Abstract
Background:
Recent trials of ribociclib, a cyclin-dependent kinase inhibitor, have shown promising results in patients with hormone receptor-positive (HR+)/HER2-negative (HER2−) breast cancer. This meta-analysis evaluates the efficacy and safety of combining ribociclib with endocrine therapy (ET) versus ET alone in this patient population.
Methodology:
A comprehensive search of PubMed, Embase, CENTRAL, Scopus, and Web of Science (up to July 2024, no language restrictions) identified randomized controlled trials (RCTs) comparing endocrine therapy (ET) with and without ribociclib for HR+/HER2− breast cancer. Bias was assessed using Cochrane’s Risk of Bias tool. Hazard ratios (HRs) for overall survival (OS) and progression-free survival (PFS) and odds ratios (ORs) for Grade III + adverse effects (neutropenia, hepatobiliary toxicity, QT prolongation, and interstitial pneumonitis) were calculated with 95% confidence intervals (CI). A random-effects model accounted for heterogeneity (I² statistic). The protocol was registered in PROSPERO (CRD42024558512).
Results:
The meta-analysis of 5 RCTs (7286 participants) showed ribociclib plus ET significantly improved OS (HR 0.76, 95% CI 0.68–0.85, P < .001) and PFS (HR 0.57, 95% CI 0.51–0.64, P < .001) compared with ET alone. Grade III + adverse effects of special interest that were more common with ribociclib plus ET include neutropenia (OR 75.76, 95% CI 35.23–162.93, P < .001), hepatobiliary toxicity (OR 2.54, 95% CI 1.32–4.90, P = .005), and QT prolongation (OR 2.95, 95% CI 1.69–5.16, P < .001). The rare grade III + interstitial pneumonitis events (OR 3.36, 95% CI 0.56–20.07, P = .18) also warrant ongoing vigilance.
Conclusion:
Ribociclib combined with ET improves OS and PFS but is associated with higher rates of adverse effects, compared with ET alone, highlighting the need for careful monitoring and management.
Funding:
None
Introduction
GLOBOCAN 2022 estimates rank breast cancer as the second most common cancer globally, the most frequently diagnosed among women, and the leading cause of cancer-related deaths in women worldwide. 1 Its burden is predicted to increase to over 3 million new cases, and 1 million deaths annually by 2040. 2 Breast cancer is classified into 4 molecular subtypes: Luminal A (40%), Luminal B (20%), HER2-enriched (10–15%), and Triple Negative (15–20%). Among these, Luminal A and Luminal B are hormone receptor-positive (HR+), collectively accounting for approximately 60% of all breast cancers. 3 Endocrine therapies, such as selective estrogen receptor modulators (SERMs) and aromatase inhibitors (AIs), have been the first-line treatment for HR+ breast cancer for decades. However, resistance to these therapies often develops over prolonged use owing to mechanisms like loss of hormone receptors, upregulation of growth factor signaling, and activation of intracellular pathways, prompting exploration into combination regimens.4,5
Cyclin-dependent kinase (CDK) 4/6 inhibitors are targeted anticancer agents that have shown promise in recent years to address this challenge, particularly in combination with endocrine therapy (ET) as the first-line treatment for HR+/HER2− advanced breast cancer. 6 Their primary mechanism involves inhibiting retinoblastoma (RB) protein phosphorylation, leading to G1 cell cycle arrest in tumor cells.7,8 Three major CDK4/6 inhibitors—ribociclib, abemaciclib, and palbociclib—have been approved and extensively studied for HR+/HER2− breast cancer. 9 Ribociclib is an orally bioavailable, highly selective inhibitor of cyclin-dependent kinases (CDK) 4 and 6, targeting the hyperactivated CDK4/6-Rb pathway associated with HR+ breast cancer.10,11 Recent trials involving ribociclib in combination with ET for HR+/HER2− breast cancer have shown promising results in terms of overall survival (OS) and progression-free survival (PFS) in both pre-menopausal and postmenopausal women. 12
Previous meta-analyses have studied CDK4/6 inhibitors overall in combination with ET in terms of safety and efficacy endpoints.13-15 With ribociclib gaining popularity in real-world practice and newer trials exploring its efficacy in different settings, this systematic review and meta-analysis synthesize the evidence for efficacy and safety of ribociclib in combination with ET, compared with ET alone, in patients with HR+/HER2− breast cancer.
