Abstract
Objectives:
We provide a systematic review of the literature to identify clinical studies assessing the effects of extracorporeal shock wave therapy (ESWT) on acutely injured tissues of human subjects, also highlighting the biological mechanisms by which the technique is proposed to promote the processes of early tissue repair. Special attention is also paid to the progress of research in animal models.
Method:
A systematic review of the literature on ESWT of acute injuries of bone and soft tissue as available in the PubMed/MEDLINE, Cochrane CENTRAL, SPORTDiscus, and CINAHL databases up to December 2017 was conducted.
Results:
A total of 10 studies were included. There is some evidence for the application of ESWT in an early postacute injury phase. Most studies report benefits with no or minimal side effects. However, different types of treated tissues and wounds (varying cause and severity) and resulting heterogeneity in study design and outcome measurement make it difficult to compare studies. The picture of knowledge remains limited by an apparent lack of data on optimal treatment timing and on tissue- and injury-specific parameters.
Conclusions:
Although the amount of studies to date is still limited, recent clinical research has presented first successful steps to introduce ESWT as a means of treatment in acute injury care. Therefore, on the basis of the analyzed data, further testing is encouraged to validate optimal timing, physical settings, and possible long-term effects to exclude potential risks.
Introduction
Focused high-energy shock waves applied from outside the body have been used as a means of medical treatment since 1980 when extracorporeal shock wave lithotripsy was introduced to enable the noninvasive disintegration of renal and gall stones. 1 Since then, the scope of disorders to which the technique is applied has been extended to various medical fields.
When directed to biological tissues, shock waves generate physical interactions and effects by formation of cavitation bubbles and shear stress, which are considered to evoke distinct biological responses in tissues, cells, and at subcellular level. 2 Energy flux density (EFD) and the number of applied impulses are thus key determinants of shock wave effects in medical use. Despite the considerable time span over which extracorporeal shock wave therapy (ESWT) has been used for medical purposes, the technique is not yet optimized for clinical practice. It has been shown that responses to shock waves are dose dependent and tissue specific.3–6 This means that clinicians and physical therapists have to handle optimal dosage while deployment of appropriate tissue-specific energy levels is required to induce a positive outcome. In addition to the optimization of energy modalities, it is particularly the timing of the therapy onset within the postinjury period which may be a further major factor of influence on the success of ESWT that has been given less attention so far.
The importance of optimal treatment timing in the postinjury phase is highlighted by considering basic biological principles of tissue repair processes. It is well established that tissue regeneration occurs in 3 distinct phases, which partially overlap in time: inflammation (in case of bleeding proceeding interdependently with haemostasis), proliferation, and remodeling. 7 Perturbations in any of these repair phases can develop into a chronic condition and failed repair, representing a prolonged, dysregulated, and maladaptive process that eventually leads to tissue destruction. 8 In orthopedics, for a large part, it is precisely those chronic conditions that are currently treated with ESWT, typically applied at least 3 months after injury, and often only after years of unsuccessful conservative treatment 9 and/or to avoid invasive, and in some cases repeated surgical intervention. 10 Under such circumstances, the main objective of ESWT is to bring the chronic condition back into a more responsive acute state, thus escaping the vicious inflammatory cycle 11 and triggering the self-repair mechanisms of the body. This inevitably raises the question of whether ESWT could also bring a benefit in acute trauma healing by supporting the regular process of regeneration. If so, ESWT could provide a beneficial noninvasive means of therapy for treatment of severe soft tissue and bone injuries, as it is often exactly those complex conditions that are at high risk to become chronic due to persistent inflammation and poor tissue perfusion,12,13 what makes their management challenging to practitioners. If ESWT, applied in the acute postinjury phase, would be able to accelerate tissue repair while accounting for nonhazardous inflammatory control, as already shown in various animal models,10,14 it would help reduce the risk of complications and may even prevent development of chronicity.
