Abstract
Objective
To report three cases of immediate brainstem hemorrhage following intravenous thrombolysis with tenecteplase (TNK) in patients with minor ischemic stroke, resulting in poor outcomes and death.
Methods
We describe the clinical characteristics, imaging findings, and outcomes of three patients who developed brainstem parenchymal hemorrhage immediately after TNK thrombolysis.
Results
All three patients had minor ischemic stroke (National Institute of Health stroke scale score ≤5) and presented with elevated blood pressure on admission. Blood pressure was lowered to guideline-recommended levels (<180 mmHg) using intravenous antihypertensives before thrombolysis. Despite rapid brain protection measures, including hematoma evacuation or external ventricular drainage in two cases and conservative management in one, all patients died due to brainstem hemorrhage.
Conclusion
These cases suggest that fatal brainstem hemorrhage may occur after TNK thrombolysis in minor stroke. For patients with a history of poorly controlled hypertension, careful risk assessment and aggressive blood pressure management are critical if thrombolysis is considered.
Plain Language Summary
Intravenous thrombolysis is commonly used to treat acute ischemic stroke and is generally considered safe in patients with mild symptoms. In this report, we describe three patients with minor ischemic stroke who developed severe bleeding in the brainstem immediately after receiving tenecteplase. All three patients had a history of high blood pressure, and their blood pressure was lowered to recommended levels before treatment. Despite prompt medical and surgical interventions, the bleeding rapidly worsened and resulted in death in all cases. These cases highlight that life-threatening brainstem hemorrhage can occur even after thrombolysis for minor stroke. Our findings suggest that patients with long-standing or poorly controlled hypertension may be at particularly high risk. Careful patient selection and strict blood pressure control are essential when considering thrombolytic therapy in this population.
Introduction
Stroke is the second leading cause of disability and death worldwide, imposing a significant burden on both individuals and society. 1 Approximately 87% of strokes are ischemic in nature, with intravenous thrombolysis using agents such as alteplase and Tenecteplase (TNK) being the cornerstone of acute treatment. 2 However, thrombolytic therapy must be administered within 4.5 hours of symptom onset. 3 This narrow therapeutic window is partly attributable to the considerable risk of symptomatic intracerebral hemorrhage (sICH) associated with thrombolysis. 4 sICH was defined according to the Heidelberg classification as either an increase of ≥4 points in the NIHSS score or death attributable to the hemorrhage, which encompasses hemorrhagic transformation within the infarcted area and parenchymal hemorrhage in brain regions remote from the infarction. 5 PH is more commonly observed in the cerebral parenchyma, while immediate brainstem hemorrhage following thrombolysis is exceedingly rare. Here, we report three fatal cases of brainstem parenchymal hemorrhage after TNK thrombolysis, aiming to broaden the understanding of this serious complication and its clinical implications.