Materials and Methods
Search strategy
This systematic review and meta-analysis were conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines. Page et al, 16 with a completed PRISMA checklist (Supplementary File 1). The PROSPERO registration number for this review is CRD42024558512.
A systematic search was performed across multiple databases, including PubMed, Embase, CENTRAL, Scopus, and Web of Science. In addition, a search for relevant studies in the gray literature was conducted, but no additional results were found. The search strategy employed a combination of MeSH terms and text words encompassing key concepts such as “breast cancer,” “ribociclib,” and “endocrine therapy.” Studies involving human subjects published up to July 2024 were included. No language restrictions were applied during the search. Two independent reviewers screened the articles, resolving disagreements through consensus with a third reviewer. The initial search identified 6642 articles, of which 123 were selected for full-text review. Ultimately, 5 trials met the inclusion criteria for the meta-analysis. The PRISMA flow diagram (Figure 1) details the selection process, and a detailed search strategy is shown in Supplementary File 2.
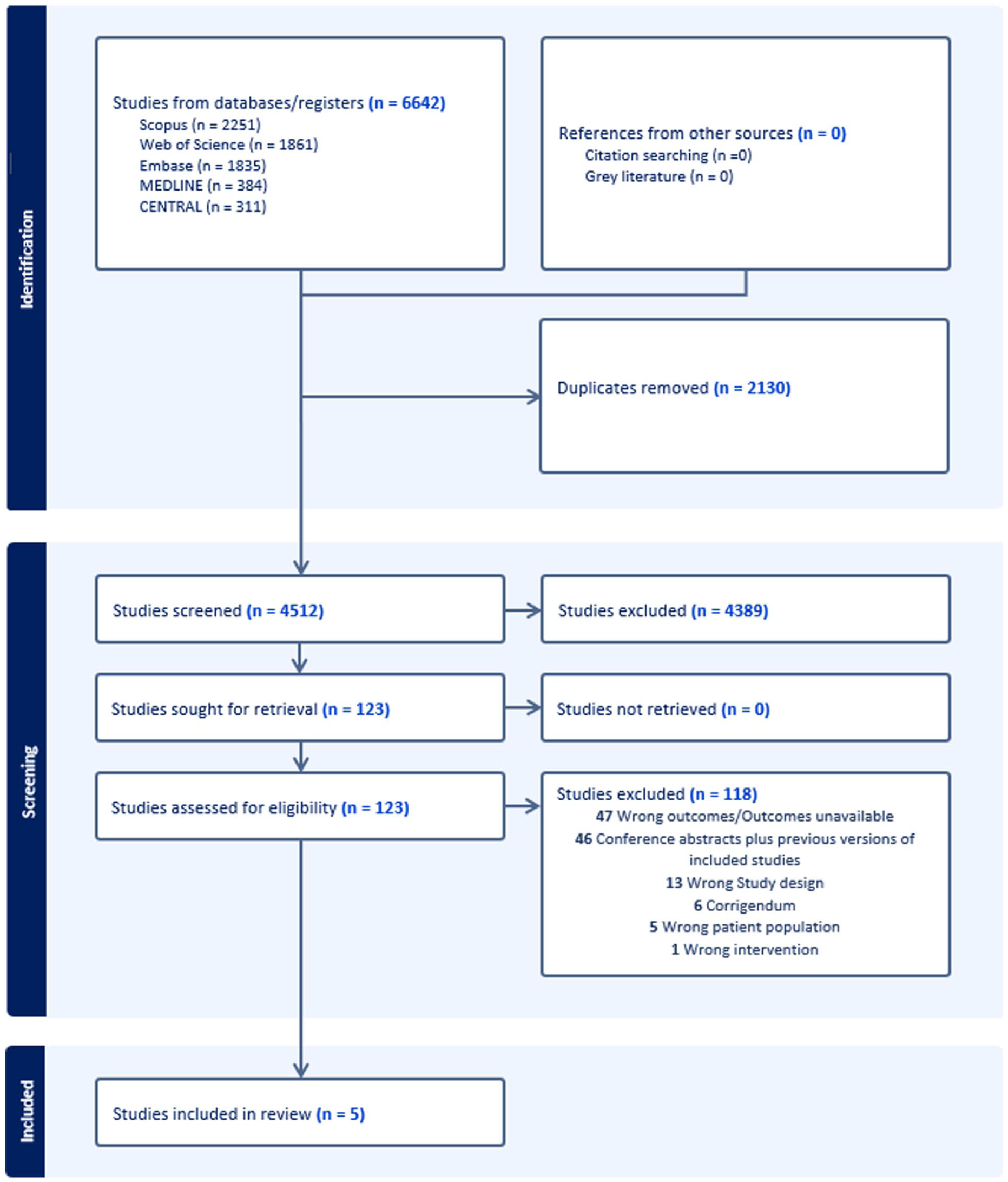
Prisma flow diagram.
