Abstract
Patients with autoimmune rheumatic conditions, particularly rheumatoid arthritis, have an increased cardiovascular risk when compared with the general population. Methotrexate is a relatively old, yet effective, immunomodulatory drug for the management of autoimmune and chronic inflammatory disorders, such as rheumatoid arthritis, particularly in terms of symptom control, quality of life, and disease progression. Recent meta-analyses have also shown that methotrexate treatment is associated with a lower risk of cardiovascular events when compared with other disease-modifying antirheumatic drugs. This suggests that methotrexate might exert specific protective effects against atherosclerosis and thrombosis. This mini-review discusses the mechanisms associated with the increased cardiovascular risk in rheumatoid arthritis, the pharmacokinetics and pharmacodynamics of methotrexate, the available evidence on the in vitro and in vivo effects of methotrexate on modifiable cardiovascular risk factors, and suggestions for future research directions.
Keywords
Introduction
Despite significant advances in diagnosis and treatment over the past 30 years, cardiovascular disease (CVD) remains the leading cause of death in middle-aged and older adults, accounting for approximately one-third of all deaths globally. 1 A significant proportion of CVD-related morbidity and mortality is attributable to acute atherothrombotic coronary or cerebrovascular events. Traditional CVD risk factors, such as age, hypertension, hypercholesterolemia, and diabetes, cannot fully predict the occurrence of CVD events. 2 Therefore, a considerable amount of research has been conducted to identify novel pathophysiological mechanisms and risk factors involved in the onset and progression of atherosclerosis and thrombosis. For example, the “inflammatory” theory of atherosclerosis, based on a large number of in vitro and in vivo studies elucidating the pathophysiological role of specific cytokines and inflammatory messengers, proposes that CVD risk factors favor the development of a pro-inflammatory and oxidative stress status both in the arterial wall and in the blood, which ultimately triggers atherothrombotic events. Notably, the presence of a pro-inflammatory state per se can alter vascular homeostasis, independently of the presence of traditional risk factors. 3 Furthermore, several studies have reported that (a) markers of inflammation, eg, C-reactive protein, are associated with an increased risk of CVD events 4 and (b) reducing inflammation by means of pharmacologic interventions, eg, statins, might reduce CVD risk. 5 Issues with the lack of a demonstrated cause-effect relationship between inflammation and CVD and data interpretation in intervention trials notwithstanding, there is good evidence that a pro-inflammatory state might promote atherosclerosis and thrombosis.
A significant number of drugs with anti-inflammatory and immunomodulatory effects are routinely prescribed for the management of musculoskeletal disorders and autoimmune conditions, such as rheumatoid arthritis (RA). The identification of novel cardiovascular protective effects and the repurposing of some of these agents for cardiovascular risk management would avoid the need for conventional drug discovery and development programs, providing at the same time significant public health benefits. This review discusses the mechanisms associated with the increased CVD risk in common autoimmune disorders, particularly RA, the recent epidemiologic data reporting a reduced risk of CVD events with the first-line disease-modifying antirheumatic drug (DMARD) methotrexate, and the evidence of in vitro and in vivo effects of methotrexate on individual CVD risk factors. Putative mechanisms accounting for the protective cardiovascular effects of methotrexate, as well as suggestions for future research directions, are also discussed.
RA and CVD
RA is a chronic disabling autoimmune condition that is characterized by local and systemic inflammation, joint pain, stiffness, and fatigue, as well as “extra-articular” manifestations affecting the skin, eye, lung, heart, kidney, blood vessels, salivary glands, nervous system, and bone marrow. 6 Despite significant advances in pharmacologic treatment, patients with RA have mortality rates that are higher than the general population. Furthermore, this mortality gap has been widening over time. Gabriel et al reported that compared with the general population, the mortality rates in RA men and women were 2.5 and 2.4 times higher in 1965, and 8.3 and 12 times higher in 2000, respectively. 7
The increased risk of death in patients with RA is primarily caused by CVD events. The increased cardiovascular risk in patients with RA, about 2-fold that of the general population, is comparable with the risk in patients with diabetes. 8 It is postulated that the presence of a chronic systemic pro-inflammatory state induces endothelial dysfunction, with consequent impaired synthesis of nitric oxide (NO), and the development of a pro-oxidant state in the arterial wall. 9 This, in turn, causes significant structural and functional alterations, particularly an increase in arterial stiffness. 10 The latter is associated with an increase in peripheral (brachial) and central (aortic) systolic and pulse pressure, left ventricular hypertrophy, and dysfunction and independently predicts adverse CVD outcomes in several patient groups.11,12 No RA-specific inflammatory factors or pathways accounting for the increased CVD risk in this patient group have been identified. However, Ajeganova et al have recently investigated the associations between 3 RA-related autoantibodies, rheumatoid factor (RF), anticyclic citrullinated peptide (anti-CCP), and anticarbamylated protein (anti-CarP), and cardiovascular mortality, assessed from national death registers, in 2331 patients with RA from 3 European cohorts. When analyzed separately, RF, anti-CCP, and anti-CarP independently predicted increased cardiovascular mortality (RF: hazard ratio [HR]: 1.62, 95% confidence interval [CI]: 1.25-2.11; anti-CCP: HR: 1.79, 95% CI: 1.37-2.34; and anti-CarP: HR: 1.66, 95% CI: 1.27-2.16). When the 3 antibodies were analyzed together, only anti-CarP independently predicted cardiovascular mortality (HR: 1.52, 95% CI: 1.04-2.21). 13
Other factors potentially contributing to the increased CVD risk in patients with RA include increased platelet activation, altered lipid profile, and insulin resistance.14–16 Furthermore, epidemiologic studies have shown an increased prevalence of hypertension, hypercholesterolemia, obesity, cigarette smoking, type 2 diabetes, metabolic syndrome, and insulin resistance in patients with RA when compared with the general population.16–20 Notably, in patients with RA, the prevalence of hypertension is significantly higher than that of other CVD risk factors.17,21 In a recent systematic review and meta-analysis, Baghdadi et al 22 observed that, similar to the general population, the presence of hypertension, type 2 diabetes, smoking, hypercholesterolemia, and obesity are associated with a significant increase in CVD morbidity and mortality in RA. Therefore, the available evidence suggests that the presence of systemic inflammation, alterations in vascular homeostasis and platelet function, and co-existing traditional risk factors, particularly hypertension, play a key role in the pathophysiology of CVD in RA.