Biological effects of ESWT in early tissue regeneration
Although the sequence of events accounting for improved healing in ESWT-treated tissues is still incompletely understood, it is now widely accepted that shock waves act through mechanotransduction, 15 which is defined as the entirety of processes by which mechanical loading triggers cellular responses. 16 Most of our present understanding of how ESWT interferes with and promotes processes of tissue repair of acute injuries was obtained from research in animal models. It has been found that the regenerative improvement involves mechanosensitive feedback responses leading to vasodilatation and increased microcirculation, induction of neo-angiogenesis, immunomodulatory action to reduce inflammation, and acceleration of stem and precursor cell recruitment for tissue replacement. An important pathway by which shock waves act on angiogenesis and inflammation is likely to be modulation of nitric oxide (NO) release.17,18 A single ESWT session applied to full-thickness soft tissue wounds in mouse skin has been shown to increase the levels of NO and endothelial nitric oxide synthase, as well as those of proangiogenic chemokines and cytokines. Similarly, in ischemic muscle of mice and rats, ESWT leads to a persistent improvement of blood perfusion, accompanied by the formation of a higher number of capillaries and arterioles.18,19 In addition, effects on the monocyte-macrophage system and phagocyte behavior were shown in ESWT-treated skin and tendon of mice and rats. Studies demonstrate a significant reduction in the levels of infiltrating inflammatory cells including neutrophils and macrophages.8,20 This has been shown to involve decreased transcript levels of pro-inflammatory cytokines and chemokines such as interleukin (IL)-1b, IL-6, IL-10, tumor necrosis factor α, and tumor necrosis factor receptor.8,21–23 Accompanying effects in injured rat tendons included reduced swelling and enhanced resolution of edema. 20 In addition, experiments in rats have shown that ESWT initially enhances the release of substance P from afferent fibers of nociceptive primary neurons. This has been thought to explain increased pain sensation during and immediately after clinical ESWT application, whereas the decreased substance P levels measured 6 weeks after treatment have been seen to correlate with the subsequent relief of pain on the long range. 24
Short-term postinjury ESWT has also been shown to induce elevated expression of tissue-specific growth factors. In animal models, this type of response was found in experimentally injured bones,25,26 tendons, 20 and skin,23,27,28 in skin affected by lymphoedema, 29 as well as in ischemic and toxin-lesioned limb muscle.30,31 Detected effects include the upregulation of vascular endothelial growth factor (VEGF), VEGF receptor protein, placental growth factor (PGF), PGF receptor protein, transforming growth factor β1, basic fibroblast growth factor, insulinlike growth factor, and bone morphogenetic protein, accompanied by an increased presence of mitotic precursor cells, the formation of new blood vessels, and accelerated healing. Observed increased union rates and enhanced recovery of mechanical properties in surgically and accidentally induced bone fractures in rabbits and dogs32–34 and higher tensile load of injured patellar tendons in rabbits 35 are in close relation to described biological ESWT effects.
To examine whether there is evidence for the benefit of ESWT applied in an acute or postacute injury phase, we systematically reviewed clinical studies of the past 10 years to search for ESWT effects on traumatic injuries of human soft tissues and bones. The key intention was to evaluate in a PICO (Patient/Population/Problem, Intervention, Comparison, Outcome) framework whether ESWT accelerates healing compared with untreated or alternatively treated controls.
Methods
An evidence-based systematic review of the literature was conducted according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) guidelines 36 to evaluate the current evidence in studies addressing clinical application of ESWT to acute bone and soft tissue injuries.
Eligibility criteria
Accelerated regeneration after ESWT application was the primary measure of treatment effect. The search criteria were formulated using a PICO framework, serving to establish whether patients with acute/traumatic injuries of bone, tendon, ligament, muscle, or skin (P) receiving ESWT in the acute postinjury phase (I), compared with subjects receiving no, or other treatment (C), exhibit enhanced recovery compared with non–ESWT-treated subjects (O). The studies were preselected electronically based on the following exclusion and inclusion criteria: peer-reviewed, published in academic journals, publishing date between January 2007 and December 2017, treatment applied to humans, English language, and full text availability.
Data source and search strategy
We electronically searched the databases PubMed/MEDLINE, CINAHL Complete, Cochrane Central Register of Controlled Trials (CENTRAL), and SPORTDiscus using a Boolean operator for “acute injury AND shockwave OR shock wave therapy OR extracorporeal shock waves OR eswt.” On completion of this evaluation, a hand search for references cited in the identified studies was performed, and an extended PubMed search was undertaken to identify further related articles using the search terms “muscle,” “ligament,” “tendon,” “bone,” “skin,” “wound,” “fracture,” “tear,” and “rupture,” variously combined with the terms “acute” and “shockwave,” “shock wave therapy,” “extracorporeal shock waves,” and “eswt.” Last search date was the December 29, 2017.
Study selection
Two reviewers screened the identified articles based on the evaluation of titles and abstracts. If it was absolutely clear from the title or the abstract that a study is not relevant, it was excluded from further evaluation. Such articles included the following: (1) studies with irrelevant content (eg, such concerning ESWT in urological or cardiovascular contexts and ESWT of chronic disorders/conditions) and studies that erroneously passed the electronically set limitation to human subjects (ie, animal and cell culture studies) and (2) articles of irrelevant form (review articles, meta-analyses, case studies, conference publications, letters to editors, journal errata and corrections, abstract collections). All studies suggesting relation to therapeutic shock wave application to acute injuries of musculoskeletal tissues and skin wounds were included. Discrepancies between the 2 reviewers regarding eligibility were discussed until a consensus was reached.
After removing duplicates, the full-text versions of the remaining studies were assessed for final eligibility. Only studies evaluating ESWT of acute or postacute injury conditions (≤6 weeks) were eventually included. Acute injury was defined as an injury directly associated with a traumatic event, causing, for example, a (skin) wound, laceration, tear, rupture, or fracture or generated by surgical intervention (cuts, reconstructions of, eg, tendons). Extracorporeal shock wave therapy could have either been used as an exclusive therapy or in combination with other standard interventions including, for example, bone fixation or wound debridement and medication, provided that the control groups received the same interventions (in the case of randomized controlled trials [RCTs]). Included studies report on bone, tendon, ligament, muscle, and skin and were chosen irrespective of whether radial or focused ESWT was applied and regardless of the number of treatment sessions and variation in other parameters (eg, EFD and number of impulses).
