Abstract
Background:
To investigate the impact of latent tuberculosis infection (LTBI) on the prognosis of non-small cell lung cancer (NSCLC) patients treated with anti-PD-1 immunotherapy and to assess the correlation between dynamic alterations in T-SPOT.TB results and prognosis.
Methods:
This retrospective cohort study analyzed clinical data from 127 patients with NSCLC who received anti-PD-1 therapy and underwent T-SPOT.TB testing at our institution between January 2020 and March 2024. Baseline imbalances between groups were addressed using inverse probability of treatment weighting (IPTW). Restricted cubic spline (RCS) modeling, Cox regression and other analyses were conducted both before and after IPTW.
Results:
Among the entire cohort, 50 patients were in the LTBI group and 77 in the Normal group. No significant differences were observed in mPFS or mOS between the two groups. RCS analysis revealed a nonlinear (U-shaped) relationship between pre-TSPOT values and OS. Patients with a T-SPOT positive (but value ⩽18) exhibited longer OS compared with the other two groups (HR = 0.13, 95% CI [0.03 ~ 0.54], P = .005; after IPTW HR = 0.21, 95% CI [0.05-0.90], P = .035). Among 63 patients monitored for dynamic TSPOT changes, 35 (55.56%) remained persistently negative, 15 (23.81%) remained persistently positive, 2 (3.17%) converted from negative to positive, and 11 (17.46%) converted from positive to negative. No significant differences in ORR, PFS, or OS across these groups.
Conclusions:
Although no statistically significant differences in treatment efficacy and prognosis were observed between the LTBI and Normal groups, this finding should not be interpreted as therapeutic equivalence, particularly given the limited sample size. Pre-treatment T-SPOT values showed a nonlinear (U-shaped) relationship with patient prognosis (OS). Lower pre-treatment T-SPOT value were associated with longer OS. The dynamic changes in T-SPOT during treatment were not significantly associated with outcomes. Four patients developed active tuberculosis during immunotherapy, with heterogeneous T-SPOT patterns, underscoring the need for TB monitoring in ICI-treated patients.
Keywords
Background
Lung cancer is one of malignant tumor with a high global incidence in both men and women, ranking as the leading cause of cancer-related deaths worldwide. 1 Non-small cell lung cancer (NSCLC), accounting for approximately 85% of lung cancer cases, is the most common subtype. 2 Unfortunately, over half of patients are diagnosed at advanced stages, with a five-year survival rate of about 5%. 3 In recent years, cancer immunotherapy has achieved significant progress, particularly through the emergence of immune checkpoint inhibitors (ICIs). By targeting PD-1 and PD-L1, ICIs have revolutionized the treatment of advanced cancers, notably improving progression-free survival (PFS) and overall survival (OS) in advanced NSCLC patients.4,5 In China, ICIs like pembrolizumab, tislelizumab, sintilimab, and camrelizumab, approved by the National Medical Products Administration (NMPA), have shown promising prognostic results. 6
The therapeutic effect of ICIs is mediated by T cells, with interferon-γ (IFN-γ) serving as a critical cytokine produced during normal T-cell activation. The capacity of T cells to release IFN-γ reflects, to a certain extent, the strength of T-cell-mediated immune function. The Mycobacterium tuberculosis (MTB) interferon-γ release assay (IGRA) leverages this principle by using phytohemagglutinin (PHA) as a non-specific stimulant to stimulate T cells and quantify the cells that release IFN-γ, serving as a positive control. Studies have shown a correlation between PHA-stimulated IFN-γ (PSIG) responses and both the disease control rate and OS in patients with advanced NSCLC undergoing chemotherapy. 7 PSIG has also been proposed as a potential biomarker for predicting responses to ICIs treatment in NSCLC patients. 8 T-SPOT.TB, commonly referred to as T-SPOT, is one of the most widely used IGRA tests. It employs an enzyme-linked immunospot (ELISPOT) assay to detect effector T cells in peripheral blood that release IFN-γ when stimulated by MTB-specific antigens. 9 This method provides quantitative results by directly measuring the number of T cells producing IFN-γ. While originally used to identify latent tuberculosis infection (LTBI), T-SPOT.TB also indirectly reflects the host’s systemic T-cell responsiveness. 10 Because ICIs rely on competent and functional T cells to mediate antitumor effects, 11 T-SPOT results may serve as a surrogate marker of T-cell activity and, by extension, ICI responsiveness. Furthermore, LTBI represents a chronic immune stimulation environment. Low-level MTB antigen presence might sustain a balanced immune tone that favors immune activation without tipping into immune exhaustion, thereby influencing cancer immunotherapy outcomes. 12 Given the high prevalence of tuberculosis in our country, T-SPOT is routinely performed in cancer patients before initiating ICIs therapy.
Studies suggest that LTBI may play a dual role in cancer progression and treatment. Components of Mycobacterium tuberculosis have been found to enhance dendritic cell (DC)-based antitumor immunotherapy by promoting antigen presentation and T-cell activation. 13 On the other hand, LTBI has been implicated in modulating systemic immunity, potentially influencing tumor immune escape and progression.12,14 However, research specifically addressing the impact of LTBI on cancer immunotherapy outcomes remains limited. In high TB burden regions like China, a significant proportion of cancer patients harbor LTBI. Most existing studies have focused on PSIG responses to chemotherapy or ICIs,7,8 while the role of T-SPOT results, particularly their dynamic changes during immunotherapy, remains underexplored. Previous studies have shown that IGRA levels are significantly correlated with high body-mass-index (BMI), high neutrophil-lymphocyte ratio (NLR), and low serum albumin (ALB) 15 while advanced age and overweight (BMI ⩾ 25 kg/m2) are significantly associated with false-negative T-SPOT results. 16 Additionally, nutritional indicators like nutrition screening 2002 (NRS2002), prognostic nutritional index (PNI), and inflammatory markers such as NLR, and monocyte-lymphocyte ratio (MLR) exhibit significant differences between elderly and younger patients. 17 To avoid the potential confounding effects of these factors on LTBI detection and cancer treatment outcomes, we conducted statistical analyses alongside our primary investigation. Thus, we conducted a retrospective study to investigate the prognostic value of pre-treatment IGRA results and post-treatment T-SPOT dynamics changes in predicting the efficacy of ICIs therapy in advanced NSCLC patients. By exploring these dynamics, we aim to clarify the potential impact of LTBI on immunotherapy outcomes and provide insights for clinical practice.