Pharmacology of Methotrexate in RA
Methotrexate, an analogue of the B-vitamin folic acid, exerts significant immunosuppressive and anti-inflammatory effects in RA and other autoimmune disorders. Methotrexate, typically administered either orally or parenterally with weekly doses ranging between 5 and 25 mg, is the only DMARD associated with beneficial survival effects in patients with RA.23–25
Methotrexate requires a carrier-mediated transport system for its absorption, distribution, and renal excretion. This involves 2 transporter superfamilies, the solute carrier (SLC) transporters and the adenosine triphosphate–binding cassette (ABC) transporters (Figure 1). 26 The enzyme folylpolyglutamate synthase (FPGS) rapidly converts intracellular methotrexate into polyglutamate forms (Figure 1). Methotrexate polyglutamates increase the intracellular retention of methotrexate, mediate its pharmacodynamic effects, and correlate with treatment response in RA.27,28 Methotrexate inhibits the activity of key enzymes involved in cell proliferation and turnover, dihydrofolate reductase (DHFR), thymidylate synthase (TYMS), and aminoimidazole carboxamide ribonucleotide (AICAR) transformylase (ATIC; Figures 1 and 2). 26 The consequent reduced synthesis of purines, pyrimidines, and DNA provides therapeutic benefits in disease states, such as RA, that are characterized by a high turnover of inflammatory cells. Another important mechanism of action of methotrexate involves the accumulation of adenosine through the inhibition of ATIC (Figures 1 and 2). 26 The consequent accumulation of the substrate AICAR and its metabolites inhibits adenosine deaminase and adenosine monophosphate (AMP) deaminase (Figure 2). 26 By reducing the catabolism of adenosine and adenine nucleotides, the concentrations of adenosine increase from AMP dephosphorylation and exert significant anti-inflammatory effects through the A2A and A3 receptors. 29 Therefore, the main immunomodulating and anti-inflammatory effects of methotrexate in RA are mediated by the reduced synthesis of purines and pyrimidines and by the accumulation of adenosine.
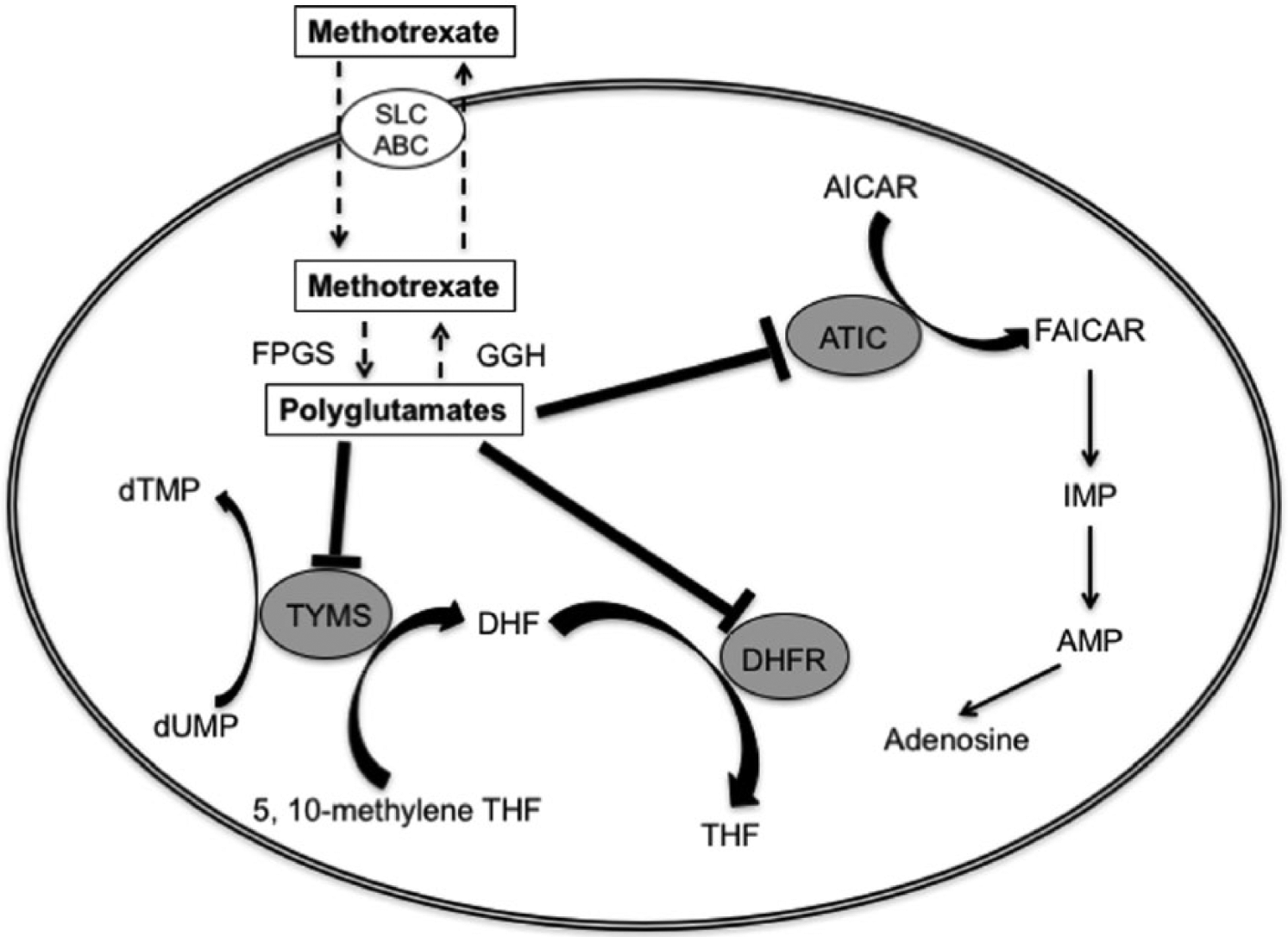
Mechanisms of action of methotrexate. ABC, adenosine triphosphate-binding cassette transporter; AMP, adenosine monophosphate; ATIC, aminoimidazole carboxamide ribonucleotide (AICAR) transformylase; dTMP, deoxythymidine monophosphate; dUMP, deoxyuridine monophosphate; DHF, dihydrofolate; DHFR, dihydrofolate reductase; FAICAR, 5-formamidoimidazole-4-carboxamide ribotide; FPGS, folylpolyglutamate synthase; GGH, gamma-glutamyl hydrolase; IMP, inosine monophosphate; SLC, solute carrier transporter; THF, tetrahydrofolate; TYMS, thymidylate synthase.
