Abstract
A 71-year-old woman with a diagnosis of normal tension glaucoma (NTG) presented with complains of progressive visual loss in the right eye. Examination revealed features consistent with compressive optic neuropathy. Although brain magnetic resonance imaging (MRI) was initially interpreted as normal, re-evaluation disclosed a compression on the right optic nerve from the right internal carotid artery. We highlight the clinical differential diagnosis between NTG and compressive optic neuropathy. This case is a reminder that a compressive optic neuropathy may be caused by anatomical variation of normal intracranial structures.
Keywords
A 71-year-old woman presented with progressive painless visual loss in the right eye (OD) for the past 10 months. Two years before, a diagnosis of bilateral (OU) Normal Tension Glaucoma (NTG) (OD worse than OS) was made and timolol .5% bid OU was prescribed. Her past general history was non-contributory, and records before glaucoma therapy showed that intraocular pressure (IOP) were never higher than 20 mmHg OU. On examination, best corrected visual acuity (BCVA) was 20/40 OD and 20/20 OS. Pupils were equal with a right relative afferent pupil defect (RAPD). Slit lamp examination was unremarkable, and intraocular pressure (IOP) was 14 mmHg OU. Central corneal thickness was 540 microns OU. Fundus examination revealed asymmetric cupping of the optic disc .5 OD with mild superior rim pallor, while the optic nerve and cup (.1) were normal OS