Materials and Methods
Study design and participants
Medical records of 450 NSCLC patients treated at Zhongshan Hospital (Xiamen), Fudan University, between January 1, 2020, and March 30, 2024, were retrospectively reviewed. Of these, 234 patients who did not receive immunotherapy were excluded. An additional 89 patients were excluded as they had not undergone T-SPOT testing before treatment. Ultimately, 127 evaluable patients were included in the study for further analysis. LTBI is defined as the absence of clinical or radiological evidence of active tuberculosis, but with a positive result on T-SPOT. 18 Patients were stratified into groups based on their LTBI status for comparative analysis. Among the 127 patients, 63 underwent repeat T-SPOT testing during the course of treatment. This subgroup was analyzed separately to evaluate the correlation between dynamic changes in T-SPOT results and patient prognosis (Supplemental Figure 1). The dynamic change in T-SPOT was calculated by subtracting the pre-treatment T-SPOT value from the post-treatment T-SPOT value. A negative value indicates a decrease in T-SPOT after treatment, while a positive value indicates an increase.
The criteria for inclusion and exclusion
Inclusion criteria: Patients aged ⩾18 years with a pathological and radiographic diagnosis of stage IV, IIIB, IIIC, or inoperable stage IIIA NSCLC, or those with extensive postoperative recurrence. They must have received at least two cycles of immune checkpoint inhibitors (ICIs) ± chemotherapy, with only first-line ICI treatments (such as pembrolizumab, tislelizumab, camrelizumab, and sintilimab) considered for those who have undergone multiple lines of ICIs therapy. T-SPOT testing was conducted prior to ICI treatment, and standard treatment and follow-up data were complete.
Exclusion criteria: Patients with SCLC, severe underlying conditions affecting the heart, brain, liver, kidneys, or other major organs; active pulmonary or extrapulmonary tuberculosis prior to treatment; autoimmune diseases or long-term use of immunosuppressive therapy; severe infections; or other conditions that significantly impair immune function.
Data collection
General and disease-specific data were obtained from the hospital information system (HIS), including age, gender, performance status (PS), pathology, smoking history, TNM stage, ICIs, combination therapies, irAEs, PD-L1 expression, T-SPOT results, and laboratory parameters (lymphocytes, neutrophils, monocytes, lactate dehydrogenase [LDH], and albumin) prior to immunotherapy initiation.
Nutritional status and inflammation markers
The NRS2002 score 19 assessed nutritional risk based on nutritional impairment (weight changes, dietary intake, BMI), nutritional needs (illness severity, 0-3 points), and age (1 point for ⩾70 years), with ⩾3 indicating high risk. The PNI 20 calculated as albumin (g/L) + 5 × lymphocyte count (×109/L), was also used to assess nutritional status. Systemic inflammation was evaluated using the NLR and MLR. 21
Assessment of T-SPOT.TB
The T-SPOT.TB test, conducted using the Oxford Immunotec kit, followed the supplier’s protocol (https://www.tspot.com/wp-content/uploads/2020/09/TB-PI-US-0001-V8.pdf). A positive result was defined as: (1) test well spots minus negative control spots ⩾6 (negative control spots: 0-5), or (2) test well spots at least twice the negative control spots (negative control spots ⩾6). The T-SPOT.TB value was calculated as antigen A + antigen B spots minus blank control spots.
Evaluation of efficiency
All patients received at least two cycles of ICIs, including pembrolizumab, camrelizumab, tislelizumab, or sintilimab, administered intravenously at 200 mg every 3 weeks. Treatments included ICIs alone, combinations with platinum-based or non-platinum chemotherapy, and regimens involving anti-angiogenic agents or radiotherapy.
The response was evaluated every 6-8 weeks using RECIST 1.1, 22 categorizing outcomes as complete response (CR), partial response (PR), stable disease (SD), or progressive disease (PD). ORR was defined as the proportion of patients achieving CR or PR. The primary endpoint, OS, is the time from ICI initiation to death from any cause, while the secondary endpoint, PFS, is the time to first PD or death. Follow-up ends on September 30, 2024.
Statistical analysis
The Shapiro-Wilk test was employed to assess the normality of data distributions. Categorical variables were summarized as counts and percentages, with group comparisons conducted using the Chi-squared test or Fisher’s exact test, as appropriate. For non-normally distributed quantitative data, medians along with interquartile ranges (Q1: first quartile, Q3: third quartile) were reported, and the Mann-Whitney U test was utilized for comparisons between two independent groups. Pearson correlation analysis was performed to evaluate relationships between variables.
Inverse probability of treatment weighting (IPTW) (WeightIt, survey and cobalt packages) was applied based on propensity scores estimated with treatment weighting under the average treatment effect on the treated (ATT) scheme. Covariate balance before and after weighting was assessed using standardized mean differences (SMDs) and visualized via Love plots. Truncated IPTW (IPTW-trunc) was additionally performed, restricting weights at the 1st and 99th percentiles to reduce the influence of extreme weights, serving as a sensitivity analysis.
Restricted cubic spline (RCS) models (splines package) were employed to examine potential nonlinear associations between continuous predictors and survival outcomes. The P for overall and P for nonlinear were reported. Based on the shape of RCS curves, threshold-effect analyses were further performed using segmented Cox regression (segmented package) to identify inflection points, with likelihood ratio tests (LRTs) comparing segmented versus linear models. Informed by these results, T-SPOT values were stratified into relevant groups, which were subsequently included as categorical exposures in Cox regression models. Survival analyses were conducted using Kaplan-Meier estimators, with differences between groups assessed via the Log-rank test. Univariate Cox regression was conducted to identify prognostic factors. Variables with a P-value less than .1 in univariate Cox proportional hazards models were included in the multivariate Cox regression analysis, while T-SPOT grouping was forced into all multivariable models regardless of significance.
Receiver operating characteristic (ROC) curves were generated, and the area under the curve (AUC) was calculated to evaluate predictive performance. The optimal cutoff value was determined by maximizing Youden’s index, balancing sensitivity and specificity. No a priori sample size calculation was performed, as this was a retrospective study. The number of patients included was based on all eligible cases meeting the inclusion criteria during the study period. A two-tailed P-value <.05 was considered statistically significant for all analyses. Data were processed and curves plotted using R 4.4.3and Python 3.12.3. This study adheres to the STROBE statement 23 (Supplemental Table 1) for reporting.