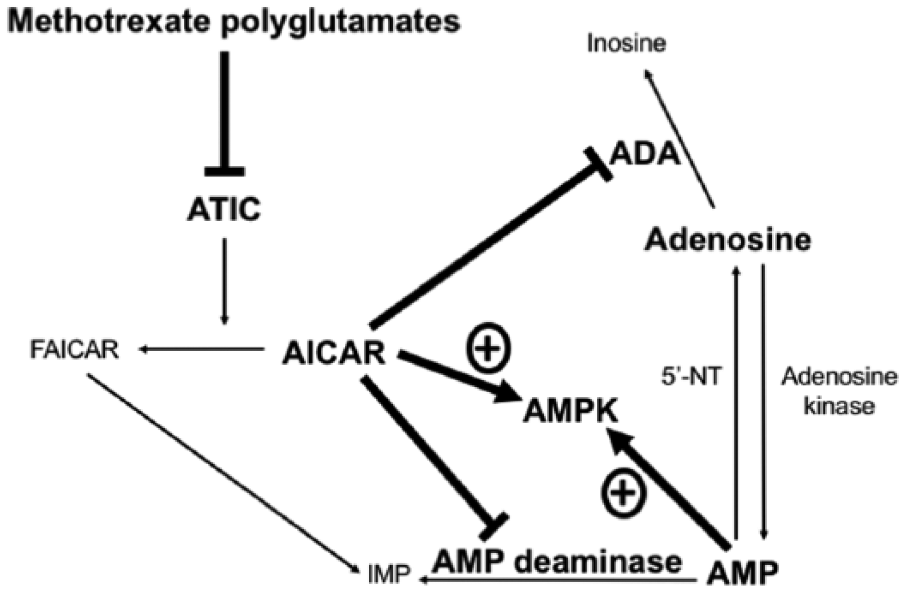
Intracellular effects of methotrexate-induced inhibition of aminoimidazole carboxamide ribonucleotide transformylase. 5′-NT, 5′ nucleotidase; ADA, adenosine deaminase; AMP, adenosine monophosphate; AMPK, 5′ adenosine monophosphate-activated protein kinase; ATIC, aminoimidazole carboxamide ribonucleotide (AICAR) transformylase; FAICAR, 5-formamidoimidazole-4-carboxamide ribotide; IMP, inosine monophosphate.
Genetic polymorphisms of transporters (SLC19A1, ABCC1-4, ABCB1, ABCG2) and target enzymes (methylenetetrahydrofolate reductase [MTHFR], DHFR, TYMS, ATIC, FPGS, and gamma-glutamyl hydrolase) can influence the efficacy and toxicity of methotrexate in RA and other autoimmune disease states (Figure 1). 26 The evidence regarding the potential impact of genetic polymorphisms on methotrexate treatment efficacy and toxicity is particularly strong for SLC19A1, MTHFR, and ATIC. 30 The nonsynonymous single-nucleotide polymorphism (SNP) in exon 2 of the SLC19A1 gene, Arg27His (80G > A), has a total minor allele frequency of 44% and is found across all ethnic groups. 31 Higher intracellular concentrations of methotrexate polyglutamates have been reported in patients with RA with the SLC19A1 AA genotype when compared with those with the GG or GA genotypes. 32 The A allele has shown associations with increased methotrexate treatment efficacy,33,34 whereas the G allele has shown associations with toxicity. 35 The nonsynonymous MTHFR SNP Ala222Val (677C > T) is also common. The homozygous TT genotype is particularly frequent in Northern China, Southern Italy, and Mexico (20%-32%). 36 However, only a small proportion of published studies have reported significant associations between the MTHFR 677C > T SNP and methotrexate efficacy or toxicity. 30 Similarly, relatively little evidence exists for an association between another MTHFR SNP, 1298A > C, the ATIC 347C > G SNP, and methotrexate efficacy or toxicity. 30
Methotrexate and CVD
In addition to its well-known anti-inflammatory and immunomodulatory effects, recent systematic reviews and meta-analyses have shown that methotrexate treatment is associated with a lower risk of CVD events.37–39 In 27 studies conducted in patients with RA, methotrexate use was associated with a reduced risk of all cardiovascular events (relative risk [RR]: 0.72, 95% CI: 0.57-0.91, P = .007) and myocardial infarction (RR: 0.81, 95% CI: 0.68-0.86, P = .01) when compared with other synthetic DMARDs. 39 These data support the hypothesis that methotrexate exhibits specific protective cardiovascular effects.
The following sections describe the available evidence on possible associations with, and/or effects of methotrexate on, traditional and new modifiable cardiovascular risk factors, particularly blood pressure, cholesterol, diabetes, insulin resistance, metabolic syndrome, endothelial dysfunction, and arterial stiffness. A PubMed literature search on in vitro and in vivo studies of RA and other autoimmune conditions was conducted from inception to May 2017, using the following terms: methotrexate, blood pressure, hypertension, low-density lipoprotein (LDL) cholesterol, high-density lipoprotein (HDL) cholesterol, diabetes, serum glucose, metabolic syndrome, insulin resistance, endothelial function, and arterial stiffness. The risk factors C-reactive protein and homocysteine were not considered because their associations with methotrexate treatment are likely to be significantly diluted by the disease activity per se (C-reactive protein) or the concomitant use of potential cardioprotective treatments such as the B-vitamin folic acid, a known homocysteine-lowering agent. 40
Cholesterol
Human studies have assessed the effects of methotrexate on serum cholesterol concentrations and cholesterol efflux capacity; a protective mechanism in cell types involved in the atherosclerotic process, particularly macrophages within the vascular wall, that reverts the LDL-induced intracellular cholesterol accumulation and favors its elimination through the liver and bile. 41 Reiss et al 42 observed that methotrexate treatment was able to prevent the inhibitory effects of cyclooxygenase-2 inhibitors and interferon gamma on the cholesterol efflux protein in human THP-1 monocyte/macrophages. In a cross-sectional study of 169 patients with RA, Rho et al 43 did not observe any significant differences in LDL and HDL cholesterol (LDL-C and HDL-C) concentrations between patients treated with methotrexate and patients treated with other DMARDs (LDL-C: 112 ± 31 vs 115 ± 38 mg/dL, P = .75; HDL-C: 48 ± 15 vs 43 ± 11 mg/dL, P = .12), after adjusting for age, sex, race, 28-joint disease activity score, hypertension status, smoking status, and use of statins. Ronda et al 44 reported that 6-month methotrexate treatment in patients with RA (n = 34) was associated with a significant increase in total (+8%), LDL-C (+9%), and HDL-C (+15%) cholesterol concentrations and cholesterol efflux capacity. Combination treatment with methotrexate and the tumor necrosis factor α (TNF-α) inhibitor adalimumab (n = 22) did not cause any significant changes in lipid profile, barring a transient increase in HDL-C concentrations. Rodriguez-Jimenez et al observed similar changes, to that reported by Ronda et al, in total cholesterol (+11%), LDL-C (+9%), and HDL-C (+6%) concentrations in patients with RA (n = 13) treated with methotrexate for 24 weeks. Combination treatment with methotrexate and the TNF-α inhibitor etanercept (n = 22) caused a reduction in total cholesterol (−3%) and LDL-C (−3%) concentrations and an increase (+18.5%) in HDL-C concentrations. 45 A greater increase in total cholesterol (+30%), LDL-C (+28%), and HDL-C (+39%) concentrations was reported by Navarro-Millàn et al after 24-week methotrexate treatment in patients with RA (n = 226). Similar changes were observed with combination of methotrexate and etanercept (n = 155) and with triple therapy with methotrexate, sulfasalazine, and hydroxychloroquine (n = 78). 46 However, the magnitude of the reported changes in the 3 lipid fractions was smaller after 2 years of follow-up. 47
In contrast to the previous reports, Ormseth et al 48 did not observe any significant changes in cholesterol efflux capacity in patients with RA receiving 6-month treatment with methotrexate (n = 23, −3% ± 20%, P = .38), adalimumab (n = 22, +2% ± 15%, P = .44), or tocilizumab, a humanized monoclonal antibody targeting the interleukin 6 (IL-6) receptor (n = 25, +4% ± 14%, P = .23). O’Neill et al did not report any significant changes in cholesterol efflux capacity in patients with RA randomized to 54-week treatment with either methotrexate alone (n = 7, +8%, P = .26) or combination treatment with methotrexate and the TNF-α inhibitor infliximab (n = 11, +4%, P = .72). Furthermore, no significant changes in total cholesterol, LDL-C, and HDL-C concentrations were observed with either treatment. 49 Park et al 50 did not observe any significant association between 1-year treatment with methotrexate and changes in total cholesterol, LDL-C, and HDL-C concentrations in newly diagnosed patients with RA (n = 37), although no specific quantitative data are presented. Similarly, Georgiadis et al reported that 1-year treatment with methotrexate and prednisolone in patients with early RA (n = 58) did not significantly change total cholesterol and LDL-C concentrations but significantly increased HDL-C concentrations (+30%). The latter was associated with a significant reduction in C-reactive protein concentrations, erythrocyte sedimentation rate, and cholesterol ester transfer protein. 51 Filippatos et al 52 observed similar effects, to those reported by Georgiadis et al, on total cholesterol, LDL-C, and HDL-C concentrations after 1-year treatment with methotrexate and prednisolone in patients with RA (n = 30).