Risk of bias assessment
Due to the small study output, we agreed to also include other study types than RCTs. For evaluation of methodological quality, included studies were divided into 2 groups: the first of which representing RCTs and the second various other types of clinical studies. All studies were collaboratively assigned to their appropriate level of hierarchy according to the National Health and Medical Research Council (NHMRC) designation of level of evidence categories. 37 For the RCTs in the first group, 2 review authors performed risk of bias assessment using the Cochrane collaboration Risk of Bias tool (RoB 2.0), 38 to assess the “internal validity.” Disagreements were resolved by discussion between these authors. Items were scored as follows: low risk of bias, high risk of bias, or moderate risk/some concern, according to the Cochrane handbook descriptions. 38
Data extraction and synthesis
Due to the heterogeneity of injury types and treated tissues, and the concomitant fundamental differences in experimental design and outcome measurement, data were not collapsed for meta-analysis but instead synthesized in a descriptive approach. Standardized data extraction forms were created in consultation with all authors. Data were extracted by 2 independent authors and cross checked for accuracy. The following data were extracted: (1) general characteristics of study design and hierarchy of evidence (according to NHMRC 37 ), aim of the study, study groups, type of treated tissue, cause of injury, number, and characteristics of study participants; (2) treatment details including time of treatment onset, type of shock waves, ESWT parameters (EFD, frequency, impulses), number of treatment sessions; and (3) main study outcomes including type of outcome and potential effects of treatment.
Studies were compared in terms of study design and outcome, also incorporating the quality of the individual studies. Appraisal of evidence across the studies was discussed taking into account strength of evidence, consistency of findings, clinical impact, and generalizability of the results based on the recommendations of the NHMRC strength of the body of evidence matrix. 37 This review does not intend to restrict the application of recommendations to a specific geographic region; therefore, the last item addressing the applicability of evidence to the Australian population was omitted.
Results
Study selection
The systematic search in the 4 examined databases yielded a total of 647 hits (for details, see Figure 1). After revision of study titles and abstracts, 618 articles were excluded leaving 28 studies for full-text evaluation. Most of the omitted articles were excluded because the titles and abstracts clearly indicated irrelevant content (eg, chronic diseases of musculoskeletal tissues, urological studies) or study design (animal or cell culture study) or format (eg, review articles). The expanded searches resulted in different numbers of hits, depending on the combination of search terms used. These hits are not included in the aforementioned total of 647, and after revision of titles and abstracts, only 1 of these remained valid for full-text revision. This study was identified by a PubMed/MEDLINE search using the search term combination “acute AND fracture AND ((((shockwaves) OR shock wave therapy) OR extracorporeal shock waves) OR eswt),” and although being indexed as a mini review, it included a trial investigating ESWT of acute fractures.

Flowchart of the literature search process and study selection according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) guidelines. 36
After exclusion of duplicates (n = 9), 20 studies were full text-assessed for final eligibility. From these, 10 studies were excluded because ESWT was not applied in an acute or postacute injury phase, or because the presented injury was not traumatic or surgical, so that eventually 10 studies were found appropriate for inclusion in this review.
Study characteristics
Included studies evaluated the effects of ESWT received by patients with accident- or surgically induced traumatic injuries in the acute or postacute phase. Types of injury included acute bone fractures, delayed-onset muscle soreness (DOMS), surgically reconstructed tendons and ligaments, and various types of skin wounds. Studies described no specific populations (eg, athletic and military), and numbers of participants varied from 15 to 100. Of the 10 studies, 7 were RCTs representing level II evidence. All of these studies were designed to compare a verum group with an untreated control group receiving standard injury care without additional ESWT. In one study evaluating muscle soreness, the verum group was compared with 2 control groups, one of which received sham treatment and the other remaining completely untreated. Two articles were level IV single-armed feasibility studies, and one article reports on a small controlled trial investigating the treatment of acute bone fractures within a larger, single-armed study investigating chronic nonunions. Because this study made no reference to a randomization, it was judged to be of level III-2 evidence. All relevant extracted data are summarized in Table 1.
Characteristics of study design, injuries, and patient population of included studies.

Abbreviations: ESWT, extracorporeal shock wave therapy; NI, no information; RCT, randomized controlled trial.
Hierarchy of evidence according to the National Health and Medical Research Council (NHMRC). 37
Percentage of men and women across whole study referring to patients with acute and chronic wounds. Mean age of patients with acute injuries is not specified.
Risk of bias assessment
The included single-armed studies42,43 were all assigned to be of high risk of bias because of the impossibility to distinguish between effects of treatment, effects of natural history (spontaneous improvement over time), and placebo effects. The small nonrandomized trial of Moretti et al 48 was also assigned with high risk of bias because of a lack of essential information in both methods and results. Details of the risk of bias of all included RCTs, assessed using the Cochrane Collaboration’s tool (RoB 2.0), 38 are presented in Figure 2 and in Supplementary Material 1.

Risk of bias assessment according to the Cochrane Collaboration’s tool (RoB 2.0) 38 for randomized controlled trials. Risk of bias to nonrandomized and/or controlled trials was judged to be of high risk of bias.