Results
Characteristics of the patients
A total of 127 NSCLC patients who treated with ICIs were enrolled. Among these, there were 114 males (89.76%) and 13 females (10.24%). A total of 63 patients were <65 years old (49.61%), while 64 were 65 years or older (50.39%). There were 54 non-smokers (42.52%) and 73 former or current smokers (57.48%). Pathologically, 60 cases of squamous cell carcinoma (47.24%) and 67 cases of non-squamous NSCLC (52.76%). There were 50 patients in stage III (39.37%) and 77 patients in stage IV (60.63%). PD-L1 expression was less than 1% in 30 patients (23.62%), positive in 65 patients (51.18%), and unknown in 32 patients (25.20%). With regards to performance status, 69 patients had an ECOG-PS of 0 (54.33%), while 58 had a score of 1 or higher (45.67%). 104 patients received first-line immunotherapy (81.89%), while 23 received second-line or later therapy (18.11%). According to the NRS2002 nutritional risk screening, 93 patients were classified as low nutritional risk (73.23%), and 34 were at high nutritional risk (26.77%). Immune-related adverse events (irAEs) occurred in 38 patients (29.92%), while 89 patients (70.08%) experienced no irAEs. Total of 63 patients achieved disease remission, resulting in an ORR of 49.61%. The pre-treatment T-SPOT results were 2.0 [0.00, 18.00], with values ranging from 8 to 80 in the LTBI group and 0 to 4 in the normal group. According to the results provided by the test kits, there were 50 cases in the LTBI group (39.37%) and 77 cases in the normal group (60.63%). Apart from smoking status (P = .013), treatment line (P = .032), and T-SPOT results (P < .001), no significant statistical differences were observed between the two groups. After IPTW under the ATT estimated, baseline covariates were well balanced between the treated and control groups, with all standardized mean differences <0.1 and all weighted comparison P-values >.05 (Table 1, Figure 1). There were 4 patients who had a confirmed history of pulmonary tuberculosis and had undergone anti-tuberculosis treatment, indicating cases of healed tuberculosis. All patients were checked to ensure they did not have active pulmonary or ex-pulmonary tuberculosis before starting the treatment.
Demographic and clinical characteristics of All patients before and after IPTW.
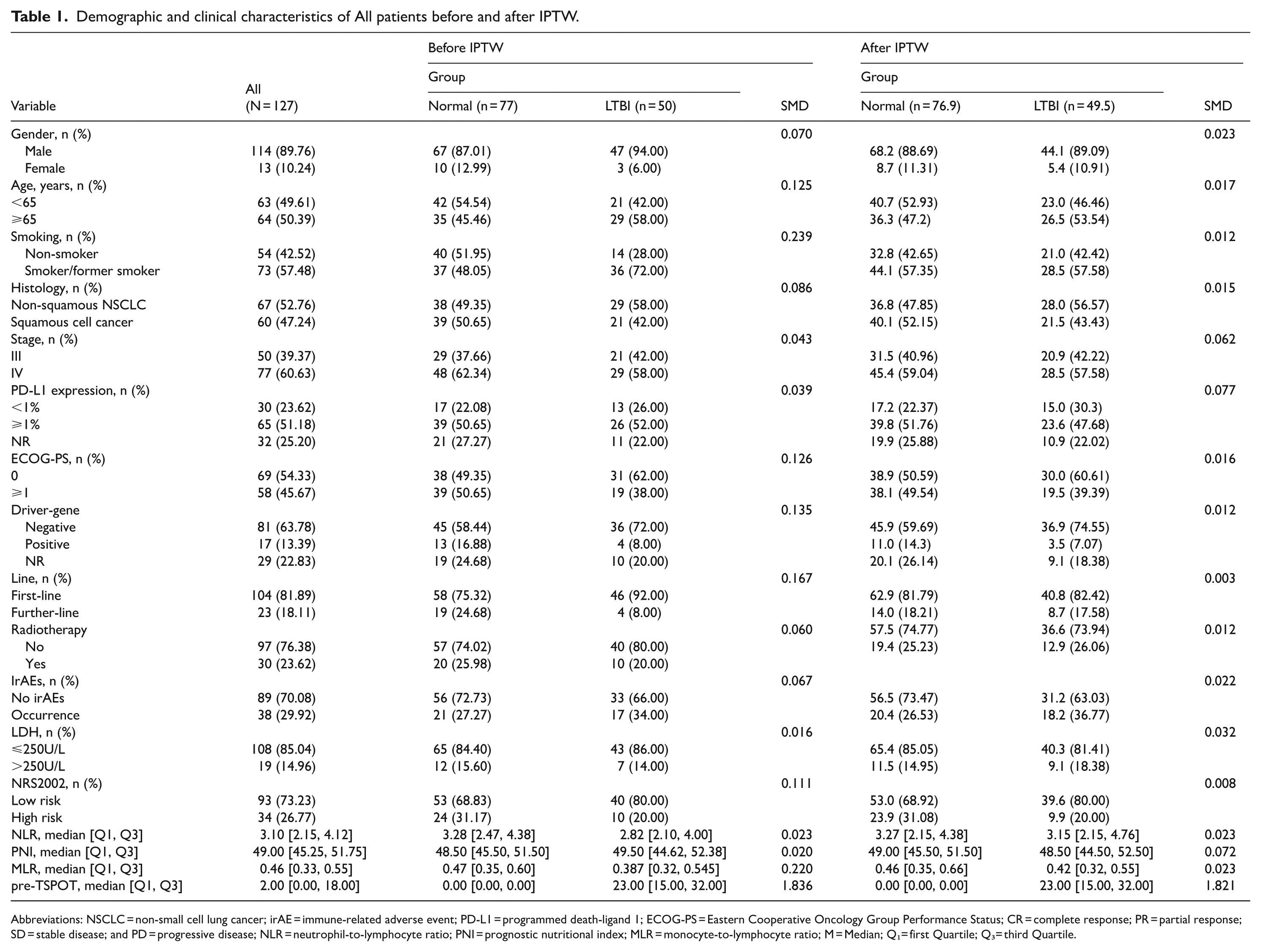
Abbreviations: NSCLC = non-small cell lung cancer; irAE = immune-related adverse event; PD-L1 = programmed death-ligand 1; ECOG-PS = Eastern Cooperative Oncology Group Performance Status; CR = complete response; PR = partial response; SD = stable disease; and PD = progressive disease; NLR = neutrophil-to-lymphocyte ratio; PNI = prognostic nutritional index; MLR = monocyte-to-lymphocyte ratio; M = Median; Q₁= first Quartile; Q₃= third Quartile.

Love plot for inverse probability of treatment weighting (IPTW).
The Pearson correlation analysis was conducted to assess the relationship between pre-treatment T-SPOT results and various clinical and biological markers, including NLR, age, NRS2002, MLR, and PNI. The analysis revealed that there was no significant correlation between pre-treatment T-SPOT results and the other assessed factors (Figure 2).
