Abstract
Background:
Adverse drug events (ADEs) are a burden to the healthcare system. Preventable ADEs, which was ADEs due to medication errors, could be reduced if medication errors can be prevent or ameliorate.
Objective:
We investigated the burden of preventable ADEs on the length of hospital stay (LOS) and costs, and estimated the national burden of preventable ADEs in pediatric inpatients in Japan.
Methods:
We analyzed data from the Japan Adverse Drug Events (JADE) study on pediatric patients and estimated the incidence of preventable ADEs and associated extended LOS. Costs attributable to extended LOS by preventable ADEs were calculated using a national statistics database and we calculated the effect of preventable ADEs on national cost excess.
Results:
We included 907 patients with 7377 patient-days. Among them, 31 patients (3.4%) experienced preventable ADEs during hospitalization. Preventable ADEs significantly increased the LOS by 14.1 days, adjusting for gender, age, ward, resident physician, surgery during hospitalization, cancer, and severe malformation at birth. The individual cost due to the extended LOS of 14.1 days was estimated as USD 8258. We calculated the annual extra expense for preventable ADEs in Japan as USD 329 676 760. Sensitivity analyses, considering the incidence of preventable ADEs and the length of hospital stay, showed that the expected range of annual extra expense for preventable ADEs in Japan is between USD 141 468 968 and 588 450 708.
Conclusion:
Preventable ADEs caused longer hospitalization and considerable extra healthcare costs in pediatric inpatients. Our results would encourage further efforts to prevent and ameliorate preventable ADEs.
Keywords
Introduction
Medications are the most frequent cause of injury due to medical care and such injuries due to medication use, which may or may not result from medication errors (MEs), are called adverse drug events (ADEs)1-5 while an adverse drug reaction (ADR), a related term, is defined as a response to a medicinal product which is noxious and unintended by the European Medicines Agency. 6 The World Health Organization announced the Third Global Patient Safety Challenge as “Medication Without Harm” at the Second Global Summit of Health Ministers on Patient Safety in 2017. 7 ADEs represent a major public health issue1,8 because they are associated with an increase in morbidity and mortality1,8-12 and cause substantial costs.13-15 Specifically, preventable ADEs, defined as ADEs due to MEs, are important because their impact on patient health and healthcare costs can be reduced if an effective strategy is developed to ameliorate and prevent MEs. 16
In pediatric settings, the risk of MEs is higher than that in adult settings because pediatric patients often have limited reserves with respect to metabolism and fluid volume and they cannot identify and describe the MEs. Previous reports have shown that pediatric inpatients are vulnerable to ADEs and MEs.17-22 Thus, the impact of preventable ADEs is larger in pediatric patients than that in adults. Several reports have shown that preventable ADEs extend the length of hospital stay (LOS) in pediatric intensive care units and increase the direct cost to emergency departments23-26; however, little data are available that identify the detailed epidemiology of preventable ADEs and quantify the additional resource utilization associated with preventable ADEs among general pediatric inpatients.18-22,27 Furthermore, most reports are from western countries and data that reveal the impact of preventable ADEs in Asian countries are scarce.23-26
Thus, we investigated the burden of preventable ADEs on LOS and costs in our cohort of pediatric inpatients in Japan. Then, we estimated the national burden of preventable ADEs based on the results in our cohort.
Materials and Methods
Study design and patient population
The primary outcome of the study was the burden of preventable ADEs, which was calculated as both LOS and cost. The LOS was estimated using the cohort of the JADE study for pediatric patients, a historical cohort study that was carried out in pediatric inpatient settings in 2 tertiary care teaching hospitals in Japan. Cost was calculated using public data from the open resource of the Statistics of Medical Care Activities in Public Health Insurance conducted by the Ministry of Health, Labour and Welfare (MHLW).28,29
The details of the JADE study for pediatric patients have been described elsewhere. 22 Briefly, patients are all admitted inpatients regardless of ADEs or not; patients aged ⩽15 years old who were admitted to any wards including neonatal intensive care units (NICUs), pediatric ICUs (PICUs), and ICUs and those aged >15 years old who were admitted to pediatric wards at 2 teaching hospitals in Japan during the 3 month study period in 2009 were included. The total number of beds in these 2 hospitals is 1754, and 152 beds among them were for pediatric inpatients.
