Abstract
Introduction:
We assessed the effectiveness of the joint use of a pH meter in combination with dietary supplements in restoring the urinary pH balance of patients with medical history of uric acid or calcium phosphate/calcium oxalate stones in real-world practice.
Methods:
An interventional, prospective, and open-label study was performed. At baseline visit, patients were assigned to a group according to the type of previous calculus and urinary pH: the alkalinizer group (uric acid stones and/or pH < 5.5) and acidifier group (calcium oxalate stones and/or pH > 6.2) received dietary supplement to increase or decrease, respectively, urinary pH. Patients were examined at baseline and after treatment for 30, 60, and 90 days. Urinary pH, type of therapy, compliance, and self-reported renal colic events were recorded at each visit.
Results:
The study included 143 patients, 45.5% in the alkalinizer group and 54.5% in the acidifier group, and the mean age was 53.60 years. Both nutraceuticals were significantly effective in normalizing urinary pH (P < 0.00001) at all follow-up visits compared with baseline, with a maximum percentage of patients who achieved nonlithogenic pH (54.9%) at day 60 (P < 0.00001). Analysis of the effect of treatment compliance at 60 days indicated that 71.8% of compliant and 45.9% of noncompliant patients achieved nonlithogenic pH (odds ratio [OR]: 3.03, 95% confidence interval [CI]: 1.29-6.66). A Cox-regression model indicated that nonlithogenic pH at 90 days (hazard ratio [HR]: 0.428, 95% CI: 0.193-0.947) and compliance at 60 days (HR: 0.428, 95% CI: 0.189-0.972) were independently associated with colic complaints-free survival.
Conclusions:
In patients with medical history of renal lithiasis, monitoring of pH in combination with dietary supplements may be useful in maintaining nonlithogenic pH values, yielding very high ratios of success, especially in compliant patients. Besides this main outcome, a reduction in self-reported colic complaints associated with pH balance was also observed.
Keywords
Introduction
Renal lithiasis is the consequence of a pathological change of conditions in the urinary tract toward those that favor crystallization. Urinary pH is one of the major determinants of kidney stone formation. 1 However, the importance of urinary pH as a major risk factor for kidney stone formation is not universally recognized by physicians and other health care providers. 1 In general, a pH below 5.5 increases the risk of uric acid stone formation, and a pH above 6.0 increases the risk of calcium stones. 2
When the urinary pH is below 5.5, insoluble uric acid crystals are formed. Therefore, uric acid renal stones are typically found in individuals with low urinary pH, even with normal excretion rate of uric acid. 3 , 4 In contrast, when the urinary pH is greater than 6.0, calcium phosphate forms hydroxyapatite or brushite crystals. 2 Formation of calcium oxalate monohydrate or dihydrate crystals is apparently unrelated to urinary pH, because the solubility of these salts hardly changes at physiological levels of urinary pH. However, calcium oxalate stone formation implies a heterogeneous nucleation process, 2 in which the presence of specific amounts of preformed uric acid and calcium phosphate crystals can act as heterogeneous nucleants of calcium oxalate crystals.5–7 Thus, a small number of these crystals may be sufficient to induce the generation of calcium oxalate crystals and ultimately calcium oxalate renal stones.
The standard treatment to prevent the formation of renal lithiasis has traditionally consisted on alkalinizing the urine, regardless of the urinary pH or type of stone. 4 So, instead of measuring and balancing urinary pH, alkalinizing therapies have been used to prevent renal stones in patients with previous renal lithiasis. However, it is particularly important to monitor and control urinary pH to prevent and treat renal lithiasis in a customized way according to the needs of each patient when dietary interventions (alkalinizing and acidifying agents) are performed. 8 , 9
Glass pH electrodes and pH meters are the gold standard systems for measuring pH due to their high accuracy and precision. 8 However, they require regular calibration and user training, so they are less widely used than dipsticks. Although dipsticks have the advantages of being inexpensive and easy to use, their precision and accuracy are insufficient for clinical decision-making in patients with renal lithiasis,10–12 according to the criteria needed to manage and prevent crystal formation.
An innovative domiciliary pH control system allows patients with renal lithiasis to perform self-monitoring and self-management of urinary pH values. 13 This system consists of a pH meter and 2 nutraceuticals, an alkalinizer or an acidifier supplement, and enables patients to control their own urinary pH. Urinary pH is adjusted using the dietary supplements, so that it is maintained within a safe range depending on the type of renal lithiasis and particularities of each patient. Although designed for urinary pH self-control, the pH measurements conducted in this preliminary study were exclusively obtained by physicians at the follow-up visits.
According to the large and robust evidence associating urinary pH and lithogenesis, we designed a real-world study to assess the effectiveness of the joint use of a pH meter and nutraceuticals, as a complementary tool of the current management of renal lithiasis, in maintaining nonlithogenic pH values in patients with previous medical history of renal lithiasis. Furthermore, we analyzed the existence of clinical symptoms of renal lithiasis and the safety of the nutraceuticals.
Materials and Methods
Study design
An interventional, prospective, and open-label study was performed between November 2014 and May 2017 at multiple health institutions in Spain. All procedures were in accordance with Declaration of Helsinki and ethical standards of the institutional and/or national research committee. Approval was obtained from Ethics Committee of the Hospital Clínico y Provincial (Barcelona), Hospital 12 de Octubre (Madrid), Hospital Universitari Germans Trias i Pujol (Barcelona), Hospital Universitario Ramón y Cajal (Madrid), Hospital de Bellvitge (Barcelona), Hospital Universitari Sagrat Cor (Barcelona), and Hospital Universitario La Paz (Madrid).
Participants
All eligible patients had medical histories of renal lithiasis due to calcium oxalate or uric acid stones and provided informed consent prior to enrollment. Participants were divided into 2 groups according to type of calculus and urinary pH: the acidifier group with calcium oxalate stones and/or pH above 6.2, and the alkalinizer group with uric acid stones and/or pH below 5.5. For all pH measurements, values were obtained from fasting spot urine samples.