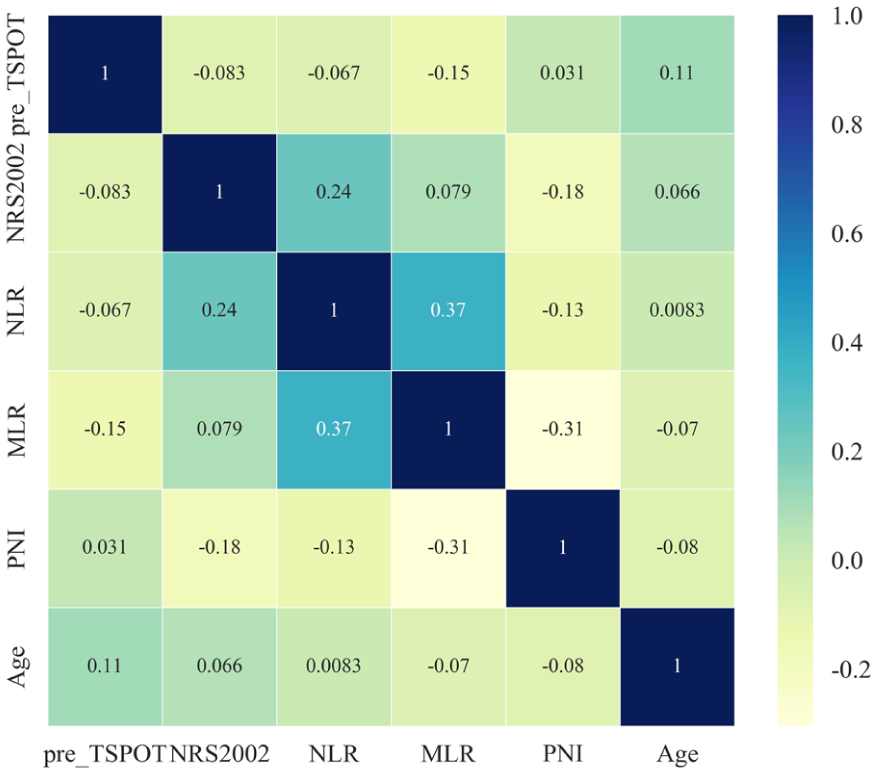
Correlation analysis for pre-SPOT with other factors.
Efficacy and prognostic evaluation of immunotherapy in the LTBI and normal groups
As of September 30, 2024, the median follow-up time for the total population was 15 months (10, 22 months). Disease progression was observed in 71 patients (55.91%), and 42 patients (33.07%) experienced all-cause of death. The ORR in the LTBI group was 54%, compared with 46.75% in the normal group, with no significant difference between the two groups (P = .425). In terms of PFS, the LTBI group had a median PFS (mPFS) of 12 months (95% CI, 10-NA), while the Normal group had a mPFS of 13.5 months (95% CI, 12-25). Regarding OS, the LTBI group had a median OS (mOS) of 35.5 months (95% CI, 26-NA), and the Normal group had a mOS of 33 months (95% CI, 20-NA). There were no statistically significant differences in mPFS or mOS between the two groups (P = .81 and P = .39, respectively). After applying IPTW, no significant difference in mOS was observed between the treatment and control groups (P > .05). A comparison of the Kaplan–Meier survival curves for the two groups is provided in Supplemental Figure 2. However, this should not be interpreted as equivalence in outcomes, especially given the limited sample size and retrospective nature of the study.
Survival analysis and linear relationship analysis
To evaluate the prognostic significance of pre-TSPOT levels, we performed RCS analysis adjusted for NRS2002, NLR, MLR, PD-L1, and LDH—factors selected a priori due to their known prognostic relevance in NSCLC patients receiving ICIs. The RCS model revealed a significant nonlinear association between T-SPOT levels and OS (P for overall = .047; P for nonlinear = 0.022 before IPTW, while P for overall = .062; P for nonlinear = .031), with a U-shaped hazard curve (Figure 3A and C).
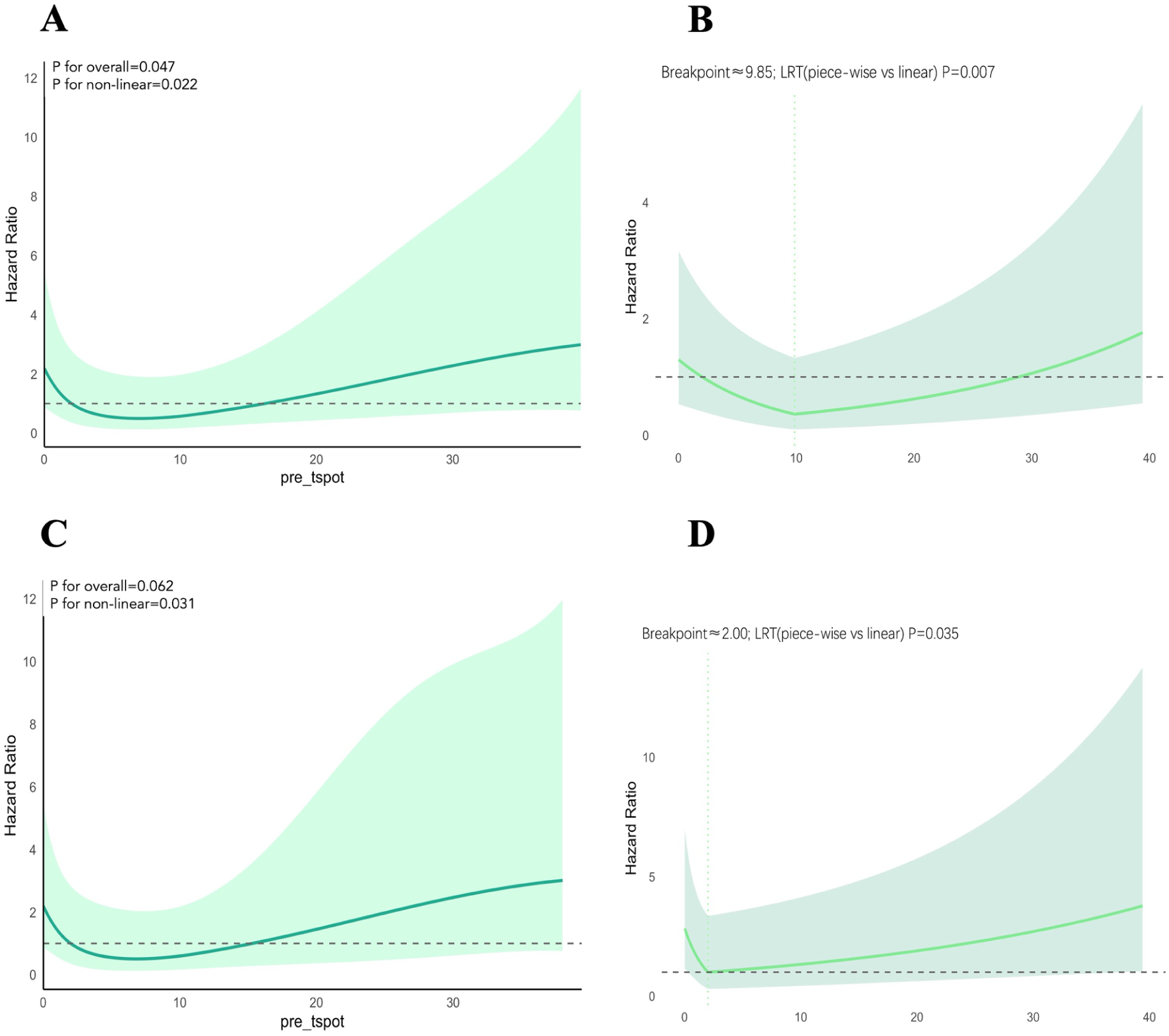
The Restricted Cubic Spline (RCS) for OS (A) and threshold-effect analysis (B) before IPTW; Restricted Cubic Spline (RCS) for OS (C) and threshold-effect analysis (D) after IPTW.
