Abstract
Extrahepatic bile duct carcinoma is a potentially malignant gastrointestinal lesion. Cancer cells spread via the lymphatic system to regional lymph nodes and help in tumor progression. However, there are no reports on the prognostic impact of extracapsular lymph node invasion and myofibroblastic activity in this cancer. Hence, we classified the histopathologic patterns of lymph nodes into 2 patterns: extracapsular lymph node invasion or not. Based on this, we investigated 32 cases of extrahepatic bile duct cancer with lymph node metastasis and classified 21 cases as positive and 11 cases as negative. The extracapsular lymph node invasion cases were associated with poor disease-free survival and overall survival. The myofibroblast density of the metastatic foci was significantly higher in the extracapsular lymph node invasion cases. This is the first study to demonstrate that extracapsular lymph node invasion cases were associated with poor prognosis and that the myofibroblast distribution contributed to malignancy.
Introduction
Extrahepatic bile duct carcinoma is an epithelial cancer that originates from the bile ducts, has features of cholangiocyte differentiation, and represents one of the most potentially malignant gastrointestinal lesions. Chemotherapy and radiation therapy are not fully effective, and surgical treatment is the only curative therapy for this type of carcinoma. 1 The spread of cancer cells via the lymphatic system to regional lymph nodes is an important factor in tumor progression. In a recent study, the median disease-specific survival rate after surgery in patients with lymph node metastasis was lower than that of patients without lymph node metastasis. 2 However, there are no reports on the prognostic impact of extracapsular lymph node invasion and myofibroblastic activity in extrahepatic bile duct carcinoma. In this study, the histopathologic patterns of lymph nodes were classified into 2 patterns: extracapsular lymph node invasion, whereby the metastatic carcinoma cells had invaded the lymph node capsule, or not. We also focused on the clinicopathological significance of the extracapsular lymph node invasion in extrahepatic bile duct carcinoma and evaluated the myofibroblast density of the lymph metastatic foci and primary cancerous lesion. Recently, the association between cancer and myofibroblasts in the tumor microenvironment has received attention. 3 Myofibroblasts are known as a component in the granulation tissue healing of wounds and as cancer stromal cells. 4 In addition, myofibroblasts act as a representative extracellular matrix (ECM) that induces cancer proliferation, invasion activities, and metastasis by stimulating angiogenesis and lymph angiogenesis.5–7 Hence, we analyzed the association between extracapsular lymph node invasion and the density of myofibroblasts using imaging analysis.
Materials and Methods
Patients
We investigated 101 consecutive patients with bile duct carcinoma who were surgically treated between January 2004 and December 2011 at the Hirosaki University Hospital. We obtained informed consent from each patient to use their clinical records and pathology specimens. Of the 101 patients enrolled, 36 patients had lymph node metastasis and 4 were excluded because they had no regional lymph node involvement; 32 cases were investigated in this study.
Survival data were obtained from hospital medical charts and the median observation period was 19 months. The series consisted of 27 men and 5 women with a median age of 71.5 (range: 52-80) years. About 10 carcinomas were perihilar and 22 were located in the distal bile duct. Curative resection and regional lymph node dissection were dependent on the location of the primary tumors. A pancreaticoduodenectomy or pylorus-preserving pancreaticoduodenectomy was performed in 22 patients and a combined hepatectomy with a bile duct resection was performed in 10 cases. The study protocol was approved by the Committee of Ethics at Hirosaki University School of Medicine.