Exclusion criteria were diagnosis of renal tubular acidosis and previous pH control therapy (alkalizing agents such as citrate, bicarbonate, and carbonated drinks; acidifying agents such as ammonium chloride, arginine chloride, and methionine). Patients with previous pH control therapy were included only if they underwent a 1-week washout period prior to the start of the study.
Treatment description
Patients were treated with Lit-Control® system (Devicare, Spain), consisting of Lit-Control® pH Meter and 2 nutraceuticals, an alkalinizer supplement and an acidifier supplement that, respectively, increase (Lit-Control® pH Up) or decrease (Lit-Control® pH Down) urinary pH value, whereas promoting the normal functioning of the body’s acid-base metabolism. The alkalinizer supplement consists of magnesium and potassium citrate, cocoa dry extract (theobromine), zinc, and vitamin A. The acidifier supplement is a combination of
Patients with previous uric acid stones and a urinary pH below 5.5 were treated with alkalinizer supplement (1 capsule every 12 h). Patients with previous calcium oxalate stones and/or pH above 6.2 were treated with acidifier supplement (1 capsule every 12 h). All participants, regardless of the treatment prescribed, were recommended to adopt the following specific dietary guidelines during the study: increase daily fluid intake, avoid salt and foods high in sodium, and avoid foods that have clear acidifying effects (excessive high-protein diets and alcoholic beverages) or alkalizing effects (oranges, tangerines, lemons, kiwis, and strict vegetarian diet). 20
Follow-up evaluation
Visits were scheduled every 30 days during the 3-month follow-up period. Fresh morning urine (<2 h from micturition), in fasting state and after consumption of the supplement, was used by physicians to measure the pH at each visit. Urinary pH of each patient was expected to be in the nonlithogenic range (5.5 < pH < 6.2).
Patients were also asked several questions at each visit to evaluate therapeutic compliance, adverse events, renal colic complaints, and consumption of food or medications that could modify urinary pH. Patients received supplements for the next 30 days at the end of each visit.
Patients were classified as “compliant” if they consumed more than 80% of the supplement and “noncompliant” if they failed to follow this indication.
Renal colic complaints since the previous visit were recorded at each visit and registered as events. Renal colic complaints from patients with more than 1 self-reported event during follow-up period were re-entered as new events (recurring cases).
Statistical analysis