Eligibility criteria
Randomized clinical trials (RCTs) were included if they evaluated ribociclib plus any form of ET (selective estrogen receptor modulator, aromatase inhibitor, or fulvestrant, with or without ovarian suppression), included patients with HR+/HER2− breast cancer, and reported survival outcomes or adverse events of interest. Trials involving concurrent mTOR inhibitors, chemotherapy, or any other anticancer agents in combination with these agents were excluded. Overlapping study populations were carefully assessed by reviewing trial inclusion periods, participating centers, and trial registration numbers; in cases where overlap was suspected, only the most complete and updated dataset was included to ensure population homogeneity. When multiple publications for a trial were available, the most recent data were prioritized. Efforts were made to contact the corresponding authors of 4 out of the 5 studies for the latest updates. Two authors responded, informing us that there were no significant updates to the data.
The included trials were MONALEESA-2, 17 MONALEESA-3 18 MONALEESA-7 19 NATALEE, 20 and MAINTAIN. 21 A brief description of these trials is provided in Table 1.
Baseline characteristics of the included studies.

This table summarizes the study population, various intervention and control regimens in each trial, phase of each trial, and disease stage eligibility criteria. It highlights that among the listed trials, only the NATALEE trial was conducted exclusively in the early-stage setting, whereas the others focused on advanced or metastatic disease. Similarly, the MAINTAIN trial studied the use of Ribociclib plus ET versus ET alone after progression on a prior CDK4/6 inhibitor.
Quality assessment and data extraction
Two independent reviewers separately performed bias analysis and data extraction, with any discrepancies resolved by the third reviewer. Bias analysis was conducted using Cochrane’s Risk of Bias (RoB 2.0) tool. 22 The major domains assessed were randomization process, deviations from intended interventions, missing outcome data, outcome measurement and selection of reported results. Data on baseline characteristics of the studies, survival outcomes (OS) and PFS, and grade III+ adverse effects of special interest (neutropenia, hepatobiliary toxicity, QT prolongation, and interstitial pneumonitis) were extracted. A detailed bias analysis is provided in Supplementary File 3.
Statistical analysis
Survival outcomes for OS and PFS were analyzed using HRs with 95% confidence interval (CI). Odds ratios (ORs) were calculated for adverse events. A random-effects model was applied to account for heterogeneity, which was evaluated using the I² statistic. Data were analyzed using RevMan 5.4
Patient and public involvement
Patients and/or the public were not involved in the design, conduct, reporting, or dissemination of this systematic review and meta-analysis. This study is based entirely on the analysis of published literature and does not include any direct involvement from patients or the public.
Results
A total of 5 randomized controlled trials (RCTs) comprising 7286 patients were included in the study. The detailed baseline characteristics of the included studies are presented in Table 1.
