Abstract
Background
Inflammation may mediate response to acute reperfusion therapy (RT) in acute cerebral ischaemia. Neutrophil-lymphocyte ratio (NLR), an inflammatory biomarker, may play an important role in acute ischaemic stroke (AIS) prognostication.
Objective
This meta-analysis sought to examine the effect of NLR on functional outcomes, mortality and adverse outcomes in AIS patients receiving RT.
Methods
Individual studies were retrieved from PubMed/Medline, EMBASE and Cochrane databases. Data were extracted using a standardised data sheet and meta-analysis on association of admission (pre-RT) or delayed (post-RT) NLR with clinical/safety outcomes after RT was conducted.
Results
Thirty-five studies (n = 10 308) were identified for the systematic review with 27 (n = 8537) included in the meta-analyses. Lower admission NLR was associated with good functional outcomes (GFOs), defined as 3-month modified Rankin scale (mRS) 0–2 (SMD = −.46; 95% CI = −.62 to −.29; P < .0001), mRS 0–1 (SMD = −.44; 95% CI = −.66 to −.22; P < .0001) and early neurological improvement (ENI) (SMD = −.55; 95 %CI = −.84 to −.25; P < .0001). Lower delayed admission NLR was also associated with GFOs (SMD = −.80; 95%CI = −.91 to −.68; P < .0001). Higher admission NLR was significantly associated with mortality (SMD = .49; 95%CI = .12 to .85; P = .009), intracerebral haemorrhage (ICH) (SMD = .34; 95% CI = .09 to .59; P = .007), symptomatic ICH (sICH) (SMD = .48; 95% CI = .07 to .90; P = .022) and stroke-associated infection or pneumonia (SMD = .85; 95% CI = .50, 1.19; P < .0001). Higher delayed NLR was significantly associated with sICH (SMD = 1.40; 95% CI = .60 to 2.19; P = .001), ICH (SMD = .94; 95% CI = .41 to 1.46; P < .0001) and mortality (SMD = 1.12; 95% CI = .57 to 1.67; P < .0001). There were variations in outcomes across RT groups.
Conclusion
Higher admission or delayed NLR is significantly associated with worse morbidity, mortality and safety outcomes in AIS patients receiving RT.
Keywords
Introduction
Stroke is a leading cause of death and disease,1,2 and identifying prognostic biomarkers for this is of great clinical interest.3-6 Acute Ischaemic Stroke (AIS) forms the vast majority of strokes and occurs when blood flow within the brain is obstructed, with subsequent parenchymal hypoperfusion leading to a central infarct core and surrounding salvageable penumbra. 7 Recently, 2 reperfusion therapies (RT) have revolutionised AIS management and outcomes: Intravenous thrombolysis (IVT) and endovascular therapy (EVT). 8 Acute RT reduce conversion of penumbral tissue to core infarct by restoring blood flow. 9 Specifically, IVT shows benefit when administered within 4.5 hours of symptom onset, and EVT up to 24 hours.10-13 There is an increasing understanding of the role of immune-inflammatory system in AIS pathogenesis and RT response, as we have outlined previously. 14 Briefly, acute ischaemia causes blood-brain barrier (BBB) damage, allowing peripheral blood cell entry. Neutrophils are the first peripheral blood cells to enter the brain, within 1 hour, increasing tissue and BBB damage, which potentiates further peripheral cell entry.15-19 Lymphocytes generally enter 1–2 days post-AIS and are also thought to have a net deleterious effect14,16,20 Both these cells interact bidirectionally with resident brain immune cells to further mediate AIS damage.21-23 Neutrophils mediate this by aggravating thrombus formation and preventing the restoration of blood flow,16,19,20 but the role of lymphocytes is still unclear in the setting of AIS following RT.14,24
Blood-based biomarkers such as neutrophil-lymphocyte ratio (NLR) have been implicated in the ongoing management and prognosis of patients in the emergency medicine context,25-27 patients with acute and chronic coronary syndromes, including those receiving RT,28-31 and specifically in AIS. 14 Previous studies have demonstrated the utility of NLR in predicting AIS-related morbidity and mortality,32-34 angiographic outcomes,35,36 symptomatic intracerebral haemorrhage (sICH),37-39 and intracerebral haemorrhage (ICH). 40 However, the level of association of NLR with clinical outcomes in AIS patients receiving RT is yet to be clearly determined, with previous primary studies limited by small sample sizes and meta-analyses limited by combination of various blood collection timepoints and thresholds.34,37,38,40-42 For translation of NLR into a routine prognostic biomarker, further validation is warranted before its clinical utility can be fully established. This study sought to investigate the association of NLR, at admission and delayed timepoints, with clinical outcomes in patients receiving RT, by performing a systematic review and meta-analysis.
Our underlying research questions are: 1. Are admission and delayed NLR associated with long-term functional outcomes? 2. Are admission and delayed NLR associated with successful recanalization? 3. Are admission and delayed NLR associated with short-term functional outcomes? 4. Are admission and delayed NLR associated with safety outcomes? 5. Are admission and delayed NLR associated with stroke-associated infection (SAI) or stroke-associated pneumonia (SAP)?
We hypothesise that in RT-treated AIS patients, due to the deleterious impacts of neutrophils and lymphocytes, lower admission and delayed NLR may be associated with more favourable outcomes.
