Abstract
Background:
Endocrine hormones influence tumor progression and the response to treatment. Despite the importance of immune checkpoint inhibitors (ICIs) as treatments for advanced non-small cell lung cancer (NSCLC), few studies have explored the effects of hormone levels in NSCLC patients on the effectiveness of ICI therapies. We thus investigated the effects of baseline blood markers in patients with advanced NSCLC on ICI treatments.
Methods:
Patients with advanced NSCLC who received programmed cell death protein 1 (PD-1)/programmed death-ligand 1 (PD-L1) inhibitors at Chungnam National University Hospital between December 2016 and November 2020 and who lacked any history of thyroid gland-related diseases were analyzed retrospectively. We collected clinical information and baseline laboratory data, including the levels of endocrine hormones, cytokines, complete blood counts (CBCs), and peripheral blood chemistry panels. We explored the relationships of hormone levels with clinical outcomes (overall survival [OS], progression-free survival [PFS], and best response), liver metastasis, and blood markers using the Kaplan–Meier method, Cox’s proportional hazards regression, and logistic regression.
Results:
A total of 113 patients were enrolled. A shorter PFS was independently associated with liver metastasis, higher cortisol levels, and lower hemoglobin (Hb) levels; a shorter OS was associated with liver metastasis, lower tri-iodothyronine (T3) levels, higher lactate dehydrogenase (LDH) levels, and lower albumin levels. Patients with low T3 levels exhibited a shorter PFS and OS, and a poorer best response. Patients with low T3 levels tended to have higher disease progression rates, lower levels of adrenocorticotropic hormone (ACTH), C-peptide, albumin, Hb, and neutrophil-to-lymphocyte ratio, and higher levels of interleukin (IL)-6, white blood cells, platelets, compared with those with normal T3 levels. We found a significant association between a low T3 level and liver metastasis.
Conclusions:
We found the baseline T3 level was associated with both prognosis and the response to ICIs in patients with advanced NSCLC, probably reflecting impaired liver function and systemic inflammation induced by the interaction of T3 with other biomarkers, such as IL-6, ACTH, cortisol, C-peptide, Hb, LDH, and albumin.
Keywords
Introduction
Immune checkpoint inhibitors (ICIs) are increasingly used to treat advanced-stage, non-small cell lung cancer (NSCLC). The side effects include immune-related adverse events (irAEs) in multiple organs, such as the skin and gastrointestinal, endocrine, hepatic, pulmonary, renal, and neurological systems. The third most common problem after skin and gastrointestinal system issues is endocrinopathy. ICIs affect the secretion and functions of hormones from the pancreas, testis/ovary, pituitary, thyroid, and adrenal glands. 1 The thyroid gland is frequently affected. Hypothyroidism or hyperthyroidism caused by thyroiditis is reported in 30% of patients using ICIs. 2 However, interestingly, patients who experience irAEs exhibit better progression-free survival (PFS) compared with those without irAEs. IrAEs, including thyroiditis and vitiligo, are associated with favorable responses to interleukin (IL)-2 therapy in melanoma patients. 3 Likewise, advanced NSCLC patients exhibiting thyroid dysfunction after ICI therapy experience prolonged overall survival (OS) and PFS. 4
Hormones influence NSCLC tumor progression and the response to treatment. Thyroxine (T4) promotes tumor growth, angiogenesis, and metastasis. 5 Estrogen increases the cancer incidence and mortality by promoting cell proliferation and transcription of specific genes. 6 Adiponectin, which is produced in adipose tissue, inhibits angiogenesis, and leptin exerts tumorigenic effects by stimulating NSCLC cell proliferation and angiogenesis. 7 However, few studies have explored the effects of endocrine hormones on NSCLC progression and prognosis in patients undergoing ICI treatment. Although the levels of certain pituitary hormones, such as prolactin, were predictive, only a few dozen patients were enrolled in the study. 7 Herein, we need more work in the role of hormones on the ICI treatment of NSCLC.