Survival Outcomes
The OS outcomes were mentioned in four of five trials.17,19,20,23 The hazard ratio (HR) for OS was 0.76 (95% CI [0.68, 0.85]) favoring Ribociclib plus ET compared with ET alone (P < .001). Similarly, progression-free survival was reported in 4 trials18,21,24,25 and favored Ribociclib plus ET with HR 0.57 (95% CI 0.51 to 0.64, P < .001). For both OS and PFS, heterogeneity across studies I2 was 0. Therefore, detailed subgroup analysis and sensitivity analyses were not done. The forest plots for OS and PFS are shown in Figure 2.
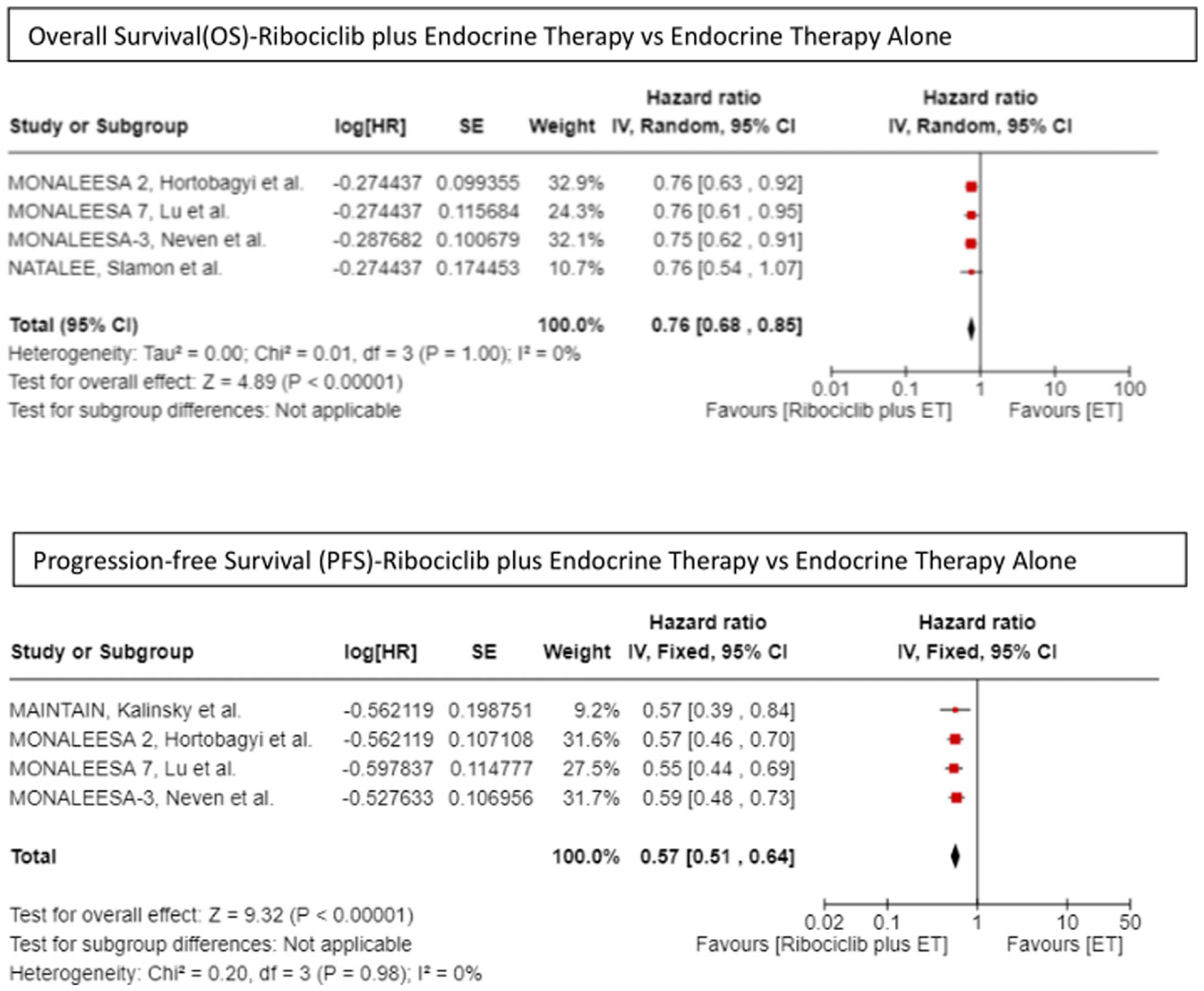
Survival outcomes: OS and PFS—Ribociclib plus endocrine therapy (ET) versus ET alone for HR+/HER2− breast cancer.