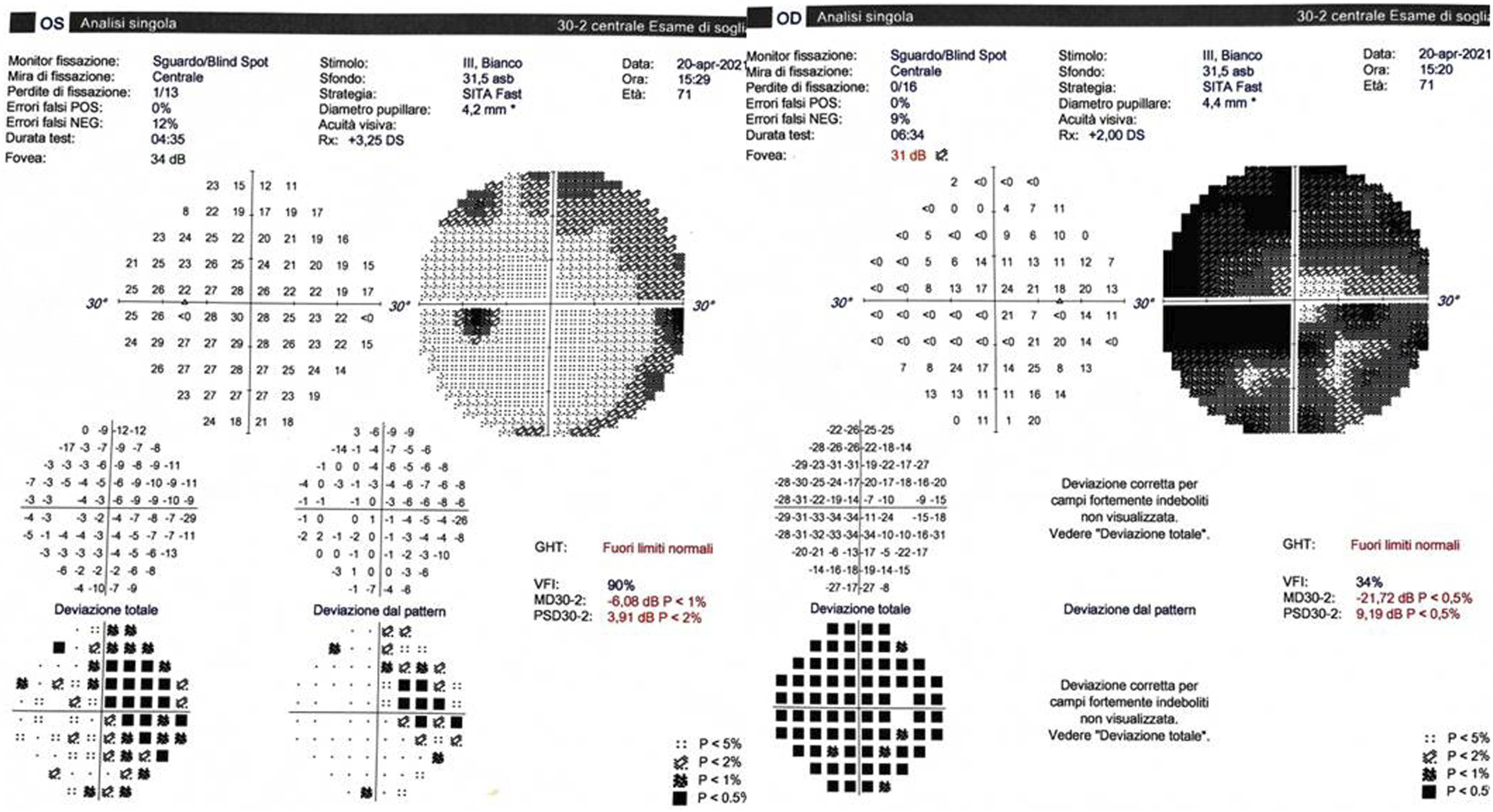
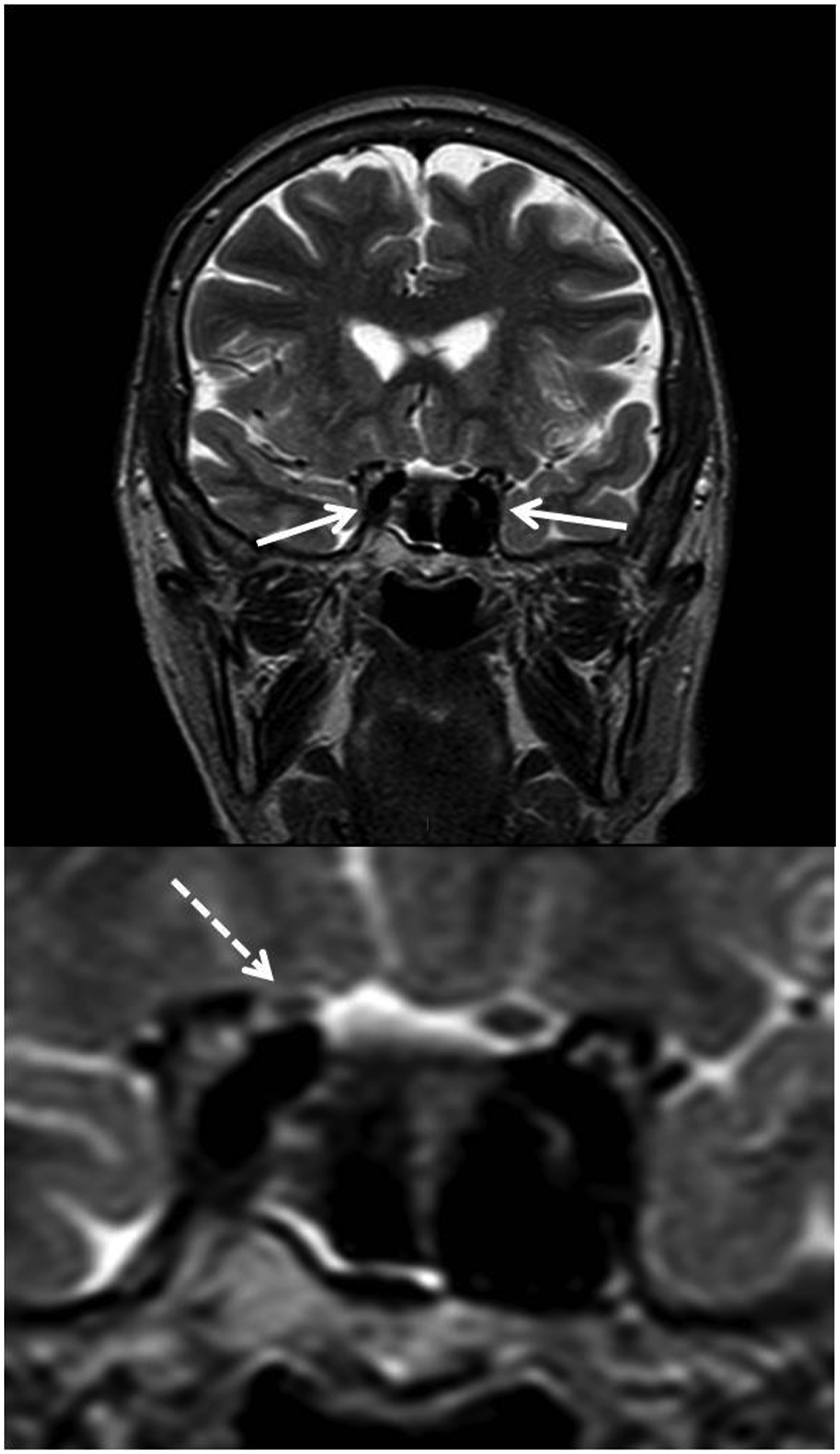
In Figure 1, standard computerized perimetry shows a diffuse defect OD with a relative central sparing and a nasal defect OS (Figure 2). Brain magnetic resonance imaging (MRI) was requested for compressive optic neuropathy (CON) and, although interpreted as normal, showed a tortuous and enlarged, dolichoectatic, right internal carotid artery (ICA). The optic nerve OD appeared thinned and elevated when compared to the contralateral side (Figure 3). A diagnosis of CON caused by ICA was made. A neurosurgical consultation was requested, but the patient was lost on follow-up. Fundus examination shows glaucomatous type cupping of the optic disc OD with a mild pallor and a normal optic disc OS. Standard computerized perimetry shows a diffuse defect OD with a relative central sparing, and a nasal defect OS. T2-weighted brain magnetic resonance imaging. Top: coronal view shows tortuous internal carotid arteries (arrows). Bottom: magnified view of the distal optic nerves shows the right optic nerve (dotted arrow) thinned and displaced superiorly when compared to the contralateral normal one.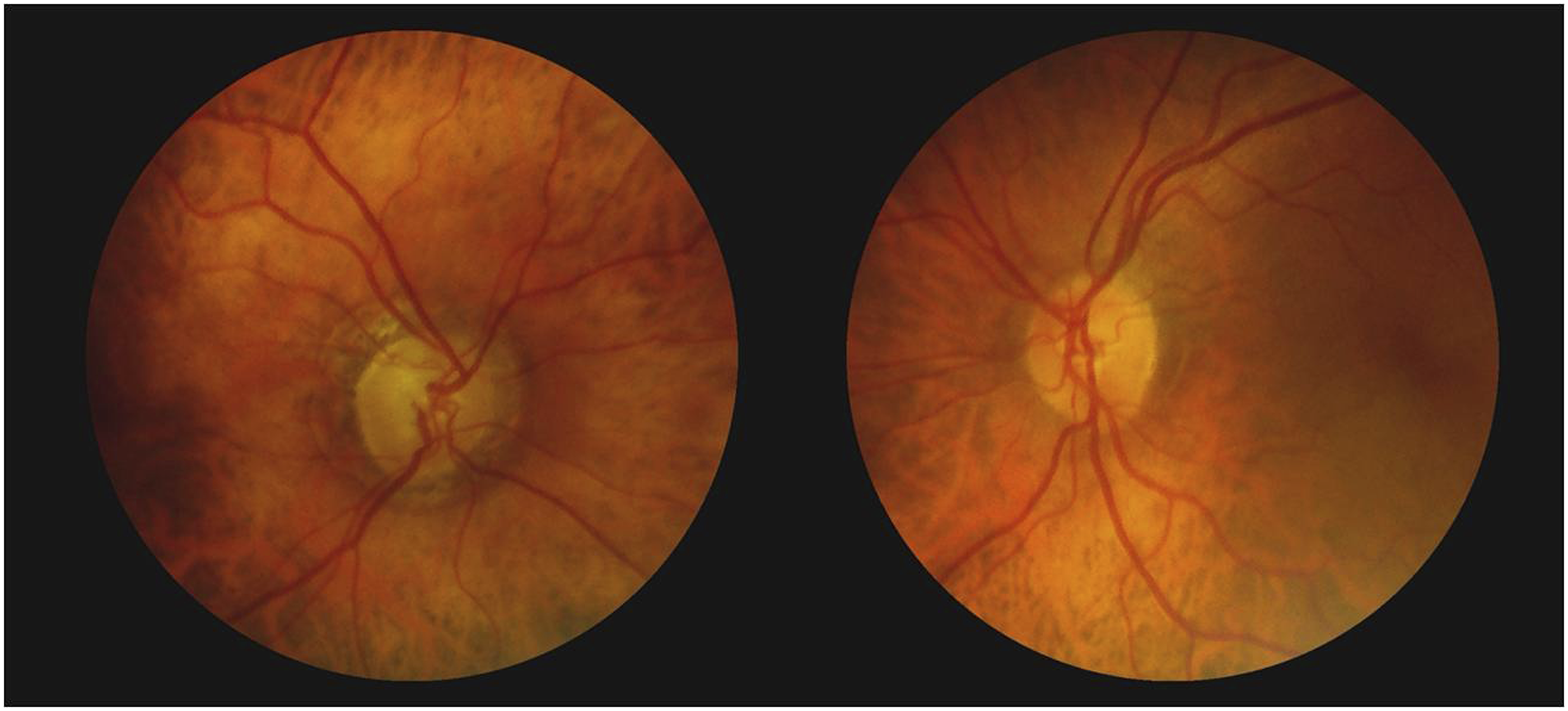
Discussion