This sub-study was limited to patients aged <15 years who were admitted to pediatric wards, emergent care units, or adult wards. We excluded inpatients admitted to NICUs, PICUs, and ICUs because these inpatients, with intensive care, may be different from those who were hospitalized in general pediatric wards in terms of disease severity, treatment, original background, and cost of medical treatment.
Then, we divided our study population above into 2 groups: patients who had preventable ADEs during their hospitalization (patients with preventable ADEs) and patients who did not have preventable ADEs during their hospitalization (patients without preventable ADEs) to estimate the burden of preventable ADEs.
The institutional review boards of the 2 participating hospitals approved the study. Because all data were obtained as part of routine daily practice, informed consent was waived by the institutional review boards.
Data from the cohort study
In the JADE study for pediatric patients, an ADE was defined as any injury due to medication use, which may or may not result from MEs such as overdose or misuse. 3 MEs were defined as any deviation from the appropriate use of a medication at any step in the medication use process including the ordering, transcribing, dispensing, administering, and monitoring. 3 If ADEs occurred that were related to such MEs, these ADEs were considered as preventable ADEs. For example, a rash in a patient receiving ampicillin without another obvious cause was considered an ADE. If a physician ordered ampicillin in a patient with known penicillin allergy, this prescription considered as a ME and this rash considered as a preventable ADE.
We categorized patients’ age into 5 age group: neonates (<1 month), infants (1 month ⩽ and <1 year), preschoolers (1 year ⩽ and <7 years), school-age (7 years ⩽ and <13 years), and teenagers (13 years ⩽ and <15 years).
We also categorized patients depending on their background: patients who had a major malformation such as cardiac malformation, meningocele, or malformation due to chromosomal abnormality were considered patients with severe malformation; patients who underwent any surgery during their hospital stay were considered patients who had surgery; and patients who had a history of cancer or those who were treated with antitumor agents were considered patients with cancer.
The JADE study for pediatric patients used a validated methodology that has been reported elsewhere. 3 Briefly, the pediatrics-trained reviewers consisted of a board-certified pediatrician, pediatric nurses, and a dietitian, and the pediatrician trained all reviewers in a standard manner. 3 These trained reviewers screened all medical charts, laboratory results, incident reports, and prescription queries by pharmacists at each participating hospital and collected potential ADEs, MEs, and preventable ADEs, and determined their onset dates, as well. The onset days of preventable ADEs was calculated by counting the days from admission date to the date when preventable ADEs occurred among patients with preventable ADEs. When multiple preventable ADEs occurred on 1 patient, we used the first one to assess the severity and to calculate the onset days of preventable ADEs.
Patient details, such as symptoms, drug names, characteristics, and related data were also collected. Two independent physicians assessed this data and determined preventable ADEs following to the validated methodology. 3 If the determination of preventable ADEs was different between physicians, they reached consensus through discussion.
Cost data
Extra costs due to preventable ADEs were estimated based on the extended LOS attributable to preventable ADEs. The national estimates of LOS attributable to preventable ADEs and the costs associated with extended LOS were calculated using public data of the estimated number of new admissions 28 and hospital medical expenditure 29 of pediatric inpatients from the open resource of the Statistics of Medical Care Activities in Public Health Insurance conducted by the MHLW.
To calculate total extended LOS and its range in our study population, we used the estimates and the 95% confidence interval (CI) shown by the regression models adjusting for the covariates such as gender, age, ward, resident physician, the presence of surgery during hospitalization, cancer, and severe malformation at birth.
L: Total extended LOS (days) in our study population = number of patients with preventable ADEs in our study population × estimated extended LOS (days) by the adjusted regression model.
Then, we calculated the total extra pediatric medical expenditure in our study population by multiplying the public data of hospital medical expenditure for pediatric patients 29 by L.
