Abstract
Background:
Accumulating evidence suggests that the use of antibiotics (ATBs) is harmful to the survival of patients with non-small-cell lung cancer (NSCLC). However, the association between the prophylactic use of these agents during surgery and patient survival has been less well studied.
Methods:
Data concerning the use of ATBs, including the cumulative defined daily dose (cDDD) and type, in stage I to III NSCLC patients were collected. The patients were subsequently divided into low or high-cDDD subgroups and ⩽2 or ⩾3 ATB-type subgroups. Differences in clinical variables, overall survival (OS), and disease-free survival (DFS) among these groups were assessed. Furthermore, differences in survival among specific ATB types (β-lactams and fluoroquinolones) were also tested. Finally, the risk factors for OS were determined using the Cox proportional hazards model.
Results:
A total of 324 patients were included. Low cDDD was more common in patients with advanced T stages, whereas ⩽2 types of ATBs were common in female patients and those with adenocarcinoma, N0 disease and stage I disease. No significant difference was found in OS among the low- or high-cDDD subgroups; however, a significant difference in OS was found between the ⩽2 and ⩾3 ATB. Similarly, patients with or without β-lactams displayed no difference in OS, whereas those with or without fluoroquinolones did. No differences were found in DFS between the subgroups. Multiple types of ATBs, rather than cDDD, were found to be risk factors for OS; however, they were not validated as independent risk factors.
Conclusions:
This study, for the first time, explored the prognostic value of ATB use during surgery in NSCLC patients and revealed that multiple types of ATBs may be associated with OS in patients with stage I to III disease. Notably, patients treated with fluoroquinolones may have inferior outcomes than those without ATB use. However, multiple types of ATBs were not validated as independent risk factors for OS. These results suggest that the use of ATBs during surgery in early-stage NSCLC is generally safe; however, caution should be taken when selecting ATB types. Multiple ATB types should be avoided, and some specific ATB types, such as fluoroquinolones, should not be administered. Nonetheless, owing to the limited sample sizes, future studies are needed to validate our results.
Introduction
Cancer is one of the deadliest, most complex and dynamic diseases affecting humans and is characterized by the uncontrolled growth and proliferation of normal cells. Many factors, such as physical, chemical, and biological carcinogens, are connected with the growth and progression of different malignancies.1-4 Infection has long been reported to play a key role in the progression of various types of cancers. Recent studies have suggested an etiological role of bacteria in these malignancies, including lung cancer,5-8 which remains a deadly disease for humans worldwide.9,10
It is well known that antibiotics (ATBs) play a key role in fighting bacteria; surprisingly, these agents are also increasingly reported to be negative factors in anticancer treatment, particularly in those who receive immunotherapy or tyrosine kinase inhibitors (TKIs). A study by Eng et al 11 that included 2737 patients who were aged 65 years or older revealed that the exposure to ATB, in particular for patients receiving fluoroquinolones before the initiation of immunotherapy, resulted in worse overall survival (OS). In addition, Tinsley et al 12 reported a study that enrolled 168 advanced stage patients and found the use of ATBs was correlated with poor progression-free survival (PFS) and OS in those who received TKIs. Similarly, these findings have been validated in non-small-cell lung cancer (NSCLC) patients.13-17 Moreover, the use of ATB can result in inferior PFS and OS for patients with locally advanced NSCLC who receive definitive concurrent chemoradiation 18 or for patients with locally advanced and metastatic disease receiving first-line chemotherapy. 19 These data suggest that the negative role of ATBs in NSCLC patient outcomes is independent of treatment; a widely accepted explanation for this phenomenon is that the use of ATBs can lead to profound dysbiosis of the gut microbiota, which has a broad function in regulating cancer cells,20-22 manipulating treatment efficacy,23-25 and potentially correlating with impaired T-cell immunity.26-28 Notably, all the aforementioned studies involved locally advanced or metastatic cases. In fact, the use of ATBs is not uncommon in early-stage cancer patients who undergo resection, and ATB use was also found to be negatively associated with survival in patients with colorectal cancer, as in our previous report and those of others.29,30 Interestingly, prophylactic use of these agents is also conventional in stage I to III NSCLC patients during surgery;31,32 however, the impact of ATB use on patient outcomes in such a scenario is still largely unknown.
