Abstract
Chemotherapy-induced premature ovarian insufficiency (POI) is one of the potential drawbacks of chemotherapy use of particular concern for newly diagnosed premenopausal breast cancer patients. Temporary ovarian suppression obtained pharmacologically with the administration of a gonadotropin-releasing hormone agonist (GnRHa) during chemotherapy has been specifically developed as a method to counteract chemotherapy-induced gonadotoxicity with the main goal of diminishing the risk of POI. In recent years, important clinical evidence has become available on the efficacy and safety of this strategy that should now be considered a standard option for ovarian function preservation in premenopausal breast cancer patients, including women who are not interested in conceiving after treatment or that would not be candidates for fertility preservation strategies because of their age. Nevertheless, in women interested in fertility preservation, this is not an alternative to gamete cryopreservation, which remains as the first option to be offered. In this setting, temporary ovarian suppression with GnRHa during chemotherapy should be also proposed following gamete cryopreservation or to women who have no access, refuse, or have contraindications to surgical fertility preservation techniques. In this article, we present an overview about the role of temporary ovarian suppression with GnRHa during chemotherapy in breast cancer patients by addressing the available clinical evidence with the aim of identifying both the best candidates for the use of this strategy and the still existing gray zones requiring further investigation.
Introduction
Breast cancer is responsible for the largest proportion of cancer diagnosis among young adult women. 1 The burden of breast cancer in young women appears to be on the rise and is of even more importance in certain parts of the world.2–4 A significant proportion of young women with early breast cancer are candidates to receive chemotherapy as part of their treatment considering both the higher incidence of developing aggressive tumor subtypes and the tendency to be diagnosed at a more advanced stage.5,6 In premenopausal women, one of the potential drawbacks of chemotherapy use is represented by its possible gonadal damage. 7 The risk of developing chemotherapy-induced premature ovarian insufficiency (POI) is of particular concern for young patients considering the potential significant negative impact on their quality of life, being associated with menopause-related symptoms, psychosocial issues, health problems, and infertility. 8 In addition, since many women are now choosing to defer motherhood, a considerable proportion of them has not completed their family plans yet at the time of cancer diagnosis, but wish to do it after treatment completion. 9 Recent data have helped to reassure patients and physicians on the safety of conceiving after prior history of breast cancer.10–13 Therefore, discussing fertility and pregnancy-related issues with all newly diagnosed young patients is now considered mandatory before the initiation of anticancer treatments.14–18
For women willing to preserve fertility, in order to have higher chances of conception after completing their treatment, embryo and oocyte cryopreservation are standard strategies and the first options to be proposed.14–18 However, these techniques do no avoid the risk of chemotherapy-induced POI with its associated psychosocial and menopause-related concerns beyond infertility. Temporary ovarian suppression obtained pharmacologically with the administration of a gonadotropin-releasing hormone agonist (GnRHa) during chemotherapy has been specifically developed as a method to counteract chemotherapy-induced gonadotoxicity with the main goal of diminishing the risk of POI. Despite the existence of more than 30 years of research, the role of this strategy has remained highly debated.19–27 In recent years, important clinical evidence has become available on the efficacy and safety of temporary ovarian suppression with GnRHa during chemotherapy and this strategy is now recommended for premenopausal breast cancer patients who are candidates to systemic cytotoxic therapy.16,18,28
In this article, we present an overview about the role of temporary ovarian suppression with GnRHa during chemotherapy in breast cancer patients by addressing the available clinical evidence with the aim of identifying both the best candidates for the use of this strategy and the still existing gray zones requiring further investigation.
Clinical Evidence on the Role of GnRHa During Chemotherapy in Breast Cancer
Following the promising pivotal experimental studies by Ataya and colleagues in the 1980s, 29 the clinical development to demonstrate the protective gonadal effect of administering GnRHa during chemotherapy rapidly started. 30 Notably, most of the evidence on the topic derives from studies conducted in the breast cancer setting, with more limited data for women with other solid tumors or with hematological malignancies. 30
Initially, the potential protective gonadal effect of GnRHa use during chemotherapy was investigated in several observational and single-arm phase II trials. 31 Overall, these studies suggested that the majority (70%-100%) of women treated with GnRHa during systemic cytotoxic therapy did not experience chemotherapy-induced POI, and more than 50 post-treatment pregnancies were described. 31 Following these promising results, several randomized clinical trials were executed and pooled in different meta-analyses.