Biomarkers that predict the response to ICI treatment include the tumor programmed death-ligand 1 (PD-L1) level, baseline tumor size, tumor mutation burden, Eastern Cooperative Oncology Group performance status, serum IL-6 level, tumor-infiltrating lymphocyte level, numbers of peripheral blood CD8+ T cells and regulatory T cells, and the neutrophil-to-lymphocyte ratio (NLR). However, only a few biomarkers have been clinically verified using standard measurement techniques. Better predictors are required.
We investigated the effects of baseline (before ICI treatment) endocrine hormone levels on the prognosis of advanced NSCLC. We sought relationships between these levels and the levels of other blood markers, including the complete blood count (CBC), inflammatory cytokine levels, and blood chemistry data. We focused on the hormones that increase the risk of liver metastasis, which predicts a poor response to ICIs in NSCLC patients.
Methods
Patients
Patients (1) over 18 years of age, (2) with advanced NSCLC, (3) who are already in stage 4 at the time of diagnosis or who have relapsed or progressed after previous chemotherapy, (4) who received programmed cell death protein 1 (PD-1)/PD-L1 inhibitors at Chungnam National University Hospital between December 2016 and November 2020 were analyzed retrospectively. We reviewed the patients’ electronic medical records (C&U Care, Daejeon, Republic of Korea). We excluded patients (1) diagnosed with carcinoma other than NSCLC, (2) with positive driver mutations (in EGFR and ALK), (3) who received more than one ICI therapy, and (4) with a history of thyroid gland-related diseases. The PD-L1 expression level was grouped into 3 categories using tumor proportion score cut-offs of 1% and 50%: no (<1%), low (1–49%), and high (⩾50%) PD-L1 expression.
ICI treatments
Patients were given intravenous nivolumab (3 mg/kg body weight every 2 weeks), pembrolizumab (2 mg/kg body weight or 200 mg every 3 weeks), or atezolizumab (1200 mg every 3 weeks) during both the first- and later-line treatments. Treatment continued until severe adverse effects were experienced or disease progression was confirmed.
At the time of the treatment of the patients included in this study, ICI could only be administered as a second-line or subsequent treatment. Previous studies had shown that ICI prolonged OS in previously treated patients with advanced NSCLC.8-10 Therefore, at the time when some patients included in this study received treatment, immunotherapy was not reimbursed as the first-line therapy, and it was used as a second-line or subsequent therapy. Also, it was difficult for some patients to administer immunotherapy immediately due to their condition. So, cytotoxic chemotherapy was administered as the first-line therapy, and then immunotherapy was administered as the second-line therapy. In addition, some patients received cytotoxic chemotherapy as the first-line treatment, and after sufficient time elapsed, the immunotherapy was administered, resulting in similar effects to those of the first-line therapy. As studies showing an increase in the survival of patients who used immunotherapy as the first-line treatment were published,11-13 in Korea, the use of ICI became reimbursable for the first-line treatment of NSCLC in March of this year. Therefore, nowadays, ICIs are being actively used as a first-line treatment. It is used as a combination therapy regardless of the PD-L1 expression level or as a monotherapy in patients with a PD-L1 expression level of 50% or more.
Data collection
We collected information on sex, age, smoking status, cell type, PD-L1 expression status, number of prior systemic treatments, liver metastasis status, and ICI type. We also recorded the baseline laboratory levels of endocrine hormones (total tri-iodothyronine [T3], thyroid-stimulating hormone [TSH], free thyroxine [fT4], adrenocorticotropic hormone [ACTH], cortisol, C-peptide, and insulin), IL-6, the CBC (white blood cell [WBC] count, hemoglobin [Hb] level, platelet [Plt] count, segmented neutrophil count [Seg. neutrophil[#]], lymphocyte count [lymphocyte(#)], and NLR), and peripheral blood markers (lactate dehydrogenase [LDH], total bilirubin, and albumin).