To further refine this relationship, we performed a threshold-effect analysis using a piecewise linear regression model. In the unweighted analysis, the algorithm identified an inflection around T-SPOT ≈ 9.85 (while no float value): the slope below 10 suggested a decrease in risk per unit increase (HR/unit = 0.88, 95% CI 0.79-0.97, P = .013), whereas ⩾10 showed an increase (HR/unit = 1.06, 95% CI 1.02-1.09, P = .002). The segmented model fit significantly better than a simple linear model (likelihood ratio P = .007). Under IPTW weighting, the data favored an earlier inflection at T-SPOT ≈ 2: <2 showed a protective slope (HR/unit = 0.59, 95% CI 0.36-0.98, P = .040), while ⩾2 was associated with increasing risk (HR/unit = 1.04, 95% CI 1.00-1.07, P = .023), and the segmented model again outperformed a linear specification (P = .034) (Table 2, Figure 3B and D).
Threshold-effect analysis and corresponding statistical values before and after IPTW.

Sensitivity analyses
To examine the robustness of our findings, we performed comprehensive sensitivity analyses under varying modeling conditions, including different spline degrees of freedom, IPTW truncation schemes, and candidate breakpoints (Table 3). Across these analyses, the direction and magnitude of the associations remained largely consistent.
Sensitivity analyses across alternative cutoff points, spline degrees of freedom, and IPTW truncation schemes.
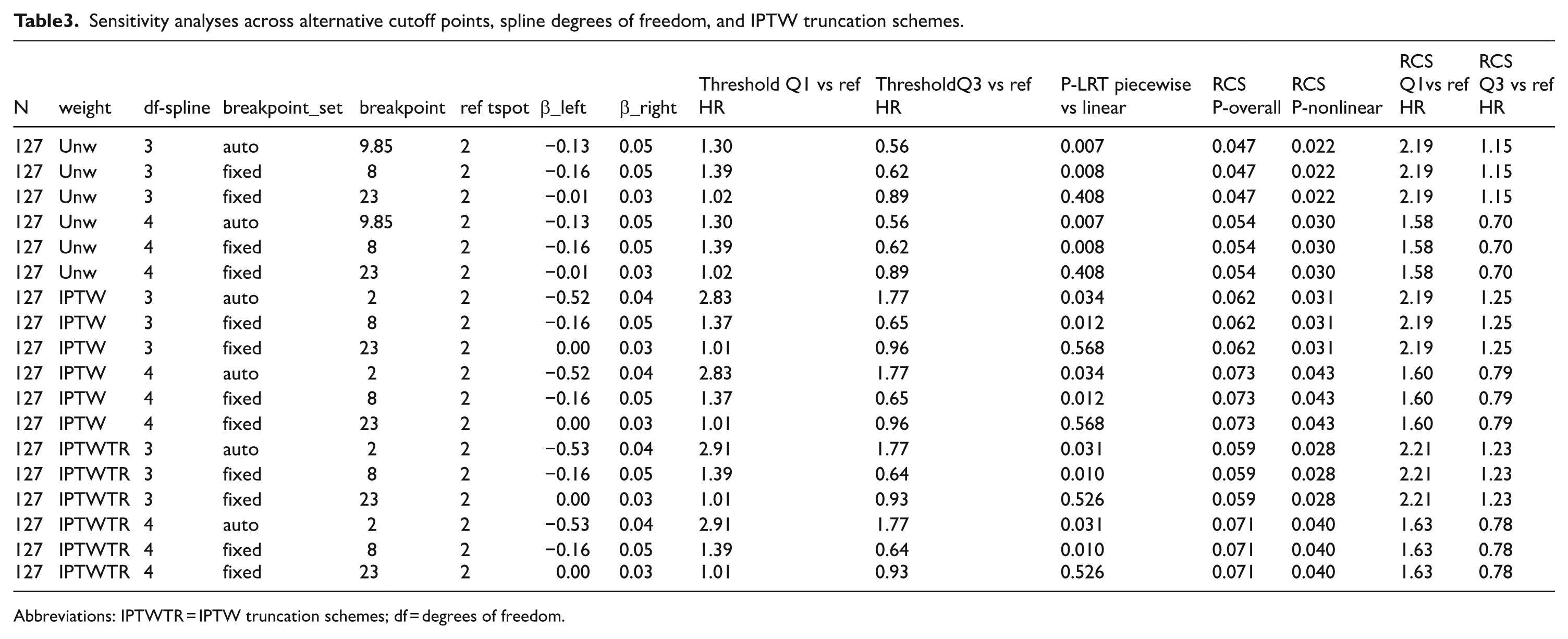
Abbreviations: IPTWTR = IPTW truncation schemes; df = degrees of freedom.
Specifically, when using cutoff values around 8 or 10, the piecewise models consistently outperformed the linear specification, confirming that values below this range were associated with significantly elevated risk (Supplemental Figure 3). In contrast, a higher cutoff at 23 did not demonstrate statistical significance across weighting schemes, although it coincided with the median level of the LTBI population. Notably, adopting the 75th percentile (Q3 = 18) as an upper threshold yielded the stable model fit, with hazard ratios remaining significant and consistent across different model specifications.
Subsequently, we conducted univariate Cox regression to screen for relevant prognostic factors. Variables with a P-value <.1 were selected for inclusion in the multivariable models. Survival analysis showed that patients in the 8 ~ 18 subgroup had significantly longer OS compared to the other groups (HR = 0.13, 95% CI [0.03 ~ 0.54], P = .005). Regarding OS, high nutritional risk (HR = 3.63, 95% CI [1.77-7.46], P < .001) and elevated LDH levels (50 U/L; HR = 2.60, 95% CI [1.18-5.70], P = .017) were associated with shorter OS. Furthermore, NLR (HR = 1.18, 95% CI [1.08-1.30], P < .001) was identified as an independent predictor of OS. After applying IPTW to further reduce potential confounding, the associations remained robust. Both high nutritional risk (HR = 3.60, 95% CI [1.70 ~ 7.63], P = .001) and elevated NLR (HR = 1.24, 95% CI [1.11 ~ 1.39], P < .001) consistently predicted worse OS, while patients in the 8 to 18 subgroup continued to derive a significant survival benefit. The hazard ratios and P-values after IPTW adjustment are provided in Table 4.
Survival analysis for all patients of OS (before and after IPTW).

Abbreviations: HR = hazard ratio, CI = confidence interval; NSCLC = non-small cell lung cancer; irAE = immune-related adverse event; PD-L1 = programmed death-ligand 1; ECOG-PS = Eastern Cooperative Oncology Group Performance Status; NLR = neutrophil-to-lymphocyte ratio; PNI = prognostic nutritional index; MLR = monocyte-to-lymphocyte ratio.