In this study, we aimed to test the role of ATB use in the prognosis of radical surgery in patients with stage I to III NSCLC.
Methods
Data collection
In this study, data were collected from consecutive patients with stage I to III NSCLC who underwent radical surgery at Hainan Hospital of Chinese PLA General Hospital during enrollment from October 2012 to December 2020. The following cases were not included: (1) patients treated with neoadjuvant therapies (any type and duration); (2) patients with in situ lesions confirmed by postoperative pathological findings; and (3) patients with follow-up problems. Clinical variables were collected as previously reported.33,34 The study was conducted following the Helsinki Declaration of 1975 and its later amendments and was approved by the ethics committee of Hainan Hospital of Chinese PLA General Hospital (ID: S2023-12). Owing to its retrospective nature, mandatory written informed consent was waived by the ethics committee according to national regulations. The study followed the STROBE guidelines for reporting. 35
cDDD calculation
Data were collected from archived documents of the digital medical system on the utilization of ATBs. The cDDD was calculated using the following method: first, the cumulative dosage of each ATB was collected and multiplied by the standard defined daily dose (DDD) according to the China Pharmacopeia; second, the sum of all the individual cDDDs for each patient was divided by the length of hospitalization. In addition, the sum of the different types of ATBs was collected for each patient.
Study endpoints
Overall survival was selected as the main endpoint because it is considered the gold standard in clinical studies. 36 In addition, disease-free survival (DFS) was selected as the secondary endpoint. Follow-up was conducted as previously described, 35 and the latest follow-up ended in October 2023.
Statistical analysis
A total of 324 patients were included (Figure 1), and they were divided into low- or high-cDDD subgroups and ⩽2 or ⩾3 ATB-type subgroups according to the median of these indices. The differences in the clinical data among these subgroups were determined using the chi-square test or Fisher’s exact test when necessary. Overall survival and DFS differences among these subgroups and those with specific ATB types (β-lactams and fluoroquinolones) were checked via Kaplan-Meier analysis. Finally, the risk factors for OS were validated using the Cox proportional hazards model. A 2-sided P < .050 was considered to indicate a statistically significant difference. All the data were analyzed using SPSS 20.0 (SPSS Inc., Chicago, IL, USA).

Flow chart of the patient recruitment process.
Results
Patient characteristics
In summary, 167 males and 157 females were enrolled. The median age of the patients was 58 (range: 23-84) years, and the median follow-up period was 53 (range: 2-128) months. By the end of the follow-up period, 14 patients in stage I, 8 patients in stage II, and 10 patients in stage III had died. The median cDDD was 3.99 (range: 0-26.76), and the median length of hospitalization was 17 (range: 6-54) days. The distribution of ATB types was as follows: none (n = 1), single (n = 134), and 2 or more (n = 189).
Clinical data differences among the low- and high-cDDD subgroups and the ⩽2 ATB-type or ⩾3 ATB-type subgroups
As shown in Table 1, a low cDDD was more common in patients with advanced T stages, whereas the use of ⩽2 ATB types was more common in females and those with adenocarcinoma, N0 disease, and stage-I disease. No other differences were found among the subgroups.
Differences in the collected clinical features among the low- or high-cDDD subgroup and the ⩽2 or ⩾3 ATB use subgroups.

Statistically significant difference.
Differences in OS and DFS among the low- or high-cDDD subgroups and the ⩽2 ATB-type or ⩾3 ATB-type subgroups
As shown in Figure 2, no significant difference was found for OS between the low- or high-cDDD subgroups (log-rank = 0.66, P = .417); however, a significant difference in OS was detected between the ⩽2 ATB-type and ⩾3 ATB-type subgroups (log-rank = 5.33, P = .021). Similarly, patients with or without β-lactams did not differ in OS (log-rank = 2.36, P = .125), but patients with or without fluoroquinolones did (log-rank = 4.86, P = .027). With respect to DFS, no significant differences were found among the low and high-cDDD subgroups (log-rank = 0.41, P = .521) or the ⩽2 ATB-type and ⩾3 ATB-type subgroups (log-rank = 1.86, P = .173). In addition, no significant differences were detected in patients with or without β-lactams (log-rank = 0.37, P = .541) or fluoroquinolones (log-rank = 1.17, P = .279) (Figure 3).