Measurements of hormones, IL-6, CBC, and blood parameters
Blood sampling was performed within 2 weeks before and after commencement of ICI treatment between 9 am and 3 pm after fasting for more than 8 h. Blood samples were transported to the laboratory within 30 min, centrifuged at 3000 r/min for 10 min at 4°C, and analyzed immediately. Serum levels of T3 and fT4 were measured by radioimmunoassay, and that of TSH was measured using an immunoradiometric assay. Plasma ACTH and serum cortisol levels were determined by radioimmunoassay. The serum levels of IL-6, C-peptide, and insulin were measured by electrochemiluminescence immunoassay. We used a hematology autoanalyzer to determine the WBC count, Hb level, Plt count, Seg. neutrophil(#), and lymphocyte(#). The serum total bilirubin and albumin levels were measured using an enzymatic oxidation method and bromocresol green dye-binding method, respectively.
Clinical outcomes
We evaluated the outcomes of ICI therapy by recording the best response, OS, and PFS. Chest computed tomography including the upper abdomen was performed every 3 cycles for patients taking pembrolizumab or atezolizumab and every 4 cycles for those taking nivolumab. The treatment response was based on the Response Evaluation Criteria in Solid Tumors version 1.1. The best response was assessed during ICI administration. OS was defined as the period from the first day of ICI administration to the date of death or the last follow-up. PFS was defined as the time from the start of ICI treatment to documented progression. The period of the follow-up was calculated using the beginning date of ICI administration to the date of death or the last visit, and the mean value was 309 days.
Statistical analysis
We present categorical variables as numbers and percentages and continuous variables as medians. Categorical variables were compared using the chi-squared test or Fisher’s exact test and continuous variables using the Mann–Whitney U-test. The Kaplan–Meier method was used to analyze PFS and OS, and survival rates were compared using the log-rank test. Univariate analyses were performed using the Kaplan–Meier method for categorical variables and Cox’s proportional hazards regression for continuous variables. All p-values were 2-sided, and values <.05 were considered statistically significant. Variables significant in univariate analyses were included in multivariate analyses (Cox’s proportional hazards regressions and logistic regression). All analyses were conducted using SPSS version 22 and Microsoft Excel 2010.
Results
Baseline characteristics and laboratory findings
This study is a retrospective cohort study (Figure 1). A total of 166 patients were initially included, of whom 9 were excluded because they had driver mutations (in EGFR or ALK) and one because of repeated ICI treatments. Next, 43 patients who were not tested for hormones were excluded. Finally, 113 patients were included, and their baseline characteristics are presented in Table 1. Patients were predominantly male (81.4%) and current or former smokers (80.1%), of median age 68 years. The major histological tumor types were squamous cell carcinoma (51.9%) and adenocarcinoma (41.7%). In terms of PD-L1 expression, 53.2% of patients evidenced high expression, 19.9% low expression, and 19.9% no expression. Most patients had been treated previously (87.2%), and did not have liver metastasis (84.6%) at the beginning of ICI therapy. All patients received ICI monotherapy, of which 53.8%, 23.1%, and 23.1% received pembrolizumab, atezolizumab, and nivolumab, respectively. The best response to ICI therapy was stable disease in 33.3%, progressive disease in 26.9%, partial remission in 25.0%, and complete remission in 3.9%. Table S1 lists the (median) baseline endocrine hormone and cytokine levels, the CBCs, and the blood data.

Study design. NSCLC indicates non-small cell lung cancer; PD-1, programmed cell death protein 1; PD-L1, programmed death ligand 1; ICI, immune checkpoint inhibitor.
Baseline characteristics.

Abbreviations: CR, complete remission; ICI, immune checkpoint inhibitor; PD, progressive disease; PD-L1, programmed death-ligand 1; PR, partial remission; SD, stable disease; TPS, tumor proportion score.
Values are presented as medians (lower and upper quartiles) or numbers (%).
Uni- and multivariate analyses of PFS and OS
Uni- and multivariate analyses were performed to determine factors affecting patient survival. Univariate analyses showed that the following factors were significantly predictive of PFS: liver metastasis and type of ICI (categorical variables) (Figure 2A) and the T3, cortisol, IL-6, WBC, Hb, Seg. neutrophil(#), NLR, LDH, and albumin levels (continuous variables) (Figure 2B). Multivariate analysis showed that an inferior PFS was independently associated with liver metastasis (hazard ratio [HR] = 8.954, P = .000), a high cortisol level (HR = 1.044, P = .017), and a low Hb level (HR = .725, P = .000) (Table S2).