To calculate the total extended LOS and its range in Japan, we used the estimates and the 95% CI shown by the regression models adjusting for the covariates above in the same manner as for L.
a: Total number of admissions in Japan = the estimated number of new admissions per day 28 × 365 days.
b: Number of patients with preventable ADEs in Japan = number of patients with preventable ADEs in our study population/total number of admissions in our study population × a.
LL: Total extended LOS (days) in Japan = b × estimated extended LOS (days) shown in the adjusted regression model.
Then, we calculated the total extra medical expenditure in Japan by multiplying the public data of daily hospital medical expenditure for pediatric patients 29 by LL.
All monetary values are expressed in Japanese yen (JPY) and United States Dollar (USD) (in September 2020, 105 JPY was equivalent to USD 1.00).
Statistical analyses
Descriptive statistics are shown as mean with standard deviations (SD) or median with interquartile ranges for continuous variables and as numbers and percentages for categorical variables. We used a t-test or a Mann–Whitney test when the data were continuous and a chi-square test or Fisher’s exact test when the data were categorical to compare patient characteristics and LOS between patients with and without preventable ADEs. When a patient was transferred from 1 ward to another, these consecutive admissions were merged into 1 hospitalization.
We used a linear regression model to assess the effects of preventable ADEs on the LOS between patients with and without preventable ADEs with 95% CIs. The model was adjusted for gender, age category (neonates, infants, preschoolers, school age children, and teenagers), ward category (pediatric ward, emergent care unit, and adult ward), doctor in charge (resident physician or not), presence of surgery during hospitalization, cancer, and severe malformation at birth. Considering that patients with shorter LOS could be less likely to have preventable ADEs, we conducted the sensitivity analysis with the same linear regression model excluding the patients whose LOS were less than 2 days.
Another sensitivity analysis was also done for showing the expected range of the pediatric medical expenses for preventable ADEs by using 95% CIs of the number of patients with preventable ADEs and the length of hospital stays due to preventable ADEs.