Pathological analysis
All surgically resected specimens were routinely fixed with 10% formalin, embedded in paraffin, and stained with hematoxylin-eosin for pathological evaluation. Metastatic carcinoma cells in the lymph node capsule are shown in Figure 1A and B, and metastatic carcinoma cells invading the lymph node capsule and spreading into surrounding adipose tissue can be seen in Figure 1C and D. To compare the presence of extracapsular lymph node invasion with the primary lesion, histologic features of the primary lesion were assessed in the largest cross-sectional tumor section as follows: depth of the invasion (T-grade), histologic type, lymphovascular invasion, perineural invasion, mode of the infiltration pattern (INF) of tumor, and lymph nodal metastasis. The degree of lymphatic, vessel, and perineural invasions was classified as follows: 0, no invasion; 1, mild invasion (1 focus at the invasive front); 2, moderate invasion (2-3 foci at the invasive front); and 3, severe invasion (more than 4 foci at the invasive front). The mode of INF was classified into 3 groups: INF-a, a cancer nest showing expansive growth and presenting a clear borderline between the tumor tissue and stroma; INF-b, intermediate patterns of growth and an invasive structure of INF-a and INF-c; and INF-c, scirrhous growth with an unclear borderline at the invasive front. These data were evaluated according to the General Rules for Surgical and Pathological Studies on Cancer of the Biliary Tract 8 with reference to the World Health Organization classification and were staged according to the TNM classification of the International Union Against Cancer (UICC [Union for International Cancer Control]). 9

(A, B) A negative extracapsular lymph node invasion lesion; the foci of the carcinoma metastasis are within the lymph node capsule: (A) ×100 and (B) ×200 (hematoxylin-eosin). (C, D) A positive extracapsular lymph node invasion lesion; the foci of the carcinoma metastasis are outside the lymph node capsule: (C) ×100 and (D) ×200 (hematoxylin-eosin).
Immunohistochemistry
Using immunohistochemical staining, we investigated the cellular proliferative potential (Ki-67 labeling index) and the myofibroblasts (α-SMA [α smooth muscle actin]) density in the primary and lymph node metastatic lesions. The immunohistochemical procedure was as follows: for a histologic examination, extrahepatic bile duct carcinoma and lymph node specimens were routinely fixed with formalin, embedded in paraffin, and thin-sectioned (4-μm thick). Sections were mounted on saline-coated glass slides and an immunohistochemical examination was performed on deparaffinized sections using the standard avidin-biotin-peroxidase complex method with an automated immunostainer (Benchmark XT; Ventana Medical Systems, Tucson, AZ, USA). We selected representative cases of primary and lymph node metastatic lesions, which were the maximum number of invasive cancer cells in a lesion. The antibodies used were Ki-67 (1:100, clone MIB-1; Dako, Glostrup, Denmark) and α-SMA (1:100, clone 1A4; Dako).
Evaluation of immunohistochemistry and image analysis
We used imaging analysis to investigate the myofibroblast density in both the primary and lymph node metastatic lesions. Images were obtained using an Olympus Microscope BX50 with a U Plan Apo objective lens (×10; Olympus, Tokyo, Japan) and a digital camera DP-70 (Olympus). Using Image J software (NIH, Bethesda, MD, USA), the α-SMA positive area was binarized to obtain objective data, and the positive area in 1 mm2 (primary lesion) and 0.25 mm2 (lymph node metastasis lesion) sections was calculated (Figure 2A to C). Nucleic immunoreactivities of the tumor were judged as positive for Ki-67. When the tumor “hot spot” was identified, the objective was focused (×20) and the tumor cells were counted (Ki-67–positive cells/500).