(A) Univariate analysis of patient characteristics associated with PFS. (B) Univariate analysis of laboratory findings associated with PFS. PFS indicates progression-free survival.
Univariate analyses showed that the following factors were significantly predictive of OS: age, liver metastasis, and type of ICI (categorical variables) (Figure 3A) and the T3, IL-6, WBC, Hb, Seg. neutrophil(#), NLR, LDH, and albumin levels (continuous variables) (Figure 3A). Multivariate analysis showed that inferior OS was independently associated with liver metastasis (HR = 3.637, P = .044) and low levels of T3 (HR = .166, P = .019), high LDH (HR = 1.002, P = .003), and albumin (HR = .264, P = .003) (Table S24).

(A) Univariate analysis of patient characteristics associated with OS. (B) Univariate analysis of laboratory findings associated with OS. OS indicates overall survival
A low baseline T3 level is prognostic
Unlike the other factors mentioned above, few studies have explored the effects of low T3 syndrome (low T3 levels but normal-to-low T4 and TSH levels) on lung cancer progression,14,15 although the effects of abnormal T4 or TSH levels have been examined. We explored whether the T3 level affected survival. We divided patients into those with low and those with normal baseline T3 levels. The normal value of total T3 in adults, 0.8–2.0 ng/mL, was defined as the reference range. If T3 level was lower than 0.8 ng/mL, it was defined as low T3. The PFS (median 66 vs 174 days; P = .008) (Figure 4A) and OS (median 204 vs 901 days; P = .000) (Figure 4B) were shorter in the former than the latter patients.

Patient survival according to the baseline T3 level (n = 113).
*Of the total 156 patients, 43 patients did not perform the hormone test, so that, the total number of patients included in this analysis was reduced to 113 and the number of patients also changed in the analysis of the subcategories presented in Table 2.
Baseline patient characteristics and laboratory findings according to the baseline T3 level (n = 113).
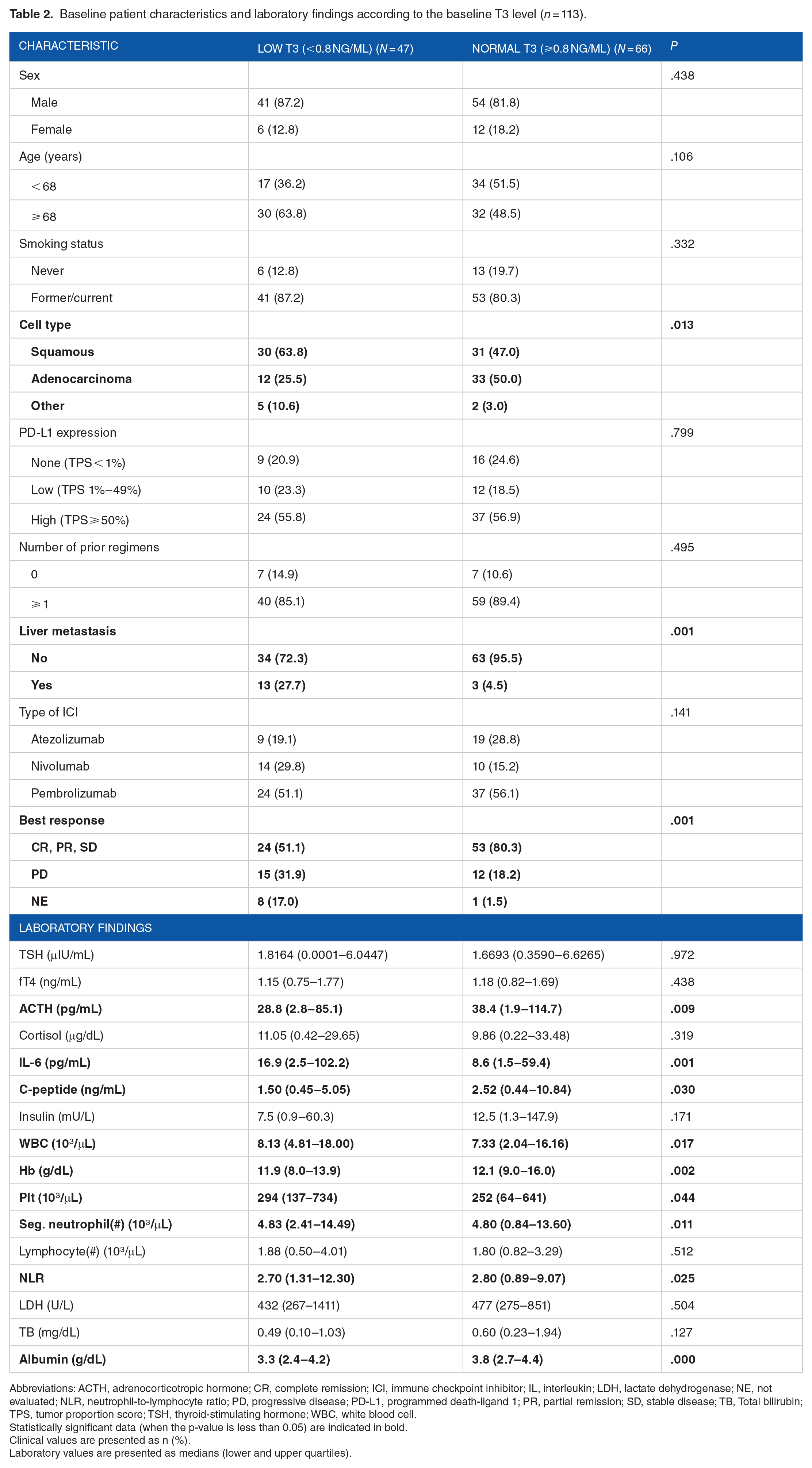