A descriptive analysis was performed for all variables collected in the case report form (CRF): sex, age, recurrence of renal lithiasis, type of calculus, time since last renal colic event, baseline urinary pH, and previous use of therapy for pH control.
Categorical variables are presented as frequencies and proportions. Quantitative variables (continuous or ordinal) are presented as central tendency and dispersion. Categorical variables were analyzed using chi-square test described by Pearson, and the effect size is presented as odds ratio (OR). For continuous variables, the effect size was calculated by Cohen d. 21 For analysis of non-normally distributed or ordinal data, the Mann Whitney U test was used. The McNemar and Wilcoxon tests were used to assess the effectiveness of the dietary supplements and the significance of the change in urinary pH.
The survival curves were chosen to compare patients with lithogenic pH and patients with nonlithogenic pH in reporting renal colic complaints during the follow-up periods.
Kaplan-Meier survival analysis was performed using renal colic events as the dependent variable. A multivariate Cox proportional hazards model was also performed to assess the independent effect of 4 factors—treatment type, previous use of pH control therapy, nonlithogenic pH, and compliance—on the presence of self-reported colic events.
All statistical analyses were performed on the intention-to-treat population using SPSS 22.0 software for Windows. Statistics for all tables, figures, and graphs were calculated from the total number of valid cases.
Results
Patient characteristics
One hundred and forty-three patients were included in the study, 45.5% of whom received the alkalinizer supplement and 54.5% of whom received the acidifier supplement (Table 1). The mean patient age was 53.45 ± 11.59 years and 61.5% of the patients were men.
Characteristics of the study population. a
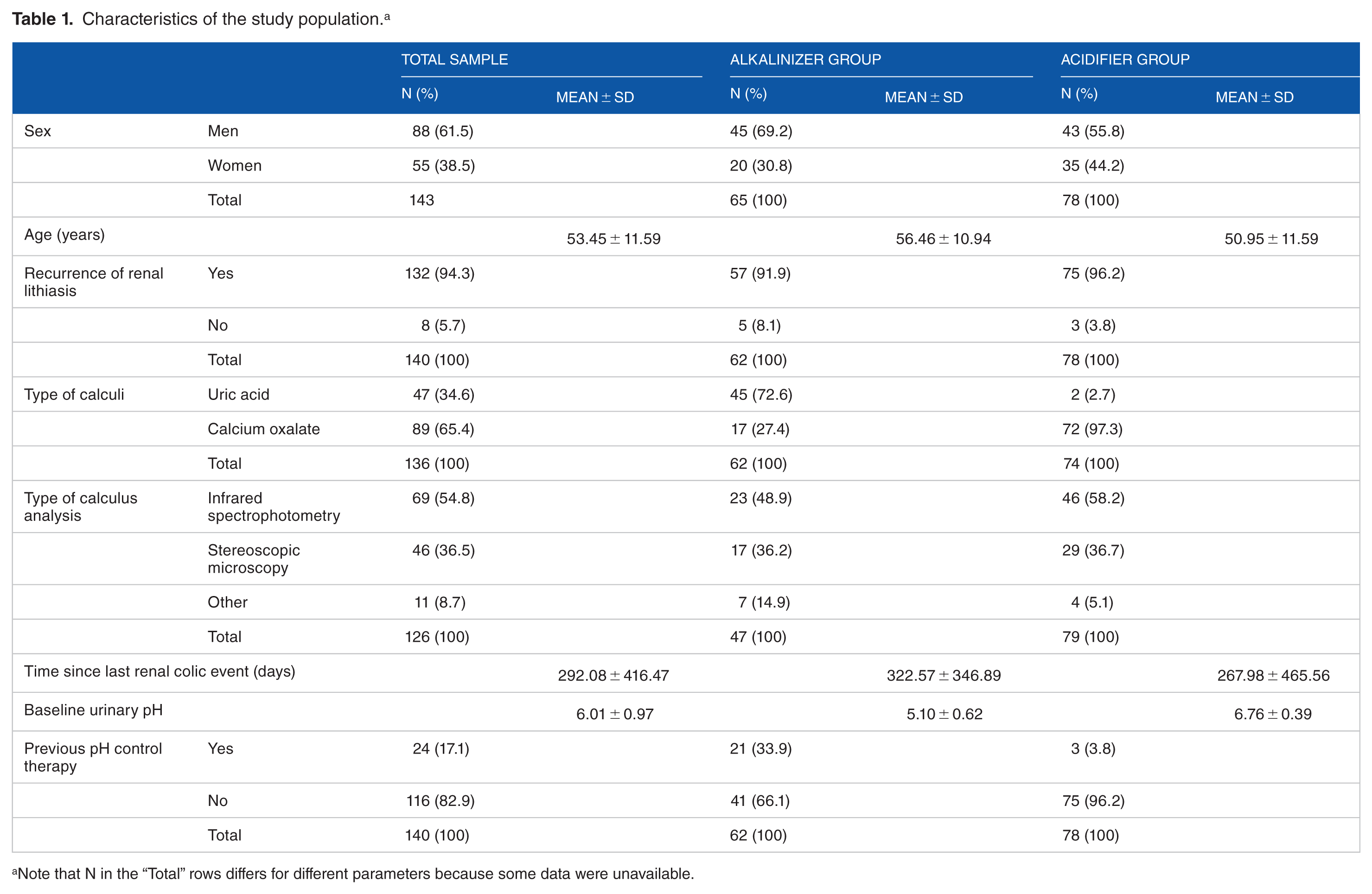
Note that N in the “Total” rows differs for different parameters because some data were unavailable.
pH meter, nutraceuticals, and pH modification
The combination of pH meter and nutraceuticals was significantly effective in normalizing urinary pH at each of the follow-up visits (30, 60, and 90 days) (P < 0.00001) (Figures 1 and 2), and the maximum change in pH from baseline was at 60 days in each group. Specifically, the pH increased by 0.77 ± 0.77 (Cohen d: 1.44) in the alkalinizer group and decreased by 0.54 ± 0.56 (Cohen d: 1.48) in the acidifier group. Pairwise comparisons indicated no significant changes between follow-up visits in either group (30 day vs 60 day, 60 day vs 90 day, 30 day vs 90 day).
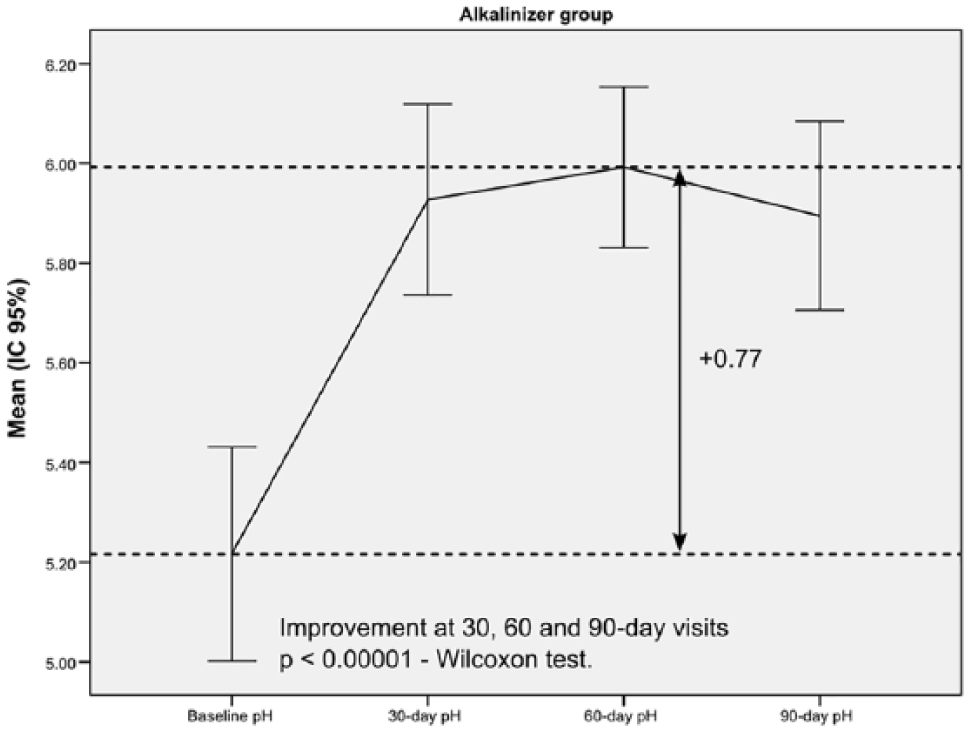
Change in urinary pH from baseline to the end of follow-up in the alkalinizer group.