Randomized clinical trials in breast cancer patients
The largest amount of data from randomized clinical trials on the clinical efficacy of temporary ovarian suppression with GnRHa during chemotherapy as a strategy to preserve ovarian function and potential fertility is available for premenopausal women with breast cancer. Fourteen randomized clinical trials have been performed in this setting (Table 1).32–47
Randomized clinical trials in breast cancer patients assessing the role of temporary ovarian suppression with gonadotropin-releasing hormone agonist during chemotherapy.
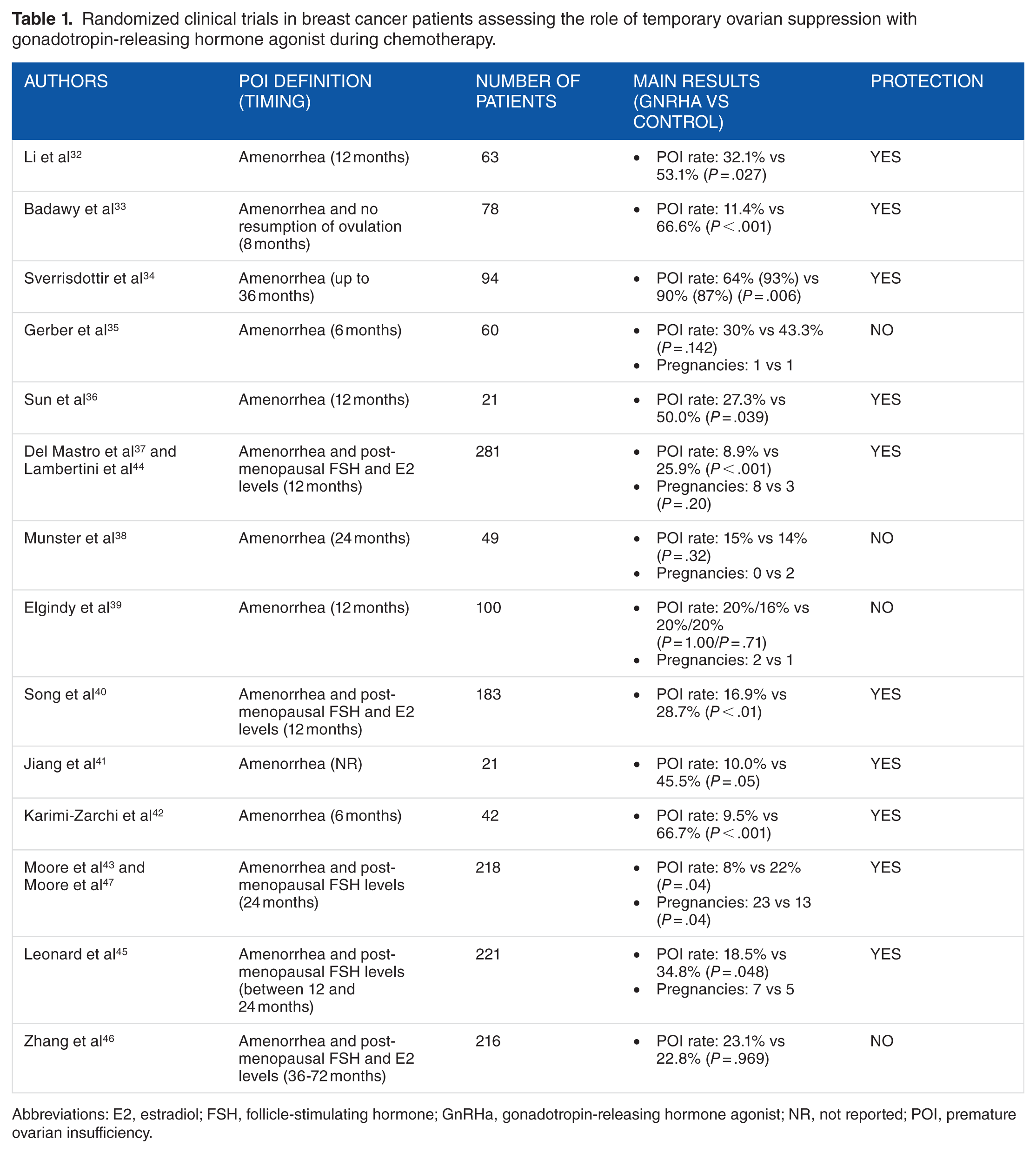
Abbreviations: E2, estradiol; FSH, follicle-stimulating hormone; GnRHa, gonadotropin-releasing hormone agonist; NR, not reported; POI, premature ovarian insufficiency.