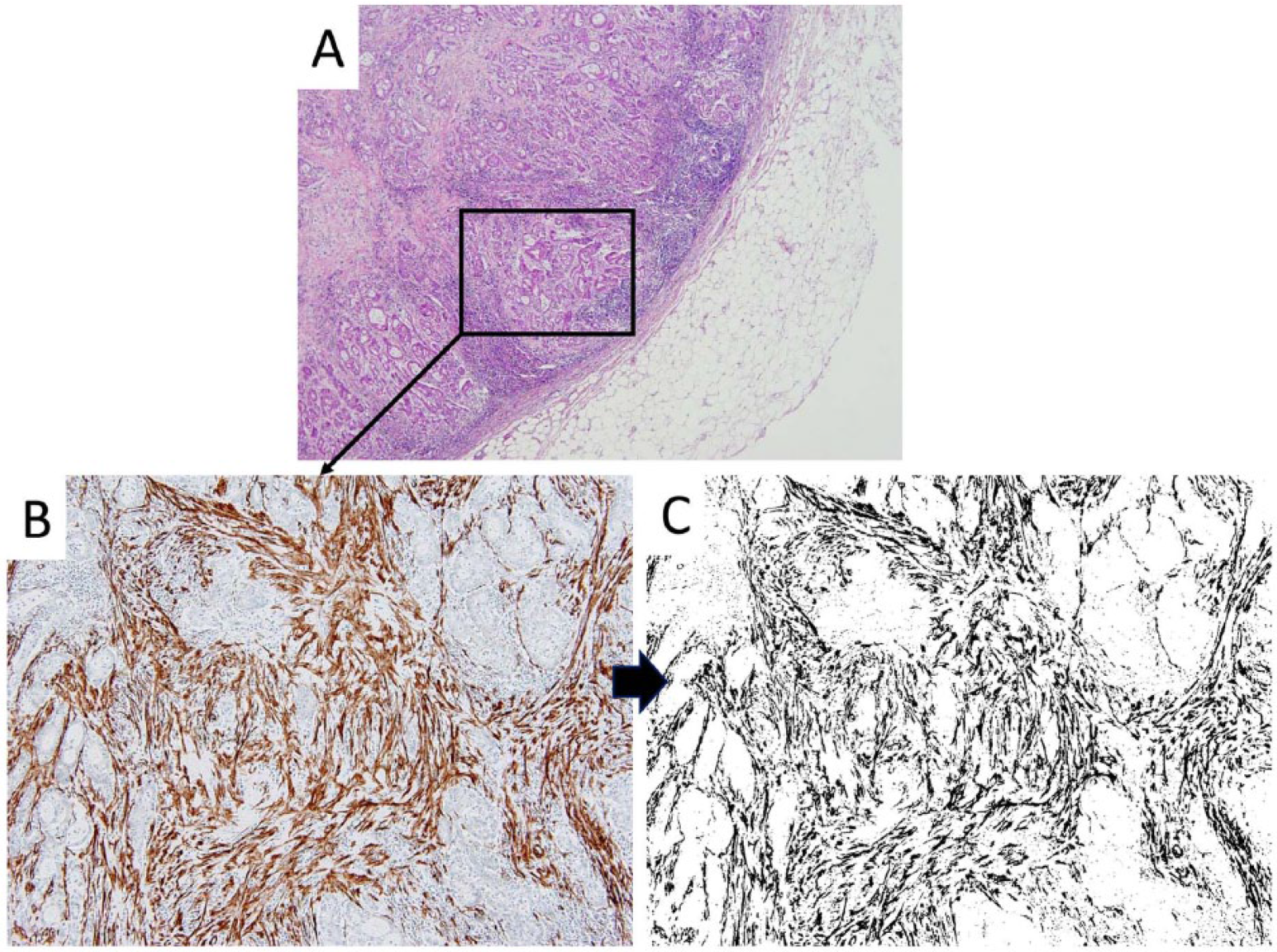
Representative case showing the foci of a metastatic lesion. (A) ×100 (hematoxylin-eosin), (B) ×400 (α-SMA staining), and (C) ×400 binarization image of α-SMA. α-SMA indicates α smooth muscle actin.
Statistical analysis
Statistical comparisons of the presence of extracapsular lymph node invasion or not were analyzed using Pearson χ2 test for categorical data and the Mann-Whitney test for continuous data. Survival curves were constructed using the Kaplan-Meier method, and differences in survival were evaluated using a log-rank test. The relative prognostic factors were analyzed using the Cox proportional hazards regression model. Differences were considered to be significant when P < .05. All statistical evaluations were performed using R (http://www.r-project.org).