The overall quality of the RCTs was moderate to high, and none was associated with high risk of bias. The studies of Fleckenstein et al, 44 Ottomann et al,39,40 and Wang et al 45 give detailed description of how the patients were randomly assigned into intervention and control groups. The other studies report randomization without describing the process in detail. Deficits include that it is not defined whether patients treated with shock waves immediately after surgery while still under anesthesia were blinded to study groups. Also, most studies do not describe whether and how the physicians and/or carers were blinded to the different study groups. It may, however, be difficult to keep the physician blinded to the change when ESWT EFD has to be reduced to (almost) 0 in cases of sham treatment. Only the study of Fleckenstein et al 44 describes details to ensure the double-blinding procedure. Outcome assessors were all blinded to the intervention received by study participants. All studies employed between-group statistical analysis. All studies except for that of Moretti et al 48 report about obtainment of ethics approvals.
Effectiveness of ESWT in acute skin wounds
Half of the reviewed articles (5 of 10) investigated the effect of ESWT on acute surface injuries, thus illustrating the current new development of ESWT in acute care (Table 2). The spectrum of these ESWT-treated wounds spans from such resulting from direct penetration or blunt trauma 42 to burn injuries39,42,43 and surgical wounds resulting from vein harvesting and skin grafting. Evidence of accelerated wound healing associated with ESWT as a primary therapy comes from 2 high-quality RCTs and from 1 RCT with moderate risk of bias. Beneficial effects of ESWT were also reported in 2 single-armed feasibility studies that were associated with a high risk of bias because of the lack of appropriate control groups.
Summary of injury characteristics, ESWT settings, and outcome data of included studies.

The RCT of Ottomann et al 39 included a total of 50 patients with second-degree burn wounds. After wound debridement, the patients of the intervention group (n = 22) received a single session of unfocused shock waves within 24 hours after burn, whereas the control group (n = 22) was left without shock wave treatment. Both groups received the same daily antiseptic therapy. The ESWT parameters were 100 impulses per square centimeter wound, using a dose of 0.1 mJ/mm2 applied with a frequency of 5 Hz. The mean time to complete wound epithelialization was found to be significantly faster in the ESWT group (9.6 ± 1.7 days vs 12.5 ± 2.2 days) when data of 44 patients are included to final calculations (P = .0005). By assuming the worst-case scenario (longest time to healing) in patients who were lost to follow-up in the intervention group and the shortest time for wound epithelialization for patients who were lost to follow-up in the control group, the significant difference (P = .003) was retained (10 ± 1.9 days vs 12 ± 2.5 days), resulting in strong evidence that a single ESWT session accelerates wound healing in acute burn wounds.
In a small RCT enrolling 28 participants, Ottomann et al 40 investigated ESWT effects on skin graft donor site wounds resulting from grafts being harvested to cover tissue excisions of burn wounds or other traumatic skin injuries. All patients received wound care according to the standards of practice. Blinded patients were randomly assigned to the intervention group (n = 13) and to the control group (n = 15). Intervention group donor sites received a single session of unfocused ESWT immediately after skin graft harvest (intraoperatively) comprising 100 impulses per square centimeter of the wound area administered with a dose of 0.1 mJ/mm2. The authors found strong evidence for accelerated graft site epithelialization after ESWT, with time to heal being on average 2.8 days shorter in treated wounds compared with untreated graft sites (13.9 ± 2.0 vs 16.7 ± 2.0; P = .0001).
The RCT of Dumfarth et al 41 investigated surgical wounds after vein harvesting for coronary artery bypass graft surgery. The study enrolled a total of 100 patients, 50 of which received a single session of unfocused ESWT immediately after vein harvesting under the same anesthesia. A total of 25 impulses per centimeter wound length were applied administering a dose of 0.1 mJ/mm2 at a frequency of 5 Hz. The remaining control group (n = 50) was left without postsurgery ESWT. All wounds were similarly closed with surgical dressings at the end of the operation. Wound healing was analyzed using the ASEPSIS score (a scoring method for postoperative wound infection allocating points for additional treatment, the presence of serous discharge, erythema, purulent exudate, separation of the deep tissues, isolation of bacteria, and duration of inpatient stay). Improved healing in the treated group was reflected by a reduced requirement for antibiotic treatment (4% vs 22% in controls; P = .015), shorter duration, and a more satisfactory progress with less disturbances (P < .05).
Although associated with high risk of bias, the 2 analyzed single-armed feasibility studies are also supporting beneficial effects of defocused ESWT to acute wounds, mainly by reporting a high rate of wound closure.
The study of Schaden et al 42 investigated shock wave effects on chronic and acute wounds of diverse causes (n = 208). Acute wounds (33.2%) caused by burning (second- or third-degree burns) or direct penetration and blunt trauma were treated with a dose of 0.1 mJ/mm2 at a frequency of 5 Hz. About 100 impulses per square centimeter wound area were applied in several sessions, initially at weekly and later at biweekly intervals. The authors reported that 86.6% of posttraumatic wounds showed complete epithelialization within a follow-up period of 6.3 weeks (mean duration). Burns showed the most rapid healing within 19. ± 18.5 days and a 100% success quotient.