Case Presentations
Case 1
Case Information

aTOAST, Trial of Org 10172 in Acute Stroke Treatment. 8
bSAO, Small artery occlusion.
cNIHSS, National institute of health stroke scale.
dONT, Onset to needle time.
eTHT, Thrombolysis to hemorrhage time.
fGlasgow coma scale.

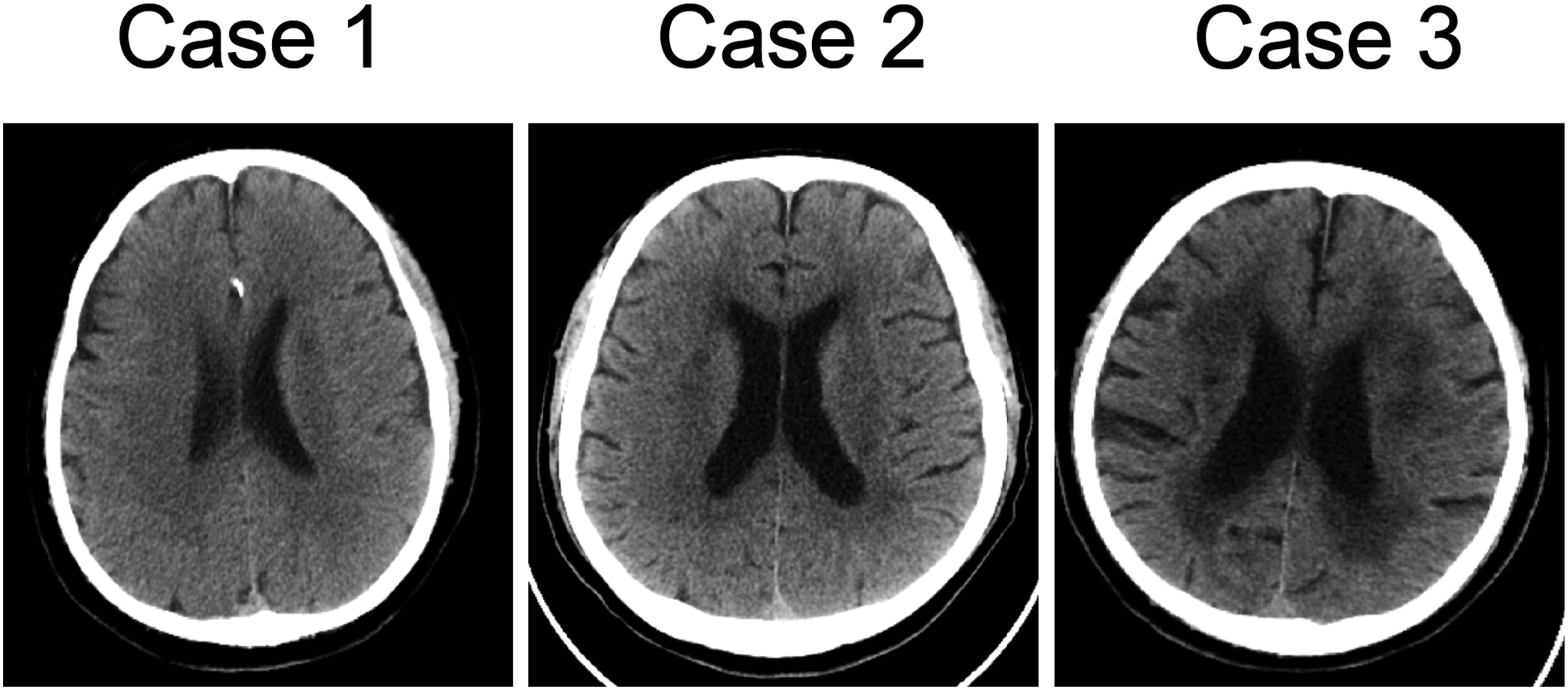
Comparison of CT before thrombolysis and after hemorrhage of three cases
Case 2
A 66-year-old male patient presented with left-sided limb weakness and speech disturbance 3 hours after symptom onset. Initial non-contrast head CT excluded intracranial hemorrhage, and ischemic stroke was diagnosed with an NIHSS score of 5. Intravenous TNK (0.25 mg/kg) was administered, with no immediate clinical improvement (post-thrombolysis NIHSS score of 5). Two hours later, the patient developed coffee-ground emesis followed by progressive stupor. Repeat CT revealed pontine hemorrhage. The family declined surgical intervention, and the patient was managed conservatively in the intensive care unit. After 4 weeks in a deep coma with no signs of recovery, the patient was discharged.
Case 3
A 59-year-old male patient presented with left-sided limb weakness and numbness 1 hour after symptom onset. Initial non-contrast head CT ruled out intracranial hemorrhage, and ischemic stroke was diagnosed with an NIHSS score of 2. Intravenous TNK (0.25 mg/kg) was administered. 2 hours post-thrombolysis, the patient developed acute left upper limb pain with paresis, progressive deterioration in consciousness, pinpoint pupils, and labored breathing. CT confirmed pontine hemorrhage. Emergency external ventricular drainage was performed. Despite surgical intervention, the patient’s condition remained critical without neurologic improvement. The family elected to withdraw medical support and life-sustaining interventions, and the patient died 1 day later.
Other possible causes of the initial neurological symptoms, including hypoglycemia, hyperglycemia, post-ictal Todd’s paralysis, and hemiplegic migraine, were carefully excluded based on clinical evaluation and laboratory findings. In all three cases, CTA was performed prior to TNK administration to exclude large-vessel stenosis, occlusion, aneurysm, and arteriovenous malformation. All patients had normal blood glucose levels at admission, no history of seizures or migraine, and their clinical features were consistent with acute ischemic stroke rather than stroke mimics, which are known to have a much lower risk of hemorrhage after thrombolysis. In addition, all patients had normal platelet counts, blood glucose, serum creatinine, fibrinogen, and INR before thrombolysis, no prior use of antiplatelet or anticoagulant agents, and no evidence of cerebral microbleeds on previous MRI examinations. None of the patients had a history of diabetes, hyperlipidemia, prior stroke, or intracerebral hemorrhage. Case 1 had a history of smoking and moderate alcohol consumption, while the other two patients denied such habits.