Patients included in these studies had a median age close to 40 years and often received anthracycline- and cyclophosphamide-based chemotherapy. Goserelin was the GnRHa administered in the majority of the studies (n = 8), followed by triptorelin (n = 5), and leuprolide acetate (n = 1). Most of these studies were also characterized by the following features: (1) small sample size (ie, less than 100 randomized patients); (2) definition of chemotherapy-induced POI based only on menstrual function after treatment; (3) variable timing for POI evaluation ranging from 6 months up to more than 5 years after chemotherapy completion. Notably, the largest trials providing the highest level of evidence on this regard (PROMISE-GIM6,37,44 POEMS/SWOG S0230,43,47 and Anglo Celtic Group OPTION 45 ) were characterized by a large sample size (more than 200 included patients) and defined chemotherapy-induced POI using a composite endpoint (ie, amenorrhea and postmenopausal hormonal levels) not earlier than 1 year after the end of chemotherapy.
With the exception of 4 trials, all the other studies showed that concurrent use of GnRHa during chemotherapy is associated with a significant reduction in POI risk. Specifically, the 3 largest trials (PROMISE-GIM6,37,44 POEMS/SWOG S0230,43,47 and Anglo Celtic Group OPTION 45 ) reported very similar results: the use of GnRHa during chemotherapy was associated with a significant 15% absolute reduction (from approximately 25% to less than 10%) in POI rates after chemotherapy. The only large trial showing no protective effect was the study by Zhang and colleagues; notably, in this trial, all patients received GnRHa and were randomized between its sequential or simultaneous administration with chemotherapy. 46 Therefore, the results on POI rates after chemotherapy should be considered with cautious also considering that patients in the two treatment arms received a different duration of GnRHa treatment (ranging between 2 and 5 years), the timepoint for the assessment of POI was highly variable, and only amenorrhea data were reported (despite the primary endpoint of POI was defined with a composite endpoint of amenorrhea and postmenopausal hormonal levels). 25
Despite the more consistent results in terms of protective effects in reducing POI risk, the evidence on the fertility preservation potential of temporary ovarian suppression with GnRHa during chemotherapy remains more limited. The POEMS/SWOG S0230 trial was the only study with post-treatment pregnancies as a pre-planned secondary endpoint.43,47 Notably, none of the studies was powered to detect differences in this outcome, wish for conceiving was not an inclusion criteria, and follow-up at the time of reporting chemotherapy-induced POI (ie, their primary endpoint) was relatively short. Therefore, they had limited possibility to assess post-treatment pregnancies considering both the inclusion of premenopausal patients older than 40 years at the time of diagnosis, as well as the fact that an adequate follow-up time to record post-treatment pregnancies is of particular importance for breast cancer patients who are often also candidates to 5-10 years of adjuvant endocrine therapy.16,48–51 Nevertheless, taking into account these limitations, the POEMS/SWOG S0230 trial was the only trial among those reporting on post-treatment pregnancies showing that the use of temporary ovarian suppression with GnRHa during chemotherapy was associated with a significant higher number of patients conceiving after the end of treatment.43,47 More post-treatment pregnancies in patients treated with GnRHa during chemotherapy were also observed in the updated analysis of the PROMISE-GIM6 trial 44 and the Anglo Celtic Group OPTION study, 45 but the absolute numbers were small and no significant difference could be detected. Notably, none of these analyses were adjusted for pregnancy desire (this information was available only for a minority of randomized patients).
When discussing the role of temporary ovarian suppression with GnRHa during systemic cytotoxic therapy in breast cancer patients and particularly in those with estrogen receptor-positive disease, two safety concerns have been raised: a potential antagonism with the concurrent administration of an endocrine agent during chemotherapy and the possible negative prognostic effect of preventing POI occurrence. 52 However, recent evidence has helped to clarify and reassure physicians and patients on these two issues.