Pending further confirmation in larger interventional studies that account for other clinical and demographic confounders, these data suggest that methotrexate might influence the concentrations of different lipid fractions in RA. Patients with RA show significantly lower plasma concentrations of total cholesterol, HDL-C, and LDL-C when compared with the general population. This suggests that systemic inflammation might reduce circulating lipid concentrations. 53 Moreover, in one study, the increased cardiovascular risk observed in patients with RA was paradoxically associated with low total cholesterol concentrations, a pattern known as the “lipid paradox.” 15 Accepting that lipid parameters generally change in parallel with suppression of inflammation, an indirect effect of methotrexate on disease activity, rather than a specific direct effect on circulating lipid fractions, might explain the paradoxical increase in several lipid fractions reported in patients with RA receiving methotrexate treatment. 54
Glucose homeostasis, metabolic syndrome, insulin resistance, and diabetes
Glucose homeostasis
In vitro studies have reported salutary effects of methotrexate on measures of glucose homeostasis. Russo et al observed that 4-week treatment with methotrexate caused a 4-fold increase in the messenger RNA expression, and a 2-fold increase in the skeletal muscle concentrations, of the GLUT-4 glucose transporter in a mouse model of diabetes. These effects, thought to be secondary to the activation of 5′ AMP-activated protein kinase (AMPK) by methotrexate-induced AICAR accumulation (Figure 2), were associated with a significant reduction in serum glucose (−44%, P < .001) and insulin (−34%, P < .001) concentrations and the homeostasis model assessment of insulin resistance index (HOMA-IR; −63%, P < .001). 55 Similarly, Pirkmajer et al 56 observed a methotrexate-induced indirect activation of AMPK, a known trigger of glucose uptake, fatty acid oxidation, and mitochondrial biogenesis, in wild-type HEK-293 cells.
Metabolic syndrome
Toms et al observed an independent association between methotrexate treatment and a reduced prevalence of metabolic syndrome (odds ratio [OR]: 0.51, 95% CI: 0.33-0.81, P = .004) in 400 patients with RA, after adjusting for age, sex, disease duration, erythrocyte sedimentation rate, Health Assessment Questionnaire score, treatment with sulfasalazine, hydroxychloroquine, leflunomide, TNF-α inhibitors, steroids, and nonsteroidal anti-inflammatory drugs (NSAIDs). In this study, no independent associations were observed between treatment with other DMARDs, steroids, and NSAIDs and the presence of metabolic syndrome. 57 Similar independent associations between methotrexate treatment and lower prevalence of metabolic syndrome (OR: 0.74, 95% CI: 0.55-0.96, P = .024), after adjusting for age, 28-joint disease activity score, erythrocyte sedimentation rate, RA duration, RF positivity, and Health Assessment Questionnaire, were reported by Dao et al 58 in 105 women with RA. An independent association between methotrexate use and reduced prevalence of metabolic syndrome (OR: 0.32, 95% CI: 0.12-0.86, P = .02), after adjusting for RA severity, NSAID use, and Framingham Risk Score, was also reported by Abourazzak et al 59 in 179 patients with RA. Furthermore, Zafar et al 60 observed a reduced prevalence of metabolic syndrome with methotrexate treatment, with (13%) or without (18%) hydroxychloroquine and sulfasalazine, when compared with no treatment (70%) or treatment with hydroxychloroquine (54%) or other DMARDs (40%, P < .001 for trend), in 384 patients with RA, although no adjustment for confounding factors was performed. Bilecik et al 61 observed, in a group of 100 women with RA, a lower prevalence of metabolic syndrome in patients treated with methotrexate when compared with those not treated with the drug (17% vs 35%, P = .046). In a retrospective study, Costa et al investigated the changes in metabolic syndrome components after 24-month treatment with methotrexate, etanercept, or adalimumab in patients with psoriatic arthritis. When compared with the other agents, methotrexate treatment was not associated with significant changes in waist circumference, prevalence of hypertension, and serum concentrations of triglycerides and glucose. However, in contrast to other reports, a significant reduction in HDL-C concentrations (−18%, P < .05) was observed. 62
Insulin resistance
Rajappa et al studied the effects of 12-week methotrexate treatment on parameters of insulin resistance in 60 patients with psoriasis. Significant reductions in insulin concentrations (−9%, P < .0001), leptin/adiponectin ratio (−41%, P < .0001), and HOMA-IR (−10%, P = .008) were observed. 63 Similarly, Bissell et al reported that 78-week combination treatment with methotrexate and methylprednisolone caused a significant reduction in HOMA-IR in 79 patients with RA. However, the effects on HOMA-IR were relatively greater when methotrexate was combined with the TNF-α inhibitor infliximab (adjusted difference: 0.58, 95% CI: 0.41-0.82, P = .003). 64 Yasar Bilge et al studied the effects of 3-month treatment with methotrexate and prednisolone on HOMA-IR and adiponectin concentrations in 65 patients with RA. Although no significant changes in HOMA-IR were observed (−23%, P = .688), treatment caused a significant increase in adiponectin concentrations (+14%, P = .032). 65 Dessein et al observed a reduction in insulin concentrations (−27%) and HOMA-IR (−51%) in 14 patients with either RA or spondyloarthropathy treated with methotrexate for 3 months, although no specific statistical analysis of the differences was presented. In this study, similar changes were observed in 8 patients not treated with methotrexate. 66
Diabetes
In a cross-sectional study of 169 patients with RA, Rho et al did not observe any significant differences in serum glucose concentrations between patients treated with methotrexate and patients treated with other DMARDs, after adjusting for age, sex, race, 28-joint disease activity score, hypertension status, smoking status, and use of statins (β = −2.91, 95% CI: 2.19 to −8.01, P = .27). 43 In a study population including 462 patients with psoriasis, psoriatic arthritis, and RA, with or without diabetes, Wu et al 67 did not observe significant changes in serum glucose concentrations and glycated hemoglobin after 8-month treatment with either methotrexate alone (n = 344, +1% and +1%, respectively) or in combination with TNF-α inhibitors (n = 118, −1% and +4%, respectively). Similarly, Rekedal et failed to observe significant reductions in glycated hemoglobin in 37 patients with RA, systemic lupus erythematosus or other autoimmune conditions, after 12-month treatment with methotrexate (−1%). In this study, the reduction in glycated hemoglobin in patients treated with hydroxychloroquine (−9%) was significantly greater than those observed with methotrexate (P = .04). 68 By contrast, Perdan-Pirkmajer et al 69 observed a significant reduction in glycated hemoglobin (−5%, P < .001) in 26 nondiabetic patients with either RA or psoriatic arthritis treated with methotrexate for 6 months.