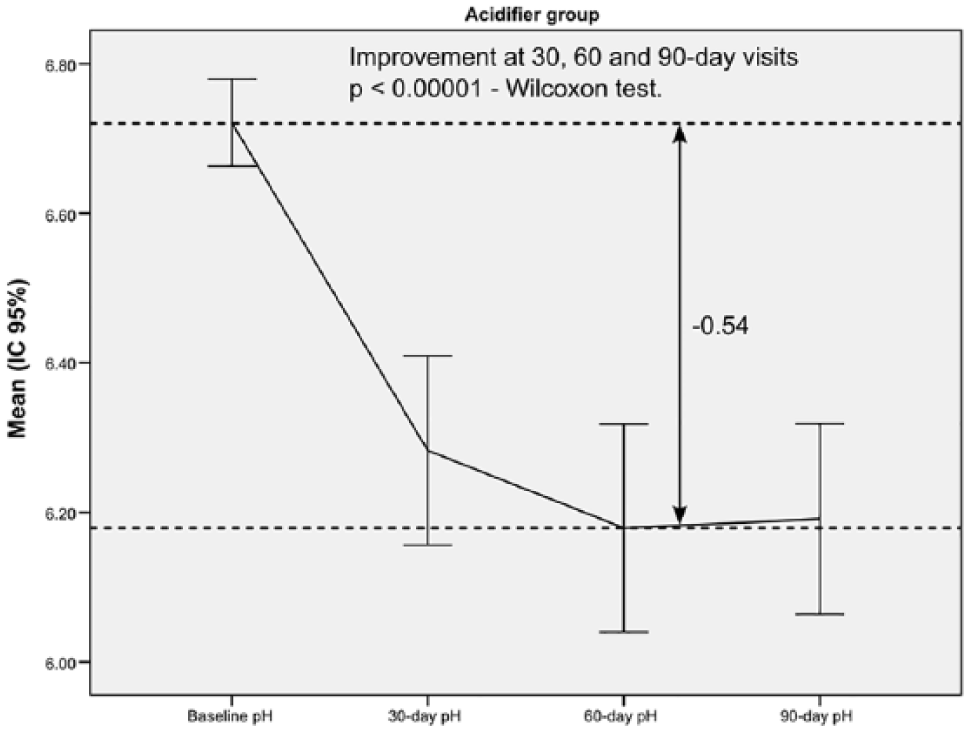
Change in urinary pH from baseline to the end of follow-up in the acidifier group.
pH meter, nutraceuticals, and nonlithogenic pH
The overall percentage of patients who had nonlithogenic urinary pH (5.5 < pH < 6.2) was significantly greater at all follow-up visits than at baseline, with a maximum of 55% at 60 days (P < 0.00001) (Figure 3A). Separate analysis of the 2 groups indicated that a maximum of 75% of individuals in the alkalinizer group had nonlithogenic pH at 60 days (P < 0.00001) and a maximum of 43.1% of individuals in the acidifier group had nonlithogenic pH at 90 days (P < 0.00001) (Figure 3B).
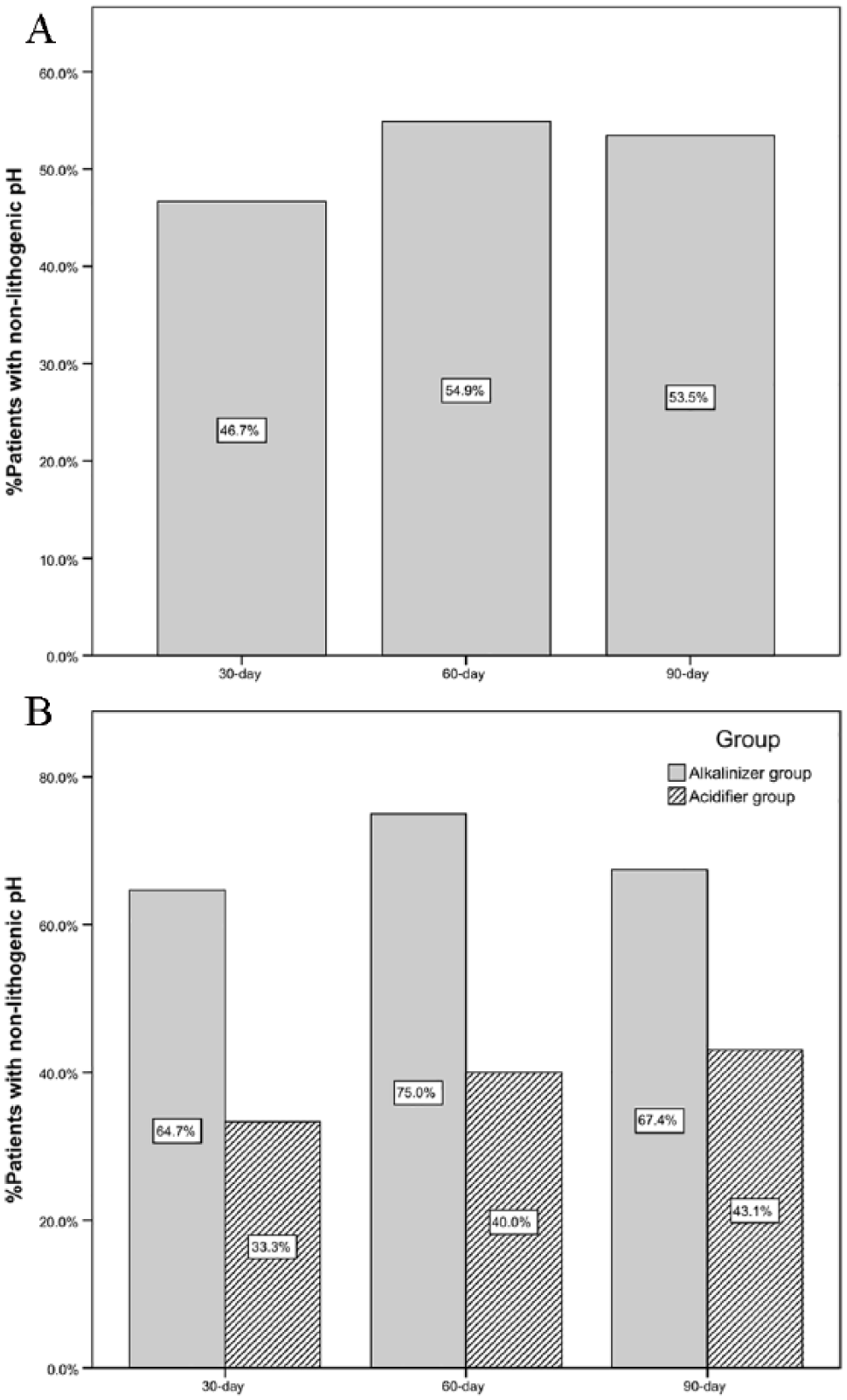
(A) Total percentage of patients who achieved nonlithogenic pH from baseline to the end of follow-up. (B) Percentage of patients in the alkalinizer group and acidifier group who achieved nonlithogenic pH from baseline to the end of follow-up.