Results
Clinicopathological findings
Based on the presence of extracapsular lymph node invasion, there were 21 positive cases and 11 negative cases in this study. When a single finding of extracapsular lymph node invasion was detected, the case was considered positive. The median total number of resected lymph nodes was 13 and the median number of lymph node metastasis was 2. Furthermore, the presence of extracapsular lymph node invasion was 2, and the absence was 1.5. The clinicopathological findings pertaining to the presence or absence of extracellular lymph node invasion are summarized in Table 1. No histologic factors were correlated with the histologic patterns of lymph node metastasis.
Clinicopathological features of significance extracapsular LN invasion.
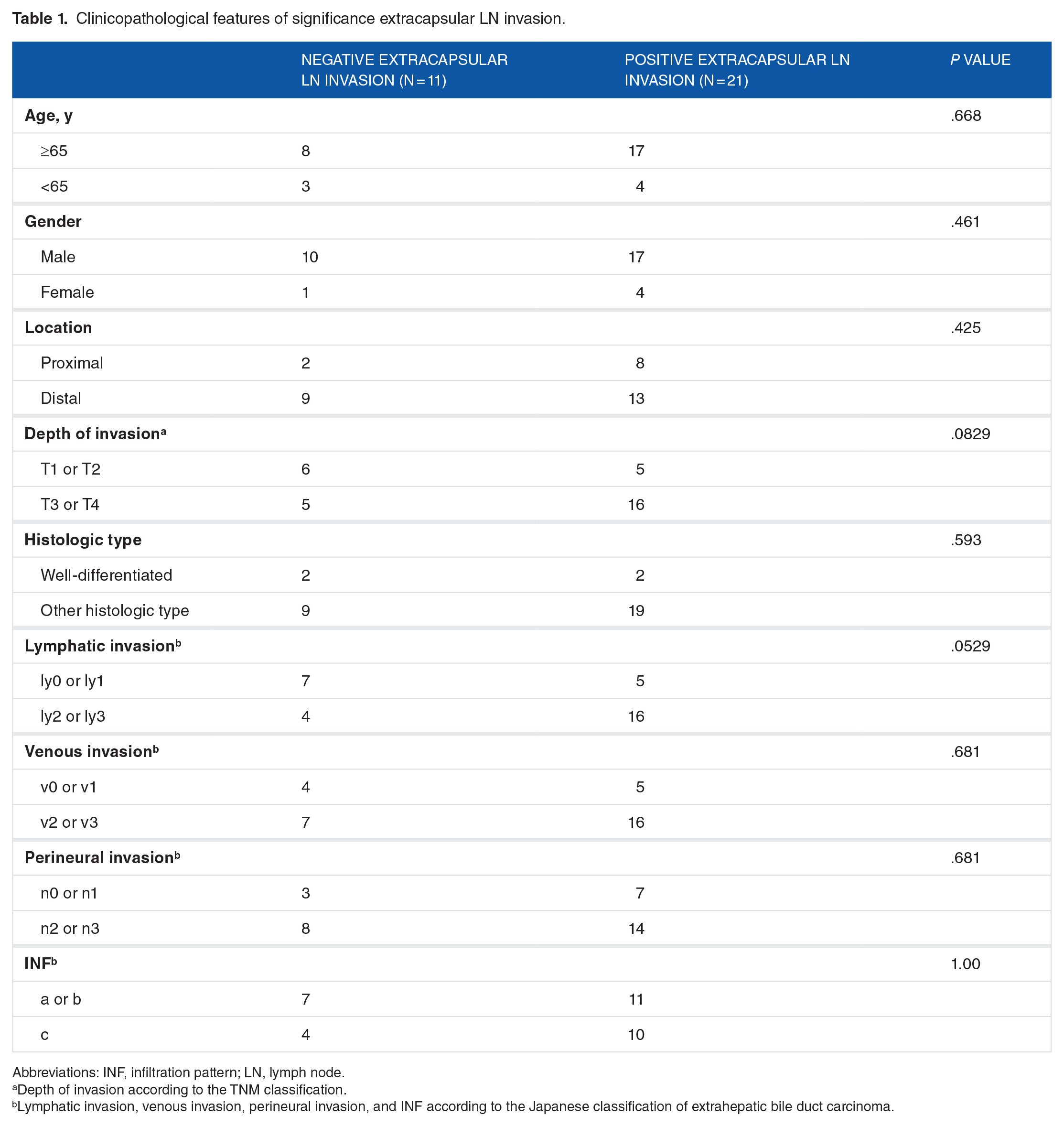
Abbreviations: INF, infiltration pattern; LN, lymph node.