In a large retrospective cohort study of 13 905 patients with RA or psoriasis, Solomon et al investigated the associations between the use of different DMARD-based treatment regimens and the risk of new-onset diabetes during a mean follow-up of 5.8 months. After adjusting for diagnosis, data source, age, sex, Charlson co-morbidity index, dermatology visits, number of different medications, number of hospital visits, prior use of hydroxychloroquine, methotrexate, nonbiologic immunosuppressive drugs, oral steroids, and year of cohort entry, the HR of diabetes with hydroxychloroquine (HR: 0.54, 95% CI: 0.36-0.80) and TNF-α inhibitors (HR: 0.62, 95% CI: 0.42-0.91), but not methotrexate (HR: 0.77, 95% CI: 0.53-1.13), was significantly lower than that with other DMARDs. 70 Similarly, a relatively lower incidence of new-onset diabetes with hydroxychloroquine (HR: 0.67, 95% CI: 0.57-0.80, P < .001) but not with combination treatment with methotrexate and other DMARDs (HR: 0.87, 95% CI: 0.72-1.05, P = .158) was reported by Ozen et al 71 in 13 669 patients with RA followed for 4.6 years, after adjusting for age, sex, RA duration, socioeconomic status, ethnicity, smoking, hypertension, Charlson co-morbidity index, body mass index, health assessment questionnaire, NSAID use, and year of study entry. Lee at al reported that an increase in 100 defined daily doses of methotrexate within 10 months was independently associated with an increased risk of diabetes (HR: 1.20, 95% CI: 1.07-1.33, P = .001) in 6611 patients with psoriasis, after adjusting for disease severity, age, dyslipidemia, hypertension, Cushing disease, treatment with psoralen plus ultraviolet A, and thiazide diuretics. 72
Blood pressure, endothelial function, vascular smooth muscle cells, and arterial stiffness
Blood pressure
In a cross-sectional study of 169 patients with RA, Rho et al observed that methotrexate treatment was associated with a trend toward a lower systolic blood pressure (SBP: 132 ± 21 vs 137 ± 18 mm Hg, P = .09) and a significantly lower diastolic blood pressure (DBP: 74 ± 11 vs 78 ± 9 mm Hg, P = .02) when compared with treatment with other DMARDs. However, the differences in SBP and DBP were no longer significant after adjusting for age, sex, ethnicity, 28-joint disease activity score, history of hypertension and diabetes, smoking status, and use of statins. 43 In 27 patients with RA without history of hypertension, diabetes, and dyslipidemia, Cuchacovich et al 73 reported that methotrexate treatment was associated with lower SBP (120 ± 15 mm Hg vs 124 ± 12 mm Hg) and DBP (76 ± 10 mm Hg vs 82 ± 11 mm Hg) when compared with treatment-naïve patients with RA, although no formal statistical analysis was presented. Gyldenlove et al 74 did not observe any significant differences in either SBP (median: 127 mm Hg, range: 95-160 vs 125 mm Hg, 95-165, P = .944) or DBP (median: 80 mm Hg, range: 50-110, vs 80, 55-95, P = .793) in 32 patients with psoriasis treated with methotrexate for 10 weeks. Tam et al investigated the effects of 6-month treatment with methotrexate, alone or in combination with infliximab, in 40 patients with RA. A reduction in SBP (−3 ± 15 mm Hg and −4 ± 13 mm Hg) and DBP (−4 ± 10 mm Hg and −1 ± 11 mm Hg) was observed with both treatments; however, no statistical analyses of changes vs baseline were presented. 75 In a repeated cross-sectional study of patients with RA treated either with methotrexate, with or without other DMARDs (n = 56), or with other DMARDs but not methotrexate (n = 30), Mangoni et al reported significantly lower clinic peripheral SBP (−8 mm Hg, 95% CI: −13 to −2, P = .006) and DBP (−6 mm Hg, 95% CI: −10 to −2, P = .001) values with methotrexate treatment, after adjusting for visit, age, sex, body mass index, folic acid use, and 28-joint disease activity score. Treatment with methotrexate was also associated with significantly lower average 24-hour ambulatory SBP (−4.6 mm Hg, 95% CI: −6.9 to −2.3, P < .001) and DBP (−2.1 mm Hg, 95% CI: −3.8 to −0.6, P = .006) values. 76 van Halm et al 77 observed a relatively lower prevalence of hypertension in patients with RA treated with methotrexate (12%) when compared with patients treated with sulfasalazine (26%), hydroxychloroquine (17%), or patients never treated with DMARDs (24%). However, no formal statistical analysis of between-group differences was presented.