Methods
The study was performed in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA)
43
(Figure 1), Standards for Reporting Diagnostic Accuracy (STARD)-2015
44
(Supplemental Table 3) and Meta-analysis of Observational Studies in Epidemiology (MOOSE)
45
guidelines (Supplemental Table 4). Flow chart of study selection according to the PRISMA diagram. The PRISMA flowchart shows the main characteristics of the included studies. Outcomes for which a meta-analysis could successfully be carried out also have the number of patients shown. Abbreviations: N = Number of Included Studies; n = number of patients; GFOs = Good Functional Outcomes; mRS = Modified Rankin Scale; sICH = Symptomatic intracerebral haemorrhage; SR = Successful Recanalization; ICH = Intracerebral haemorrhage; ENI = Early Neurological Improvement; DENI = Dramatic Early Neurological Improvement; END = Early Neurological Deterioration; SAI = Stroke Associated Infection; SAP = Stroke Associated Pneumonia.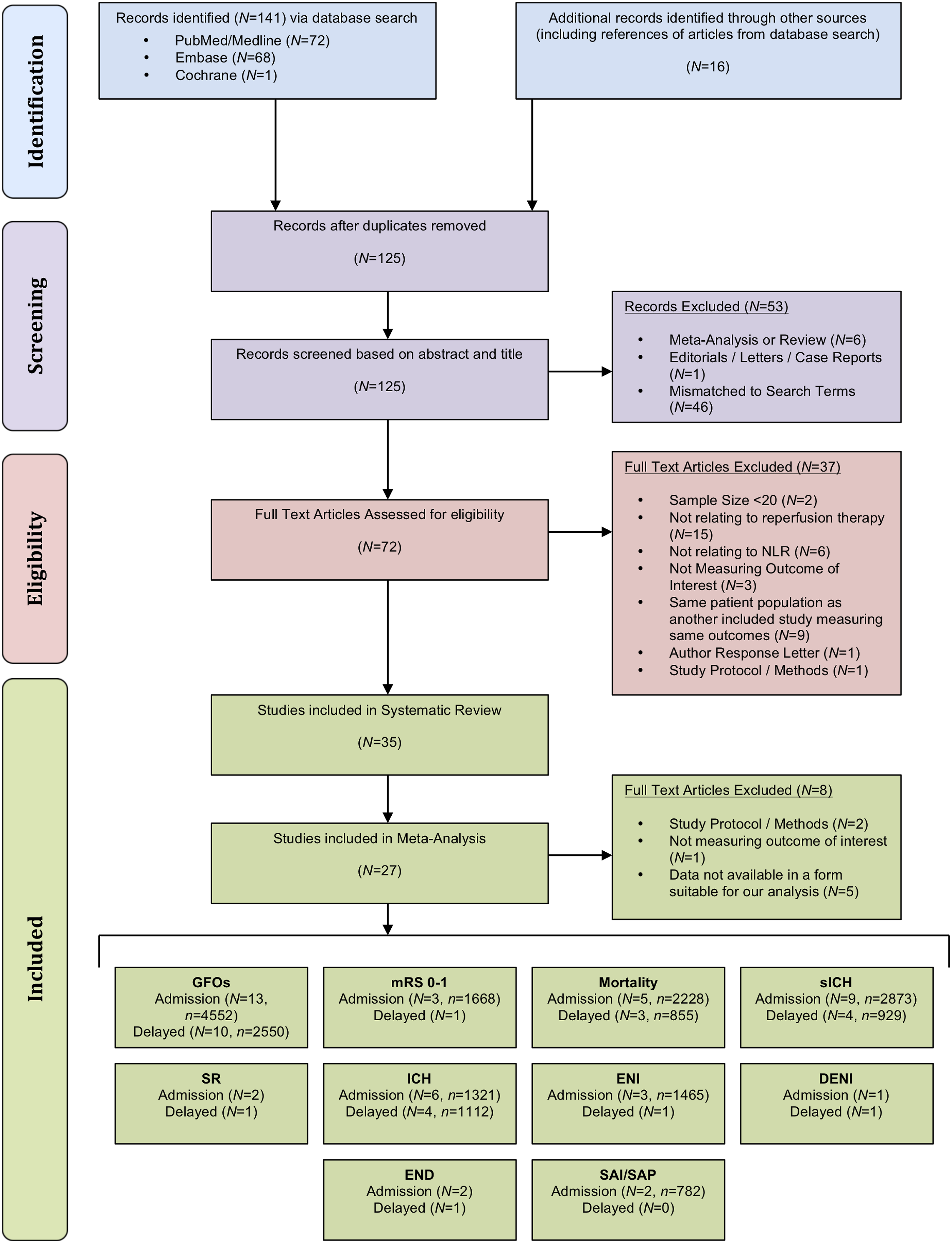
Literature Search: Identification and Selection of Studies
Published studies were retrieved from the following databases: Embase, PubMed/Medline and Cochrane Library until 6th July 2021, with no limits imposed upon the starting period. Keywords used in the search included terms or a combination of terms including: ‘acute stroke’, ‘cerebrovascular accident’, ‘brain ischemia’, ‘reperfusion’, ‘endovascular therapy’, ‘thrombectomy’, ‘thrombolysis’, ‘NLR’ and ‘neutrophil-lymphocyte ratio’. Full search strategies are provided in the Supplementary Information
Inclusion and Exclusion Criteria
Studies were eligible if they met the following criteria: (1) patients aged 18 or above; (2) patients diagnosed with AIS, (3) patients receiving RT; (4) studies with good methodological design (including sufficient sample size, determined to be > 20 patients in each group, and presence of a control population). The exclusion criteria were: (1) animal/preclinical studies; (2) duplicated publications; (3) where multiple studies from overlapping centres with varying study periods reporting similar outcomes were present, studies with smaller sample size or shorter study period were rejected; (4) full-text article not available; (5) systematic reviews, conference abstracts, meta-analyses, letters and case reports or series; and (6) studies presented in abstract form, with relevant data on NLR or control group not available or associated outcomes not reported.
Data Extraction
The title and abstracts were first reviewed using Endnote to rule out articles mismatched to the eligibility criteria. The remaining articles were examined thoroughly to determine whether they should be included for the systematic review or meta-analysis according to the eligibility criteria. Reviews, former meta-analyses and opinions were kept separately for further discussion in the manuscript. The screening was conducted independently by 2 authors. Disagreements were discussed and consulted until a consensus was made. Data from each study/trial was extracted independently using a standardised data extraction sheet to obtain the following information on: (1) baseline demographics: author, country and year of publication; (2) study population: age of patients, sample size, characteristics of AIS patients and RT type; (3) Neutrophil, Lymphocyte Count, NLR; (4) time of collection: admission (pre-intervention) and delayed (post-intervention); (5) outcome measures: primary outcome and secondary outcomes; functional outcomes, mortality, angiographic outcomes and diagnosis; and (6) adverse effects/safety outcomes. NLR was defined as either admission (pre-intervention) or delayed (post-intervention), with the timepoint closest to 24 hours selected for the latter in the case of multiple values. The primary outcome was defined in terms of morbidity: long-term functional outcomes (defined as good functional outcomes (GFOs) for modified Rankin scale (mRS) score of 0–2, and excellent outcome for mRS score of 0–1) and mortality at 3 months. Prognosis of good and excellent outcomes may be useful for decision-making and hence we included data on both these outcome variables as and when they were available. Short-term functional outcomes were: early neurological improvement (ENI) and dramatic ENI (DENI), defined as improvement in National Institutes of Health Stroke Scale (NIHSS) score 46 by 4 and 8 points respectively, across all studies, or complete recovery or drop to NIHSS 0 or 1, the latter varying between studies, as well as early neurological deterioration (END), conversely defined as NIHSS score worsening across all studies, with this being by 4 points across most studies. Successful recanalization (SR) was defined as mTICI ≥ 2b across all included studies. Considering safety outcomes, across all studies sICH was determined by neurological decline along with imaging confirmation, and ICH as any radiological evidence of bleeding, with individual variations in study definitions summarised in Supplemental Table 5.