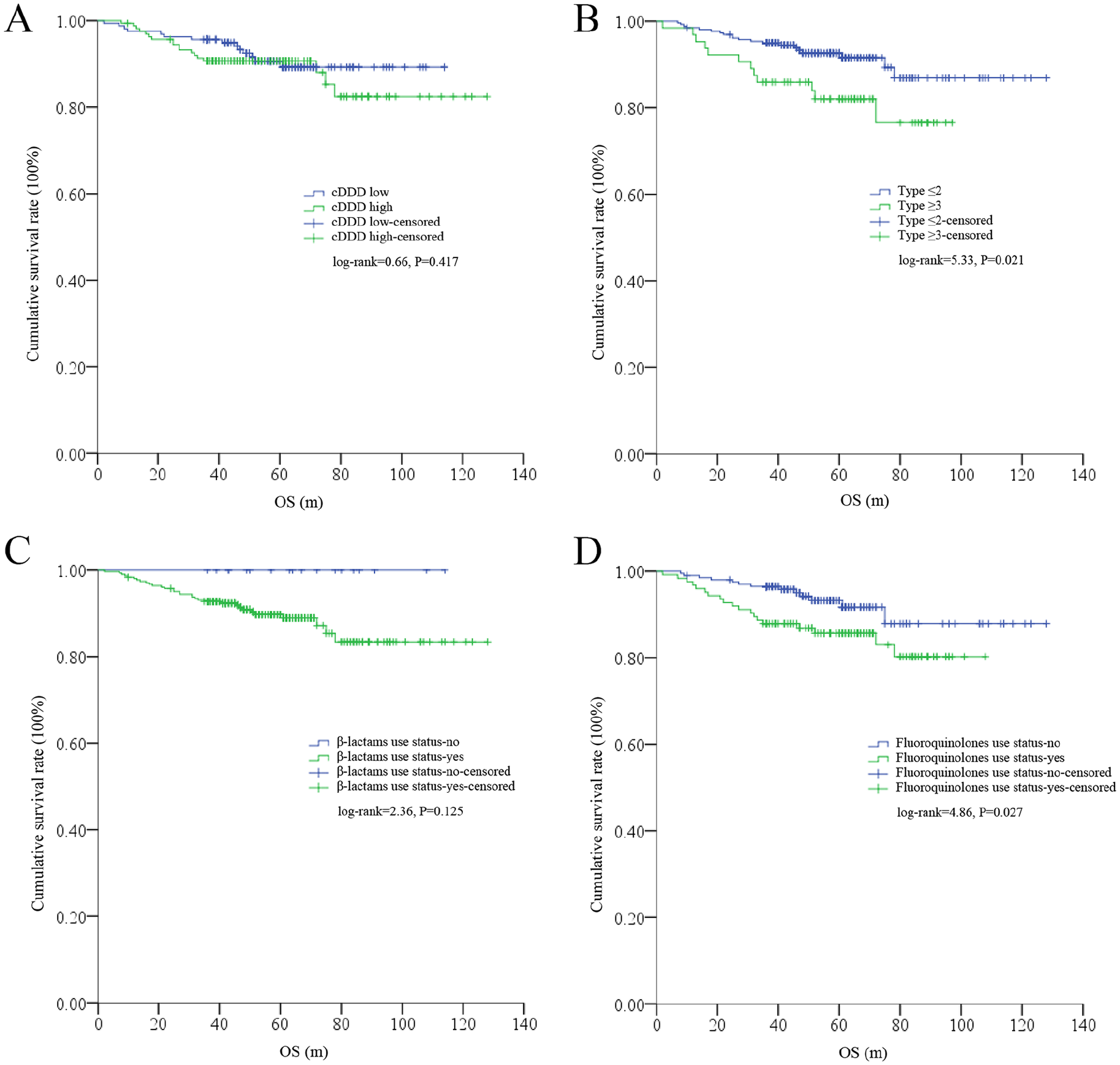
Differences in OS among different subgroups (n = 324). (A) No significant difference in OS was detected among the low- (n = 161) or high- (n = 163) cDDD subgroups. (B) A significant difference in OS was detected among the ⩽2- (n = 260) or ⩾3 (n = 64) ATB-type subgroups. (C) No significant difference in OS was detected among patients with (n = 306) or without (n = 18) β-lactams. (D) A significant difference in OS was detected among patients with (n = 124) or without (n = 200) fluoroquinolones.