All analyses were performed using JMP Pro 13.1.0 (SAS Institute Inc., Cary, North Carolina, USA) software. Two-tailed comparisons were used and P-values <.05 were considered statistically significant.
Results
Patient characteristics
From 1189 admissions in the JADE study for pediatric patients, we included 907 patients in this study (Figure 1). Among these, 511 (56%) were male and the median age was 2 (Interquartile range 0-7) years. In total, 50% of the patients were preschoolers. Patients with cancer accounted for 2.4% and those who had severe malformation at birth accounted for 16% of patients. Twelve percent of patients were mainly cared for by resident physicians (Table 1).

Flowchart of patients.
Patient backgrounds and comparison between patients with preventable adverse drug events and without.
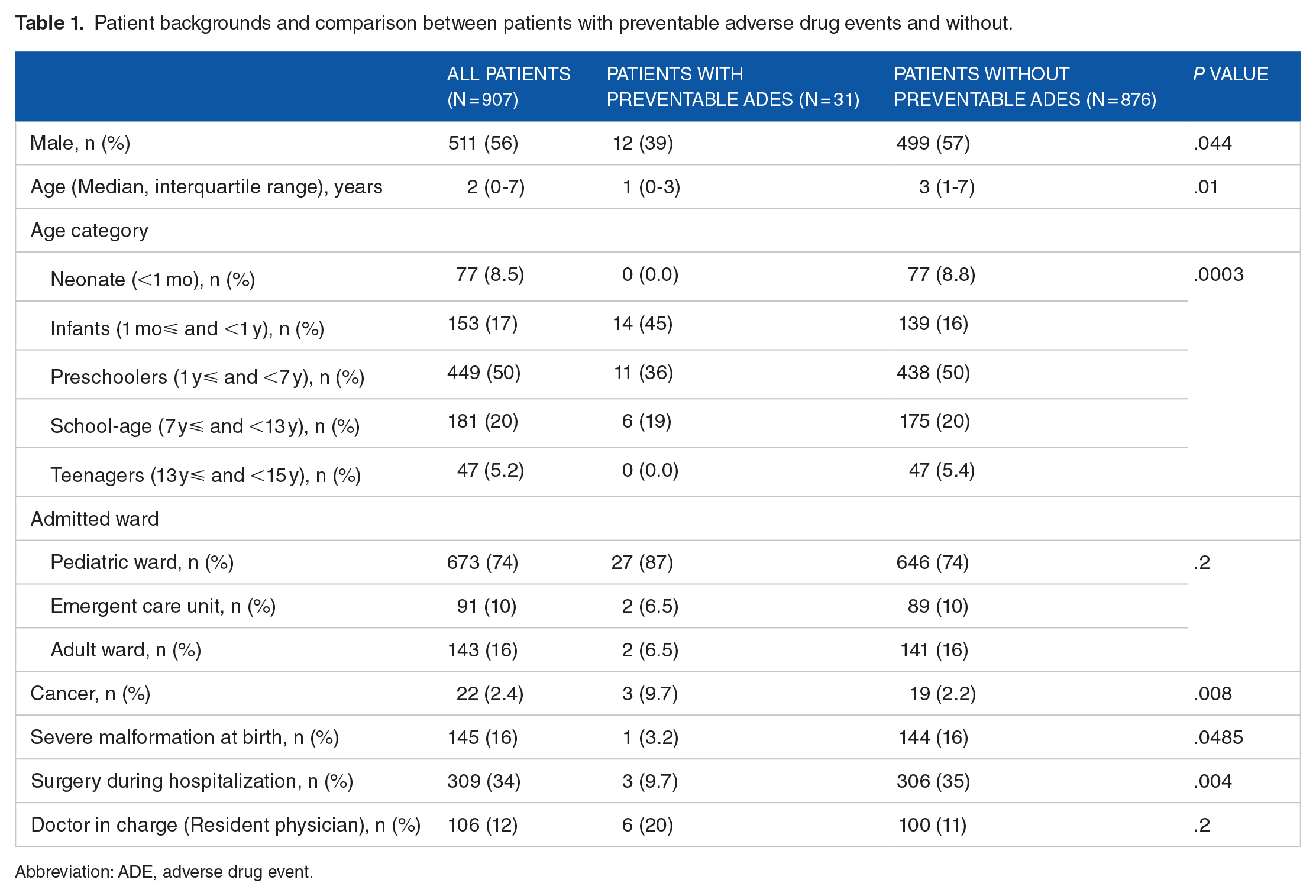
Abbreviation: ADE, adverse drug event.
Preventable ADEs
Among the 907 patients, 31 (95% CI 20-42) patients had at least 1 preventable ADE, whereas 876 patients had no preventable ADEs during their hospitalization. The rate of preventable ADEs was 3.4 (95% CI 2.2-4.6) per 100 admissions. Infants and patients with cancer were more likely to have preventable ADEs, whereas patients with severe malformation and patients who had surgery during their hospitalization were less likely to have preventable ADEs, than the rest of the study population (Table 1). Regarding the severity of these 31 preventable ADEs, 1 was fatal or life-threatening, 3 are serious, and 27 were significant, and the median onset days of these preventable ADEs after admission was 3 days (Interquartile days; 2-4).
The influence of preventable ADEs on LOS
The mean LOS of patients with preventable ADEs was 24.3 (95% CI 7.3-41.3) days, whereas the mean LOS of those without preventable ADEs was 7.6 (95% CI 6.7-8.4). A linear regression model for the LOS showed that preventable ADEs significantly extended the LOS by 14.1 (95% CI 9.4-18.7) days, even after adjusting for gender, age category, ward category, resident physician as doctor in charge, surgery during hospitalization, history of cancer, and severe malformation at birth (Table 2). Sensitivity analysis with 867 patients (LOS ⩾ 2 days) showed that the preventable ADEs extended the LOS by 14.0 (9.2-18.8) days, which is consistent with the original analysis.
Influence of preventable adverse drug events on length of hospital stay.

Abbreviations: ADE, adverse drug event; LOS, length of stay.