Relationship of compliance with pH changes and nonlithogenic pH
Analysis of the effect of treatment compliance at 60 days indicated that 71.8% of compliant and 45.9% of noncompliant patients achieved nonlithogenic pH (OR: 3.03, 95% confidence interval [CI]: 1.29-6.66) (Figure 4). Separate analysis of the acidifier group indicated that 61% of compliant and 29.5% of noncompliant patients reached nonlithogenic pH after 60 days (OR: 3.85, 95% CI: 1.29-11.11); however, compliance had no significant effect on the development of nonlithogenic pH in the alkalinizer group (data not shown).
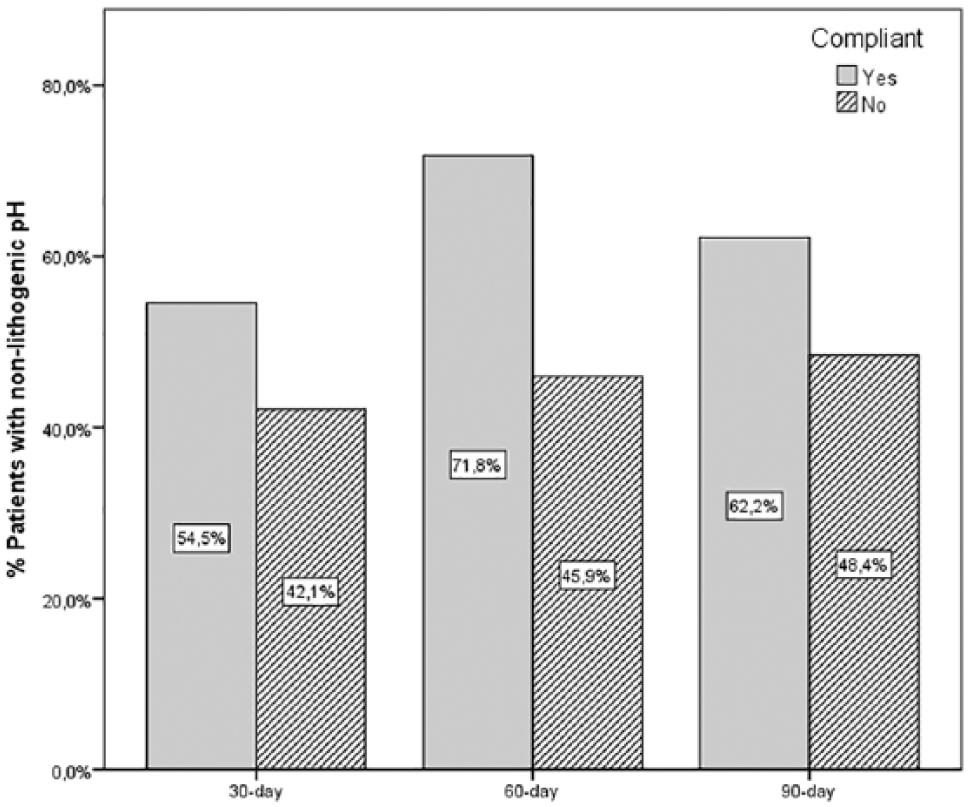
Total percentage of compliant and noncompliant patients who achieved nonlithogenic pH from baseline to the end of follow-up.
Furthermore, the pH change from baseline to 60 days in the acidifier group was 0.80 ± 0.70 in compliant patients, but 0.47 ± 0.61 in noncompliant patients (P < 0.05); the alkalinizer group and the whole sample had no significant differences in the changes of pH between compliant and noncompliant patients (data not shown).
Control of pH and self-reported colic events
Our survival analysis contained 159 observations, 43 events (self-reported renal colic complaints at each visit), and a median follow-up time of 94.5 days. Alkalinizer and acidifier groups had similar numbers of events (20 and 23, respectively). A total of 22 patients suffered more than 1 event during the study, 14 patients in the alkalinizer group and 8 patients in the acidifier group.
Kaplan-Meier survival analysis indicated that patients with nonlithogenic pH at the last visit had a censor rate of 77.2% and a mean event-free survival duration of 205.8 days (Figure 5). Patients with lithogenic pH had a censor rate of 64.2% and a mean event-free survival duration of 175.1 days (P = 0.096).