All patients received intravenous thrombolysis after providing written informed consent, during which the physicians fully explained the potential risks (including intracerebral hemorrhage), expected benefits, and alternative treatment options such as dual antiplatelet therapy. Given that all patients presented within the 4.5-hour therapeutic window and had no contraindications, thrombolysis was performed in accordance with local clinical practice and patient preference. These three patients shared several common characteristics. All of them had a history of chronic hypertension, and their systolic blood pressures upon admission were remarkably high, reaching 188, 251, and 174 mmHg, respectively. Intravenous nicardipine was used to rapidly lower blood pressure to the guideline-recommended target (systolic BP <180 mmHg) before thrombolysis, and continuous BP monitoring was performed during and after TNK administration. Before thrombolysis, their systolic blood pressures were managed to below 180 mmHg, while post-thrombolysis systolic blood pressure rebounded in two cases (Figure 2). The onset of hemorrhage occurred rapidly in all cases, within 3 hours after thrombolysis. Additionally, pre-thrombolysis CT scans for all three patients revealed periventricular low-density areas, indicating the presence of white matter hyperintensity (Figure 3). Changes in patients’ systolic blood pressure over the course of the disease. Transfer: transferring patients from the emergency ward to the neurology ward Patient’s CT on admission, suggesting white matter hyperintensity
Discussion
Brainstem hemorrhage, a rare and fatal complication of thrombolysis, is considered a form of remote intracerebral hemorrhage. Previous studies have reported a case of pontine hemorrhage associated with low-molecular-weight heparin treatment for basilar artery occlusion 9 and a case of cerebral parenchymal and brainstem hemorrhage occurring during t-PA treatment for acute myocardial infarction. 10 To the best of our knowledge, this is the first report of brainstem hemorrhage occurring after TNK intravenous thrombolysis in three patients with ischemic stroke. All three patients had an NIHSS score of ≤5 prior to thrombolysis, indicating mild strokes (two patients had an NIHSS score of ≤3), but they developed fatal brainstem hemorrhage, a serious complication, after thrombolysis. It should be noted that this report does not imply a direct causal relationship between TNK administration and brainstem hemorrhage. Rather, our intention is to highlight the need for careful decision-making regarding intravenous thrombolysis in patients with poorly controlled hypertension and mild ischemic stroke, and to emphasize the importance of more refined blood pressure management after thrombolysis when large vessels are patent. A previous registry-based study has identified several independent risk factors for remote or symptomatic intracerebral hemorrhage after intravenous thrombolysis, including baseline NIHSS score, serum glucose, systolic blood pressure, age, treatment delay, antiplatelet use, and a history of hypertension. 4 These established factors provide context for our observations, as all three patients in our series had chronic hypertension and poor blood pressure control prior to TNK administration.
Common causes of brainstem hemorrhage include chronic hypertension, cavernous malformations, and arteriovenous malformations. 11 In our reported cases, brain vascular CT angiography did not identify cavernous malformations or arteriovenous malformations as potential causes of secondary brainstem hemorrhage. However, all 3 patients had a history of chronic hypertension and poor blood pressure control. Notably, the hemorrhages occurred in the pons, a common site for hypertensive brainstem hemorrhage, as the pontine perforating vessels are more vulnerable to the effects of hypertension. 12 Research has shown that high systolic blood pressure is an independent risk factor for sICH after thrombolysis. 4 Although hemorrhagic transformation of an evolving infarct could not be completely excluded, the hemorrhage locations in our cases were remote from the initial ischemic lesions on imaging, suggesting primary parenchymal brainstem hemorrhage rather than secondary transformation within infarcted tissue. These findings suggest that for patients with chronic hypertension and poor blood pressure control, especially those with mild strokes, if intravenous thrombolysis is considered and no large vessel occlusion is present, it is crucial to actively control blood pressure. Systolic blood pressure should be maintained below 140 mmHg, when possible, to reduce the risk of post-thrombolysis hemorrhage and improve patient outcomes. 13 Additionally, for patients with an NIHSS score of ≤3, thrombolysis should be carefully considered in the absence of large vessel occlusion, as the benefit of thrombolysis in such cases remains uncertain. 14