Quality Assessment of Included Studies
The methodological quality of each study was assessed independently by 2 researchers using the modified Jadad scale.47,48 The scale evaluates study quality based on the following evaluation criteria: randomisation, blinding, withdrawals, dropouts, inclusion/exclusion criteria, adverse effects and statistical analysis. The total score for each study ranged from 0 to 8 points and using the 8 items, the trials/studies were divided into 2 levels. Trials/studies were considered of low quality if they achieved 0–3 points, and of high quality if they achieved 4–8 points. A double-blind got a score of 1 and single-blind .5.
The risk of funding bias in included studies was evaluated independently from the quality assessment through the declaration of funding sources and conflicts of interest using the scoring test developed by Saunders et al. (2017). 49 A score of 1–2 was considered to indicate a moderate potential for bias. The absence of industry funding was not taken to signify an absence of bias, but the presence of industry funding or conflicts of interest was assumed to be an indicator of bias.
Statistical Analysis
All statistical analyses were performed using STATA (Version 13.0, StataCorp LLC, College Station, Texas, USA). Forest plots were generated to present the standardised mean difference (SMD), 95% confidence intervals (CI), percentage weight and heterogeneity between studies included in the meta-analysis. Meta-analyses were split by admission NLR (pre-intervention) and delayed NLR (post-intervention). In cases where there were multiple delayed NLR timepoints, the timepoint closest to 24 hours was taken (Table 2). The I2 statistics and P-values were used to assess heterogeneity between studies, with <40%, 30–60%, 50–90% and 75–100% representing low, moderate, substantial and considerable heterogeneity, respectively. 50 A fixed-effects model was used for heterogeneity <50%, and random-effects model used for heterogeneity > 50%, across all subgroup analyses, with subgroup analyses performed for patients based on which was the primary treatment method, and which was the adjunct: IVT ± EVT, and EVT ± IVT. Where there was only IVT used, this is indicated on the forest plot. Baseline characteristics of patient populations were synthesised from all included studies. Where applicable, median and interquartile ranges were converted to mean and standard deviation using the method described by Wan et al. (2012), median and ranges were converted to mean and standard deviation using the methods described by Luo et al. (2018) 51 and Wan et al. (2014), 52 respectively, and for studies where SD was not available the method proposed by Walter and Yao (2007) was used to calculate SD, assuming the data was normally distributed. 53 Combined means were calculated where applicable. Where graphical representations of results were provided, numerical values were retrieved by 2 researchers independently. A (Begg’s) funnel plot was used to visually detect the presence of publication bias in the meta-analysis. Asymmetry on either side of the funnel plot is indicative of the presence of publication bias. This was also confirmed using Egger’s test of effect sizes for publication bias. Where possible, the command ‘metainf’ was used in STATA to determine the impact of individual studies on the overall meta-analysis (Supplemental Figure 1). P-values < .05 were considered statistically significant.
Results
Description of Included Studies
Overall summary of baseline clinical characteristics.
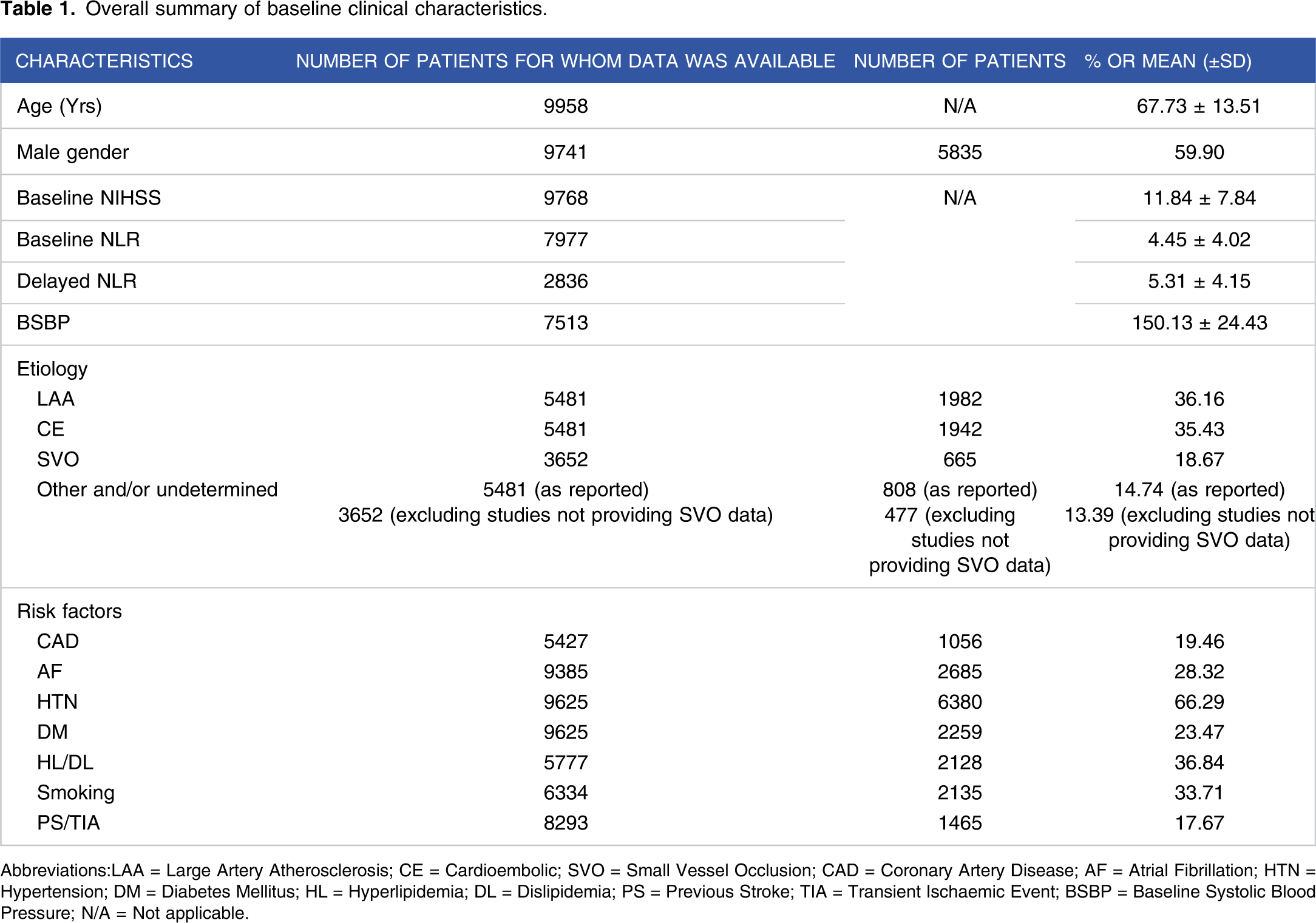
Abbreviations:LAA = Large Artery Atherosclerosis; CE = Cardioembolic; SVO = Small Vessel Occlusion; CAD = Coronary Artery Disease; AF = Atrial Fibrillation; HTN = Hypertension; DM = Diabetes Mellitus; HL = Hyperlipidemia; DL = Dislipidemia; PS = Previous Stroke; TIA = Transient Ischaemic Event; BSBP = Baseline Systolic Blood Pressure; N/A = Not applicable.