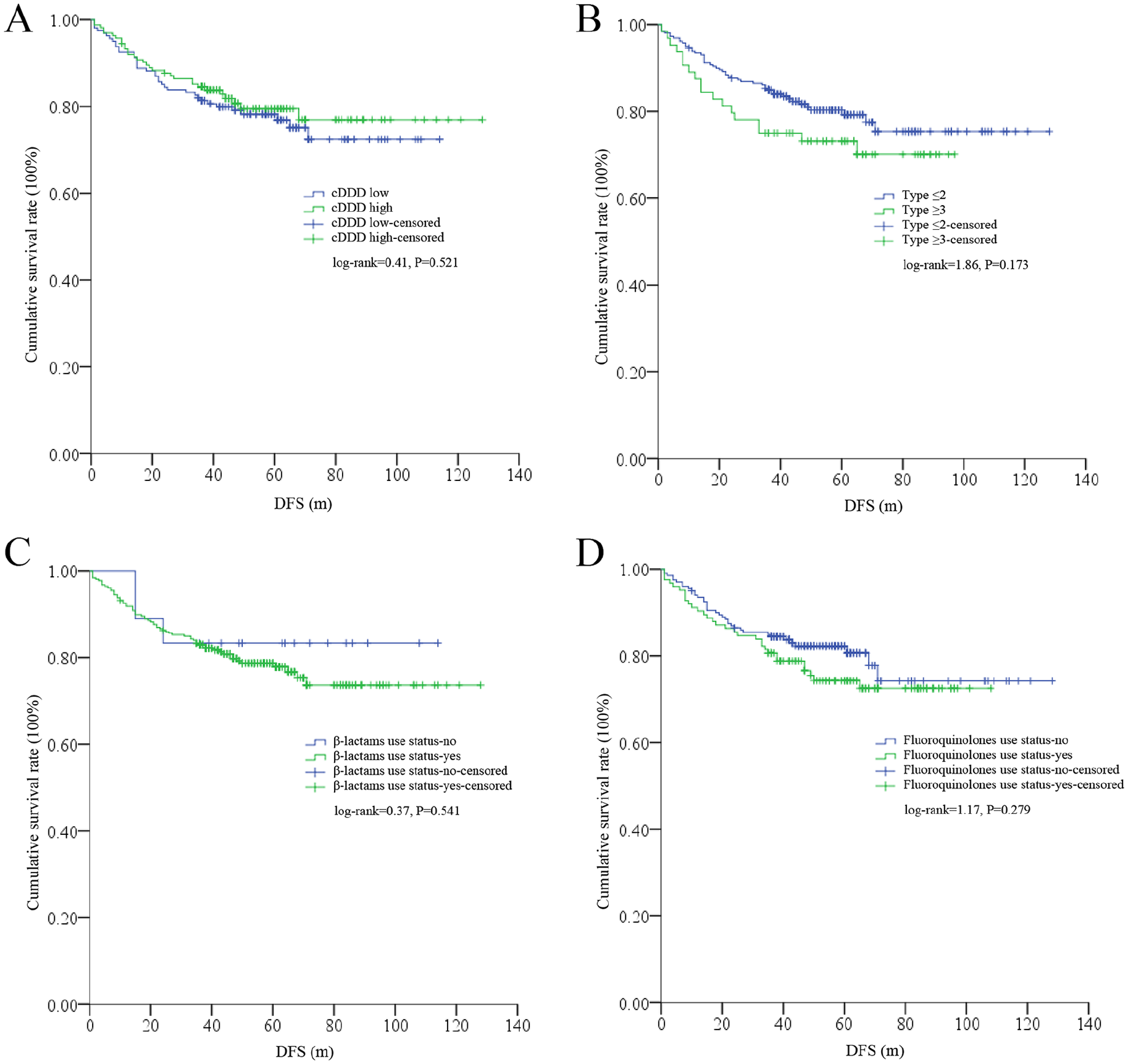
DFS differences among different subgroups (n = 324). (A) No significant difference in DFS was detected among the low- (n = 161) or high- (n = 163) cDDD subgroups. (B) No significant difference in DFS was detected among the ⩽2- (n = 260) or ⩾3 (n = 64) ATB-type subgroups. (C) No significant difference in DFS was detected among patients with (n = 306) or without (n = 18) β-lactams. (D) No significant difference in DFS was detected among patients with (n = 124) or without (n = 200) fluoroquinolones.