Regarding the potential antagonism between endocrine agents and chemotherapy, it should be noted that this has been shown only for tamoxifen in preclinical studies, 53 but it has not been confirmed in clinical trials. 54 No data are available to suggest a potential negative interaction between ovarian suppression and chemotherapy. On the contrary, results from randomized clinical trials did not demonstrate any difference in the prognosis of patients who received chemotherapy with or without concurrent pharmacological or surgical ovarian suppression.55–57 This has recently been confirmed in the TEXT and SOFT trials showing similar survival outcomes with concurrent or sequential administration of GnRHa in premenopausal breast cancer patients with estrogen receptor-positive disease. 58
Regarding the second safety issue, it is known that chemotherapy-induced POI has a strong positive prognostic value in premenopausal women with estrogen receptor-positive breast cancer.59,60 As shown in the SOFT trial, prolonging ovarian suppression for a total duration of 5 years in this setting is beneficial, 61 and it is endorsed by currently available guidelines.16,49,51 This concern can be addressed with the possibility to prolong the administration of GnRHa as part of adjuvant endocrine treatment. The two randomized clinical trials that investigated temporary ovarian suppression with GnRHa during chemotherapy, also in patients with estrogen receptor-positive breast cancer and with sufficient follow-up to assess survival outcomes, confirmed the lack of negative prognostic impact for GnRHa administration during systemic cytotoxic therapy.44,46 However, in both studies, the majority of the patients with estrogen receptor-positive disease received GnRHa as part of their adjuvant endocrine therapy in the case of ovarian function resumption after chemotherapy.44,46 Reassuring data on the safety of administering GnRHa during chemotherapy also in women with estrogen receptor-positive breast cancer have been reported in other large retrospective studies.62–64
Therefore, based on all these findings, it can be concluded that the administration of GnRHa during chemotherapy does not negatively interact with systemic cytotoxic therapy. However, it is preferable to prolong its use up to 5 years after diagnosis as adjuvant endocrine therapy in women with estrogen receptor-positive breast cancer.16,49,51
Meta-analysis of randomized clinical trials including breast cancer patients
Over the past years, the results from the available randomized clinical trials have been summarized in several meta-analyses (Table 2).65–82 Notably, the first meta-analyses included also prospective non-randomized studies. Eight of these meta-analyses were restricted to breast cancer trials while the others also included patients with autoimmune diseases and/or hematological malignancies and/or ovarian cancer.
Meta-analyses including randomized clinical trials in breast cancer patients assessing the role of temporary ovarian suppression with gonadotropin-releasing hormone agonist during chemotherapy.

Abbreviations: CI, confidence interval; GnRHa, gonadotropin-releasing hormone agonist; HL, Hodgkin lymphoma; IRR, incidence rate ratio; NHL, non-Hodgkin lymphoma; OR, odds ratio; POI, premature ovarian insufficiency; RCT, randomized clinical trial; RR, relative risk/risk ratio/rate ratio.
Five out of 16 were RCTs
Three out of 11 were RCTs
Data from breast cancer patients with hormone receptor-negative disease only.
Data from the original publication 37 and the updated analysis 44 of the PROMISE-GIM6 trial were considered twice instead of as from the same study.
Meta-analysis based on individual patient-level data.
A protective effect of temporary ovarian suppression with GnRHa during chemotherapy therapy in reducing POI risk was observed in all but two meta-analyses with a more pronounced and clearer benefit observed in those that included only the trials conducted in the breast cancer setting. The most recent meta-analyses including a higher number of patients and the largest trials showed not only a reduction in the risk of chemotherapy-induced POI, but also a significantly higher rate of post-treatment pregnancies in premenopausal women treated with GnRHa during chemotherapy.