Clinical characteristics and outcomes of studies included in the meta-analysis.

All values provided to 2 decimal places. Where data was not available, this was left blank. Definitions of outcomes were as outlined in text unless specified otherwise. Further detail around individual study definitions of sICH and ICH outcomes are found in Supplementary Table 5.
*Only included in the systematic review.
**Definition not clearly specified.
***Data only available for 23 patients.
#Data only available for 276 patients.
##Value from tables provided.
###Definition via Thrombolysis in Brain Ischemia Scale.
&Definition via eTICI scale.
&&Defined this as any increase in NIHSS score from baseline.
Abbreviations: ENI = Early Neurological Improvement; DENI = Dramatic ENI; END = Early Neurological Decline; GFOs = Good Functional Outcomes; NLR = Neutrophil-Lymphocyte Ratio; sICH = symptomatic intracerebral haemorrhage; SR = Successful Recanalization; SAI = Stroke-Associated Infection; SAP = Stroke-Associated Pneumonia; NS = Not Specified; IVT = Intravenous Thrombolysis; EVT = Endovascular Therapy; USA = United States of America; UK = United Kingdom; IVT = Intravenous Thrombolysis; RT = Reperfusion Therapy.
Neutrophil-lymphocyte ratio (NLR) values stratified by outcome.
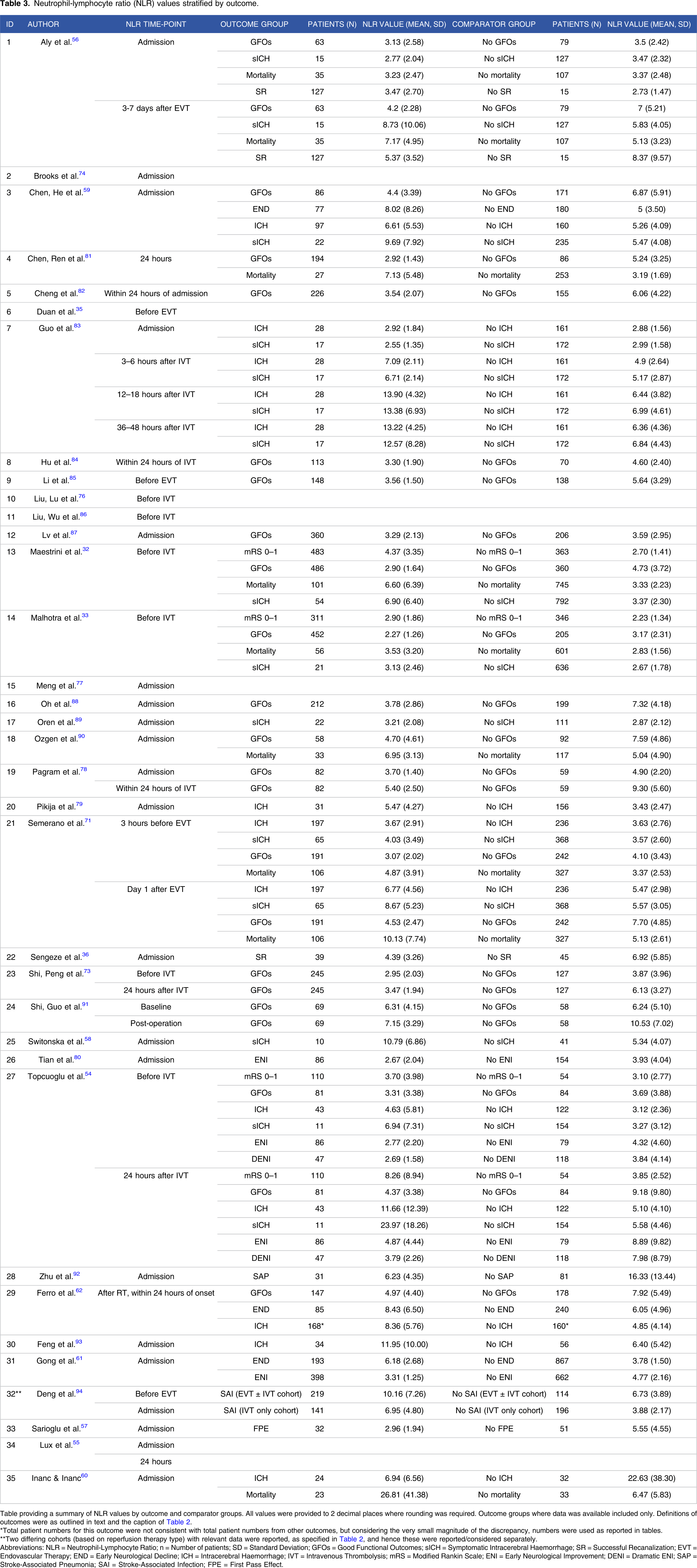
Table providing a summary of NLR values by outcome and comparator groups. All values were provided to 2 decimal places where rounding was required. Outcome groups where data was available included only. Definitions of outcomes were as outlined in text and the caption of Table 2.
*Total patient numbers for this outcome were not consistent with total patient numbers from other outcomes, but considering the very small magnitude of the discrepancy, numbers were used as reported in tables.
**Two differing cohorts (based on reperfusion therapy type) with relevant data were reported, as specified in Table 2, and hence these were reported/considered separately.
Abbreviations: NLR = Neutrophil-Lymphocyte Ratio; n = Number of patients; SD = Standard Deviation; GFOs = Good Functional Outcomes; sICH = Symptomatic Intracerebral Haemorrhage; SR = Successful Recanalization; EVT = Endovascular Therapy; END = Early Neurological Decline; ICH = Intracerebral Haemorrhage; IVT = Intravenous Thrombolysis; mRS = Modified Rankin Scale; ENI = Early Neurological Improvement; DENI = Dramatic ENI; SAP = Stroke-Associated Pneumonia; SAI = Stroke-Associated Infection; FPE = First Pass Effect.
Association of NLR With 90-day GFOs
There were 13 studies looking at admission NLR (pre-intervention), involving 4552 patients, and 10 studies reporting delayed NLR (post-intervention), comprising of 2550 patients.