Endothelium
In vitro studies have provided contrasting results on the effects of methotrexate on endothelial structure, function, and NO synthesis. Merkle et al 78 reported a reduction in bovine pulmonary artery endothelial cell integrity, consisting in the development of gaps between cells and endothelial cell death, after 4-day exposure to methotrexate. Similarly, Fuskevag et al 79 observed endothelial cell swelling and membrane disruption in rats receiving methotrexate as a bolus, followed by a continuous infusion more than 8 hours. However, as reported by Hirata et al, the antiproliferative effects of methotrexate on endothelial cells, observed in a model of corneal neovascularization, might be beneficial in the pathophysiology of RA and other autoimmune disorders, by preventing synovial neovascularization. 80 In this context, Yamasaki et al studied the effects of low-dose methotrexate on cell growth and the TNF-α–induced expression of intercellular adhesion molecule 1 (ICAM-1) and vascular cell adhesion molecule 1 (VCAM-1), key steps in the neovascularization process, in human umbilical vein endothelial cells (HUVEC). Methotrexate treatment inhibited cell proliferation without exhibiting cytotoxic effects. This was associated with a concomitant reduction in VCAM-1 and, particularly, ICAM-1 expression. 81 Furthermore, Bulgarelli et al 82 reported that methotrexate treatment significantly reduces TNF-α gene expression in HUVEC. Thornton et al observed that methotrexate activates AMPK phosphorylation, and the expression of the cytoprotective genes manganese superoxide dismutase and heme oxygenase, in HUVEC and in human aortic endothelial cells. These effects were maintained after pretreatment with TNF-α. In this study, methotrexate treatment also reduced endothelial cell apoptosis induced by glucose deficiency. 83 Although the full biological and pathophysiological roles of AMPK remain to be elucidated, studies suggest that its activation exerts beneficial effects on vascular homeostasis. 84
In a Wistar rat–isolated perfused kidney model, El-Gowilly et al 85 observed that 3-day treatment with methotrexate significantly impaired endothelium-dependent (acetylcholine-mediated), but not endothelium-independent (nitroprusside-mediated), vasodilatation, suggesting a specific detrimental effect on endothelial function. By contrast, Galarraga et al 86 did not observe significant changes in skin vascular responses to iontophoresis of either acetylcholine or nitroprusside after 4-month therapy with either methotrexate (n = 21) or TNF-α inhibitors (n = 30) in patients with RA. Furthermore, Gyldenlove et al 74 did not observe any significant changes in the reactive hyperemic index, an indirect marker of endothelial function, in 32 patients with psoriasis treated with 10-week methotrexate. By contrast, Hjeltnes et al 87 reported that 6-month treatment with methotrexate, but not when combined with a TNF-α inhibitor, significantly increased the reactive hyperemic index (+15%, P = .018) in 55 patients with RA.
Vascular smooth muscle cells
A role for vascular smooth muscle cells (VSMCs) undergoing phenotypic switching in atherosclerosis and vascular damage in rheumatic autoimmune diseases, such as RA and systemic sclerosis, has been proposed.88,89 Switched VSMCs exhibit potential for proliferation, migration, and secretion of extracellular matrix components. However, it has been recently suggested that VSMC proliferation might play a reparative role in plaque formation and atherogenesis, whereas VSMC senescence and death might favor accelerated atherogenesis.90,91 In this context, the observation that methotrexate did not inhibit the intimal proliferation of VSMCs in a murine model of atherosclerosis might be an argument supporting its beneficial role in the atherosclerotic process. 92
Arterial stiffness
In a repeated cross-sectional study of patients with RA treated with either methotrexate, with or without other DMARDs (n = 56), or with other DMARDs but not methotrexate (n = 30), Mangoni et al 76 reported that treatment with methotrexate was associated with a significantly lower pulse wave velocity (PWV: −0.2 m/s, 95% CI: −0.3 to −0.1, P < .001), the gold standard marker of arterial stiffness. Tam et al observed a significant reduction in PWV after 6-month combination treatment with methotrexate and infliximab, but not with methotrexate treatment alone (absolute change −0.78 ± 1.13 m/s vs +0.18 ± 1.59 m/s, P = .044) in 40 patients with RA. There was also a nonsignificant reduction in the augmentation index (AIx), a marker of wave reflection and arterial stiffness, in both treatment groups (−18% and −14%, respectively). 75 Similarly, in 18 patients with RA treated with methotrexate for 12 weeks, Vassilopoulos et al did not observe any significant changes in PWV (8.9 ± 1.9 vs 8.4 ± 2.2 m/s, P = .29) or AIx (30.1 ± 13.8 vs 28.5% ± 12.3%, P = .59). In this study, treatment with the TNF-α inhibitor adalimumab, with or without other DMARDs, significantly reduced PWV (8.2 ± 2.0 vs 7.0 ± 1.8 m/s, P = .00006), but not AIx (27.2 ± 12.9 vs 26.5% ± 9.6%, P = .75). 93
Interpretation of the Available Evidence
Despite the conflicting results reported in a number of studies, the available evidence suggests that methotrexate treatment might exert salutary effects on cardiovascular risk markers, particularly glucose homeostasis, insulin resistance, and metabolic syndrome (Table 1). Furthermore, the proapoptotic effects on endothelial cells reported in some studies might prevent the excessive angiogenesis and neovascularization in synovial and other tissues, typical of RA. However, a number of issues curtail the interpretation of the studies investigating the associations and/or effects of methotrexate treatment on modifiable cardiovascular risk factors:
Human studies were often conducted in patients with different autoimmune disease states, severity, and duration, cardiovascular risk factors and follow-up. These differences might independently impact on the effects of methotrexate, and other DMARDs, on cardiovascular end points.94,95
In the absence of a placebo group, the effects of methotrexate treatment were generally compared with those of combination treatment with methotrexate and other DMARDs or with other DMARDs. However, in several studies, there was no formal statistical analysis of changes vs baseline, between-group differences, or the independent effects of treatment after adjusting for other potential clinical and demographic confounders, including other drugs prescribed for cardiovascular risk management such as statins, antidiabetic, and antihypertensive drugs.
Virtually no study has investigated the potential role of methotrexate polyglutamate concentrations, ie, intracellular exposure, and/or genetic polymorphisms of transporters and target enzymes in mediating the effects of methotrexate on cardiovascular end points.
Given the absence of preliminary pilot data presented, it is not possible to establish whether studies were adequately powered to test the effects of methotrexate on cardiovascular end points.
The concomitant treatment with folic acid, routinely coadministered by many rheumatologists as a result of the known antifolate effects of methotrexate, might have affected several end points. Folic acid treatment per se has been shown to exert salutary effects on blood pressure, endothelial function, and arterial stiffness in patients at high cardiovascular risk.96–98
Summary of the evidence from published studies on the relationship between methotrexate and modifiable cardiovascular risk factors.