Dolichoectatic, but even normal, intracranial blood vessels can cause dysfunction of cranial nerves (e.g., trigeminal or facial nerve) when situated in an aberrant location. Even the optic nerve(s) may be affected by vascular compression with subsequent damage and visual loss. In fact, although compressive optic neuropathy is usually caused by intracranial lesions such as brain tumors and aneurysms, 1 it can be associated with intracranial arteries. Since the first report in 1932 by Caramazza, 2 an increasing number of CONs caused by abnormal intracranial vessels have been described thanks to our ability to diagnose vascular compression through neuroimaging. Visual loss from compression of the anterior visual pathways by dolichoectatic vessels is well recognized; however, even normal-appearing ICA has the potential to compress the optic nerve(s).1,3,4 Optic nerve compression from ICAs has been suggested to be one of the possible causes of visual field defects in patients with optic neuropathy, and in patients with NTG, 4 a condition of typical glaucomatous damage of the optic nerve head and visual field loss in spite of normal IOP. 5 Nishioka and co-workers reported a case whose visual function was improved by surgical release of a compression of the optic nerve from normal ICA, thus supporting (and proving) the concept that the optic nerves can be damaged by the compression of normal appearing ICAs that do not show atherosclerotic or aneurysmal changes. 6 Several studies have tried to clarify the pathogenesis of NTG, and neuroimaging has revealed important findings that could help shed light on a disease still unclear. Gutman et al 7 performed CT of the base of the skull in 62 consecutive patients with LTG in the attempt to focus on carotid artery pathology which may be related to, or at least contribute to, the optic nerve cupping. They found pathology of the intracavernous carotid arteries adjacent to the intracranial opening of the optic canal in more than 90% of the patients. They concluded that the close proximity of the carotid artery to the optic nerve at this location may have resulted in compressive neuropathy with subsequent glaucomatous cupping of the disc. More recently, Ahmed and co-workers 8 performed brain MRI in order to determine if there was an increased prevalence of intracranial compressive lesions in patients with clinically diagnosed NTG compared with a group of patients with progressive primary open-angle glaucoma (POAG). The result was that 4 of 62 (6.5%) patients with NTG had intracranial compressive lesions involving the anterior visual pathway, compared with none of the 70 patients with POAG (P = .039). A recent study of Jain et al 9 represents the largest cohort of patients with potential vascular compression of the anterior visual pathways (AVP). It also provides detailed clinical and radiological correlations as well as longitudinal data.