Analysis of pre-TSPOT, post-TSPOT, and TSPOT changes
In this study, a total of 127 patients were initially enrolled. However, due to the follow-up period, only 63 patients completed both pre-treatment and post-treatment T-SPOT tests, and therefore, the final sample size for T-SPOT change analysis was 63 patients. A comparative analysis was conducted between the baseline characteristics of the 63 patients who underwent re-testing and the 127 initial patients. The analysis revealed no significant differences in key clinical features such as gender, age, disease stage, and other baseline characteristics (Supplemental Table 2 and Supplemental Table 3). This indicates that the 63 patients who underwent follow-up testing were representative of the initial cohort. The follow-up TSPOT tests were completed within 3 to 6 months after treatment.
Among the 63 patients who underwent dynamic T-SPOT monitoring, 35 patients (55.56%) remained persistently negative, 15 patients (23.81%) remained persistently positive, 2 patients (3.17%) converted from negative to positive, and 11 patients (17.46%) converted from positive to negative. No significant differences were found in the ORR, PFS or OS across the four groups. The P-values for ORR, PFS, and OS were .654, .913, and .701, respectively (Table 5).
The efficacy or prognosis for different dynamic changes in T-SPOT.
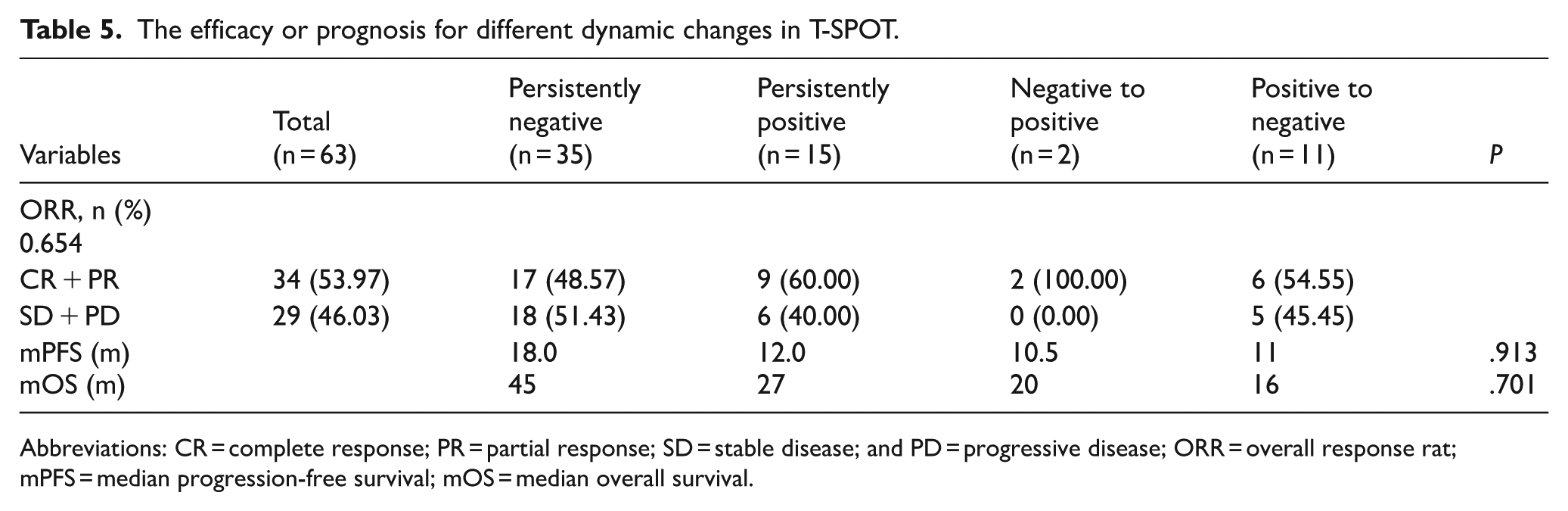
Abbreviations: CR = complete response; PR = partial response; SD = stable disease; and PD = progressive disease; ORR = overall response rat; mPFS = median progression-free survival; mOS = median overall survival.
ROC curve analysis for the T-SPOT change revealed an AUC of 0.659 (95% CI: 0.562-0.765) for predicting survival (death or survival). The optimal cutoff value for T-SPOT change was determined using the maximum Youden’s index, which was -9. Further survival analysis was performed using KM curves and the log-rank test. The results showed no significant difference in the mPFS or mOS between the two groups when comparing patients with a T-SPOT change ⩾-9 to those change<-9 (mPFS: 18 vs 11 months; P = .234, mOS: 45 vs 26 months; P = .055, respectively), (Figure 4).

ROC curve of T-SPOT dynamic changes and survival (A); KM curve for PFS (B) and OS (C).
Tuberculosis reactivation and T-SPOT dynamics
During follow-up, four patients developed microbiologically or radiologically confirmed active tuberculosis. Among them, one patient had persistently negative T-SPOT results, two showed T-SPOT conversion from positive to negative, and one remained persistently positive but with a declining T-SPOT value. Of these four cases, three had reached OS endpoints.
Discussion
Comprehensive meta-analysis has demonstrated that cancer patients face a notably higher risk of developing tuberculosis compared with the general populace, with lung cancer patients experiencing a sixfold increase. 24 Lung cancer and tuberculosis often present with overlapping symptoms, and it is not uncommon for both to be diagnosed concurrently or to influence each other’s development. The chronic inflammation, genomic alterations, and fibrosis typical of tuberculosis are significant factors that may contribute to cancerous processes. Similarly, the progression of lung cancer and the treatments aimed at combating it can activate previously dormant tuberculosis infections. 25
Numerous case reports have confirmed that the use of ICIs can lead to the recurrence of tuberculosis.26,27 A meta-analysis has indicated that treatment with anti-PD-1/PD-L1 therapies disrupts the immune control of LTBI, thereby increasing the activity of Mycobacterium tuberculosis and the incidence of active tuberculosis disease. The mortality rate associated with tuberculosis in patients undergoing PD-1/PD-L1 blockade therapy is extremely high. 28 Interestingly, research conducted by Tezera et al 29 has shown that hypoxia within tuberculosis lesions actually leads to an upregulation of the PD-1/PD-L1 axis proteins. This finding is particularly significant given that most current studies generally agree that the efficacy of ICIs in treating NSCLC, especially in patients with adenocarcinoma, is correlated with the levels of PD-L1 expression. 30 This suggests a complex interplay between the tumor microenvironment, infectious disease factors like tuberculosis, and the immunological responses influenced by therapies targeting PD-1/PD-L1. 12
In this study, we analyzed a population of NSCLC patients undergoing treatment with ICIs, with 39.37% (50/127) individuals identified as having LTBI. Although no significant differences were observed in ORR, PFS, or OS between the LTBI and normal groups receiving ICI therapy, this does not imply therapeutic equivalence. Given the sample size and retrospective design, the possibility of a Type II error (false negative) should be considered. Another Chinese cohort study 31 included 45, 21, and 32 cancer patients with active, latent, and old tuberculosis, with NSCLC accounting for 79.4%. The ORR to anti-PD-L1 treatment were 34.2%, 35.5%, and 41.2%, and mPFS were 8, 6, and 6 months, respectively, with no significant differences. 31 However, the study had only 21 LTBI cases and did not compare outcomes with a non-tuberculosis population. Additionally, our multivariable analysis indicated that long-term survival in NSCLC patients receiving ICIs, particularly OS, is associated with factors such as NRS2002, inflammatory markers like NLR, and LDH levels. Previous research has consistently highlighted the relationship between tumor survival and factors such as nutritional status 32 and systemic inflammation markers. 33 Our study corroborates these findings, underscoring the significant impact that both nutritional health and inflammatory markers have on the outcomes of tumor diseases. This reaffirms the importance of addressing nutritional needs and managing inflammation as part of comprehensive cancer care to optimize patient survival. Notably, the U-shaped relationship between pre-treatment T-SPOT.TB values and overall survival may reflect an immune “Goldilocks zone” hypothesis. Moderate T-SPOT levels (approx. 8 ~ 18) may represent an optimal immune tone 34 —sufficient T-cell function without excessive chronic stimulation—allowing effective ICI-mediated tumor control. In contrast, very low values may indicate immunosuppression or T-cell anergy, 35 while very high values may reflect chronic MTB antigen exposure, leading to T-cell exhaustion or IFN-γ–driven inflammation that hinders antitumor immunity. 36 Similar U-shaped prognostic patterns have been reported for immune biomarkers such as NLR and IL-6 in patients treated with ICIs. 37 These findings support the idea that both immune hypo- and hyper-responsiveness can impair immunotherapy outcomes, and pre-TSPOT may serve as a surrogate marker of this balance.