Univariate and multivariate tests to validate the risk factors for OS
Considering that both cDDD and ATB type were not significantly associated with DFS, risk factor analysis was only conducted for OS by using the Cox proportional hazards model. As shown in Table 2, age, sex, smoking, drinking, T stage, N stage, tumor node metastasis (TNM) stage, and type of ATB (hazard ratio [HR] = 2.28, 95% confidence interval [CI]: 1.11-4.67, P = .025) were found to be risk factors for OS in univariate tests, whereas only smoking, T-stage, and N-stage were validated as independent factors in multivariate tests (Table 2).
Validation of the risk factors for OS by univariate and multivariate analyses.

Statistically significant difference.
Discussion
To the best of our knowledge, this report is the first on the prognostic value of ATB use during surgery in patients with NSCLC. We found that multiple types of ATBs during surgery, rather than cDDD, were correlated with OS in patients with stage I to III NSCLC; in particular, patients who received ⩾3 ATB types were likely to have an inferior OS. However, neither cDDD nor ATB type was found to be an independent risk factor for OS; these results are partly in line with those of previous studies.29,30
Although the prognostic role of ATB use has been well documented in locally advanced or metastatic NSCLC patients, the definition of ATB use or exposure in these reports is highly inconsistent. For example, different time points are accepted for the use of ATBs in patients who receive immunotherapies.14,15,37-42 Notably, these studies did not consider the spectra or the dose of the ATBs. Similarly, in a study of patients who underwent treatment with TKIs, ATB use was defined as ATB concurrently or within 180 days with TKIs. 14 However, there are different algorithms to define ATB use or exposure; in a study with 90 advanced patients who received nivolumab (an agent of immunotherapy), the use of ATBs was considered to have a duration of ⩾3 days within 30 days before the start of the treatment, 43 and a duration ⩾7 d or ⩾6 days within 30 days18,19 was also adopted in patients who received first-line chemotherapy or definitive concurrent chemoradiation. Interestingly, Galli et al 44 introduced a new index called the “ATB-immunotherapy exposure ratio (AIER),” which was calculated by “days of ATB /days of immunotherapy” during the whole treatment period and found that patients with a relatively high AIER have poor PFS and OS. Geum et al 45 introduced DDD, which was recommended by the World Health Organization, for the first time to explore the use of ATB in the prognosis of NSCLC. Nonetheless, taking into consideration the importance of the dose‒response relationship between the use of ATBs and cancer46,47 and the fact that patients can be administered multiple types of ATBs in real clinical practice (over half [189/324] of patients received more than one type of ATB in our study, which is similar to the findings of a previous study with patients who received TKIs [27/44]) 17 . The cDDD, which is derived from the DDD, can be considered an alternative index for predicting patient prognosis in these studies. In our study, although cDDD was not significantly correlated with OS in stage I to III patients, its value in locally advanced or metastatic settings still needs further validation.
Indeed, previous observations have indicated that specific types of ATBs may play different roles in the prognosis of cancer patients. For example, our previous study in colorectal cancer indicated that quinolones and macrolides were adversely correlated with survival. 29 Qiu et al 13 studied 148 advanced patients with NSCLC and reported that the use of β-lactams (only penicillins rather than carbapenems and cephalosporins) was significantly associated with worse PFS and OS in patients, whereas the use of fluoroquinolones had no such association. However, patients experienced the worst PFS and OS when they received β-lactams and fluoroquinolones synchronously, 13 which indicated a potential synergistic effect among ATB. Moreover, Ochi et al, 15 in a report with 531 advanced patients, also reported that β-lactams correlated with obviously worse OS; however, owing to the small sample size for other ATBs, no such correlations were further validated. In our study, we found that patients with or without the use of fluoroquinolones presented significantly worse OS but not with or without β-lactams, which was not in line with previous studies.13,15 However, cases without β-lactams were limited (n = 18), and it is more plausible that a synergistic effect was detected, as in Qiu et al’s 13 study. Nonetheless, as our study was conducted in patients who underwent radical resection, our speculation needs to be validated in the future.