The burden of extended LOS associated with preventable ADEs on pediatric medical expenses
As 31 patients had at least one preventable ADE and preventable ADEs in hospitalized patients extended their LOS by a mean of 14.1 (95% CI 9.4-18.7) days (Table 2), the estimate of the total extended LOS was 437.1 days among our study population of 907 patients over 3 months. The average cost of a hospitalized pediatric patient per day was JPY 61 497 (USD 585.69) (Table 3); thus, we estimated that the individual extra cost of extended hospitalization of a patient with preventable ADEs was JPY 867 108 (95% CI JPY 578 072-1 149 994), which is USD 8258.23 (95% CI USD 5505.49-10952.40). The total extra cost of extended hospitalization of patients with preventable ADEs over the study period in our study population was JPY 26 880 339 (95% CI JPY 17 920 226-35 649 811), which is USD 256005.10 (95% CI USD 170670.07-339524.49) (Table 4).
Government statistical data in pediatric care.

Abbreviations: JPY, Japanese yen (Japanese currency); USD: United States Dollar (1 USD = 105 JPY).
Estimation of total extra pediatric medical expenditure due to preventable adverse drug events on admissions.

Abbreviations: ADE, adverse drug event; JPY, Japanese yen (Japanese currency); LOS, length of hospital stay; USD, United States Dollar (1 USD = 105 JPY).
n = number of patients.
Because the estimated total number of new admissions of pediatric patients to hospital per day in Japan was 3200 patients (Table 3), we estimated the annual extra pediatric medical expenses for preventable ADEs in Japan as JPY 34 615 806 492 (95% CI JPY 23 077 204 328-45 908 906 482), which is USD 329676759.91 (95% CI USD 219784506.61-437230880.16) (Table 4). Figure 2 shows the estimate of extra pediatric medical expenses (USD) for preventable ADEs in Japan and its expected ranges calculated using 95% CIs of the number of patients with preventable ADEs and length of hospital stays due to preventable ADEs.
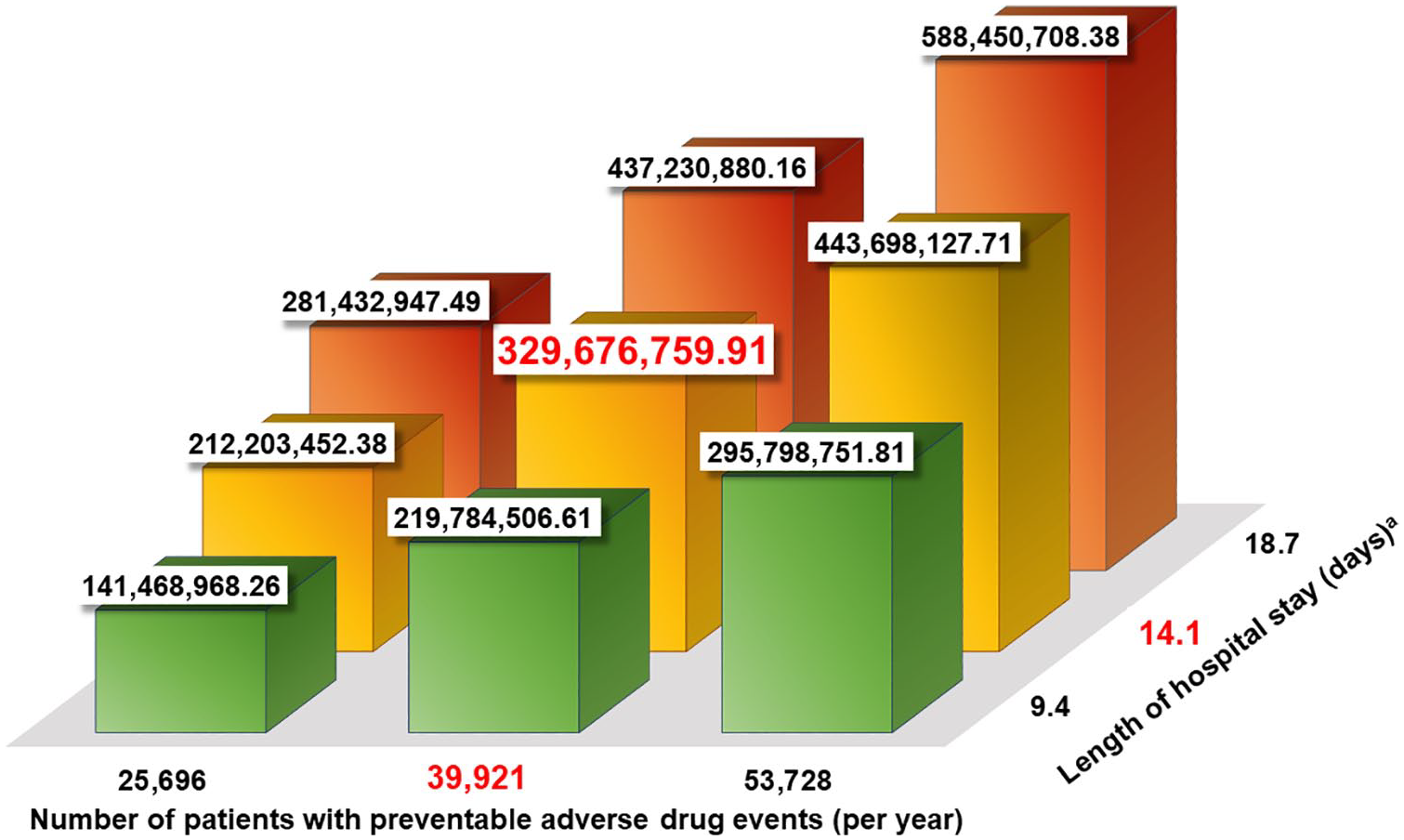
The estimate and ranges of extra pediatric medical expenses (USD) for preventable ADEs in Japan.
Discussion
Summary of main findings
We showed that 3.4% of our study population experienced at least one preventable ADE during their hospitalization and preventable ADEs significantly increased the LOS by 14.1 days with adjusting for gender, age, ward, the presence of surgery, cancer, severe malformation at birth, and resident physician. The total cost of extended hospitalization associated with preventable ADEs in our study population was estimated to be USD 256.005. USD 141 million to USD 588 million was the estimated additional cost nationwide per year, calculated by implementing our results with national statistics. We also found that