Depth of invasion according to the TNM classification.
Lymphatic invasion, venous invasion, perineural invasion, and INF according to the Japanese classification of extrahepatic bile duct carcinoma.
Association between histological findings of the lymph node pattern and survival rates
To clarify the prognostic impact of extracapsular lymph node invasion, we investigated the prognosis. Survival curves based on univariate survival analysis demonstrated an association between patients with extracapsular lymph node invasion and a poor prognosis (Figure 3A; P = .014) and poor disease-free survival (Figure 3B; P < .001). To clarify potential prognostic indicators, we analyzed various pathological factors (Table 2). The univariate analysis revealed that only extracapsular lymph node invasion was correlated with a poor prognosis (relative risk: 0.2683; 95% confidence interval, 0.103-0.701; P < .01).

Patient prognosis using Kaplan-Meier survival curves. Patients positive for extracapsular LN invasion had a (A) poor disease-free survival (P = .014) and (B) poor overall survival (P < .001). LN indicates lymph node.
Univariate analyses of prognostic factors of survival.
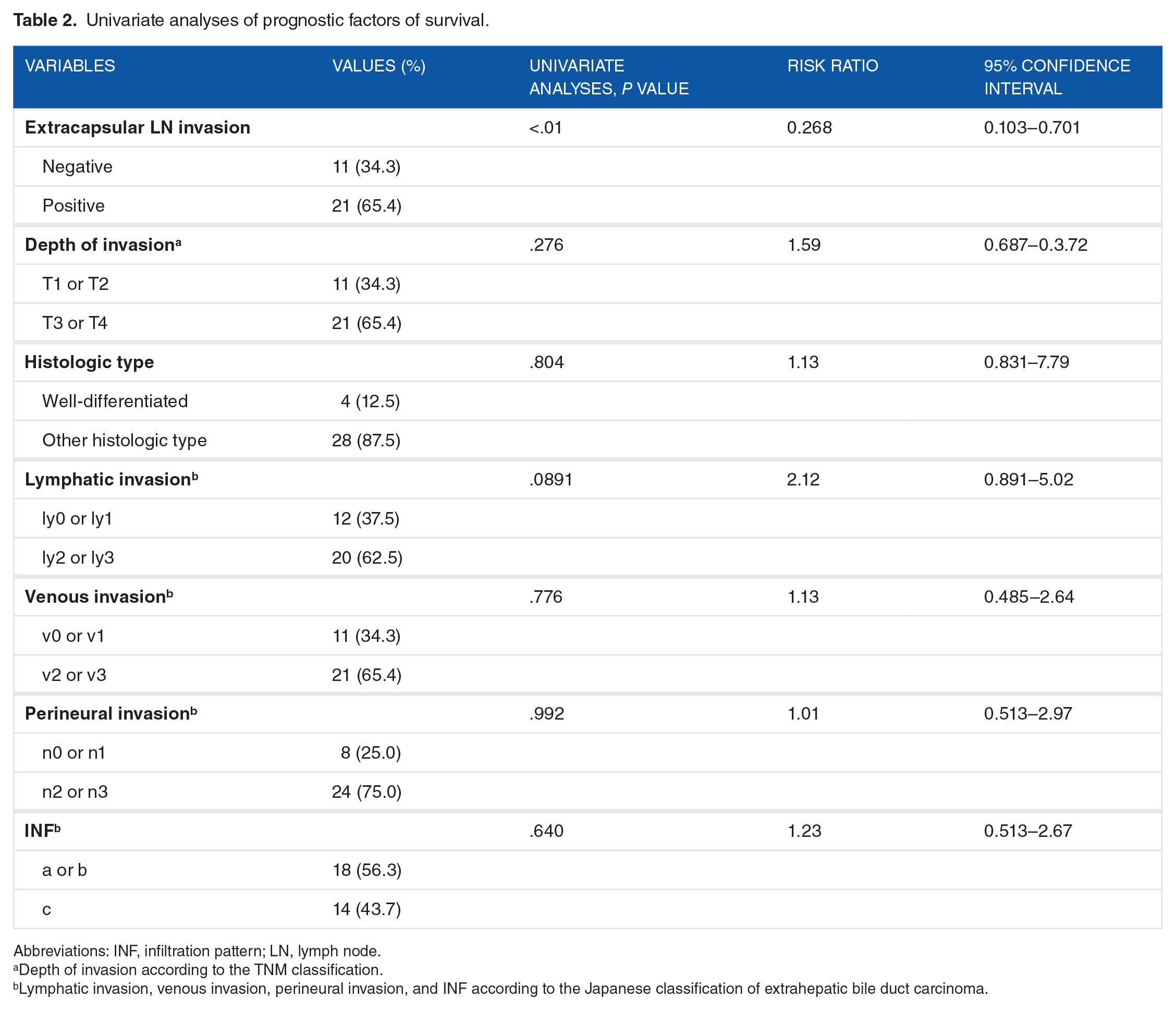
Abbreviations: INF, infiltration pattern; LN, lymph node.
Depth of invasion according to the TNM classification.
Lymphatic invasion, venous invasion, perineural invasion, and INF according to the Japanese classification of extrahepatic bile duct carcinoma.
Immunohistochemical analysis