Notably, large randomized trials comparing TNK with alteplase have shown comparable safety profiles with no significant difference in symptomatic intracerebral hemorrhage, indicating that the bleeding risk is not uniquely drug-driven.15,16 Chronic hypertension induces profound structural and mechanical remodeling of cerebral microvessels, leading to persistent vascular fragility even after blood pressure normalization. Experimental studies have demonstrated that long-term hypertension causes rupture of the internal elastic lamina and loss of mural integrity, resulting in irreversible weakening of the vessel wall. 17 Similarly, sustained hemodynamic stress produces bioengineering fatigue and destruction of mural components such as elastin and collagen, reducing tensile strength and predisposing arteries to rupture during abrupt pressure fluctuations. 18 Recent evidence indicates that cerebral arterioles undergo rapid thickening, increased wall-to-lumen ratio, and reduced elasticity during hypertension, and that while some parameters recover when arterial pressure normalizes, inward remodeling often persists, suggesting incomplete vascular recovery. 19 At the molecular level, disruption or downregulation of vascular endothelial-cadherin weakens endothelial junctions, impairing vascular stability and increasing permeability. 20 Moreover, arterial stiffness, a hallmark of vascular aging, creates an adverse feedback loop in which elevated pulsatile load and small-artery remodeling exacerbate microvascular fragility and increase susceptibility to hemorrhage, particularly within deep brain regions such as the pons. 21 Evidence from germinal-matrix hemorrhage models also underscores that immature or fragile vasculature is highly vulnerable to fluctuations in cerebral blood flow. 22 Collectively, these findings support a pathophysiologic model in which rapid blood-pressure reduction combined with TNK-induced fibrinolysis in patients with chronically fragile cerebral vessels may precipitate rupture of pontine perforating arteries, resulting in brainstem hemorrhage. WMH is considered a surrogate marker of small vessel disease and is commonly observed in ischemic stroke. 23 Hypertension is one of the key factors contributing to WMH. It is also a major cause of spontaneous intracerebral hemorrhage, particularly affecting the deep small penetrating arteries of the brain, including those in the basal ganglia/thalamus, brainstem, and cerebellum. Previous studies have reported that WMH is an independent risk factor for remote intracerebral hemorrhage following intravenous thrombolysis in acute ischemic stroke. 24 In our study, all three patients had evidence of white matter lesions on CT prior to thrombolysis, and all developed remote brain hemorrhage post-thrombolysis, suggesting the presence of small vessel disease as a contributing factor to post-thrombolysis hemorrhage.25,26 Therefore, when CT scans reveal WMH, we should consider the possibility of underlying small vessel disease and carefully weigh the benefits and risks of thrombolysis treatment in such patients.
This study has several limitations. First, it is a case report series focusing on immediate brainstem hemorrhage after thrombolysis, and the number of such cases in our hospital is extremely limited. Therefore, we were unable to perform comparative analysis between brainstem and non-brainstem hemorrhage cases to identify differential risk factors. Second, as a retrospective descriptive report, potential selection bias cannot be fully excluded. Third, MRI was not performed due to the patients’ sudden clinical deterioration after thrombolysis, which limited our ability to exclude underlying cerebral amyloid angiopathy or other microvascular pathologies. Future large-scale or multicenter studies are warranted to further investigate the underlying mechanisms and risk profiles associated with this rare but fatal complication.
Conclusion
In conclusion, our study underscores the occurrence of fatal brainstem hemorrhage immediately following intravenous thrombolysis with TNK in three patients with minor ischemic stroke and poorly controlled hypertension. These cases highlight the importance of heightened caution when considering thrombolysis in patients with a history of chronic hypertension and WMH. Careful risk assessment and strict blood pressure management may be critical in reducing the risk of post-thrombolysis hemorrhage. Given the uncertain benefits of thrombolysis in patients with minor strokes and no large vessel occlusion, a personalized approach to treatment is essential. Further research is needed to elucidate the mechanisms and predictors of brainstem hemorrhage associated with TNK and to optimize clinical guidelines for managing high-risk patients.
Footnotes
Acknowledgment
The authors thank the patients’ relatives for their consent to publish these case reports.
Ethical Considerations
Our study was conducted in accordance with the Declaration of Helsinki. The study protocol was approved by the Ethics Committee of Zhongnan Hospital of Wuhan University (Approval No. 2025005K).
Consent to Participate
Informed consent was obtained from patients’ family members.
Consent for Publication
All authors have read and approved the final version of the manuscript and consent to its submission for publication. Written informed consent for publication of the clinical details was obtained from the patients’ family members.
Author Contributions
Bin Mei and Nao Yan contributed to conceptualization and supervision. Lei Shen contributed to manuscript writing and data analysis. Niao Yang contributed to data analysis. Dong Sun contributed to data collection. All authors read and approved the final version of the manuscript and agreed to its submission for publication.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (Grant No.82301376).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Data Availability Statement
All de-identified data in this study are available from the corresponding author upon reasonable request.