infants and patients with cancer were high risk for preventable ADEs and that 13% of the preventable ADEs resulted in the most severe or relatively severe harm. These results suggest that the intensive monitoring focusing on these high risk groups and introducing alert systems for early detection of ADEs on electronic medical records might be effective to reduce or ameliorate preventable ADEs and decrease the extra cost due to preventable ADEs.
Comparisons and implications
This is the first study that identified that preventable ADEs extended LOS in pediatric inpatients in general wards, and it seems to be consistent with previous reports in ICU settings, which have shown that adverse drug reactions increase the mean ICU LOS 3.8-fold (2.4 days vs 15.9 days) in pediatric patients 30 and that medication prescribing error increases the median PICU LOS (7 days vs 3 days) and median length of mechanical ventilation (31 hours vs 2.7 hours). 23
Considering the impact of preventable ADEs on LOS in pediatric inpatients, the LOS of 14.1 days shown in this study is longer than the LOS of 6.1 days in adult inpatients. 16 Though the higher risk of MEs in pediatric inpatients is less likely to be associated with actual harm in pediatric inpatients than in adults, 18 this study shows that the impact of preventable ADEs on LOS is greater in pediatric patients than in adults once MEs result in actual harms.
Our estimates of the total extra direct cost of preventable ADEs was USD 141 million to USD 588 million, and our point estimate of the individual extra cost of extended hospitalization of a patient with preventable ADEs was USD 8258. Previous studies which reported the cost related to preventable ADEs were mostly from Western countries and data from other regions are scarce.24-26 For example, the study in New Zealand showed that the cost related to preventable ADEs which caused hospital admission or prolonged hospital stay in pediatric setting is USD 6722 per patient (1 NZD = USD 0.68 as of September 2020). 26 Other studies reported that the direct cost related to preventable drug-related problems (DRPs) in 187 emergency department attendances in Australia is about USD 174 per a patient (1 AUD = USD 0.73 as of September 2020), 24 and the total direct cost associated with preventable DRPs of 38 hospital admissions in Australia is USD 2089 per a patient (1 GBP = USD 1.29 as of September 2020). 25 To implement the evidences to improve medication safety in various situations in each area or countries, aggregating evidences from various settings is crucial although it is difficult to compare the results due to the diversity in the methods used to calculate the costs, definition of preventable ADEs, and systems of medical insurance.
