Abstract
Background:
This analysis evaluated patient-reported outcomes (PROs) to assess health-related quality of life (HRQoL) in the phase III MONALEESA-7 trial, which previously demonstrated improvements in progression-free survival (PFS) and overall survival (OS) with ribociclib (cyclin-dependent kinase 4/6 inhibitor) + endocrine therapy (ET) compared with placebo + ET in pre- and perimenopausal patients with hormone-receptor-positive, HER2-negative (HR+/HER2−) advanced breast cancer (ABC).
Methods:
The European Organisation for Research and Treatment of Cancer (EORTC) Quality of Life questionnaire C30 (QLQ-C30) and the EQ-5D-5L were used to evaluate HRQoL.
Results:
EORTC QLQ-C30 assessments were evaluable for 335 patients in the ribociclib arm and 337 patients in the placebo arm. Adherence rates at baseline and ⩾1 postbaseline time point were 90% and 83%, respectively. Patients treated with ribociclib + ET had a longer time to deterioration (TTD) ⩾ 10% in global HRQoL {hazard ratio (HR), 0.67 [95% confidence interval (CI), 0.52–0.86]}. TTD ⩾ 10% in global HRQoL was delayed in ribociclib-treated patients without
Conclusion:
HRQoL was maintained longer in patients who received ribociclib + ET
Introduction
Endocrine therapy with ovarian suppression plus a cyclin-dependent kinase 4/6 (CDK4/6) inhibitor is a recommended first-line treatment for premenopausal women with hormone-receptor-positive (HR+)/human epidermal growth factor receptor 2-negative (HER2−) advanced breast cancer (ABC)1,2; however, treatment resistance and disease progression ultimately occur. In addition to these potential complexities of breast cancer in premenopausal women, maintaining quality of life (QoL) in a younger patient population presents a unique treatment challenge. ABC is associated with reduced QoL; however, treatment may improve or at least stabilize global QoL and specific disease-related symptoms. 3
MONALEESA-7 is a phase III trial of first-line endocrine-based therapy with ribociclib or placebo with goserelin and a nonsteroidal aromatase inhibitor (NSAI; letrozole or anastrozole) or tamoxifen in premenopausal patients with ABC.
4
This is the only phase III trial to prospectively assess a CDK4/6 inhibitor in combination with endocrine therapy in a population of exclusively premenopausal patients with HR+/HER2− ABC. In MONALEESA-7, progression-free survival (PFS) was prolonged with ribociclib
Because QoL is among the most important considerations for patients with cancer, one of the predefined secondary objectives of the MONALEESA-7 study was evaluation of patient-reported outcomes (PROs) for health-related QoL (HRQoL). We report these data, which were analyzed from the recent final data cutoff used to evaluate OS.
Methods
The details of the MONALEESA-7 trial design and participants have been described previously. 4 Written informed consent was obtained from all patients at enrollment. The study was approved by each participating site’s institutional review board or independent ethics committee (Supplemental Table S1). The trial was performed in accordance with the Good Clinical Practice guidelines and the Declaration of Helsinki. This study is registered [ClinicalTrials.gov identifier: NCT02278120]. The data cutoff date for this analysis was 30 November 2018.
Patient-reported outcomes
Patients completed questionnaires in person at the beginning of each visit. PROs were evaluated based on the European Organisation for Research and Treatment of Cancer (EORTC) Quality of Life questionnaire core 30 (QLQ-C30) and Breast cancer module (QLQ-BR23). The EQ-5D-5L was used to evaluate PRO measures of HRQoL, functioning, disease symptoms, and treatment-related side effects.
Time to deterioration (TTD) ⩾ 10% in the global health status/QoL scale and secondary PRO variables of the EORTC QLQ-C30 were analyzed based on the 10-point threshold, which is considered a reference for clinical meaningfulness.6,7 Definitive 10% deterioration was defined as a worsening in QoL score by ⩾10% compared with baseline, with no later improvement above this threshold observed during the treatment period, or death due to any cause.