In this study, 37 patients with evidence of intracranial artery AVP compression demonstrated on MRI were identified by a retrospective review of case records over a period of 7 years. Internal carotid artery-optic nerve compression was the most frequent followed by chiasmal compression by the anterior cerebral artery and a combination of optic nerve and chiasmal compression. Visual acuity and visual fields were stable on follow-up (mean, 4 years) in 93% of cases. They concluded that AVP compression by intracranial arteries may be a causative factor in unexplained visual dysfunction. The visual defects are mostly non-progressive.
The importance of such findings (compression of AVP caused by ICA or other vessels) cannot be overemphasized.
In fact, as our case shows, the phenomenon of optic nerve compression can potentially be confused with NTG, as IOP may not be raised and visual field defects can resemble glaucomatous ones.
The clinical differential diagnosis between POAG and compressive optic neuropathy is usually straightforward.
Generally, optic atrophy caused by these lesions does not resemble glaucoma as the concomitant cupping is usually lacking or minimal, and the nerve head is excessively pale. However, the optic nerve in patients with compressive optic neuropathy may lack pallor and appear to be cupped to such a degree that NTG is suspected.
Bianchi Marzoli et al 10 demonstrated an increased median cup-to-disc area in patients with compressive lesion. Trobe et al, 11 moreover, studied whether glaucomatous and non-glaucomatous optic disc cupping could be distinguished by ophthalmoscopy alone. Unfortunately, even trained experts can misdiagnose glaucomatous cupping on isolated stereo photographs.
Visual field examination is also important in the differential diagnosis of NTG.
Respect of the vertical meridian suggests an anterior junctional, chiasmal, or posterior retrochiasmal syndrome; however, the topographical anatomy and horizontal respect of the meridian of the nerve fiber bundles remains preserved all the way up to the junction of the anterior chiasm. Thus, compression of the intracranial optic nerve up to the junction of the anterior optic chiasm can produce accurate visual field defects resembling glaucoma with sparing of central vision. 12
Clinicians should be aware of these distinguishing and differentiating features of non-glaucomatous optic neuropathy and consider neuroimaging to exclude a compressive lesion mimicking glaucoma.
Prescribing neuroimaging in all cases of NTG is a discussed issue. Some authors consider it unnecessary as compression is an uncommon finding. 13 However, other studies have shown that up to 8% of patients diagnosed with NTG actually have compressive lesions of the anterior visual pathways (optic nerve or chiasm). 14 In our opinion, neuroimaging should be reserved to those cases in whom history and clinical examination suggest an alternative diagnosis. In particular, younger patients (< 50), visual field defects with respect of the vertical meridian, mismatch between cupping and visual field loss, rapid deterioration of visual acuity, and neuroretinal rim pallor more evident as compared to the degree of cupping may increase the likelihood of identifying an intracranial compressive lesion.
In conclusion, neuroimaging should be considered for patients with features atypical of NTG. This clinical case is a reminder that ICA, and not only tumors or aneurysms, may compress the optic nerve(s), causing a compressive optic neuropathy that mimics NTG.
Footnotes
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent:
the patient provided written informed consent to publish anonymous images of fundus, visual field and neuroimaging as they are reported in the paper.