Adverse Effects of Special Interest
The adverse effects of special interest, as consistently reported across trials and included in the analysis, were grade III + neutropenia, grade III + hepatobiliary toxicity, grade III + QT prolongation, and grade III + interstitial pneumonitis.
Grade III + neutropenia
Grade III+ was significantly higher in the ribociclib plus ET arm compared with ET-alone, with an OR of 75.76 (95% CI 35.23–162.93, P < .001). Heterogeneity across studies was high (I² = 76%). Sensitivity analysis was performed to investigate the cause of heterogeneity. On excluding the MONALEESA-7 trial, heterogeneity was reduced to 0, and the OR was recalculated as 101.53 (95% CI 69.05–149.28, P < .001). MONALEESA-7 differs from other trials in that it exclusively involves pre- and peri-menopausal women (Table 1). However, this variability due to MONALEESA-7 is not observed for other adverse effects, so no definitive conclusions can be made.
Grade III + hepatobiliary toxicity
Ribociclib plus ET was associated with a higher incidence of Grade III + hepatobiliary toxicity compared with ET alone, with an OR of 2.54 (95% CI 1.32–4.90, P = .005). Heterogeneity was high (I² = 83%). Sensitivity analysis revealed a considerable reduction in heterogeneity (to 57%) when the NATALEE trial was excluded. The subsequent analysis still favored the treatment arm, but with a reduced OR of 2.07 (95% CI 1.20–3.56, P = .009). Notably, the NATALEE trial used a lower dose of ribociclib (400 mg/day) compared with 600 mg/day in other trials, which should typically suggest fewer side effects, contrary to what is observed in our analysis. However, no significant differences in patient characteristics or treatment regimens were found that could explain the observed hepatobiliary toxicity, implying that the variability may stem from other unmeasured factors.
Grade III + QT prolongation
Grade III + QT prolongation was a rarer outcome but was reported more frequently in the ribociclib plus ET arm. The OR was 2.95 (95% CI 1.69–5.16, P < .001), with no heterogeneity across studies (I² = 0).
Grade III + interstitial pneumonitis
Interstitial pneumonitis, though rare, was reported only in the ribociclib plus ET arm. Across 3 studies, the ribociclib plus ET arm showed a greater incidence of Grade III + interstitial pneumonitis, with an OR of 3.36 (95% CI 0.56–20.07). However, this finding was not statistically significant (P = .18), and heterogeneity across studies was low (I² = 0).
The forest plots of all Grade III+ adverse effects of special interest are presented in Figure 3. Publication bias was not assessed, as there were only five eligible studies.

Adverse effects of special interest—ribociclib plus endocrine therapy (ET) versus ET alone for HR+/HER2− breast cancer.
Discussion
This meta-analysis aimed to evaluate the efficacy and safety of ribociclib in combination with ET for the treatment of HR+/HER2− breast cancer. Specifically, our study focused on comparing OS and PFS between ribociclib plus ET and ET alone, while assessing the incidence of Grade III + adverse events of special interest that were consistently reported in trial updates. By synthesizing data from published studies, we sought to provide a more comprehensive understanding of ribociclib’s effectiveness and safety profile, helping to inform clinical decisions for managing HR+/HER2− breast cancer with ribociclib-based combination therapy.
As observed in our meta-analysis, ribociclib combined with ET significantly improved survival outcomes compared with ET alone, with a hazard ratio of 0.76 for OS and 0.57 for PFS. These results confirm its role as a cornerstone treatment in HR+/HER2− breast cancer. However, ribociclib’s use is associated with a significantly higher incidence of Grade III + adverse events, as illustrated in results, such as neutropenia, hepatobiliary toxicity, and QT prolongation. The increased frequency of these adverse effects necessitates more rigorous monitoring, such as regular blood counts, liver function tests, electrocardiogram (ECG) monitoring, and vigilance for clinical signs and symptoms. Although Grade III+ interstitial pneumonitis is rare, it is a side effect unique to the ribociclib arm, and clinicians should remain vigilant for any signs of respiratory distress.