Abbreviations: ACTH, adrenocorticotropic hormone; CR, complete remission; ICI, immune checkpoint inhibitor; IL, interleukin; LDH, lactate dehydrogenase; NE, not evaluated; NLR, neutrophil-to-lymphocyte ratio; PD, progressive disease; PD-L1, programmed death-ligand 1; PR, partial remission; SD, stable disease; TB, Total bilirubin; TPS, tumor proportion score; TSH, thyroid-stimulating hormone; WBC, white blood cell.
Statistically significant data (when the p-value is less than 0.05) are indicated in bold.
Clinical values are presented as n (%).
Laboratory values are presented as medians (lower and upper quartiles).
Variables associated with short survival in patients with low T3 levels
We compared the baseline characteristics and laboratory findings of the low and normal T3 groups (Table 2). Patients with a low T3 level (<0.8 ng/mL) tended to have more squamous cell cancers (63.8% vs 47.0%; P = .013) and liver metastases (27.7% vs 4.5%; P = .001) compared with those with a normal level. The PD rate during ICI therapy was significantly higher in the low than normal T3 group (31.9% vs 18.2%; P = .001). The low T3 group had lower ACTH (P = .009), higher IL-6 (P = .001), lower C-peptide (P = .030), higher WBC (P = .017), lower Hb (P = .002), higher Plt (P = .044), higher Seg. neutrophil(#) (P = .011), lower NLR (P = .025), and lower albumin (P = .000) levels compared with the normal T3 group (Table 2).
Association between the baseline T3 level and liver metastasis
Liver metastasis is negatively predictive in NSCLC patients treated with ICIs. 16 We sought an association between the T3 level and liver metastasis. Patients with liver metastasis exhibited a shorter PFS (median 36 vs 120 days; P = .040) (Figure S1A) and a shorter OS (median 85 vs 575 days; P = .007) compared with those without liver metastasis (Figure S1B). Univariate analysis showed that liver metastasis was significantly associated with the T3, ACTH, insulin, Hb, LDH, and albumin levels (Table 3). Multivariate analysis showed that liver metastasis was independently associated with a low T3 level (odds ratio [OR] = 0.002, P = .021) (Table 4). Thus, we confirmed a significant association between the T3 level and liver metastasis.