The largest meta-analysis performed to summarize the results of the breast cancer trials showed positive results. 76 When pooling the results from 12 randomized clinical trials including 1231 breast cancer patients, the use of temporary ovarian suppression with GnRHa during chemotherapy was associated with both reduced POI rates (15% absolute reduction [from 34% to 19%]; 64% relative reduction [odds ratio [OR] 0.36, 95% confidence interval [CI] 0.23-0.57]) and increased chances of post-treatment pregnancies (33 vs 19; OR 1.83, 95% CI 1.02-3.28). 76 More recently, a meta-analysis including individual patient-level data from 873 patients randomized in the 5 major breast cancer trials showed similar results. 82 Chemotherapy-induced POI rate was 14.1% in patients who received GnRHa and 30.9% in the control group (adjusted OR 0.38, 95% CI 0.26-0.57). The protective effect of GnRHa administration was observed irrespectively of patients’ age at the time of treatment (including those older than 40 years), estrogen receptor status (including those with estrogen receptor-positive disease), type and duration of chemotherapy. In terms of fertility rates, 37 and 20 patients had at least one post-treatment pregnancy in the GnRHa and control groups, respectively (incidence rate ratio [IRR] 1.83, 95% CI 1.06 to 3.15). In addition, concurrent use of GnRHa during chemotherapy was associated with no impact on disease-free survival (hazard ratio [HR], 1.01; 95% CI 0.72-1.42) and a non-significant trend toward better overall survival (HR, 0.67; 95% CI 0.42-1.06). The safety of this approach was observed irrespectively of tumor estrogen receptor status. 82
When considering the results from the meta-analyses that were not restricted to breast cancer trials, the protective effect of temporary ovarian suppression with GnRHa during chemotherapy was present in the overall population, but could not be observed for women with hematological malignancies. The largest meta-analysis, that also included lymphoma trials, pooled the results from 13 studies and a total of 1208 patients with breast cancer (n = 1099) and hematological malignancies (n = 109). 80 Globally, GnRHa administration was associated with a significantly reduced risk of chemotherapy-induced POI (POI rate: 20% vs 34%; relative risk [RR] 0.60, 95% CI 0.45-0.79), but this benefit did not persist in the subgroup analysis restricted to lymphoma patients (POI rate: 19% vs 32%; RR 0.70, 95% CI 0.20-2.47). More women treated with GnRHa had a post-treatment pregnancy (57 vs 42; RR 1.43, 95% CI 1.01-2.02), but the benefit was not observed in the subgroup of women with hematological malignancies (17 vs 18; RR 1.13, 95% CI 0.66-1.93). 80
The Still Missing Evidence in the Field
Despite all the research efforts conducted over the past 30 years and the evidence on the protective effect of temporary ovarian suppression with GnRHa during chemotherapy shown in the recently published large trials, several gray zones remain in the field both overall and specifically for breast cancer patients.
First, it should be highlighted that the mechanisms by which GnRHa administration can protect ovarian function during chemotherapy are not yet fully elucidated.30,83 None of the biological hypotheses including gonadotropin suppression, decrease of follicular recruitment, reduction in ovarian blood flow or direct effects on the ovaries have been clearly demonstrated by experimental studies. 30 Notably, the majority of the experimental data has been obtained with in vivo studies in rodents (ie, the most studied model in reproductive biology). However, extrapolating the results obtained from these studies to humans remains hazardous. Therefore, additional well-conducted research efforts in the field including species other than rodents are warranted in the next years. 30
Second, despite the consistent results observed in the trials conducted in the breast cancer setting, limited evidence exists on the role of this strategy to counsel women diagnosed with other tumors. The four randomized trials performed in women with hematological malignancies showed no protective effect for temporary ovarian suppression with GnRHa during chemotherapy.84–88 However, it should be highlighted that all these studies had a small sample size with a total of approximately 150 patients when considered all together. Other large retrospective or prospective series have shown a potential protective effect of temporary ovarian suppression with GnRHa during chemotherapy in preserving ovarian function and potential fertility also in women with hematological malignancies.89–93 In women with solid tumors other than breast cancer, only one small randomized trial has assessed the role of temporary ovarian suppression with GnRHa during chemotherapy in 30 young patients with ovarian cancer. 94 The study showed a significant reduction in the risk of chemotherapy-induced POI with the use of GnRHa during chemotherapy, but no information on post-treatment pregnancies was reported. 94
Third, limited evidence exists on the role of administering GnRHa during chemotherapy on patients’ ovarian reserve and on its long-term protective effect. Amenorrhea alone, as used in the majority of the randomized clinical trials that investigated this strategy, is not an optimal surrogate for defining POI development. 95 In fact, it has been shown that the use of chemotherapy can have a negative impact on a woman ovarian reserve leading to infertility and early menopause beyond the risk of acute POI. 96 Irrespectively of the primary endpoint definition of chemotherapy-induced POI used in the different randomized clinical trials conducted in this setting, none of them have reported age at menopause for patients who received chemotherapy with or without concurrent GnRHa. Long-term follow-up from the currently available randomized trials would be crucial to capture this important information. In addition, there is paucity of data on the actual protective effect of GnRHa treatment on patients’ ovarian reserve. A prospective study including 88 premenopausal breast cancer patients has recently shown that antral follicle count recovered faster and to a greater degree among women who received GnRHa during chemotherapy. 