Admission NLR with 90-day GFOs
The meta-analysis demonstrated significantly lower admission NLR in patients with GFOs (mRS 0–2) in comparison to those with poor functional outcomes (mRS 3–6) (SMD = −.46; 95% CI = −.62 to −.29; P < .0001; Figure 2). This significant effect was seen in both IVT ± EVT (SMD = −.41; 95% CI = −.62 to −.20; P < .0001) and EVT ± IVT patients (SMD = −.50; 95% CI = −.76 to −.23; P < .0001). There was non-significant heterogeneity between groups (P = .604), but substantial to considerable overall heterogeneity (I2 = 84.8%, P < .0001). No evidence of publication bias was observed by visual inspection of the funnel plot (Figure 5), and this was confirmed by the Egger’s test (Supplemental Table 2 & Supplemental Figure 2). The meta-analysis was repeated with further stratification of the IVT ± EVT group (Figure 2) into IVT only (SMD = −.28; 95% CI = −.50 to −.06; P = .011) and IVT ± EVT (SMD = −.62; 95% CI = −.74 to −.49; P < .0001) studies. There was significant heterogeneity between groups (P = .035). Forest plot of meta-analysis on the association of neutrophil-lymphocyte ratio (NLR) with functional outcomes and mortality at 90 days in acute ischaemic stroke patients receiving reperfusion therapy. Abbreviations: mRS = Modified Rankin Scale; NLR = Neutrophil-Lymphocyte Ratio; IVT = Intravenous Thrombolysis; EVT = Endovascular Thrombectomy. Note: Studies, where only IVT was used with no EVT adjunct, are denoted with an asterisk (*) unless these have been already split up into TPA and IVT ± EVT.
Delayed NLR with 90-day GFOs
The meta-analysis demonstrated significantly decreased NLR values, collected at delayed timepoints, in patients with 90-day GFOs (SMD = −.80; 95% CI = −.91 to −.68; P < .0001). All patients in the IVT ± EVT group received IVT only, and the forest plot reflected this (Figure 2). The significant effect was seen in patients receiving IVT only (SMD = −.87; 95% CI = −1.03 to −.71; P < .0001), EVT ± IVT (SMD = −.74; 95% CI = −.89 to −.59; P < .0001) and 1 study that had all treatment combinations (SMD = −.59; 95% CI = −.81 to −.36; P < .0001). There was a non-significant heterogeneity between groups (P = .119), and moderate overall heterogeneity (I2 = 48.7%, P = .041). No major evidence of publication bias was observed by visual inspection of the funnel plot (Figure 5), and this was confirmed by the Egger’s test (Supplemental Table 2 & Supplemental Figure 2).
Association of NLR With 90-day mRS 0–1
Considering mRS 0–1, there were 3 studies looking at admission NLR (pre-intervention), with 1668 patients, and only 1 study reporting delayed NLR with 165 patients; a meta-analysis could not be performed for the latter.
Admission NLR with 90-day mRS 0–1
The meta-analysis demonstrated that patients with 90-day excellent outcomes (mRS 0–1) had significantly lower admission NLR relative to those without (mRS 2–6) (SMD = −.44; 95% CI = −.66 to −.22; P < .0001; Figure 2). All studies contained patients receiving IVT ± EVT. There was substantial to considerable heterogeneity between studies (I2 = 75.0%, P = .018). No evidence of publication bias was observed from visual inspection of the funnel plot (Figure 5), and this was confirmed by the Egger’s test (Supplemental Table 2 & Supplemental Figure 2).
Delayed NLR with 90-day mRS 0–1
Only Topcuoglu et al. 54 provided relevant data, reporting that NLR was statistically significantly lower in the mRS 0–1 group.
Association of NLR With 90-day Mortality
There were 5 studies looking at admission NLR (prior to intervention), with 2228 patients, and 3 studies reporting delayed NLR with 855 patients.
Admission NLR with 90-day mortality
The meta-analysis demonstrated significantly increased admission NLR in patients with 90-day mortality (SMD = .49; 95% CI = .12 to .85; P = .009; Figure 2). This effect was seen in patients receiving IVT ± EVT (SMD = .74; 95% CI = .08 to 1.41; P = .028) but did not reach statistical significance in those receiving EVT ± IVT (SMD = .31; 95% CI = −.02 to .65; P = .067). There was a non-significant heterogeneity between groups (P = .259), but substantial to considerable overall heterogeneity (I2 = 88.2%, P < .0001). No evidence of publication bias was observed by visual inspection of the funnel plot (Figure 5), and this was confirmed by the Egger’s test (Supplemental Table 2 & Supplemental Figure 2).
Delayed NLR with 90-day mortality
The meta-analysis showed that higher delayed NLR values in patients with 90-day mortality (SMD = 1.12; 95% CI = .57 to 1.67; P < .0001). All patients in the IVT ± EVT group received IVT only, and the forest plot reflected this (Figure 2). The significant effect was seen in the 1 study containing patients receiving IVT only (SMD = 1.69; 95% CI = 1.27 to 2.11; P < .0001) and both studies where patients received EVT ± IVT (SMD = .86; 95% CI = .29 to 1.42; P = .003). There was significant heterogeneity between groups (P = .020), and substantial to considerable overall heterogeneity (I2 = 87.1%, P < .0001). No evidence of publication bias was observed by visual inspection of the funnel plot (Figure 5), and this was confirmed by the Egger’s test (Supplemental Table 2 & Supplemental Figure 2).
Association of NLR With SR
Admission NLR grouped according to SR status (SR vs no-SR) was only reported in 2 studies (n = 226), and only 1 study reported delayed NLR (n = 142) grouped as such. Thus, a meta-analysis could not be performed for either due to an insufficient number of studies. For this outcome, 2 studies also considered temporal NLR changes from admission to 24 hours, with mixed results, as Lux et al. reported that these were not significantly associated with SR, 55 but Aly et al. reported statistically significant smaller temporal changes in the SR group. 56
Admission NLR with SR
The systematic review indicated mixed results, with Aly et al. 56 reporting a higher NLR in the SR group and Sengeze et al. 36 a lower NLR. Differences between groups were statistically significant in both studies. Lux et al. and Duan et al. did not provide groupwise data by outcome but reported conflictingly that admission NLR was not significantly correlated with SR and NLR > 7 was significantly associated with higher SR rates, respectively.35,55
Delayed NLR with SR
Again, the systematic review indicated mixed results. Aly et al. reported a statistically significantly higher NLR in the SR group. Lux et al. did not provide groupwise data but reported that admission NLR was not significantly associated with SR. 55 One study also considered the first pass effect (FPE), where complete recanalization (mTICI 3) is achieved with a single pass and reported a lower NLR in the FPE group. 57
Association of NLR With sICH
There were 9 studies looking at admission NLR (prior to intervention), with 2873 patients, and 4 studies reporting delayed NLR with 929 patients.