Univariate analyses of factors associated with liver metastasis.

Abbreviations: ACTH, adrenocorticotropic hormone; CI, confidence interval; ICI, immune checkpoint inhibitor; IL, interleukin; LDH, lactate dehydrogenase; NLR, neutrophil-to-lymphocyte ratio; PD-L1, programmed death-ligand 1; TB, total bilirubin; TSH, thyroid-stimulating hormone; WBC, white blood cell.
Statistically significant data (when the p-value is less than 0.05) are indicated in bold.
Multivariate analysis of factors associated with liver metastasis.

Abbreviations: CI, confidence interval; LDH, lactate dehydrogenase.
Statistically significant data (when the p-value is less than 0.05) are indicated in bold.
Discussion
We found that a low T3 level was a negative predictor of PFS and OS in NSCLC patients treated with ICIs. A low T3 level was associated with liver metastasis; both a low T3 and liver metastasis were negative predictors of the response to ICIs. Recently, low T3 syndrome was found to usefully predict the survival of multiple myeloma patients. 17 We are the first to show that a low T3 level is associated with shortened survival in NSCLC patients on ICIs. Moreover, we confirmed that many blood biomarkers and liver metastasis were associated with lower T3 levels. As thyroid hormones (THs) stimulate physiological growth, maturation, and metabolism, 18 we assumed that the hormones might affect tumor growth and progression. Previous studies revealed that hypo/hyperthyroidism at the time of cancer diagnosis affected prognosis. 5 However, the diagnoses of hypo/hyperthyroidism are determined based on the free T4 and TSH levels; T3 has received relatively little attention. Moreover, a low T3 level was associated with liver metastasis, a known negative predictor of ICI treatment outcomes.
Non-thyroidal illness syndrome (NTIS) is associated with abnormal TH levels secondary to a systemic illness, such as sepsis or a long-term disease. NTIS does not reflect a defect in the hypothalamus–pituitary–thyroid gland axis. The T3 level decreases, but those of T4 and TSH do not change. 19 Systemic illness associated with production of inflammatory cytokines, such as IL-6 and tumor necrosis factor-alpha (TNF-α), and cytokine-mediated oxidative stress reduce T4–T3 conversion and the tissue/cellular uptake of T4 and T3. 20 The extent of reduction in the T3 level is proportional to the severity of illness. 21 NSCLC is associated with systemic inflammation caused by IL-6, TNF-α, and other cytokines; this may reduce the T3 level. In a previous study, we showed that the serum IL-6 level before ICI treatment predicted the responses of NSCLC patients to ICIs and that systemic inflammation per se affected ICI efficacy. 22
We divided all patients into those with low and those with normal T3 levels and compared their characteristics. Patients with low T3 levels tended to have liver metastases at initiation of ICI therapy, to exhibit PD after treatment, and to exhibit significantly higher serum IL-6 and lower ACTH levels compared with patients with normal levels. The IL-6 level is frequently elevated in patients with infections or inflammation, major trauma, or systemic diseases and in those who undergo surgery. IL-6 reduces T3 conversion and activity by inhibiting deiodinases 1 and 2. 23 Thus, IL-6 is associated with changes in thyroid function similar to those seen in NTIS patients. 24 Furthermore, when deiodinase 3 activity decreases because of IL-6-mediated oxidative stress and reactive oxygen species production, reverse T3 production and activity increase, promoting tumor cell proliferation. 25 As T3 inhibits activation of signal transducer and activator of transcription 3 (STAT3) via IL-6 or lipopolysaccharide, 26 tumor growth and metastasis cannot be well controlled when the T3 level is low. 27 Also, IL-6 plays a critical role in augmentation of adrenal cortex function in response to immune system activation, and a higher IL-6 concentration is associated with a higher cortisol response to ACTH stimulation. 28 This is most apparent in the context of corticotropin-releasing hormone deficiency 29 ; the ACTH level is lowered via negative feedback from an increased cortisol level.