97 Nevertheless, so far, data on the dynamic of anti-mullerian hormone (AMH, a promising biomarker of chemotherapy-induced gonadal damage98–101) during and after treatment are limited and mostly negative. Specifically, among the breast cancer trials with available information on patients’ hormonal profile, only a minority evaluated post-treatment AMH, which did not differ between women who received chemotherapy alone or with concurrent administration of GnRHa.35,39,45 In women with hematological malignancies, the only exception was the trial by Demeestere and colleagues that showed higher post-treatment AMH levels in patients who received GnRHa during chemotherapy at 1-year follow-up (P = .040), 87 but no difference after 5 years (P = .520). 88 Ongoing prospective studies are currently investigating the hormonal profile including AMH in women receiving temporary ovarian suppression with GnRHa during chemotherapy.102–104
Fourth, particularly important for breast cancer patients, there is lack of data on the efficacy and safety of temporary ovarian suppression with GnRHa during chemotherapy in patients with hereditary cancer syndromes, such as those with pathogenic germline BRCA mutations. 105 To our knowledge, the only piece of information on this regard derives from the case series by Wong and colleagues in which out of 4 BRCA-mutated breast cancer patients receiving GnRHa during chemotherapy, 3 resumed menstrual function before undergoing prophylactic gynecological surgery. 106 Preclinical evidence suggests that the presence of BRCA mutations can be associated with decreased ovarian reserve as well as increased risk of fertility-related problems and primary ovarian insufficiency. 105 In breast cancer patients, although not confirmed by other studies,107–109 some of the available data suggest the possible presence of reduced baseline ovarian reserve in BRCA-mutated patients with subsequent potential higher risk of developing chemotherapy-induced POI and reduced efficacy of fertility preservation procedures.110–112 To acquire evidence on the protective effect of GnRHa administration during chemotherapy in this setting within the currently available randomized clinical trials would be of particular importance. However, these women are candidates to receive prophylactic gynecological surgery before the age of 40-45 years due to the significant risk of developing ovarian cancer. 113 Therefore, temporary ovarian suppression with GnRHa during chemotherapy is not an optimal strategy in this setting, and it should be offered only to women diagnosed years before the age of recommended prophylactic gynecological surgery. 105
Conclusions
More than 30 years have passed since the publication of the first preclinical data suggesting a possible protective role of administering GnRHa during chemotherapy in order to preserve ovarian function and potential fertility in premenopausal cancer patients through systemic cytotoxic therapy. 29 In the last years, results from the largest randomized clinical trials conducted to assess the efficacy and safety of this strategy in premenopausal women with newly diagnosed early breast cancer have supported its protective role.43–45,76,82 Therefore, recent guidelines support the use of temporary ovarian suppression with GnRHa during chemotherapy in this setting.16,18,28 Indeed, this strategy is now available and covered in many countries.28,114,115
Premenopausal patients interested in reducing the risk of developing chemotherapy-induced POI are the best candidates for this strategy irrespectively of their pregnancy desire and their age at diagnosis. In other words, temporary ovarian suppression with GnRHa during chemotherapy should be considered a standard strategy for ovarian function preservation in breast cancer patients. This strategy will impact on reducing the risk of menopausal signs and symptoms including loss of bone density in the long-term; these are issues of crucial importance also in premenopausal patients including women not interested in conceiving after treatment or not candidates to fertility preservation strategies because they are older than 40 years at diagnosis.
On the other hand, the role of this option as a strategy for fertility preservation is to be considered with more caution. In fact, although the most recent studies showed a significantly higher number of post-treatment pregnancies in the group of women who received GnRHa during chemotherapy,43,76,82 more limited data are available as compared to those on POI so that no strong conclusions on this endpoint can be drawn to date. Therefore, in these patients, gamete cryopreservation remains the first option to be discussed, but temporary ovarian suppression with GnRHa during chemotherapy should be also proposed following this strategy or to women who have no access, refuse, or have contraindications to surgical fertility preservation techniques. For women who receive gamete cryopreservation followed by temporary ovarian suppression with GnRHa during chemotherapy, the type of agent to be used and the best timing for GnRHa administration (considering its potential use as trigger of follicular maturation instead of chorionic gonadotropin or short-acting GnRHa) 82 are important to be clarified in the coming years.
Footnotes
Acknowledgements
ML acknowledges the support from the European Society for Medical Oncology (ESMO) for a Translational Research Fellowship at the Institut Jules Bordet in Brussels (Belgium). FR acknowledges the support from Les Amis de Bordet for a post-doctoral fellowship at the Institut Jules Bordet in Brussels (Belgium).
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
ML served as a consultant for Teva and received honoraria from Theramex outside the submitted work. All remaining authors declared no conflicts of interest.
Author Contributions
All the authors participated in the writing of this article.