Admission NLR with sICH
The meta-analysis revealed higher admission NLR in sICH patients relative to non-sICH (SMD = .48; 95% CI = .07 to .90; P = .022; Figure 3). However, subgroup analyses failed to reach statistical significance in both IVT ± EVT (SMD = .50; 95% CI = −.14 to 1.13; P = .123) and EVT ± IVT groups (SMD = .28; 95% CI = −.33 to .89; P = .376), with a study where all treatment combinations were reported forming a third group (SMD = 1.16; 95% CI = .43 to 1.89; P = .002). There was a non-significant heterogeneity between studies (P = .180), but substantial to considerable overall heterogeneity (I2 = 88.0%, P < .0001). Evidence of publication bias was observed by visual inspection of the funnel plot (Figure 5), and this was confirmed by the Egger’s test (Supplemental Table 2 & Supplemental Figure 2). Forest plot of meta-analysis on the association of neutrophil-lymphocyte ratio (NLR) with bleeding complication outcomes in acute ischaemic stroke patients receiving reperfusion therapy. Abbreviations: ICH = Intracerebral Hemorrhage; sICH = Symptomatic Intracerebral Hemorrhage; NLR = Neutrophil-Lymphocyte Ratio; IVT = Intravenous Thrombolysis; EVT = Endovascular Thrombectomy. Note: Studies, where only IVT was used with no EVT adjunct, are denoted with an asterisk (*) unless these have been already split up into IVT and IVT ± EVT.
Further stratification of the IVT ± EVT group into IVT only (SMD = .57; 95% CI = −.29 to 1.44; P = .191) and IVT ± EVT (SMD = .44; 95% CI = −.53 to 1.40; P = .376) still yielded statistically insignificant results for each subgroup (Figure 3), and a non-significant heterogeneity between groups (P = .328). Removing the study will all treatment combinations, Switonska et al., 58 caused statistical significance to be lost (SMD = .41; 95% CI = −.02 to −.85), but this was also observed for the removal of 3 other studies32,54,59 (Supplemental Figure 1). Inanc & Inanc, 60 included in the systematic review but not meta-analysis, also reported an increased NLR in sICH patients as opposed to non-sICH.
Delayed NLR with sICH
The meta-analysis showed significantly higher delayed NLR in sICH patients relative to non-sICH (SMD = 1.40; 95% CI = .60 to 2.19; P = .001 Figure 3). This was seen in both IVT ± EVT (SMD = 2.11; 95% CI = .52 to 3.70; P = .009) and EVT ± IVT treated groups (SMD = .83; 95% CI = .58 to 1.08; P < .0001). There was non-significant heterogeneity between groups (P = .118), but considerable overall heterogeneity (I2 = 91.1%, P < .0001) was observed. No evidence of publication bias was observed by visual inspection of the funnel plot (Figure 5), and this was confirmed by the Egger’s test (Supplemental Table 2 & Supplemental Figure 2). Notably, the overall effect became markedly weaker with the exclusion of Topcuoglu et al. but was still statistically significant (SMD = .93; 95% CI = .57 to 1.27; Supplemental Figure 1).
Association of NLR With ICH
Six studies comprising of 1321 patients reported admission NLR data, whilst 4 studies reported relevant delayed NLR data, in 1112 patients.
Admission NLR with ICH
The meta-analysis demonstrated significantly higher admission NLR in patients with ICH relative to non-ICH (SMD = .34; 95% CI = .09 to .59; P = .007; Figure 3). Patients treated with IVT ± EVT showed no significant effect (SMD = .24; 95% CI = −.15 to .62; P = .229), but a significant effect was observed in EVT ± IVT treated patients (SMD = .40; 95% CI = .05 to .75; P = .024). Non-significant heterogeneity between groups (P = .537), albeit substantial overall heterogeneity (I2 = 73.0%, P = .004), was observed. Possible publication bias was observed by visual inspection of the funnel plot (Figure 5), and this was supported by Egger’s test (Supplemental Table 2 & Supplemental Figure 2).
Delayed NLR with ICH
Significantly increased NLR collected at delayed time points was observed in patients with ICH (SMD = .94; 95% CI = .41 to 1.46; P < .0001; Figure 3). Patients treated with IVT ± EVT showed this significant effect (SMD = 1.40; 95% CI = .42 to 2.39; P = .005), as did EVT ± IVT treated patients (SMD = .34; 95% CI = .15 to .53; P < .0001) and patients for studies where all treatment combinations were used (SMD = .70; 95% CI = .41 to 1.46; P < .0001), albeit with only 1 study included in both these groups. There was significant heterogeneity between groups (P = .012), and considerable overall heterogeneity (I2 = 93.0%, P < .0001). Possible publication bias was observed by visual inspection of the funnel plot (Figure 5), but this was not supported by Egger’s test (Supplemental Table 2 & Supplemental Figure 2).
Association of NLR With ENI
There were 3 studies reporting admission NLR values with 1465 patients, and one study reporting relevant delayed NLR data, with 165 patients; a meta-analysis could not be carried out for the latter.
Admission NLR with ENI
The meta-analysis showed that patients with ENI have significantly lower admission NLR relative to those without ENI (SMD = −.55; 95% CI = −.84 to −.25; P < .0001; Figure 4). All patients were treated with IVT ± EVT, and hence this was split into IVT only (SMD = −.40; 95% CI = −.60 to −.19; P < .0001) and IVT ± EVT (SMD = −.78; 95% CI = −.91 to −.65; P < .0001) groups, the latter having only 1 study. There was significant heterogeneity between groups (P = .002) and substantial to considerable overall heterogeneity (I2 = 79.8%, P = .0007). No evidence of publication bias was observed by visual inspection of the funnel plot (Figure 5), and this was supported by Egger’s test (Supplemental Table 2 & Supplemental Figure 2). Notably, the omission of Gong et al.