Ribociclib is one of the three main CDK4/6 inhibitors that act by preventing retinoblastoma (RB) protein phosphorylation, halting the cell cycle in the G1 phase. 26 In recent years, it has been extensively studied in the treatment of HR+/HER2− breast cancer.10-12,17-21 The MONALEESA trials demonstrated its efficacy across various clinical scenarios: MONALEESA-7, focusing on pre-menopausal and peri-menopausal women, and MONALEESA-2 and MONALEESA-3, focusing on postmenopausal women, showed its benefits in improving OS and PFS.17-19 The MAINTAIN trial highlighted ribociclib’s role in managing disease progression even after prior use of CDK4/6 inhibitors, establishing its utility in later lines of therapy. 21 Most recently, the NATALEE trial explored ribociclib in early-stage breast cancer, using a lower dose to balance efficacy and safety, and demonstrated improved invasive disease-free survival (iDFS) in patients with stage II and III disease. 20 These trials underscore ribociclib’s broad applicability across early and advanced stages.
Two other CDK4/6 inhibitors used for the treatment of HR+/HER2−breast cancer are palbociclib and abemaciclib. Although no direct head-to-head comparison between the agents has been conducted, the results are conflicting. The PALOMA-2 trial, which evaluated palbociclib, failed to improve OS significantly, whereas other studies conclude that no single agent is superior to another.27-29 Furthermore, abemaciclib, although notable for activity in brain metastases, incurs higher costs than ribociclib.30,31 These factors led ribociclib to be the primary focus of our study.
Limitations and recommendations
In our meta-analysis, we tried our best to consolidate the evidence for ribociclib’s overall safety and efficacy, although our study has its limitations. The small number of eligible studies could impact the statistical power and generalizability of the findings. Data unavailability further limited a comprehensive analysis of all adverse effects, and efforts to obtain unpublished data from corresponding authors provided limited updates. In addition, this analysis was limited to ribociclib, and comparisons with other CDK4/6 inhibitors, such as palbociclib and abemaciclib, were beyond its scope.20-22 Although statistical heterogeneity across studies was minimal, there was some heterogeneity in the choice of ET and patient population (early/late stage and pre-/postmenopausal). However, separate subgroup analysis was not possible due to the limited studies included. In local settings, the choice of endocrine backbone will likely depend on institutional policies, cost considerations, and availability of drugs. Also, future research should focus on head-to-head comparisons of CDK4/6 inhibitors, detailed subgroup analyses, and the exploration of ribociclib in neoadjuvant settings to optimize its clinical utility and address specific patient needs. The real-world evidence is necessary for further optimizing safety, efficacy, and cost-effectiveness.
Conclusion
Ribociclib, when combined with ET, improves both OS and PFS, showing efficacy in early as well as advanced HR+/HER2− breast cancer. However, its use is associated with a higher risk of Grade III + or higher adverse events, including common toxicities such as neutropenia and hepatobiliary effects, as well as less frequent but serious events such as Grade III + QT prolongation and interstitial pneumonitis. In real-world clinical practice, these risks emphasizes the importance of vigilant monitoring, including regular blood counts, liver function tests, ECG assessments, and careful observation for respiratory symptoms. In conclusion, while ribociclib provides substantial therapeutic benefits, proactive management of its adverse effects is essential to ensure patient safety and optimize treatment outcomes.