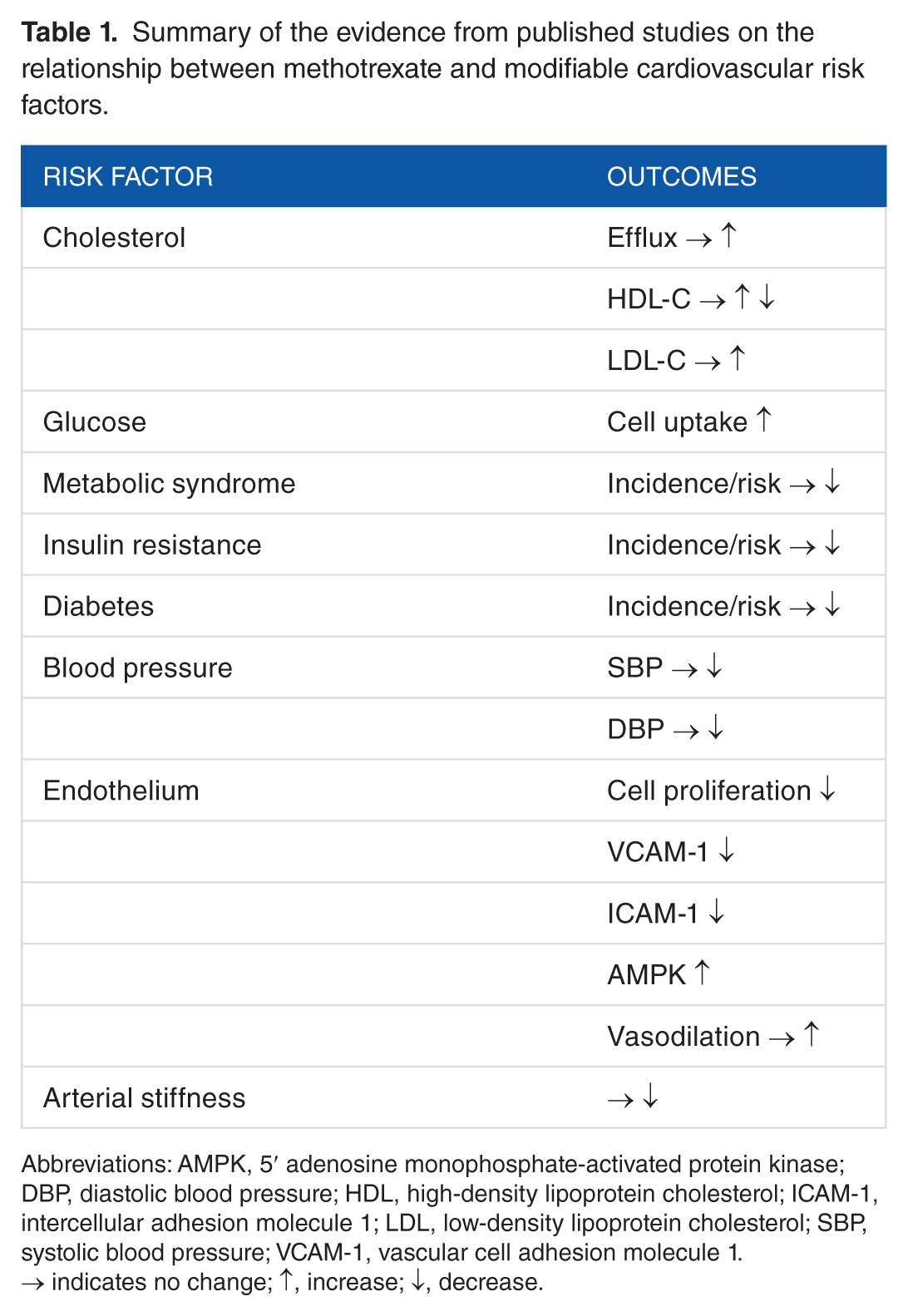
Abbreviations: AMPK, 5′ adenosine monophosphate-activated protein kinase; DBP, diastolic blood pressure; HDL, high-density lipoprotein cholesterol; ICAM-1, intercellular adhesion molecule 1; LDL, low-density lipoprotein cholesterol; SBP, systolic blood pressure; VCAM-1, vascular cell adhesion molecule 1.
→ indicates no change; ↑, increase; ↓, decrease.
The above issues should be accounted for in future studies investigating the potential cardioprotective effects of methotrexate either in animal models or in patients with or without autoimmune disease states.
Possible Mechanisms Involved in the Protective Cardiovascular Effects of Methotrexate
Based on the known pharmacodynamic effects and the recent evidence in experimental models of CVD, a number of mechanisms might account for the potential cardioprotective effects of methotrexate (Figure 3).
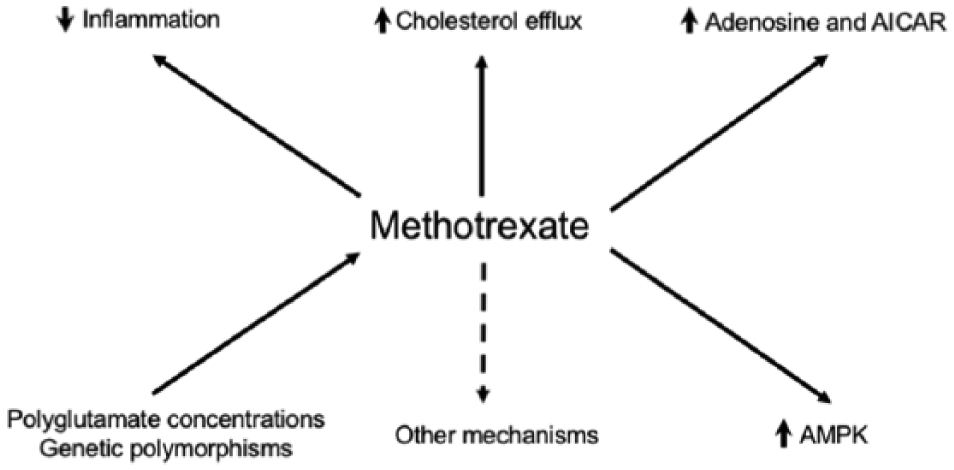
Putative mechanisms mediating the cardioprotective effects of methotrexate. AICAR indicates aminoimidazole carboxamide ribonucleotide; AMPK, 5′ adenosine monophosphate-activated protein kinase.
Reduced inflammation
The anti-inflammatory activity of methotrexate, eg, by targeting the nucleotide-binding domain, leucine-rich family (NLR), pyrin-containing 3 gene (NLRP3) inflammasome, and/or key cytokines, such as TNF-α, interleukin 1 (IL-1), and IL-6, either directly or indirectly, through the activation of adenosine receptors and/or AMPK, might theoretically provide cardioprotective effects.99,100 The latter would include enhanced NO synthesis and endothelium-dependent vasodilatation, reduced arterial stiffness and blood pressure, plaque stabilization, reduced platelet aggregation, and insulin resistance. 101 However, the lower risk of cardiovascular events reported with methotrexate in recent systematic reviews and meta-analyses, when compared with other DMARDs that exert comparable anti-inflammatory effects,37–39 suggests that other mechanisms are likely to be involved.