Interestingly, the use of ATBs during surgery in stage I to III NSCLC patients may share a similar scenario as that used in locally advanced or metastatic settings, since a high frequency of circulating tumor cells (CTCs) was detectable in these patients (although R0 resection was reached and the lesion was completely removed);48,49 however, the underlying mechanisms for the negative association between ATB use and patient outcomes in NSCLC are still largely unknown. On one hand, it has been reported that some ATBs can have positive anticancer effects. For example, levofloxacin can suppress cell proliferation and induce apoptosis in lung cancer; 50 some fluoroquinolones also present anti-proliferative activity and induce apoptosis in these cells; 51 and erythromycin can suppress cell migration. 52 On the other hand, the use of ATBs in NSCLC patients could result in significant dysbiosis of the gut microbiota, which could have a broad effect on regulating the development of the disease.20-22 In particular, although it has not been validated in lung cancer, the dysbiotic microbiota was found to play an important role in promoting epithelial-mesenchymal transition (EMT) 53 and regulating the Hedgehog signaling pathway. 54 Coincidentally, CTCs with EMT phenotypes were found to be associated with inferior recurrence-free survival (RFS) and OS in NSCLC.55,56 In addition, some of the CTCs in NSCLC patients are cancer stem-like cells 57 (a small cluster of cells with a high capacity for self-renewal and resistance to treatment, which is considered the ultimate reason for cancer initiation and treatment failure)58-60, and the Hedgehog signaling pathway plays a key role in promoting the EMT and maintenance of these cells.61-63 On the basis of these data, it is plausible that the use of ATBs during surgery, although prophylactic and transient, could also have long-term effects on the gut microbiota 64 and is associated with poor outcomes in patients. In our study, multiple types of ATBs were identified as risk factors for survival but not independently, which may indicate that the influence of multiple types of ATBs on the gut microbiota overwhelms their individual role in anticancer effects; accordingly, its heterogeneous function in cancer may also be an explanation for its absence as an independent factor in the outcome.
On the basis of these data, we suggest that the use of ATBs during surgery in early-stage NSCLC is generally safe; however, caution should be taken in selecting the type of ATB; multiple types of ATBs should be avoided, and some specific types, such as fluoroquinolones, should not be administered.
Our study had several limitations. First, it was conducted retrospectively in a single hospital with relatively small samples, which did not undergo calculation and justification; in particular, cases for stages II to III are rare. Second, most of the patients suffered from adenocarcinoma, and other pathological features were rare. Third, a relatively high proportion of the cases were lost due to follow-up problems. All these limitations may result in potential biases and impair the conclusions. We believe that this effect can be attenuated only with larger sample sizes in the future. Finally, some patients may have had a history of ATB use before surgery (days or months before) because their pulmonary lesions may not be distinguishable from inflammation-related disorders by imageological examination (commonly computed tomography scan); detailed information on the use of ATBs during these periods was unavailable, which may also have affected the results, and these subpopulations should be excluded in future investigations.
Conclusion
Our study explored the prognostic value of ATB use during surgery in NSCLC patients for the first time and revealed that multiple types of ATBs, rather than cDDD, may be associated with OS in stage I to III patients. Interestingly, patients treated with fluoroquinolones may have poorer outcomes than those not treated with them. However, multiple types of ATBs were not validated as independent risk factors for OS. These results suggest that the use of ATBs during surgery in early-stage NSCLC is generally safe; however, caution should be taken when selecting ATB types. Multiple ATB types should be avoided, and some specific ATB types, such as fluoroquinolones, should not be administered. Nonetheless, owing to the limited sample sizes, future studies are needed to validate our results.
Footnotes
Acknowledgements
None.
Data Availability Statement included at the end of the article
Ethical Considerations
The study was approved by the ethics committee of Hainan Hospital of Chinese PLA General Hospital (ID: S2023-12) and mandatory written informed consent was not necessary owing to its retrospective nature according to the national regulations.
Authors’ Contributions
BY was responsible for conception of the study and wrote the manuscript. QY and BY obtain the data. MC, ML, and BY analyzed the data, and LL critically revised the article. All authors have read and approved the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated or analyzed during the current study are available from the first author (Bing Yan) upon reasonable request.