Higher serum cortisol and lower Hb levels were associated with a shorter PFS and a higher LDH level, and a lower albumin level was associated with a shorter OS. A higher concentration of cortisol may inhibit DNA repair. 30 A low pretreatment Hb level was correlated with shorter survival 31 because anemia compromises the T cell response, inducing immunosuppression in patients with late-stage tumors. 32 Furthermore, hypoxia induced by a reduced Hb level stimulates tumor growth and decreases tumor drug-sensitivities. 33 As cancer cells consume high levels of glucose, increasing lactate production, a high serum LDH level is associated with poor prognosis in patients with multiple solid tumors. 34 LDH promotes tumor growth by upregulating PD-L1 in lung cancer cells, which may in turn reduce survival. 35 The serum albumin level reflects nutritional status and is affected by inflammation and liver dysfunction. A decreased serum albumin level is associated with poorer survival. 36
We examined the relationships between the levels of T3 and other blood markers (shown in Table 2). The Hb level appeared lower in patients with low T3 levels, perhaps because reduced intracellular TH levels disturb the balance between the proliferation and differentiation of erythrocytic progenitors, in turn triggering anemia. 37 We found that patients with low T3 levels also had low albumin levels, consistent with the results of a study confirming an independent positive relationship between T3 and albumin levels. 38 Another interesting finding was that the C-peptide level was significantly lower in the low than normal T3 group. The C-peptide hormone is synthesized and released by the pancreas, along with insulin. Therefore, the concentration of C-peptide reflects the concentration of endogenous insulin, with a decrease in the blood C-peptide level indicating a decrease in insulin secretion. THs alter the functions of other endocrine glands. In the pancreas, THs play a role in physiological development and affect insulin secretion and function. 39 T3 promotes human embryonic stem cell differentiation into β-cells in the pancreas (where insulin and C-peptide are synthesized and released), thus increasing the number of β-cells. 40 The mechanisms inducing Type 2 diabetes in patients with thyroid dysfunctions may include disturbances in the expression levels of many genes, combined with physiological abnormalities that predispose to impaired glucose utilization and disposal by muscles, hepatic glucose overproduction, and enhanced splanchnic glucose absorption. 41 Therefore, lower TH levels can trigger pancreatic disorders, even if the low levels are caused by factors other than thyroid dysfunction per se. As we also found, others showed that islet insulin secretion was positively correlated with the serum T3 and T4 concentrations. 42
Liver metastasis in patients on immunotherapy is very important in clinical terms. Metastases exert different impacts on treatment outcomes depending on the site of the metastasis. Liver metastasis disproportionately and negatively impacts survival and the responses to immunotherapy. The liver regulates the response. Anticancer responses are thus affected by liver metastasis, 43 which induces systemic loss of antigen-specific T cells and a response to PD-1 blockade. Therefore, liver metastasis triggers acquired therapeutic resistance, associated with poor prognosis. 44
We explored the relationships between liver metastasis and blood parameters. As shown in Table 3, liver metastasis was significantly associated with the T3, ACTH, insulin, Hb, LDH, and albumin levels. The liver is the principal organ involved in TH conversion (from T4 to T3) and secondary release of T4 and T3 into the circulation; abnormal TH levels are often evident in patients with liver disease. 45 Liver metastasis may decrease the levels of THs including T3. Also, a lower T3 level increases liver metastasis. When blood-borne tumor cells from other organs arrive in the liver, Kupffer cells (liver-specific macrophages, ie, the first-line liver defense mechanism) detect and remove them to prevent metastasis. 46 As T3 promotes Kupffer cell hyperplasia and hypertrophy, 26 the reduced numbers and functionality of such cells caused by T3 deficiency compromise cancer cell phagocytosis and elimination in the liver. Lung cancer that has metastasized to other organs, such as the liver, imposes a high disease burden and is associated with systemic inflammation, accompanied by elevated TNF-α levels that reduce ACTH secretion by the pituitary gland,47,48 explaining the decrease in the ACTH level in patients with liver metastases. The role of insulin in carcinogenesis involves principally insulin receptor overexpression by tumor cells.49,50 When the receptor is activated by insulin, 51 the ligand binding receptor transduces the signal into cells via autophosphorylation, in turn activating the PI3K/Akt/mTOR signaling pathway, which triggers abnormal cell proliferation and inhibits apoptosis and carcinogenesis.52,53 This explains the positive correlation between the insulin level and liver metastasis.