The second uncontrolled pilot trial (the study by Arnó et al 43 ) applied unfocused shock waves to second- and third-degree burns after wound treatment with semiocclusive dressings. Patients received 2 sessions of ESWT (0.15 mJ/mm2, 100 impulses per square centimeter wound area) on the third and fifth day after injury to nondebrided wounds. About 80% of wounds were found to reepithelialize spontaneously in a mean period of 15.6 ± 4.9 days. Despite their potential risk of bias, these studies provide important additional information about the safety of ESWT application to skin injuries. Both studies clearly state that the therapy was well tolerated by the patients and no adverse effects were experienced. Cardiac, neurologic, thermal, and allergic reactions were also not detected. Of 15 persons, 3 reported some pain during treatment, but visual analog scale (VAS) score remained below 3.
Altogether, these studies allow the conclusion that ESWT exerts positive effects on the healing of acute and postacute wounds. However, further support by high-quality large-scale studies is required to corroborate this conclusion and to disclose possible benefits for a broader variety of wounds.
Effectiveness of ESWT in acute muscle injuries
The systematic search revealed 1 RCT that evaluated the effectiveness of ESWT on an acute muscle condition (Table 2). Analyzing the effects of a single ESWT session on experimentally induced DOMS, 44 this study is of continuous high quality, however, with a rather small sample size (15 treated subjects, 15 controls). Focused ESWT was applied immediately after DOMS induction to 7 predefined individual points along the biceps brachii muscle. In total, 1400 impulses were applied (200 per spot) with an EFD between 0.06 and 0.09 mJ/mm2. Although they found no significant acceleration of recovery, the authors concluded that the relief from pain intensity, pressure pain, and daily impairments felt by the patients were of clinical relevance and might serve to enhance return to play in injured athletes. In surprising contrast to the high incidence of sports-related muscle injuries, we found no current studies that had investigated ESWT effects on acute muscle strain or contusion injury. This certainly indicates a considerable need for further research to determine the scope of ESWT in treatment of acute muscle damage.
Effectiveness of ESWT in acute tendon and ligament injuries
Sports-related activities have a high potential to generate injuries of tendons and ligaments due to repetitive motion, excessive short-time force and strain, static overload, and unnatural postures of body and limbs, with factors often acting in a cumulative manner (Table 2). 49 Present information on the influence of ESWT on acute ligament and tendon conditions in humans is limited. The systemic search identified 2 RCTs investigating ESWT in this field, one analyzing effects after anterior cruciate ligament reconstruction and the second after arthroscopic rotator cuff repair. Both studies were associated with moderate risk of bias and methodological quality, mainly because a lack of details of the randomization process.
The study of Wang et al 45 investigated the effects of ESWT to tendon bone junctions in patients receiving hamstring autograft anterior cruciate ligament reconstruction. A total of 52 patients were enrolled and randomly divided into an intervention group (n = 26) and a control group (n = 27). The patients of the intervention group received a single session of ESWT applied to the bone tunnel immediately after surgery under the same anesthesia. All other treatment was similar for all participants. ESWT parameters were set as follows: 1500 impulses applied to the middle third of the tibia tunnel, with an EFD of 0.298 mJ/mm2. The treatment was judged to have an improving influence in tendon to bone healing. Compared with the control group, ESWT group patients showed improved Lysholm functional scores in 1- and 2-year follow-up examinations (P < .001), decreased anterior-posterior laxity of the knee (assessed with KT-1000 arthrometer; P = .027 in 2 years’ follow-up), and decreased tibia tunnel sizes (P = .018 at the middle 1/3) and tibia tunnel enlargement ratios (P < .05). No difference between groups was found in bone mineral density and in IKDC (International Knee Documentation Committee) subjective score (assessing function in daily living).
The study of Kim et al 46 investigated the effects of ESWT after arthroscopic rotator cuff repair. 6 weeks after operation, patients, randomly assigned to an intervention group (n = 35 vs n = 36 in the control group), received a single session of ESWT to the area of the greater tuberosity of the proximal humerus. The dose was set to 0.12 mJ/mm2, and 1500 impulses were applied with a frequency of 10 Hz. The authors report a significant decrease in the VAS scores although with no difference between intervention group and control group, and a significant increase in constant score and UCLA (University of California Los Angeles) shoulder rating scale, but again with no difference between control and intervention groups at 1-year follow-up. They also found no difference in the range of motion and cuff integrity between the 2 groups at this time.
As with muscle injuries above, these differences in outcome again indicate that further research effort is required to determine the full scope of ESWT in this therapeutic area.