Cholesterol efflux capacity
Despite the contrasting findings reported in the studies previously described, the potential effects of methotrexate on cholesterol efflux might provide significant protection against atherosclerosis and thrombosis. This mechanism allows the transport of cholesterol out of macrophages in the atherosclerotic plaque onto HDL particles, facilitating plaque size reduction and stability. 102 There is good evidence of an inverse association between cholesterol efflux capacity and vascular damage and atherosclerosis in experimental and human studies in patients with different cardiovascular risk profiles. 103 Furthermore, epidemiologic studies have reported that an increase in cholesterol efflux capacity is associated with a significant reduction in the risk of cardiovascular events during follow-up. 104
Adenosine accumulation
As previously discussed, the methotrexate polyglutamate forms inhibit the catabolism of adenosine, leading to its accumulation (Figures 1 and 2). In addition to its anti-inflammatory effects, adenosine lowers blood pressure by direct vasodilatation, increased NO synthesis, and central nervous system–mediated mechanisms.105–107 Activation of the A2B receptor in endothelial cells reduces the expression of adhesion molecules, such as E-selectin, P-selectin, and ICAM-1, with consequent reduced adhesion of leukocytes and monocytes to the vascular wall, a key step in the atherosclerotic process. 108 Activation of the A2A receptor in macrophages is associated with an increase in anti-inflammatory cytokines, such as IL-10, and a concomitant reduction in pro-inflammatory cytokines, such as TNF-α, and platelet aggregation. 109 Furthermore, the pharmacologic activation of the A2B receptor prevents the formation of atherosclerotic lesions and reduces the plasma concentrations of cholesterol and triglycerides. 110 A proposed mechanism for the effects on plasma lipid concentrations involves the reduced activation of the transcription factor sterol regulatory element–binding protein 1 in the liver.110,111 Experimental and clinical studies have also demonstrated an important role of adenosine receptors in modulating glucose homeostasis, although the pathophysiological and clinical applications of these findings remain to be established. 112
Activation of 5′ AMPK
The indirect activation of AMPK, previously described (Figure 2), might also account for the potential cardioprotective effects of methotrexate. The AMPK activation is known to stimulate cellular glucose uptake, a process mediated by GLUT-1 and GLUT-4 transporters, and glycolysis, through phosphorylation of 2 isoforms of the enzyme 6-phoshofructo-2-kinase: fructose-2,6-biphosphatase, with beneficial effects on glucose homeostasis.113–116 There is also evidence that AMPK activation protects endothelial cells from the deleterious effects of chronic exposure to high concentrations of glucose and fatty acids, which in turn favors oxidative stress, inflammation, and endoplasmic reticulum stress.117,118 The AMPK-mediated protective effects on endothelial cells have also been demonstrated with the oral antidiabetic drug metformin, the angiotensin receptor antagonist telmisartan, and the angiotensin-converting enzyme inhibitor ramipril.119–121 These drugs have been shown to reduce cardiovascular morbidity and/or mortality in large randomized clinical trials.122–124 However, it remains to be established whether AMPK-mediated effects play a significant role in the reported benefits in these studies. Notably, AICAR, another metabolite that accumulates during methotrexate treatment, as a result of the inhibition of ATIC (Figure 1), is also an AMPK activator (Figure 2).26,125 Experimental studies have shown that AICAR prevents leukocyte adhesion, limits the size of atherosclerotic lesions, stimulates NO synthesis, exerts vasodilatory effects through endothelium-dependent and endothelium-independent mechanisms, prevents vessel re-stenosis, and reduces blood pressure in animal models of hypertension.126–132
Directions for Future Research
Further experimental and clinical studies are required to support the potential role of methotrexate in cardiovascular risk management and prevention. Experimental studies should investigate the effects of methotrexate on the NLRP3 inflammasome, TNF-α, IL-1, and IL-6–mediated pathways and on surrogate cardiovascular end points in animal models of dyslipidemia, obesity, diabetes, metabolic syndrome, and hypertension. The investigation of the specific effects on glucose homeostasis would also benefit from studies in isolated adipose and skeletal muscle cells. Furthermore, appropriately powered and controlled human studies, using either placebo or other DMARDs as comparator, should investigate the short-term and long-term effects of methotrexate treatment on different surrogate end points, such as blood pressure, lipid profile, and glucose homeostasis. Comparisons with other DMARDs are required to confirm, or refute, the hypothesis that methotrexate has specific cardioprotective effects and that these effects are, at least in part, independent of inflammation. These studies should investigate the role of methotrexate polyglutamate concentrations, a robust marker of methotrexate intracellular exposure, and genetic polymorphisms of methotrexate transporters and target enzymes as possible factors mediating the possible cardioprotective effects of methotrexate. 30 Furthermore, whenever possible, the effects of concomitant folic acid administration should be taken into account, given the salutary effects exerted by this B-vitamin on several cardiovascular risk factors. Human studies should be conducted in participants with different degrees of cardiovascular risk, and risk factor combinations to identify which specific risk factors and/or patient populations are more likely to benefit from methotrexate treatment. The results of the Cardiovascular Inflammation Reduction Trial (CIRT; clinicaltrials.gov identifier NCT01594333), an ongoing randomized placebo-controlled trial investigating whether treatment with low-dose methotrexate reduces a composite primary end point of myocardial infarction, stroke, and cardiovascular death in 7000 patients with type 2 diabetes or metabolic syndrome and stable coronary artery disease, should be available in 2020. 133 Similarly, the results of the Inflammation and Coronary Endothelial Function in Patients With Coronary Artery Disease (clinicaltrials.org identifier NCT02366091), an ongoing interventional randomized placebo-controlled study investigating the effect of methotrexate, colchicine, and their combination, on coronary endothelial function, and the results of the Effects of Methotrexate Therapy on ST Segment Elevation Myocardial Infarctions trial (TETHYS; clinicaltrials.gov identifier NCT01741558), investigating the role of methotrexate in reducing infarct size when administered within the first 6 hours of admission for ST-elevation myocardial infarction, 134 are eagerly awaited. Therefore, the potential cardiovascular protective effects of methotrexate, and their effect size, are likely to be better elucidated over the next 5 years.
Conclusions
The evidence generated from experimental and human studies mainly conducted over the past decade suggests that methotrexate, an anchor drug in the treatment of RA and other autoimmune disorders, might be repurposed for the management of cardiovascular risk. However, further research is warranted to confirm, or refute, the potential role of methotrexate in cardiovascular protection in appropriately designed studies that investigate surrogate as well as hard clinical end points.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AAM conceived and designed the manuscript and wrote the first draft of the manuscript. AZ, SS, CC, and GLE contributed to the writing of the manuscript. AAM, AZ, SS, CC, and GLE jointly developed the structure and arguments for the paper and made critical revisions and approved final version. All authors reviewed and approved the final manuscript.
Disclosures and Ethics
As a requirement of publication, author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality, and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. The external blind peer reviewers report no conflicts of interest.