Statistical analyses
TTD was compared between the two treatment arms using a stratified log-rank test at a 1-sided 2.5% level of significance. Survival distributions were analyzed using the Kaplan–Meier method. A stratified Cox regression was used to estimate the HR for TTD, along with 2-sided 95% CI. Descriptive statistics were used to summarize the scores from study PRO assessments at each scheduled assessment time point. Change from baseline in all subscales was analyzed using a linear mixed-effects model that included the following factors: treatment, stratification factors, and baseline score; this analysis only included assessments up to the time point at which ⩾50 patients were evaluable in each treatment arm.
Results
Patient disposition
As previously reported at the data cutoff of 30 November 2018, there were 116 patients (34.6%) continuing treatment in the ribociclib arm and 57 patients (16.9%) continuing treatment in the placebo arm. 5 At data cutoff, 219 patients (65.4%) in the ribociclib arm and 280 patients (83.1%) in the placebo arm had discontinued treatment. The main cause of treatment discontinuation among these patients was progressive disease (51.6% and 68.2% in the ribociclib and placebo arms, respectively).
Quality of life
QoL was assessed at baseline and throughout treatment; the EORTC QLQ-C30 was completed at baseline and ⩾1 postbaseline time point by 90% and 83% of patients in the ribociclib and placebo arms, respectively. TTD ⩾ 10% in global HRQoL was significantly delayed with ribociclib
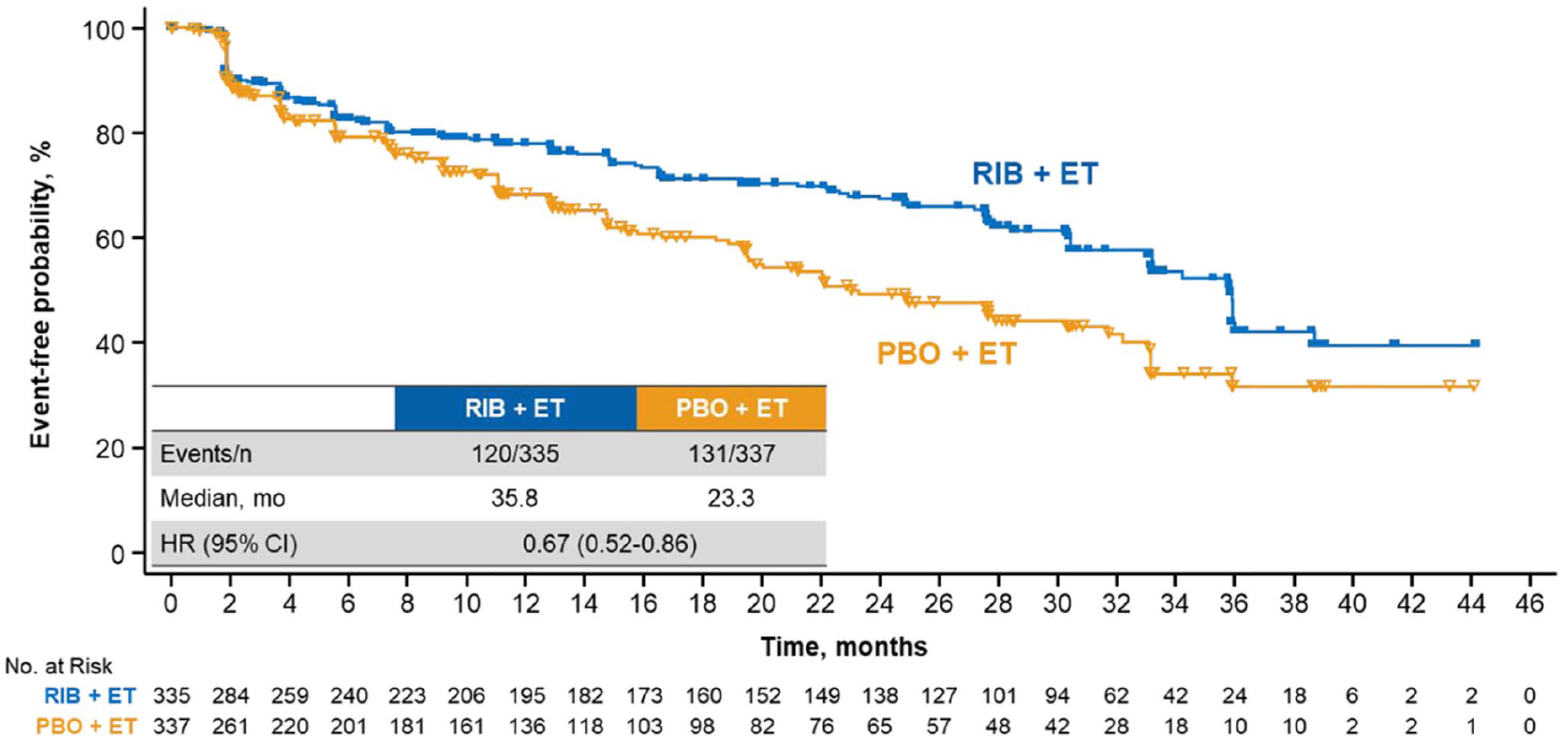
TTD ⩾10% in global HRQoL in patients treated with RIB

TTD ⩾10% in global HRQoL in patients treated with RIB with
Global health status was also assessed as change from baseline in a linear mixed-effects model that accounted for treatment, stratification factors, and baseline health status score (Supplemental Figure S2). The baseline global health status scores of 64.7 in the ribociclib arm and 65.1 in the placebo arm were similar to the 62.9 observed in a large general (i.e., not cancer specific) population of women aged 40–49 years in North America and Europe.