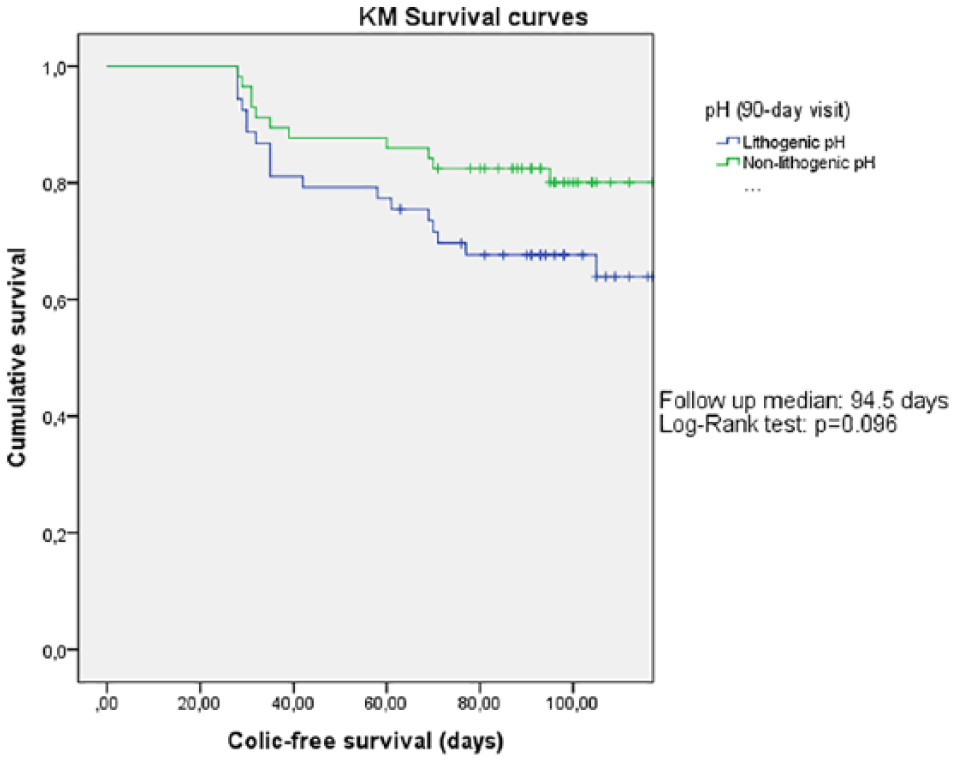
Kaplan-Meier analysis of renal colic-free survival of patients who achieved and did not achieve pH control.
Multivariate analysis using the Cox proportional hazards model showed that nonlithogenic pH at the 90 day visit (hazard ratio [HR]: 0.435, 95% CI: 0.193-0.985) and compliance in all the study (HR: 0.311, 95% CI: 0.12-0.84) were independently associated with the self-reported events (Figures 6 and 7, and Table 2). Neither of the other covariates included in the analysis (prescribed treatment and previous pH control therapy) showed significant effects.
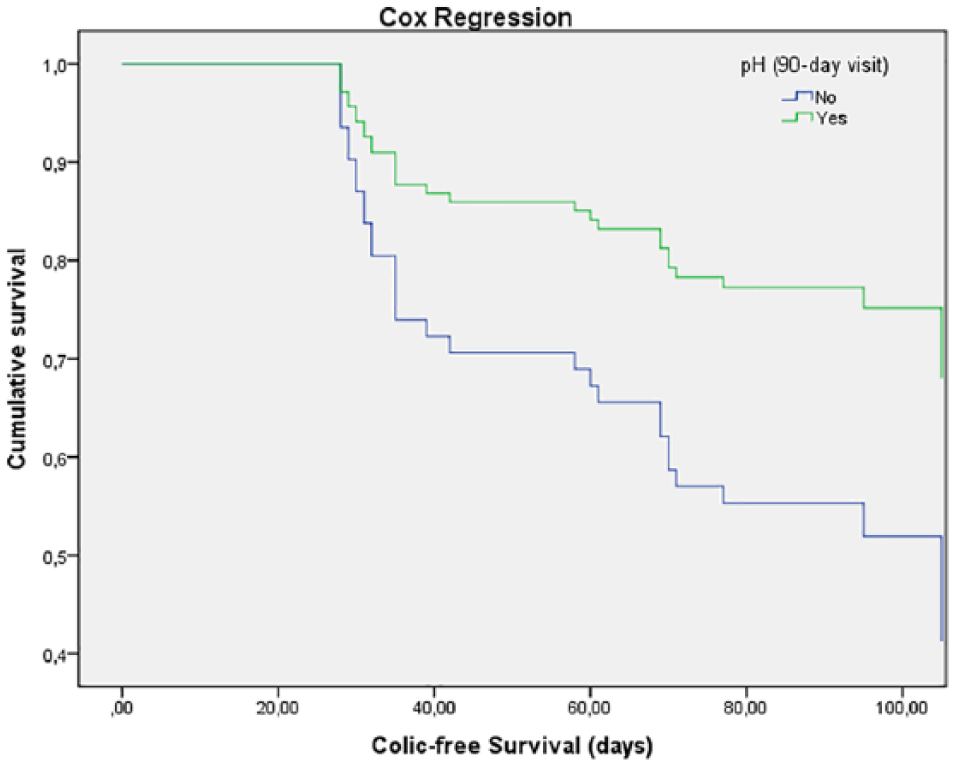
Cox proportional hazards model of renal colic-free survival of patients with and without pH control.