Beyond statistical significance, clinical relevance is essential in interpreting survival outcomes. After IPTW, the adjusted hazard ratio of 0.21 (95% CI: 0.05-0.90) for OS in the T-SPOT positive ⩽18 group indicates a notable reduction in mortality risk. According to the MCID framework recently proposed by Horita et al, 38 this effect size likely exceeds the threshold for clinical importance. Together, these results underscore the prognostic value of pre-treatment T-SPOT levels in advanced NSCLC patients receiving ICIs.
The low burden of tuberculosis antigens may also relate to a state of immune equilibrium post-MTB infection, which could influence the outcomes in these patients. The observed survival differences based on varying TB antigen loads could be linked to the need for a delicate balance between the immune system’s control of latent tuberculosis infection caused by MTB and the suppression of pathogen growth based on adequate immune cell activation. This balance is crucial to prevent excessive inflammation and the associated pathological damage.39,40 Effective control of latent TB involves regulating immune responses to prevent both uncontrolled bacterial proliferation and overwhelming inflammatory damage, which could compromise patient health and impact the outcomes of other conditions such as cancer. This balance might explain why patients with lower TB antigen loads, reflecting a potentially more stable immune environment, appear to have better survival outcomes. T-SPOT.TB can quantify IFN-γ production by peripheral T cells, potentially predicting the response of NSCLC patients to ICI treatment. Serving as a method to evaluate T-cell-mediated immune responses, 41 it offers a distinctive perspective on how NSCLC patients respond to ICIs. 8 Another study mentioned that patients with higher pre-treatment IGRA values, compared with those with lower levels (ELISA, with a cutoff value of 7.06 IU/ml), appeared to achieve longer mPFS. 10
A prospective study in Japan retested lung cancer patients treated with ICIs for T-SPOT before treatment and at 6 and 12 months post treatment. 42 The study found a 3.3% conversion rate (negative to positive) for T-SPOT and a 1.6% incidence of active tuberculosis. However, among the study population, only 18 cases were positive for pre-treatment TSPOT.TB, and the study did not further explore the relationship between T-SPOT.TB results and the efficacy and prognosis of immunotherapy. In our study, 2 patients exhibited conversion from negative to positive, and 11 patients converted from positive to negative.
Tuberculosis reactivation was observed in four patients, its relationship with T-SPOT dynamics remains unclear. Interestingly, three of these patients had experienced either persistently negative or declining T-SPOT values. This could suggest that loss of MTB-specific T-cell surveillance might predispose to reactivation, but the small number of cases limits interpretation. Previous studies have reported TB reactivation as a rare but serious complication of ICIs, particularly in TB-endemic regions. Our findings support the need for ongoing vigilance and longitudinal immune monitoring, especially in LTBI-positive patients undergoing immunotherapy.
Although there were no significant differences in efficacy and survival among those with persistent positivity, persistent negativity, conversion from negative to positive, or conversion from positive to negative, three of the four patients with active tuberculosis died, reflecting the complex interaction between ICI treatment and TB infection.
The efficacy of ICIs is mediated by T cells. Prolonged treatment with ICIs may lead to both activation and exhaustion of T-cell functions.11,43 Since the T-SPOT test also relies on T cells to measure the release of IFN-γ, the dynamic changes in T-SPOT can, to a certain extent, represent the in vivo efficacy of T-cell activity. This suggests that the fluctuations in T-SPOT results could serve as an indicator of the immunological changes occurring during ICI therapy, potentially reflecting both the enhancement of immune response early in the treatment and the subsequent T-cell exhaustion. In the study, although there were no significant differences in survival between groups with changes in T-SPOT results <-9 vs ⩾-9, we observed a trend toward prolonged OS in patients with smaller decreases or increases in post-treatment T-SPOT (change ⩾ -9) (P = .055). Although a study from Japan indicated that patients with stable IGRA levels (using ELISA, value <10 IU/ml) had better efficacy and longer treatment duration compared with those with lower pre-treatment or post-treatment IGRA values. 44 Due to the limited sample size, it remains uncertain whether this trend can manifest as a significant difference, and further research with expanded sample sizes and analyses is required.
This study has several limitations. Its retrospective, single-center design introduces potential selection bias and limits generalizability. No pre-specified sample size calculation was performed. Subgroup analyses of T-SPOT dynamics (n = 63)—particularly conversions (e.g., n = 2)—were clearly underpowered, and nonsignificant findings should be interpreted with caution. Additionally, tuberculosis reactivation occurred in four patients with heterogeneous T-SPOT patterns, but the low event rate limits the interpretability of these findings.
Overall, these findings should be considered exploratory. Larger, prospective studies with rigorous confounder adjustment (e.g., propensity score methods) are needed to validate the prognostic value of T-SPOT levels and their dynamics in ICI-treated NSCLC patients.