To investigate the association between myofibroblast density and extracapsular lymph node invasion, we calculated the percentage of α-SMA expression at the invasive front. Furthermore, we investigated the association with the cell proliferation rate using the Ki-67 labeling index. Positive extracapsular lymph node invasion lesions had a higher percentage of α-SMA expression than negative (Figure 4A; P < .001) and primary lesions (Figure 4B; P < .01). However, there was no significant difference in the Ki-67 labeling index between the lymph node (Figure 4C; P = .747) and primary lesions (Figure 4D; P = .920).
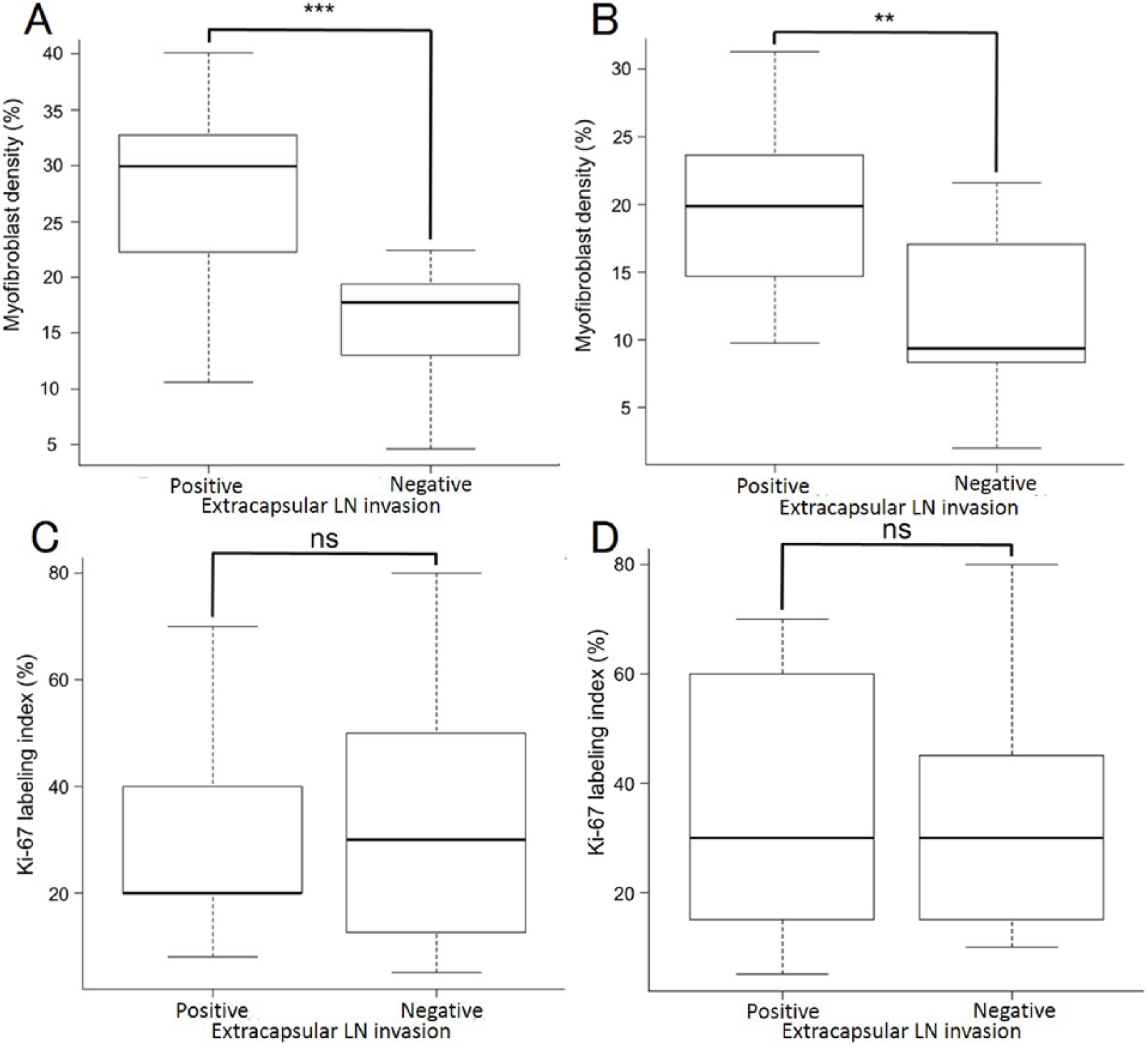
(A, B) The median myofibroblast density and (C, D) Ki-67 labeling index in primary and lymph node metastasis lesions. The association between positive and negative extracapsular LN invasion in (A, C) lymph node metastasis and (B, D) primary lesions. LN indicates lymph node.