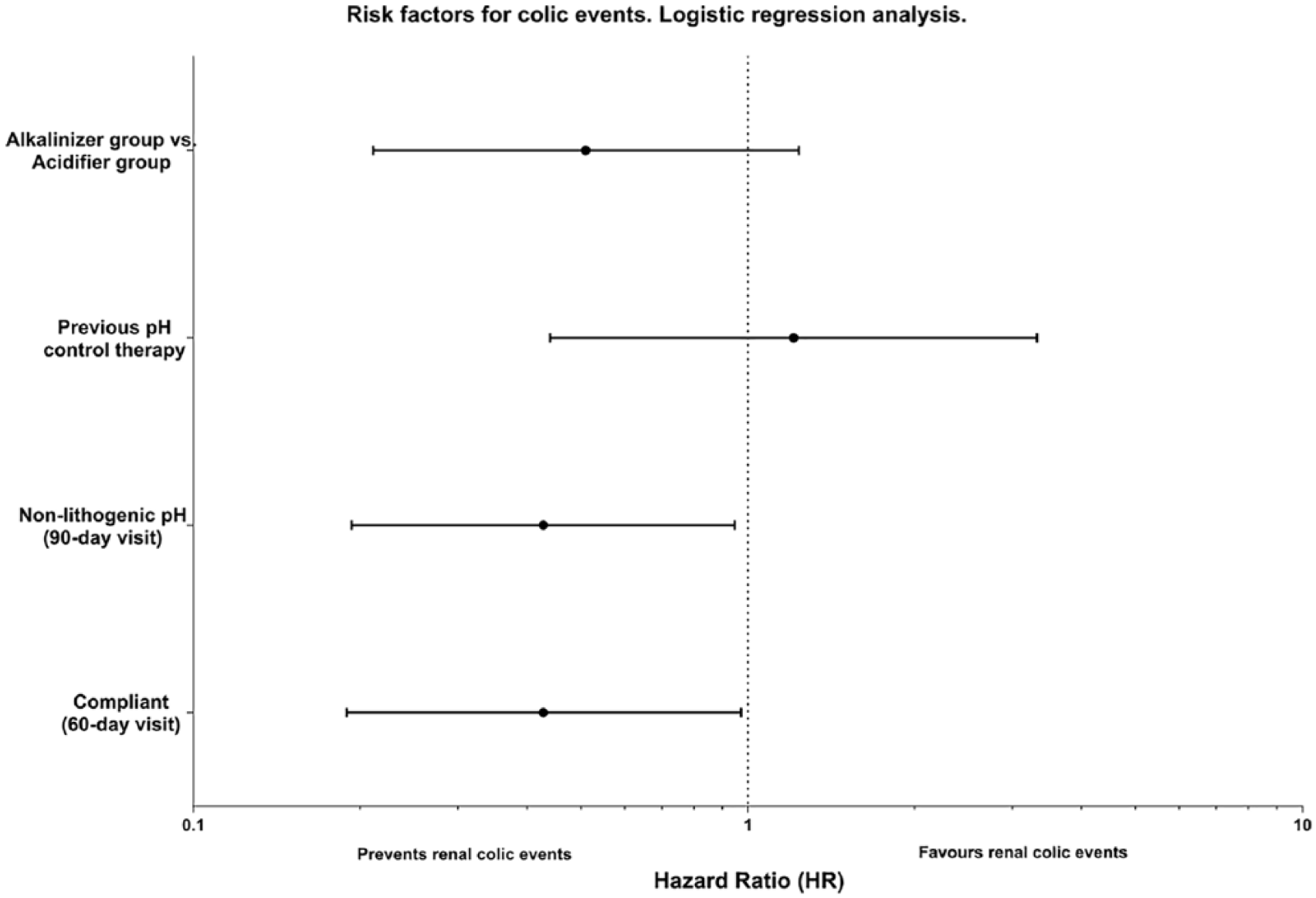
Logistic regression analysis of factors associated with renal colic-free survival.
Multivariable Cox regression analysis of factors associated with renal colic events.
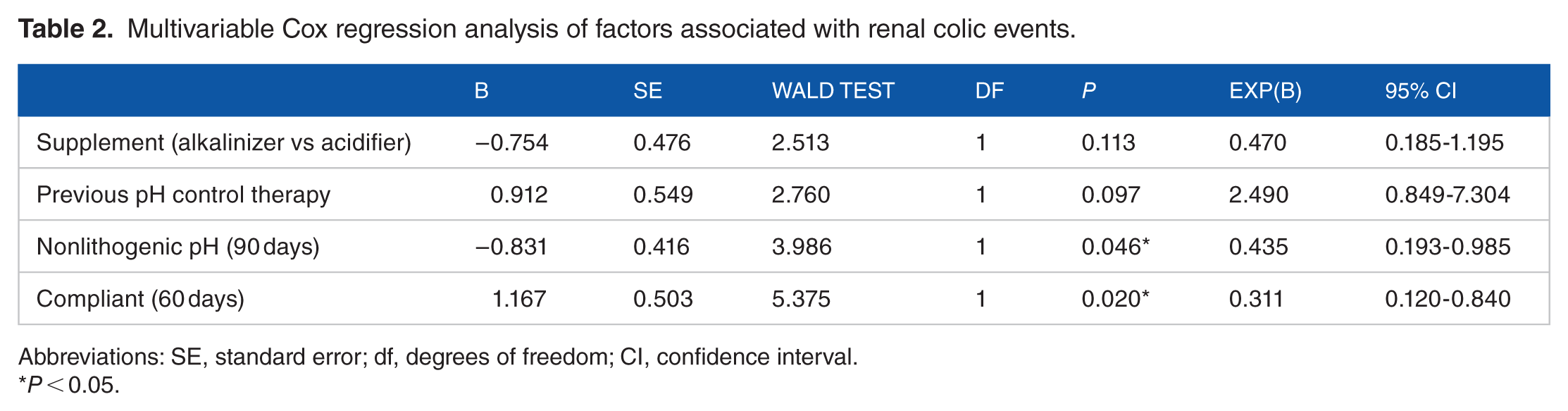
Abbreviations: SE, standard error; df, degrees of freedom; CI, confidence interval.
P < 0.05.
Safety
Both supplements were well tolerated, and during the 90-day study, there were only 8 adverse events in the alkalinizer group (diarrhea, insomnia, yellow urine, heartburn, constipation, and nocturnal polyuria) and 5 adverse events in the acidifier group (headache, mucus, and heartburn). None of these events required treatment cessation.
Discussion
This study, conducted in a real-world setting of medical assistance, showed that the use of a pH meter in combination with nutraceuticals was effective in normalizing urinary pH in patients who previously had renal lithiasis.