Effectiveness of ESWT in acute bone fractures
Similar to skin wounds, broad experience from nonunion treatment in the chronic range,50,51 just as from acute fracture treatment in animal studies (eg, the works by Wang et al 25 and Oktaş et al 52 ) and clinical veterinary medicine,33,34 suggests that ESWT should have a positive perspective if applied to fresh fractures of human bone (Table 2). However, regardless of these indications, evidence supporting that the animal-based assessment might be transferable to fresh fracture healing in humans is still limited. One study on this topic is the RCT of Wang et al. It involved a total of 56 patients with open or closed fractures of the femur and tibia, 28 of which received a single session of high-energy (0.62 mJ/mm2, 6000 impulses) ESWT immediately after open or closed surgical fracture reduction and internal fixation under the same anesthesia. The control group (28 patients) received similar treatment except for ESWT. Evaluation parameters included clinical assessment of pain (VAS score), weight-bearing status of the affected leg, and serial radiographs to assess fracture alignment, callus formation, and bony union across the fracture site. Although this study had to be associated with moderate risk of bias because of improper description of the randomization process, it provides concrete hints on ESWT’s influence on fracture healing in various human bones. The authors report significantly shortened healing times (89% of fractures vs 80% at 12-month follow-up; P < .001), a reduced nonunion rate (11% vs 20%; P < .001), lower pain scores, and a better weight-bearing status in the intervention group as compared with the control group at 3, 6, and 12 months postinjury. Receiving ESWT reduced the risk of nonunion by 46%.
Although assessed as being of poor methodological quality, a second study treating chronic and acute bone fractures with focused ESWT (the study by Moretti et al 48 ) is at least in general support of these observations. As part of a larger investigation also including treatment of nonunions and pseudoarthroses, 16 cases of postacute fractures of the tibia received a single ESWT session (0.07-0.17 mJ/mm2) applied within 1 month from external fixation. The authors compared the outcome data with a control group and measured enhanced healing of the cortical domains in about 80% of the ESWT-treated bones. The study does not provide group details such as number of patients and randomization procedures.
Overall, the evidence analyzed above, while certainly indicating positive effects, does still not suffice to enable definitive judgment on the reliability of ESWT to promote acute human bone fracture healing. Further high-quality research is required to determine the scope of ESWT in this field.
Discussion
The main goal of the present review was to inquire to which extent the use of ESWT in human medicine is developing beyond the field of application to chronic disorder after failure of conservative treatment. The present review of the literature to date confirms that the advances made in the understanding of mechano-inductive effects of shock waves, together with the availability of cheaper and more flexible treatment devices, 9 has, over the past 10 years, enabled and facilitated attempts to test ESWT as a primary therapy in acute clinical care. However, the present systematic search shows also clearly that the amount of valid literature on the application of ESWT to accident- or surgery-induced acute or postacute injury is still rather limited and heterogeneous in topic and scientific quality. Thus, the 10 studies that passed the filtering criteria of this review focused on a highly diverse spectrum of tissue damage, including severe burn wounds, surgical wounds after grafting, DOMS, surgically reconstructed ligaments and tendons, and acute bone fractures. Only 7 of the included studies comprise randomized controlled clinical trials. Commonly addressed outcome parameters included pain, joint function, subjective satisfaction with treatment, and radiologic status. Specific aspects of the studies are discussed in more detail below.
Overall body of evidence
There is good to satisfactory evidence base for the effective treatment of skin wounds, muscle and tendon injuries, and bone fractures treated with ESWT. The consistency in outcome reporting is excellent for skin, but poor for the other tissues, mainly because of the lack of a sufficient amount of studies or heterogeneous outcome effects. Clinical impact varies between investigated tissues, whereas patient populations studied reflect those usually treated in clinical practice. Considering all studies, the overall effectiveness of ESWT applied during acute care treatment must be rated as moderate (grade C) (Table 3). This classification is mainly a consequence of the high heterogeneity of the treated tissues, resulting in very small numbers of studies investigating similar injury conditions. However, as none of the studies reported significant side effects to enrolled participants, the present evaluation provides clear support for the conclusion that the procedure is safe.
Overall body of evidence.