Discussion
In this study, we clarified the clinicopathological characteristics of extracapsular lymph node invasion. In particular, tumor cells that had invaded the lymph node capsule resulted in a shorter life expectancy and disease-free survival compared with patients who did not have an invading lymph node metastasis. Furthermore, there was an increase in the α-SMA expression on fibroblasts at the invasive front of extracapsular lymph node invasion lesions and in primary lesions. The association between the α-SMA expression ratio of a primary lesion and a lymph node metastasis lesion was significantly different when comparing positive and negative extracapsular lymph node invasion cases. However, no correlation was observed between the Ki-67 labeling index of positive and negative extracapsular lymph node invasion lesions. This is the first report to describe the association between clinicopathological characteristics of extracapsular lymph node invasion of metastatic lymph nodes and α-SMA expression surrounding the metastatic cancer cells in extrahepatic bile duct carcinoma. Recently, the malignancy of a tumor was not defined in the cancer cells itself, but in the ECM. In particular, cancer-associated fibroblasts (CAFs) have been noted. 10
Cancer-associated fibroblasts are expressed in the α-SMA, generate myofibroblasts, and induce a desmoplastic reaction. 11 Furthermore, they enhance the angiogenesis and biogenesis of the ECM. The contraction between cancer cells and CAFs is closely associated and leads to the release of cytokines, such as transforming growth factor β (TGF-β) and hepatocyte growth factor. These cytokines induce the invasive and migratory capacity of the cancer cells. Furthermore, there are several factors that influence the interaction between CAFs and cancer cells. For example, cancer cells release platelet-derived growth factor that attracts more macrophages, and these macrophages produce TGF-β which activates the fibroblast. 12 Cancer-associated fibroblasts stimulate vascular endothelial cells and pericytes to produce a growth factor and chemokine, which promote the epithelial cells to cancer cells.13,14
In this study, we elucidated cancer malignancy, focusing on the histology of a lymph node metastatic lesion. Cancer cells that invade the lymph node capsule were indicative of a poor prognosis. Furthermore, we focused on the histology of the surrounding cancer cells. When focusing on the surrounding cancer cells at the invasive edge of extracapsular lymph node invasion lesions, a greater desmoplastic reaction was observed compared with the absence of such lesions. The desmoplastic reaction was induced by the myofibroblasts, and myofibroblasts with stained α-SMA were significantly increased in positive compared with negative extracapsular lymph node invasion lesions. Interestingly, the extracapsular lymph node invasion status was not correlated with the proliferation of cancer cells based on the Ki-67 labeling index. The same tendency was observed for primary lesions; that is, an increased expression of CAFs in a lymph node metastasis site was also indicated by increasing CAFs in the primary lesion. The desmoplastic changes consisted of a proliferation of fibroblasts and collagen fibers that were not only found in the primary lesion but also in parts of the lymph node metastasis. These changes were thought to be associated with an epithelial-mesenchymal transition that may contribute to the aggressive growth of an invasive cancer. The results of the univariate analysis revealed that if cancer cells had invaded the extra capsule of a lymph node metastasis site, the prognosis would have been significantly poorer. An important clinical issue is that tumor cell invasion of the lymph node capsule indicates a malignant potential with lymph node metastasis. Another study showed that squamous cell carcinoma of the vulva with an extranodal extension of lymph node metastases had significantly higher rates of all-cause mortality, cancer-specific mortality, and recurrence. 15 Our results indicate the same tendency. However, extracapsular lymph node involvement in extrahepatic bile duct carcinoma has no impact on the survival of patients with surgically treated extrahepatic bile duct carcinoma, 16 which is inconsistent with our results. One reason for this is the differences in the ratio of perihilar bile duct cancer and distal bile duct cancer; the proportion of perihilar bile duct cancer was high in the previous study, whereas perihilar bile duct cancer was low in this study. The prognosis of perihilar bile duct cancer is worse than distal bile duct cancer 17 ; hence, there was no significant difference in the previous study.
Our present findings should be considered within the limitations of this study. The most important limitation is the small number of cases; more cases may be needed to confirm the statistical significance detected in this study.
This study clearly showed that tumor cells invading the capsule of a lymph node resulted in an overall shorter life expectancy and disease-free survival compared with patients who did not have invading lymph node metastasis. Furthermore, we showed that the myofibroblasts surrounding the lesion play an important role in its malignant potential and poor prognosis.
Footnotes
Peer review:
Six peer reviewers contributed to the peer review report. Reviewers’ reports totaled 1529 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Grants-in-Aid for Science from the Ministry of Education, Culture, Sports, Science, and Technology in Japan and a Grant for Hirosaki University Institutional Research.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Study conception and design: TY and HK. Acquisition of data: TY, KI and KH. Ananysis and interpretation of data: TY, KS, TH, HS, YW, SM and HK. Drafting and revision of manuscript: TY.