The total annual medical care cost in inpatient settings was estimated to be JPY 16.21 trillion (USD 154.40 billion) in 2017 by the MHLW in Japan. 31 JPY 654.2 billion (USD 6.2 billion; 4.0%) was estimated as the cost of pediatric inpatients (<15 years old). 31 If 10% of preventable ADEs could be reduced by computerized physician order entry 32 and clinical pharmacist interventions, 33 the reduction in cost from the annual medical care expenditure may be USD 33.0 million, which accounts for 0.5% of the annual medical care expenditure for pediatric inpatients; thus, these strategies may have a large impact on medical care expenditure.
The effects and importance of vaccinations in terms of cost, as well as public health, is well known worldwide. Previous reports have estimated that the development of the pneumococcal conjugate and Hepatitis B vaccines can reduce the annual medical care cost in Japan by JPY 2.9 billion (USD 28 million).34,35 The impact of reducing 10% of preventable ADEs on medical care expenditure is similar, or even larger, than that of these vaccines.
Thus, ensuring medication safety for pediatric inpatients and further efforts for reducing preventable ADEs is one of the most important issues from the perspectives of cost effectiveness and public health. Therefore, we believe that further study into how to prevent preventable ADEs in pediatric inpatients is important.
Limitations
There were several limitations to our study. First, we calculated the estimates of extra hospitalization costs by approximating national medical expenses, which did not include direct non-medical costs (such as transportation expenses) or indirect costs (such as extra treatments for ADEs and loss of working opportunities for caregivers). Therefore, our results may provide conservative cost estimates. Second, some preventable ADEs may not have been noted in the charts and may thus have not been detected. Therefore, our estimate of preventable ADEs might be lower bound and it would make the cost estimates lower bound as well. Third, the JADE study for pediatric patients was originally conducted in 2009, which might influence on our cost estimates. However, the data from the JADE study for pediatrics is still the most reliable data of preventable ADEs in pediatric settings in Japan. Fourth one interpretation of extended hospitalization associated with preventable ADEs could be that patients with a longer hospitalization were more likely to experience a preventable ADE. However, the median onset days of preventable ADEs after admission was 3 and shorter than the LOS of the patients without preventable ADEs. It implies that preventable ADEs increased LOS rather than that longer LOS resulted in the occurrence of preventable ADEs during the hospitalization.
Conclusion
We showed, for the first time to the best of our knowledge, that preventable ADEs caused extended hospitalization and considerable extra healthcare costs in pediatric inpatients in Japan with sensitivity analyses, considering the incidence of preventable ADEs and length of hospital stay. Our results should encourage further efforts to prevent and ameliorate preventable ADEs.
Footnotes
Acknowledgements
The JADE study for pediatric inpatients was conducted by the following investigators: Tsukasa Nakamura, Eriko Waki, Sanae Ohashi, Masako Shibuya, Takashi Kawakubo, Kaori Yamamoto, Makiko Ohtorii, Ai Mizutani, Mika Sakai, Yuko Ueo, Hitomi Ito, Chikako Senou, Megumi Hara, Tomohiro Sonoyama, and Taro Saito.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Education, Culture, Sports, Science and Technology (MEXT) of Japan [grant numbers 15K08862, 24689027, 22790494, 20K10364, 18H03032]; and the Pfizer Health Research Foundation.
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Hitoshi Iwasaki is an employee of Novartis Pharma KK. Mio Sakuma, Hiroyuki Ida, and Takeshi Morimoto declare that there is no conflict of interest.
Author contributions
Hitoshi Iwasaki performed the statistical analyses and drafted the manuscript; Mio Sakuma contributed to the conception, design, and funding acquisition of this study, performed whole project administration, collected the data, performed the statistical analyses, and drafted the manuscript; Hiroyuki Ida critically reviewed the manuscript and supervised the whole study process; Takeshi Morimoto contributed to the conception, design, and funding acquisition of this study, critically reviewed the manuscript and supervised the whole study process. All authors read and approved the final manuscript.