8
After 25 cycles, which corresponds with the median treatment duration of 2 years in the ribociclib arm,
5
the mean score changes from baseline in the ribociclib arm (
Ribociclib treatment led to a longer maintenance than placebo in key subdomains of the EORTC QLQ-C30 questionnaire, including pain and fatigue. The ribociclib arm demonstrated a delay in TTD ⩾ 10% in pain

TTD ⩾10% in pain in patients treated with RIB
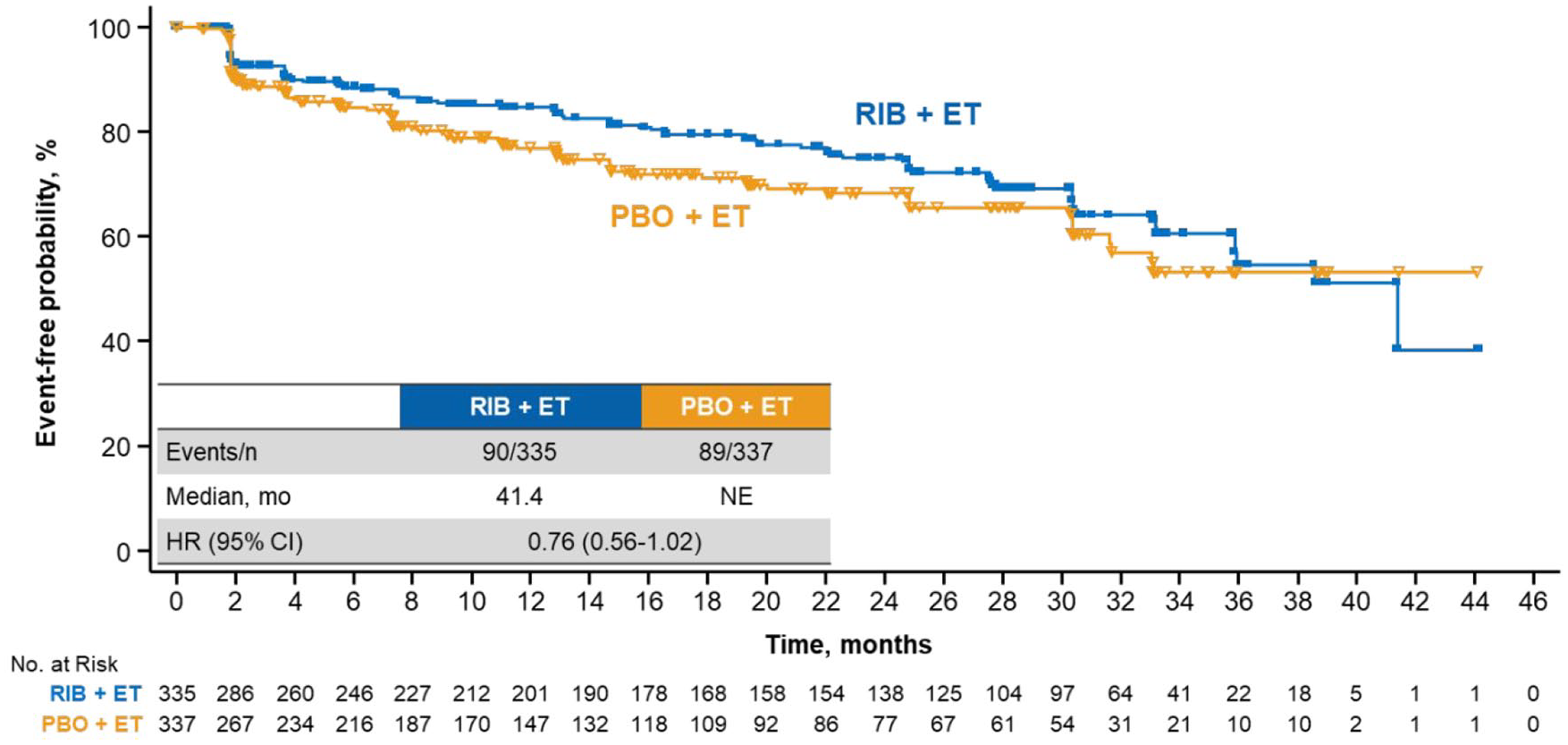
TTD ⩾10% in fatigue in patients treated with RIB
Ribociclib is approved for use in combination with an NSAI in premenopausal patients; therefore, analyses were performed on global health status and subdomains of the EORTC QLQ-C30 questionnaire in the MONALEESA-7 NSAI cohort (ribociclib arm,

TTD ⩾ 10% in global HRQoL in patients treated with RIB

TTD ⩾ 10% in pain in patients treated with RIB
Discussion
The primary efficacy report and a more recent OS report of the MONALEESA-7 trial have demonstrated statistically significant benefits in PFS and OS with ribociclib plus endocrine therapy over placebo plus endocrine therapy, as well as a manageable safety profile,4,5 supporting the clinical benefit of the regimen. Another key aspect of clinical benefit is HRQoL, which was assessed
In MONALEESA-7, the longer PFS in the ribociclib arm over the placebo arm was accompanied by an improvement in global health status over placebo. That is, patients receiving ribociclib had an improved QoL during treatment as well as a longer time without disease progression, which are likely the key factors driving the longer TTD ⩾ 10% in global health status with ribociclib.
Clinical benefit is generally thought to be based on the three pillars of efficacy, safety, and HRQoL. PFS benefits have been observed with ribociclib combinations in all three MONALEESA trials.4,10–12 In the MONALEESA-3 and MONALEESA-7 trials, ribociclib combinations also demonstrated statistically significant benefits in OS5,13 (OS results from MONALEESA-2 had not been presented as of this writing because the data were not