Conclusions
No significant differences in ORR, PFS, or OS were observed between the LTBI and normal groups receiving ICI therapy. High-risk NRS2002 and higher LDH (>250 U/L) were also linked to shorter OS. Pre-TSPOT levels showed a nonlinear (U-shaped) relationship with prognosis, suggesting a potential “immune balance” zone favoring ICI benefit. Lower pre-TSPOT values (positive but ⩽18) were associated with longer OS. NLR was an independent predictor for OS. After IPTW, both high nutritional risk and elevated NLR consistently predicted worse OS, while patients in the 8 to 18 subgroup continued to derive a significant survival benefit. Dynamic changes in T-SPOT during treatment were not significantly associated with outcomes, likely due to limited subgroup size. Four patients developed active tuberculosis during ICI therapy, but varied T-SPOT patterns and low event frequency preclude firm conclusions. These findings are exploratory and require prospective validation in larger cohorts.
Supplemental Material
sj-docx-4-onc-10.1177_11795549251394955 – Supplemental material for Impact of Latent Tuberculosis Infection and T-SPOT.TB Dynamics Alterations on Prognosis in Advanced NSCLC Treated With ICIs——IPTW-Based Retrospective Study
Supplemental material, sj-docx-4-onc-10.1177_11795549251394955 for Impact of Latent Tuberculosis Infection and T-SPOT.TB Dynamics Alterations on Prognosis in Advanced NSCLC Treated With ICIs——IPTW-Based Retrospective Study by Yijiao Xu, Jianying Liu, Qingwei Zhang, Yijun Song, Shuwen Yang, Haiyan Chen, Congyi Xie, DaWei Yang, Zhisheng Chen and Hongni Jiang in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-5-onc-10.1177_11795549251394955 – Supplemental material for Impact of Latent Tuberculosis Infection and T-SPOT.TB Dynamics Alterations on Prognosis in Advanced NSCLC Treated With ICIs——IPTW-Based Retrospective Study
Supplemental material, sj-docx-5-onc-10.1177_11795549251394955 for Impact of Latent Tuberculosis Infection and T-SPOT.TB Dynamics Alterations on Prognosis in Advanced NSCLC Treated With ICIs——IPTW-Based Retrospective Study by Yijiao Xu, Jianying Liu, Qingwei Zhang, Yijun Song, Shuwen Yang, Haiyan Chen, Congyi Xie, DaWei Yang, Zhisheng Chen and Hongni Jiang in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-6-onc-10.1177_11795549251394955 – Supplemental material for Impact of Latent Tuberculosis Infection and T-SPOT.TB Dynamics Alterations on Prognosis in Advanced NSCLC Treated With ICIs——IPTW-Based Retrospective Study
Supplemental material, sj-docx-6-onc-10.1177_11795549251394955 for Impact of Latent Tuberculosis Infection and T-SPOT.TB Dynamics Alterations on Prognosis in Advanced NSCLC Treated With ICIs——IPTW-Based Retrospective Study by Yijiao Xu, Jianying Liu, Qingwei Zhang, Yijun Song, Shuwen Yang, Haiyan Chen, Congyi Xie, DaWei Yang, Zhisheng Chen and Hongni Jiang in Clinical Medicine Insights: Oncology
Supplemental Material
sj-jpg-1-onc-10.1177_11795549251394955 – Supplemental material for Impact of Latent Tuberculosis Infection and T-SPOT.TB Dynamics Alterations on Prognosis in Advanced NSCLC Treated With ICIs——IPTW-Based Retrospective Study
Supplemental material, sj-jpg-1-onc-10.1177_11795549251394955 for Impact of Latent Tuberculosis Infection and T-SPOT.TB Dynamics Alterations on Prognosis in Advanced NSCLC Treated With ICIs——IPTW-Based Retrospective Study by Yijiao Xu, Jianying Liu, Qingwei Zhang, Yijun Song, Shuwen Yang, Haiyan Chen, Congyi Xie, DaWei Yang, Zhisheng Chen and Hongni Jiang in Clinical Medicine Insights: Oncology
Supplemental Material
sj-jpg-2-onc-10.1177_11795549251394955 – Supplemental material for Impact of Latent Tuberculosis Infection and T-SPOT.TB Dynamics Alterations on Prognosis in Advanced NSCLC Treated With ICIs——IPTW-Based Retrospective Study
Supplemental material, sj-jpg-2-onc-10.1177_11795549251394955 for Impact of Latent Tuberculosis Infection and T-SPOT.TB Dynamics Alterations on Prognosis in Advanced NSCLC Treated With ICIs——IPTW-Based Retrospective Study by Yijiao Xu, Jianying Liu, Qingwei Zhang, Yijun Song, Shuwen Yang, Haiyan Chen, Congyi Xie, DaWei Yang, Zhisheng Chen and Hongni Jiang in Clinical Medicine Insights: Oncology
Supplemental Material
sj-jpg-3-onc-10.1177_11795549251394955 – Supplemental material for Impact of Latent Tuberculosis Infection and T-SPOT.TB Dynamics Alterations on Prognosis in Advanced NSCLC Treated With ICIs——IPTW-Based Retrospective Study
Supplemental material, sj-jpg-3-onc-10.1177_11795549251394955 for Impact of Latent Tuberculosis Infection and T-SPOT.TB Dynamics Alterations on Prognosis in Advanced NSCLC Treated With ICIs——IPTW-Based Retrospective Study by Yijiao Xu, Jianying Liu, Qingwei Zhang, Yijun Song, Shuwen Yang, Haiyan Chen, Congyi Xie, DaWei Yang, Zhisheng Chen and Hongni Jiang in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
The authors acknowledge and express their deepest gratitude to the participants of this study.
Ethical considerations
This study was approved by the Ethics Committee of Zhongshan Hospital (Xiamen), Fudan University (No. B2022-068, approved on February 16, 2023) and conducted in accordance with the principles of the Declaration of Helsinki.
Consent to participate
As it is a retrospective observational study, the ethics committee waived the requirement to obtain informed consent.
Author contributions
Yijiao Xu: Conceptualization; Methodology; Project administration; Resources; Validation; Writing—original draft; Writing—review & editing
Jianying Liu: Conceptualization; Formal analysis; Validation; Visualization; Writing—original draft; Writing—review & editing.
Qingwei Zhang: Conceptualization; Investigation; Methodology; Validation; Writing—review & editing.
Yijun Song: Investigation; Resources; Validation; Writing—review & editing.
Shuwen Yang: Investigation; Resources; Validation; Writing—review & editing.
Haiyan Chen: Investigation; Resources; Validation; Writing—review & editing.
Congyi Xie: Investigation; Resources; Validation; Writing—review & editing.
DaWei Yang: Conceptualization; Investigation; Methodology; Project administration; Resources; Validation; Writing—review & editing.
Zhisheng Chen: Conceptualization; Investigation; Methodology; Project administration; Resources; Validation; Writing—review & editing.
Hongni Jiang: Conceptualization; Methodology; Project administration; Resources; Validation; Writing—original draft; Writing—review & editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from Medical and health guidance project of Xiamen science and technology bureau (3502Z20244ZD1115), Natural Science Foundation of Fujian province (No. 2023J011690), and Fujian Province Department of Science and Technology (2022D014).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets used and/or analyzed during this study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