Supplemental Material
sj-docx-1-onc-10.1177_11795549251402955 – Supplemental material for Efficacy and Safety of Ribociclib Plus Endocrine Therapy Versus Endocrine Therapy Alone in HR-Positive/HER2-Negative Breast Cancer: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Supplemental material, sj-docx-1-onc-10.1177_11795549251402955 for Efficacy and Safety of Ribociclib Plus Endocrine Therapy Versus Endocrine Therapy Alone in HR-Positive/HER2-Negative Breast Cancer: A Systematic Review and Meta-Analysis of Randomized Controlled Trials by Reechashree Dhungana, Parikshit Prasai, Bishal Paudel, Kamal Ranabhat, Simit Sapkota and Sunil Shrestha in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-2-onc-10.1177_11795549251402955 – Supplemental material for Efficacy and Safety of Ribociclib Plus Endocrine Therapy Versus Endocrine Therapy Alone in HR-Positive/HER2-Negative Breast Cancer: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Supplemental material, sj-docx-2-onc-10.1177_11795549251402955 for Efficacy and Safety of Ribociclib Plus Endocrine Therapy Versus Endocrine Therapy Alone in HR-Positive/HER2-Negative Breast Cancer: A Systematic Review and Meta-Analysis of Randomized Controlled Trials by Reechashree Dhungana, Parikshit Prasai, Bishal Paudel, Kamal Ranabhat, Simit Sapkota and Sunil Shrestha in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-3-onc-10.1177_11795549251402955 – Supplemental material for Efficacy and Safety of Ribociclib Plus Endocrine Therapy Versus Endocrine Therapy Alone in HR-Positive/HER2-Negative Breast Cancer: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Supplemental material, sj-docx-3-onc-10.1177_11795549251402955 for Efficacy and Safety of Ribociclib Plus Endocrine Therapy Versus Endocrine Therapy Alone in HR-Positive/HER2-Negative Breast Cancer: A Systematic Review and Meta-Analysis of Randomized Controlled Trials by Reechashree Dhungana, Parikshit Prasai, Bishal Paudel, Kamal Ranabhat, Simit Sapkota and Sunil Shrestha in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-4-onc-10.1177_11795549251402955 – Supplemental material for Efficacy and Safety of Ribociclib Plus Endocrine Therapy Versus Endocrine Therapy Alone in HR-Positive/HER2-Negative Breast Cancer: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Supplemental material, sj-docx-4-onc-10.1177_11795549251402955 for Efficacy and Safety of Ribociclib Plus Endocrine Therapy Versus Endocrine Therapy Alone in HR-Positive/HER2-Negative Breast Cancer: A Systematic Review and Meta-Analysis of Randomized Controlled Trials by Reechashree Dhungana, Parikshit Prasai, Bishal Paudel, Kamal Ranabhat, Simit Sapkota and Sunil Shrestha in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
None.
Ethical considerations
As this is a systematic review and meta-analysis, no direct interaction with participants was required. However, this review strictly adhered to PRISMA guidelines.16
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author Contributions
The study was conceptualized by R.D., who also conducted the literature searches, screening, bias analysis, and data extraction, performed the statistical analysis, and drafted and finalized the manuscript. P.P. contributed to literature screening, bias analysis, and data extraction. B.P. provided critical inputs on study design, contributed to data synthesis, and reviewed the manuscript. K.R. assisted with methodology refinement, offered public health insights, and critically reviewed the manuscript. S.Sa. contributed to the interpretation of oncological data and critically reviewed the manuscript for clinical relevance. S.Sh. supervised the study as the senior author, contributed to literature searches, screening, bias analysis, and data extraction, and critically reviewed and approved the final manuscript. All authors reviewed and approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All data underlying this meta-analysis were extracted from previously published articles, which are available in the public domain. No new raw data were generated.
Data Availability Statement
All data used in this systematic review and meta-analysis were extracted from previously published studies. No new participant data were collected for this work. The extracted data are available from the corresponding author upon reasonable request.
Declaration of generative AI and AI-assisted technologies in the writing process
While preparing this manuscript, the authors utilized ChatGPT and Grammarly to improve grammar, spelling, readability, and sentence structure. These tools were limited to these specific tasks, and no generative AI was employed for literature writing or reference generation. After utilizing these tools, the authors thoroughly reviewed and edited the content to ensure its accuracy and integrity. The authors take full responsibility for the content and the final manuscript.
PROSPERO registration number
CRD42024558512.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