An elevated LDH serum level is thought to reflect enzyme leakage from hepatocytes destroyed by metastasis. The LDH level increase may also indicate increased enzyme production attributable to hypoxia associated with malignancy. LDH is an essential enzyme of anaerobic glycolysis54,55; hepatocytes increase the LDH production under hypoxic conditions until the cells become necrotic. 56 Commonly, monitoring serum LDH levels is viewed as non-informative because LDH is produced by various organs and its specificity for detecting liver disease is thus low. However, it has been found that LDH production increases more than alanine aminotransferase (ALT) production under hypoxic conditions.56,57 The serum albumin level seems to reflect systemic inflammation, nutritional deficiency, and liver synthetic function and may even protect against cancer growth. 58 Although the mechanism remains unclear, serum albumin may inhibit a signaling pathway involved in cancer cell proliferation. 58 In some cancers, including lung cancer and hepatocellular carcinoma, a high serum albumin level is a major indicator of a favorable prognosis. 59
Recent studies have explored the roles played by THs in the oncogenic signaling involved in lung cancer development and progression and in the development of cancer- or patient-specific biomarkers. 60 Also, some anticancer interventions are focused on TH. In a murine model, T3 injection reduced metastasis, while T4 treatment promoted tumor growth.61,62 Hence, TH signaling may be excitatory or inhibitory depending on the TH (T3 or T4), ratio of T3–T4, thyroid receptor type/isoform, TH concentration, and the activated molecular pathway. 60 More clinical data are required, and studies on humans are needed.
This is the first report that the T3 level can predict the prognosis of patients on ICI therapy. However, our study had certain limitations. As this was a retrospective study, some hormone and cytokine data were missing. TH levels were missing in approximately 25% of patients, and insulin and C-peptide levels were available in less than half of all patients. Thus, further studies with larger numbers of patients and blood samples are required. Also, the effects of systemic treatment, such as cytotoxic chemotherapy, as a confounding factor on the actions of ICIs were not considered in detail. Further research is required.
Conclusion
We found that the baseline T3 level was associated with the prognosis and the ICI response of patients with advanced NSCLC. A low baseline T3 level in patients on ICI therapy was associated with poorer survival (PFS and OS). The specific mechanism probably involves liver impairment by the metastasis, and systemic inflammation induced by interactions of T3 with other biomarkers, such as IL-6, ACTH, cortisol, C-peptide, Hb, LDH, and albumin.
Supplemental Material
sj-docx-1-onc-10.1177_11795549221139522 – Supplemental material for The Tri-iodothyronine (T3) Level Is a Prognostic Factor for Patients With Advanced NSCLC: Receiving Immune Checkpoint Inhibitors and Is Associated With Liver Metastasis
Supplemental material, sj-docx-1-onc-10.1177_11795549221139522 for The Tri-iodothyronine (T3) Level Is a Prognostic Factor for Patients With Advanced NSCLC: Receiving Immune Checkpoint Inhibitors and Is Associated With Liver Metastasis by Yoonjoo Kim, Jeong Suk Koh, Seong-Dae Woo, Song-I Lee, Da Hyun Kang, Dongil Park, Chaeuk Chung, In-Sun Kwon and Jeong Eun Lee in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and Technology (grant nos. NRF-2019R1C1C1008864 and NRF-2021R1A2C2011603) and Korean Association for the Study of Targeted Therapy (KASTT) affiliated with Korean Association for Lung Cancer (grant no. KASTT-20190211).
Author Contributions
YK, JK, SW, SL, DK, DP, CC, and IK made a substantial contribution to the concept and design of the work and acquisition, analysis, and interpretation of data. JL drafted the article or revised it critically for important intellectual content. All authors approved the version to be published.
Ethics Approval and Consent to Participate
This study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines, and was approved by the institutional review board of Chungnam National University Hospital (2018-04-014 at CNUH).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
