.org/licenses/by/4.0/.
By contrast, differentiating hemiplegic migraine from seizure using ASL is more challenging, as both entities present with a non-territorial pattern of perfusion changes.
66
During the ictal phase of a seizure, the increased glucose and oxygen demand in the epileptogenic focus results in a regional hyperperfusion, that can be detected on ASL. It is difficult to distinguish the ictal phase of seizure from the headache phase of migraine on ASL, given that they both present with a non-territorial pattern of hyperperfusion.
67
As epileptogenic activity continues, and the regional hyperperfusion can no longer meet the elevated metabolic demands of the focus, cytotoxic edema ensues, seen as areas of reversible diffusion restriction and FLAIR hyperintensity on structural imaging (Figure 6).
68
In the post-ictal phase, decreased electrical activity in the region is paralleled by hypoperfusion on ASL, which can be difficult to differentiate from the aura phase of a migraine attack on ASL.
69
Careful correlation with structural imaging and electroencephalographic findings for an underlying anatomic abnormality, the reversibility of DWI and FLAIR changes in seizure can be helpful in differentiating hemiplegic migraine from seizure presenting with sensorimotor deficits. Ictal state seizure with Todd’s paralysis. 56-year-old male with seizure and focal right arm weakness and aphasia. (a) FLAIR image shows hyperintensity in the left temporoparietal lobes (arrow). (b) DWI image is negative for restricted diffusion. (c) Grayscale qualitative ASL image and quantitative color ASL map show area of increased signal corresponding to the area of hyperintensity on FLAIR image. Reprinted from Daftari Besheli L et al. Arterial Spin Labeling technique and clinical applications of the intracranial compartment in stroke and stroke mimics - A case-based review. Neuroradiol J. 2022 Aug; 35 (4):437-453. https://doi.org/10.1177/19714009221098806. http://creativecommons.org/licenses/by/4.0/.
In the pediatric population, ASL is now the first line perfusion modality for evaluating these three entities due to its lack of ionizing radiation, non-contrast technique, and its ability to quantify CBF in ml/100 g/min. 70 About 30% of pediatric migraineurs present with aura, and the spectrum of sensorimotor deficits is often difficult to distinguish from ischemic stroke and seizures. The non-territorial distribution of perfusion deficits, together with a lack of signal abnormality on DWI and FLAIR sequences and the dynamic perfusion changes on aura and headache in genetically susceptible individuals can increase the diagnostic accuracy of hemiplegic migraine in the pediatric population. 71
Limitations of ASL in Migraine
Although highly valued for its noncontrast technique, ASL is subject to limitations that have partly hindered its widespread clinical application in migraine imaging. The first limitation is the relatively longer acquisition time (∼3-5 minutes) as compared to other perfusion techniques eg, DSC perfusion imaging, which makes ASL more susceptible to motion artifact. Patient motion, in turn, can cause misregistration of control and label images, with erroneous subtraction and poor masking of background signal. 61 Apart from patient motion, ASL is also subject to a few technical artifacts, the most notable being faulty labeling of protons in arterial blood water, resulting in signal drop-off in the right-left, anterior-posterior or superior-inferior directions. Faulty labeling may be the result of coil sensitivity or gradient shim issues, but may also result from inappropriate head positioning, arterial tortuosity and external lines/tubes that distort the magnetic field. This artifact is particularly confounding in cases of typical and hemiplegic migraine, during the aura phase of the attack, when asymmetric perfusion is a key element in establishing the diagnosis. Prior administration of gadolinium will also cause a rapid decay in T1 relaxation time, resulting in the labeled protons losing most of their signal prior to entering the imaging slice of interest. 72 Lastly, geometric distortions from tissue-air interfaces, mineralization and blood products can cause regional signal loss and hamper the analysis of perfusion asymmetries on ASL in migraine patients. 73
Conclusions
ASL is a reliable, noninvasive method of quantifying perfusion in migraine cases. This imaging modality can help us to better understand the underlying pathophysiology of this complex disorder and potentially guide the development of future migraine subtype–specific treatments. Our comprehensive literature review revealed that perfusion is altered in most types of migraine headaches. However, the exact nature, pattern, timing, and duration of these perfusion changes have not been thoroughly studied or understood, most likely due to the practical challenges of imaging large numbers of cases during the aura, interictal period, and then after the headache attacks. More importantly, although migraine headache is considered 1 clinical entity, recent data confirmed that it is a heterogenous disorder with many categories that may have different patterns of perfusion changes and various pathophysiologies. Many previous studies did not separate cases based on whether their symptoms were chronic or episodic, which can also influence the results across cohorts and explain the conflicting results.
In migraine without aura, the current literature shows conflicting results, ie, perfusion is either increased or unchanged. Therefore, large cohort studies are needed to evaluate perfusion changes during and after the headache phase, separating cases based on types of headaches (ie, chronic vs episodic), with particular attention to perfusion in the dorsolateral pons, ipsilateral to the side of the attack. In contrast, during the interictal period, further evaluation of the ventrolateral prefrontal and anterior temporal regions via ASL imaging may help validate the findings of earlier studies.
Migraines with typical aura are characterized by hypoperfusion during the aura phase involves the cerebral hemisphere contralateral to the side of neurologic deficit, with a posterior predominance. More studies establishing the range of changes in perfusion during the aura phase and headache phase may help us differentiate migraine with aura from other cerebrovascular events. More importantly, to understand the underlying pathophysiology of migraine and its subtypes, it will be crucial to investigate the association between these perfusion changes and connectivity in various brain regions during the different phases of migraine. The changes in hemiplegic migraine are more dramatic, with hypoperfusion involving the entire cerebral hemisphere contralateral to the side of the deficit. Regarding the more uncommon migraine forms––migraine with brainstem aura and retinal migraine––studies of altered functional connectivity and ASL images may be particularly useful for developing a more comprehensive approach to the diagnosis and management of these disorders. Also, using ASL to differentiate hemiplegic migraine from stroke and focal seizures, particularly in the pediatric population, is a valuable resource, especially in the emergency setting. However, awareness of the limitations and technical artifacts of ASL is also important when interpreting findings of asymmetric perfusion. More widespread clinical application of this noninvasive imaging tool will help improve the diagnostic accuracy of these acute conditions.
Footnotes
Author contributions:
Alexander Lerner MD: editing, conceptualization. Daniel Phung MD: data collection, editing. Giuseppe Barisano MD: Collection of Images, Data. Wilson Xu BA: data collection, editing. Brendon Chou BS: Data Collection, Editing. Nasim Sheikh-Bahaei MD MRCP FRCR PhD: conceptualization, editing.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