61
caused this effect to weaken (SMD = −.60; 95% CI = −.40 to −.19) most, as compared to the omission of other studies (Supplemental Figure 1). Forest plot of meta-analysis on the association of neutrophil-lymphocyte ratio (NLR) with early neurological improvement and stroke-associated infection outcomes in acute ischaemic stroke patients receiving reperfusion therapy. Abbreviations: ENI = Early Neurological Improvement; SAI = Stroke-Associated Infection; SAP = Stroke-Associated Pneumonia; NLR = Neutrophil-Lymphocyte Ratio; IVT = Intravenous Thrombolysis; EVT = Endovascular Thrombectomy. Note: Studies, where only IVT was used with no EVT adjunct, are denoted with an asterisk (*), unless these have been already split up into IVT and IVT ± EVT. Funnel plots of meta-analyses studies on effect of neutrophil-lymphocyte ratio (NLR) on clinical and safety outcomes in acute ischemic stroke patients receiving reperfusion therapy. A: Admission NLR association with Good Functional Outcomes; B: Delayed NLR association with Good Functional Outcomes; C: Admission NLR association with mRS 0–1; D: Admission NLR association with Mortality; E: Delayed NLR association with Mortality; F: Admission NLR association with sICH; G: Delayed NLR association with sICH; H: Admission NLR association with ICH; I: Delayed NLR association with ICH; J: Admission NLR association with ENI; K: Admission NLR association with stroke-associated infection/pneumonia (SAI)/(SAP). Note: Funnel plots for each meta-analysis.

Delayed NLR with ENI
Only Topcuoglu et al. 54 provided relevant data, reporting that NLR was statistically significantly lower in the ENI group.
Association of NLR With DENI
Only 1 study with 165 patients reported relevant NLR data for this outcome; thus, a meta-analysis was not carried out. However, in this study both admission and delayed NLR were lower in the group with DENI, but this was not statistically significant. 54 Considering the systematic review, Inanc & Inanc looked at correlation between NIHSS scores and median admission NLR 60 was not statistically significant.
Association of NLR With END
Two studies reported admission NLR data, with a total of 1317 patients, and only 1 study for delayed NLR data, with 325 patients.
Admission NLR association with END
The systematic review indicated that higher admission NLR was associated with END, with both studies reporting statistically significant groupwise data reflecting this.59,61
Delayed NLR association with END
Only Ferro et al. 62 provided relevant data, reporting a lower NLR in the END group, and a significant association of delayed NLR with END on multivariate analysis.
Association of NLR With SAI And/or SAP
For this meta-analysis, SAI and stroke-associated pneumonia (SAP) were combined. Only admission NLR values were reported, in 2 studies, with 782 patients, although one of these studies reported 2 cohorts based on treatment regimen, and thus a meta-analysis was successfully conducted.
Admission NLR with SAI or SAP
The meta-analysis found significantly higher admission NLR in patients with SAI or SAP (SMD = .85; 95% CI = .50 to 1.19; P < .0001; Figure 4). This significant effect was maintained in EVT ± IVT (SMD = .87; 95% CI = .18, 1.56; P = .014) and IVT only (SMD = .87; 95% CI = .65, 1.10; P < .0001) groups, though the latter contained only 1 study. There was non-significant heterogeneity between the groups (P = .996), but substantial to considerable overall heterogeneity (I2 = 77.7%, P < .011). No evidence of publication bias was observed by visual inspection of the funnel plot (Figure 5), and this was confirmed by the Egger’s test (Supplemental Table 2 & Supplemental Figure 2).
Discussion and Conclusions
This study investigated the association of NLR with clinical and safety outcomes in AIS patients receiving RT. We demonstrate that both admission and delayed NLR are significantly associated with 90-day GFOs (mRS 0–2), and that lower admission NLR is associated with 90-day excellent outcomes (mRS 0–1). Higher admission NLR was significantly associated with SAI or SAP. Notably, we also show that delayed NLR has a larger SMD for GFOs than admission NLR demonstrating better prognostic utility. Increased admission and delayed NLR were both associated with ICH, sICH and mortality. As such, we clearly show that there is a role for NLR in both prognostication and improved clinical safety outcomes, underscoring the clinical implications of NLR in AIS patients receiving RT.
Inflammation is known to play a role in the pathophysiology of AIS, and NLR has been established as a marker for this. 63 This potentially explains our finding that lower admission and delayed NLR is associated with better outcomes and is supported by previous meta-analyses37,41,42 which demonstrate a predictive role of NLR for predicting clinical outcomes. By imposing more stringent criteria for NLR collection timepoints, we demonstrated that delayed NLR has a larger SMD for GFOs than admission NLR. This could be related to underlying pathophysiology; as lymphocyte entry into ischaemic tissue is thought to occur some 1–2 days after initial cerebral ischaemia, 16 and subsequently causes further pro-inflammatory cytokine release and damage9,16,19,20; measuring NLR at this timepoint may improve prognostication for functional outcomes. 14 This contradicts earlier findings by Song et al., 34 who reported that admission NLR was better at predicting 3-month mRS outcomes than delayed NLR timepoints. However, our meta-analysis includes more than twice the number of studies, including numerous more recent studies, and we impose a more stringent inclusion criteria, as they also included studies with good functional outcomes defined as mRS 0–1, as well as mRS assessments at times other than 90 days. Additionally, our finding that admission NLR is lower in patients with 90-day GFOs could help stratify patients and guide initial management decisions. 14 Repeating this analysis also showed that NLR SMD was even lower in patients receiving IVT ± EVT than those receiving IVT only. This may have clinical decision-making implications with regards to treatment selection, and shows that there is a role for admission NLR in prognostication for patients receiving adjunct EVT, despite previous authors having hypothesised that EVT may modify outcomes and hence admission NLR would not have predictive value. 55 Our meta-analysis also showed that lower admission NLR was significantly associated with excellent outcome at 3-month (mRS 0–1), with a similar SMD to patients with GFOs, suggesting that NLR may be able to predict both mRS 0–1 and GFOs, and hence could potentially inform treatment stratification and follow-up.