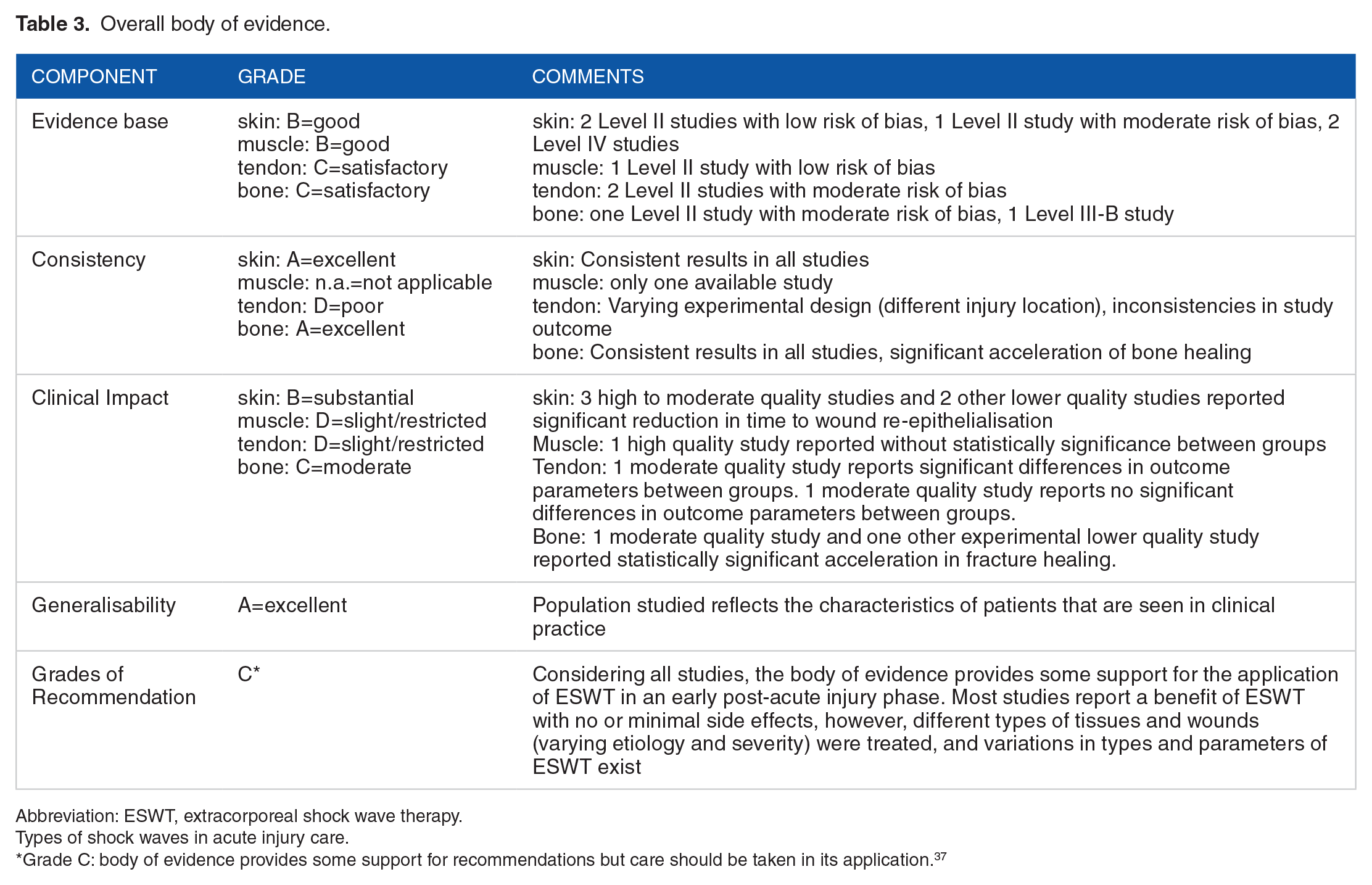
Abbreviation: ESWT, extracorporeal shock wave therapy.Types of shock waves in acute injury care.
Grade C: body of evidence provides some support for recommendations but care should be taken in its application. 37
Types of shock waves in acute injury care
All studies within this review that investigate effects of ESWT on muscle, tendon, and bone injury44–48 relied on ESWT in focused form which is able to deliver energy to targets located deeper within the tissue. In addition to this, unfocused radial ESWT has recently gained importance for treatment of superficial target areas. 53 Due to its technical principle, 54 radial ESWT has turned out to have advantages in the management of surface wounds. This is reflected in the select list of the present review in that radial ESWT was the method of choice in all work on treatment of skin injury.39–43
A more ambiguous situation exists regarding the application of different shock waves types for treatment of muscle injury. A large part of studies evaluating ESWT of muscle stiffness, myofascial trigger points, and muscle stroke reports on the effects of focused ESWT,55–57 as did Fleckenstein et al. 44 Applied to muscles with DOMS, focused ESWT resulted in improved relief of pain, better force generation, and higher living comfort, but not in a significant acceleration of the healing process. Some recent work has reported that also radial ESWT may be beneficial in certain conditions of muscle disorders.58,59 A recent systematic review found no difference in efficiency between focused and radial ESWT 54 in various orthopedic conditions including tendinopathies. Regarding bone fractures, there is as yet only 1 recent study that evaluates effects of radial ESWT on fracture nonunions in superficial bones. 60 Because of the limited amount of available evidence, it is hardly possible to formulate any advice regarding the application of the 2 shock wave types in further research of acute muscle, tendon, and bone injuries. Aspects such as specific anatomical situation, size of surface area, and depth of target region may all be critical for developing improved ESWT modalities, including choice between focused and radial ESWT.
Time of treatment onset after injury
Current practice of ESWT in the acute postinjury phase relies on 2 slightly different approaches. In most cases, shock waves are applied as early as possible after tissue damage. This is especially done in cases including surgery. An alternative approach favors a delayed application of ESWT after a resting period to allow for scar tissue formation. In skin wound treatment, these resting periods comprise a few days, in tendon treatment several weeks. Both approaches have been tested in regeneration studies using other types of mechanotherapeutic intervention, such as controlled loading, specific exercises, or compression. Irrespective of whether applied immediately after injury or after periods of rest, these methods were likewise shown to accelerate regeneration in tendon, muscle, cartilage, and bone injury. 16 It is presently unclear whether ESWT would lead to similar responses.