yet mature). The tolerability profile of ribociclib has been well established, and adverse events are generally manageable. Consistent with the maintenance of QoL with ribociclib in the MONALEESA-7 trial, separate analyses of PROs demonstrated that QoL was also maintained throughout treatment with the addition of ribociclib to letrozole in the MONALEESA-2 trial and the addition of ribociclib to fulvestrant in the MONALEESA-3 trial.14,15
In this analysis of MONALEESA-7, HRQoL was maintained for a longer duration with ribociclib than with placebo, with a HR of 0.67 (95% CI, 0.52–0.86) for TTD in EORTC QLQ-C30 global health status. To place these data into context, it should be noted that the HRQoL benefit of adding CDK4/6 inhibitors to endocrine therapy was less pronounced in other phase III trials, although cross-trial comparisons must be made with caution. For example, the HR for abemaciclib over placebo for time to sustained deterioration in EORTC QLQ-C30 global health status [based on a ⩾10% decrease (similar to TTD in this analysis) followed by all subsequent scores meeting the minimally important difference criteria compared with baseline] in the MONARCH-2 trial was 0.80 (95% CI, 0.63–1.02). 16 Also, although results for TTD in EORTC scores had not been reported for MONARCH-3 as of this writing, an analysis of HRQoL revealed that the addition of abemaciclib to an NSAI did not result in statistically significant and clinically meaningful differences in patient-reported global health status. 17 Additionally, while QoL results for palbociclib plus letrozole in the PALOMA-2 study did not include EORTC QLQ-C30 global health status, results for TTD in Functional Assessment of Cancer Therapy-Breast questionnaire scores were similar between the palbociclib and placebo arms [HR, 0.88 (95% CI, 0.67–1.16)]. 18 The results for MONARCH-3 and PALOMA-2 are particularly relevant because these trials are also investigating the addition of a CDK4/6 inhibitor to an NSAI in the first-line setting, albeit primarily in postmenopausal patients.