Considering mortality, higher admission NLR was seen in patients with 90-day mortality. This is consistent with previous findings, thought to reflect the more pronounced immune response a higher NLR denotes.34,37,40 Notably, this effect did not reach statistical significance in EVT ± IVT treated patients, which may be related to the selection cohort of patients eligible for EVT; patients offered EVT may be more likely to benefit from their treatment in comparison to patients receiving IVT, due to the more stringent inclusion criteria for the former. 55 Interestingly, the 3 studies for mortality with a patient cohort receiving EVT ± IVT had higher baseline NIHSS scores than patients treated with IVT ± EVT, which may support the notion that treatment differences may play a role in mortality outcomes. We also showed that delayed NLR was associated with 90-day mortality. As with GFOs, a larger SMD was observed with delayed NLR than admission NLR for 90-day mortality, which may relate to the pathophysiological mechanisms outlined earlier. 14 Infections, a major cause of in-hospital mortality, were also examined as combined SAI or SAP, and we found that higher admission NLR was seen in patients with infections. This could be related to prior exposure to a pathogen such as LPS, or potential concurrent stimulation of other molecules such as matrix metalloproteinases, which may cause increased autoimmune responses, and resultantly raise NLR.21,64 In the setting of Coronavirus Disease 2019 (COVID-19), there is a particular role for biomarkers in triaging, prognosticating and stratification of treatment in patients with COVID-19, considering reports of increased predisposition to and incidence of AIS, as well as concurrent use of multiple biomarkers to form more accurate prognostic nomograms.14,65,66 This could be of particular utility if point-of-care instrumentation incorporating NLR were to be successfully developed during and beyond the pandemic,14,67 considering the strain on healthcare systems and increasing uptake of telemedicine.66,68
Our meta-analysis demonstrated that overall increased admission NLR was significantly higher in sICH patients, but interestingly, this was not true for the subgroup analyses stratified by RT type. The detected publication bias may have played a role in this. The overall effect observed is consistent with the meta-analysis by Zhang et al. (2017) which indicated a prognostic role of NLR for sICH. However, stratification by timepoint and treatment type was not performed unlike our study.14,38 In contrast, delayed NLR values showed a higher SMD for sICH, and this significant effect was preserved regardless of the treatment administered, though the SMD was larger in the IVT ± EVT group than the EVT ± IVT group. Given that higher NLR is associated with larger infarct volume 69 and IVT can increase sICH risk in large infarcts, 70 there may be a role for NLR in guiding treatment decision-making pertaining to suitability for IVT. 14 Additionally, underlying factors such as patients’ collaterals status 71 or a history of coronary artery disease, which may manifest as increased anti-platelet use, 39 may have influenced the results observed, but very few studies reported on this and hence we could not account for these. Both admission and delayed NLR were found to be significantly higher in patients with ICH, although in the former group, there was an evidence of publication bias. Notably in subgroup analyses, EVT ± IVT patients showed both significantly lower admission as well as delayed NLR, but this was not seen in IVT ± EVT patients for the admission NLR group. This differs from previous findings by Zhang et al. (2019), who performed a meta-analysis stratified by RT type similar to our study and found NLR to have significant associations with ICH in patients receiving primary IVT but not EVT.14,40 This difference may be because we include a greater number of more recent studies and impose stricter criteria for NLR time points. Additionally, the difference between associations of NLR with ICH in IVT ± EVT and EVT ± IVT patients in our study might be related to a potential role of IVT in exacerbating blood-brain barrier (BBB) dysfunction, resulting in delayed (post-intervention) NLR having more prognostic value in patients receiving IVT ± EVT than admission (pre-intervention) NLR. 54 Nonetheless, the role of the detected publication bias cannot be discounted, and hence there is a pressing need for further prospective studies considering admission NLR in IVT ± EVT patients developing ICH.
There is a scarcity of studies focussing on SR outcomes. Our systematic review indicated mixed results and thus further research is necessary given the potential role of neutrophils in no-reflow injury.14,16 With regards to short-term functional outcomes, we could only carry out a meta-analysis to evaluate the association of admission NLR with ENI, which revealed significantly lower admission NLR in patients with ENI. There are very few studies linking NLR with other short-term functional outcomes, although all included studies showed prognostic benefit. This could be related to NLR being a marker of inflammation and hence being associated with the degree of immune response and neurological outcomes.14,20,56,72,73 Resultantly, it is critical that further studies be carried out to ascertain this, as well as the role of temporal NLR variations.
The major strengths of our study are use of SMD to account for the continuous nature of NLR, whereas previous meta-analyses have combined various thresholds without such standardisation.37,38,41,42 Additionally, we looked at admission and delayed NLR separately, which is poignant due to the pathophysiological considerations owing to delayed entry of lymphocytes and subsequent pro-inflammatory cytokine release.9,16,19,20 By considering IVT ± EVT and EVT ± IVT treated patients separately, our meta-analysis provided comparison of the utility of NLR in either of these patient groups, which can benefit further prognostication, stratification and treatment selection. Finally, we also incorporated the most recent evidence.
Our work is not without limitations. Most of our included studies were retrospective, and there was resultantly high heterogeneity; we sought to minimise this through random-effects modelling. Additionally, varying NLR timepoints were reported; whilst we attempted to address this by defining admission and delayed NLR using pre- and post-intervention NLR, with the timepoint closest to 24 hours in each study used for the latter, NLR’s dynamic profile means there may be some impact on results observed for delayed NLR. Very few studies looked at the dynamic NLR profile and hence this could only be considered in the systematic review pertaining to SR. Some studies failed to exclude patients with underlying or acute inflammatory conditions, which can impact NLR,32,33,39,60,71,73-80 along with the site of blood procurement, which was not specified in several studies. Racial and ethnic differences in NLR have been reported previously,34,38,40,79 but data was not available for these, and thus we could not account for these. Our study population was AIS patients receiving RT, and thus, should not be extrapolated beyond this group. A further high quality RCT is recommended to corroborate our findings, with specific attention to standardisation of the timepoints and sites at which blood is collected, and appropriate exclusion of patients with infections or acute or chronic inflammatory conditions.
In conclusion, NLR is an important prognostic biomarker in AIS patients receiving RT. Our meta-analysis shows a role for both admission and delayed NLR in predicting long-term functional outcomes and mortality, especially in patients receiving EVT, and for delayed NLR in predicting bleeding complications (ICH and sICH). Our findings indicate a role of NLR in treatment selection and post-RT prognostication.
Supplemental Material
Supplemental Material - Role of Neutrophil-Lymphocyte Ratio in the Prognosis of Acute Ischaemic Stroke After Reperfusion Therapy: A Systematic Review and Meta-analysis
Supplemental Material for Role of Neutrophil-Lymphocyte Ratio in the Prognosis of Acute Ischaemic Stroke After Reperfusion Therapy: A Systematic Review and Meta-analysis by Divyansh Sharma, Kevin J. Spring and Sonu M. M. Bhaskar in Journal of Central Nervous System Disease
Footnotes
Acknowledgments
Funding for the NSW Brain Clot Bank (Chief Investigator: Dr SMM Bhaskar) from the NSW Ministry of Health (2019-2022) is acknowledged. The funding body has no role in the study design, data collection, analysis, interpretation of findings, and manuscript preparation. The content is solely the responsibility of the authors and does not necessarily represent the official views of the affiliated/funding organization/s.
Authors’ Contributions
SMMB conceived the study, contributed to the planning, draft, and revision of the manuscript; supervision of the student. SMMB encouraged DS to investigate and supervised the findings of this work. SMMB and DS wrote the first draft of this paper. All authors contributed to the revision of the manuscript. All authors approved the final draft of the manuscript.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the NSW Brain Clot Bank (Chief Investigator: Dr Sonu M. M. Bhaskar) from the NSW Ministry of Health (2019-2022) is acknowledged.
Data Availability Statement:
The original contributions presented in the study are included in the article and Supplementary Information online, further inquiries can be directed to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