ESWT parameters in acute injury care
A favorable situation seems to prevail for skin wounds. Despite the diverse nature of these wounds, largely identical ESWT settings were successfully applied in the reviewed studies, using 100 impulses per square centimeter (in 1 case, 25 per centimeter wound length) at an EFD of 0.1 to 0.15 mJ/mm2 and a frequency of 5 Hz. These settings are comparable with those successfully used to treat wounds with delayed healing and chronic ulcers.61,62 Altogether, this may be consistent evidence that this dosage is safe and appropriate for acute wound treatment. However, larger well-controlled randomized trials are required for further corroboration.
A clearly higher demand for treatment dosage optimization is evident for ESWT application to acute tendon and bone injuries. The 2 reviewed studies on treatment of tendon injury differ greatly, not only in anatomical region (shoulder vs knee) but also in relation to the EFDs applied. Wang et al 45 used an EFD of 0.298 mJ/mm2 (1500 impulses), whereas Kim et al 46 administered an EFD of only 0.12 mJ/mm2 (1500 impulses). Comparing these studies, it may be hypothesized that ESWT dosage correlates with the outcome, as particularly the study with the higher dosage resulted in a significantly improved healing success in the ESWT group. This hypothesis is also supported by findings in chronic tendinopathies. 54 However, as there is only limited information from studies that did not investigate similar injury conditions, no truly valid statement can be made in this context.
High EFDs are commonly also applied to bony nonunions,50,51 with the aim to indirectly induce neo-vessel formation and osteogenesis via generation of microfractures. Although this mode of intervention might seem not be the best choice under acute state conditions, in the study of Wang et al, 47 significantly improved reunion of acute fractures was achieved with a high EFD of 0.62 mJ/mm2. However, as suggested in the uncontrolled study of Moretti et al, 48 testing whether lower energy levels acting via NO-induced stimulation of angiogenesis might be also useful under acute state conditions. Similarly, it remains to be tested whether low-energy shock waves are able to interfere with a still existing endosteal vascularization in the fracture area.
Another parameter that needs to be addressed is the number of ESWT sessions required for treatment success. In this respect, the fact that all RCTs reviewed in this study used only a single session may indicate that 1 application is sufficient to induce a positive outcome under a variety of different conditions.
Safety of ESWT in acute injury care
All studies except for that on muscle soreness of Fleckenstein et al 44 monitored possible occurring ESWT-induced side effects. None of the studies detected serious complications. For example, no cardiac, neurologic, dermal, or allergic reactions were found in association with skin wound treatment.39,40,42 No hematoma formation, skin lacerations, or enhanced postoperative pain was reported after treatment of surgical wounds. 41 Arnó et al 43 reported that 3 of 15 enrolled patients felt pain during ESWT treatment of skin burns (EFD: 0.15 mJ/mm2, 100 impulses/cm2), but VAS scores remained below 3. Similarly, no local or systemic complications were found for the application of ESWT to tendon grafts.45,46 Regarding bone fracture treatment, Wang et al 47 reported no device-related systemic or local complications, whereas Moretti et al 48 mentioned minor side effects in the form of local petechiae and hematoma that resolved spontaneously. In summary, this suggests that ESWT is without any major side effects when used for the treatment of various acute injuries.
Limitations
The results of this study cannot be generalized to all patients with similar acute injuries, as patients with certain histories and/or conditions (eg, dialysis-dependent renal failure, insulin-requiring diabetes mellitus, systemic neurologic disorders, coagulation disorders, and drug addiction) and pregnant women were excluded from individual studies. Because of the inhomogeneities among the included studies regarding types of injury, anatomical location of injury, treated tissues, etc, in turn leading to immense differences in experimental design and modes of outcome measurement, no meta-analysis was performed and direct comparisons between studies are complicated.
Although the authors attempted to identify all relevant studies, it is possible that some may have been missed (eg, due to nonavailability in searched databases or incompatible terminologies). Publication bias is also possible from the fact that the review only includes literature in English language.
Conclusions and Implications for Future Research
The overall data obtained in this study support the conclusion that ESWT improves the healing of acute injuries while highlighting several limitations. With the current data available, there is substantial evidence that ESWT applied as a primary therapy can accelerate the healing of several types of skin wounds. However, considering the extremely limited evidence, it is presently not possible to provide a true estimate of how shock waves interfere with the healing of acute injuries of muscle, tendon/ligament, and bone. In this regard, the results of the present evaluation rather provide an incitement into further research. Aspects to be considered will be to obtain larger numbers of high-quality studies on individual tissue types and study designs. A wide open field of uncertainty has also remained regarding optimizing the timing of postinjury administration and determining the usefulness and appropriate intervals of repeated application. In addition, there is as yet no information about how ESWT compares with other noninvasive treatment approaches. It therefore also remains to be tested whether ESWT could, for example, be an alternative to corticosteroids and nonsteroidal anti-inflammatory drugs, which, although routinely administered, have both been shown to implicate ambiguous effects and delayed or irreversibly disturbed healing. 63
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge financial support by the Open Access Publication Fund of the University of Salzburg.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Study conception and design: all authors; literature search and acquisition of data: AZ, SP; analysis and interpretation of data: AZ, SP, WS; drafting of manuscript: AZ, WS; critical revision: AMS.