In particular, this intervention was found to be effective in patients with calcium oxalate calculi or uric acid calculi after 30, 60, and 90 days. The normalization of urinary pH in both treatment groups was remarkable, readily apparent at the first follow-up visit (30 day) and reached maxima at 60 days. There were no significant changes in urinary pH from day 30 to day 90 analysis, suggesting that the benefit observed at 30 days was sustained during the follow-up period. More than half of the patients in each group achieved nonlithogenic pH status. More specifically, 3 out of 4 patients in the alkalinizer group and 4 out of 10 patients in the acidifier group achieved nonlithogenic pH, and these changes were readily apparent at the 30-day follow-up visit.
After analyzing the effects of patient compliance to treatment, 7 out of 10 compliant and 4 out of 10 noncompliant patients reached nonlithogenic pH. This finding emphasizes the importance of compliance and indicates it should be considered when studying the effectiveness of a pH meter combined with nutraceutical intervention in maintaining nonlithogenic pH values in renal lithiasis patients.
Our outcomes point to the direct role of pH in the formation of uric acid stones and the indirect role of pH in the promotion of heterogeneous nucleation processes and the formation of calcium oxalate stones. 2 On this regard, it is important to take into account that both products confer renal lithiasis inhibition by the presence of phytate (acidifier supplement) and theobromine (alkalinizer supplement). Both molecules are crystallization inhibitors, and they work synergistically with pH correctors, reducing the nucleation and crystallization.22–27
In addition, the survival analyses showed that, once the urinary pH became nonlithogenic, free-survival rates of self-reported colic events improved. Although this result should be interpreted with caution, it shows a relationship between the reduction in renal lithiasis-related events and a nonlithogenic pH. Specifically, patient compliance to treatment together with nonlithogenic pH significantly and independently decreased the likelihood of reporting colic events. Moreover, although patients in the alkalinizer and acidifier groups had similar numbers of colic complaints, the alkalinizer group had almost twice as many self-reported recurring events as the acidifier group (14 vs 8 cases).
According to these results, the difference in pH between patients who achieved nonlithogenic pH (5.5 < pH < 6.2) and those who did not, could imply that a slight change in pH (<0.5) would reduce the clinical manifestation of renal lithiasis, even in individuals with history of calcium oxalate stones (which are not directly related to urinary pH). This emphasizes the clinical relevance of controlling urinary pH in patients prone to renal lithiasis.
This study has some limitations. First, it was conducted in real-world context and had an open-label and uncontrolled design. However, at the same time, this situation allows to analyze and to understand the actual patient behavior and how the product is used in daily medical practice and in general population. Following the routine clinical practice, physicians gave patients recommendations about fluid and dietary intakes during the study, but the patient compliance with those recommendations was not collected. In the same line, the exclusion criteria were not conservative and some patients with renal tubular acidosis may have been included in the study population. Moreover, although the present results suggest a link between pH normalization and the prevention of renal lithiasis, the endpoint in the survival analysis carried out was the patient self-reported colic complaints, not the objective diagnosis of renal lithiasis.
Despite its limitations, this study proposes that the joint use of a pH meter in combination with nutraceuticals may be a useful tool to be combined with the current management of renal lithiasis patients. A simple 3-month intervention (monitoring of pH in combination with dietary supplements) could have a significant positive effect on urinary pH yielding higher ratios of success in compliant patients. Furthermore, and according to the currently accepted knowledge, it might help to reverse and/or slow the growth of new stones, reducing the clinical manifestations associated with renal lithiasis. 28
In conclusion, this study may be used as a basis for the development of large, randomized, controlled, and high-powered studies to assess the efficacy of urinary pH control on the prevention of renal lithiasis.
Footnotes
Acknowledgements
Prevent-Lit Group, integrated by the investigators involved in the clinical trial, is gratefully acknowledged. The authors take full responsibility for the content of the paper and thank M. Antonella Mollica of Clever Instruments S.L., Spain, for writing assistance in the preparation of the manuscript (supported by Devicare S.L.).
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Devicare’s own resources.
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: X.P.-N. and J.C.-C. are Devicare employees. J.A.G.-L., C.T.-O., and M.P.L.-G. declare no conflict of interest.
Authors’ Note
The Prevent-Lit Group: Enrique Argüelles Salido, Hospital Universitario Virgen del Rocio, Sevilla; Montserrat Arzoz Fàbregas, Hospital Germans Trias i Pujol, Badalona; Jose María Banús Gassol, ICUN, Barcelona; Joan María Benejam Gual, Hospital de Manacor, Islas Baleares; Alberto Budía Alba, Hospital La Fe, València; Enrique Cao Avellaneda, Hospital de Santa Lucía de Cartagena, Murcia; Antonio Conte Visús, Policlínica Miramar, Palma de Mallorca; Isabel Díaz Sánchez, Instituto Médico Tecnológico, Barcelona; Juan Antonio Galan Llopis, Hospital Universitario de Vinalopo, Elche; Juan Alberto Lancina Martin, Hospital Juan Canalejo, A Coruña; Maria Pilar Luque Gálvez, Hospital Clinic de Barcelona, Barcelona; Félix Millán Rodríguez, Fundació Puigvert, Barcelona; Jose Francisco Morera Martínez, Hospital Universitario Dr. Peset, Valencia; Paola Pardo, Hospital Universitario Dr. Peset, Valencia; Manuel Carlos Reina Ruiz, Hospital Universitario Valme, Sevilla; Miguel Ángel Rodríguez Cabello, Hospital Sanitas, Madrid; Carlos Torrecilla-Ortiz, Hospital Universitario de Bellvitge, L’Hospitalet de Llobregat.
Author Contributions
JA G-L, CT-O and MPL-G were responsible for the design and leadership of the clinical study. The Prevent-Lit group corresponds to the main Medical Doctors involved in patient’s recruitment and monitoring together with data collection. XP-N contributed in the study coordination and writing the manuscript. JC-C led the study coordination and the writing of the manuscript.