Given the substantial clinical benefit in terms of PFS and OS improvements seen in MONALEESA-7, it is noteworthy that ribociclib in combination with endocrine therapy also improves QoL in this patient population, which may include some women experiencing side effects from ovarian suppression. These data support the overall clinical and HRQoL benefits of CDK4/6 inhibitor-based combination therapy in treating premenopausal and perimenopausal patients with HR+/HER2− ABC.
Supplemental Material
Harbeck_TAMO,_Supplemental_Material_ML-7_PRO_Post-Review_22June2020 – Supplemental material for Health-related quality of life in premenopausal women with hormone-receptor-positive, HER2-negative advanced breast cancer treated with ribociclib plus endocrine therapy: results from a phase III randomized clinical trial (MONALEESA-7)
Supplemental material, Harbeck_TAMO,_Supplemental_Material_ML-7_PRO_Post-Review_22June2020 for Health-related quality of life in premenopausal women with hormone-receptor-positive, HER2-negative advanced breast cancer treated with ribociclib plus endocrine therapy: results from a phase III randomized clinical trial (MONALEESA-7) by Nadia Harbeck, Fabio Franke, Rafael Villanueva-Vazquez, Yen-Shen Lu, Debu Tripathy, Louis Chow, Govind K Babu, Young-Hyuck Im, David Chandiwana, Anil Gaur, Brad Lanoue, Karen Rodriguez-Lorenc and Aditya Bardia in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors thank the patients who participated in the study and their families. The study was approved by each participating site’s institutional review board or independent ethics committee and was conducted in compliance with the International Council for Harmonisation Good Clinical Practice Guideline and the general ethical principles of the Declaration of Helsinki. This study was sponsored by Novartis, which also provided financial support for medical editorial assistance by John McGuire of MediTech Media Ltd.
Authors’ note
Some of the data in this manuscript were presented in a poster discussion at ESMO 2019 (Lu Y-S, et al. Poster 308PD) and as a poster at SABCS 2019 (Harbeck N,
Contributors
All authors contributed to the conception or design of this analysis. All authors were involved in acquisition, analysis, or interpretation of the data; writing or reviewing and editing the manuscript; and approved the final version for submission. Study design was performed in conjunction with the study steering committee members and the sponsor. Data collection and analysis were completed by trial sponsor representatives. The corresponding author had complete access to all study data and had final responsibility for the decision to submit for publication.
Conflict of interest statement
NH reports personal fees from AstraZeneca, Novartis, Eli Lilly, Pfizer, all outside the submitted work; FF has nothing to disclose; RV-V reports personal fees from Novartis, Pfizer, Roche, outside the submitted work; Y-SL reports personal fees from Novartis, Pfizer, Boehringer Ingelheim, grants from Novartis, Roche, Merck Sharp & Dohme, Pfizer, GlaxoSmithKline, other from Novartis, outside the submitted work; DT reports personal fees from Novartis, Pfizer, grant from Novartis, outside the submitted work; LC has nothing to disclose; GKB has nothing to disclose; Y-HI has nothing to disclose; DC reports other from Novartis, during the conduct of the study; BL reports other from Novartis, during the conduct of the study; KR-L reports personal fees and other from Novartis, during the conduct of the study; AB reports personal fees from Biothemostics Inc, Pfizer, Novartis, Genentech, Merck, Radius Health, Immunomedics, Spectrum Pharma, Taiho, Sanofi, Daiichi Pharma, Puma, grants from Genentech, Novartis, Pfizer, Merck, Sanofi, Radius Health, Immunomedics, Mersana, Innocrin, Biothemostics Inc., outside the submitted work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded and designed by Novartis Pharmaceuticals Corporation.
Data sharing
A data sharing statement provided by the authors is included with the supplemental material.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
