Abstract
Background:
Bile duct carcinoma (BTC) is an uncommon malignant tumor of the gastrointestinal tract. Management is limited after the progress of first-line treatment. Immune checkpoint inhibitors (ICIs) have been proven popular in solid tumors. Immunotherapy plus chemotherapy has been a standard scheme in the management of multiple types of cancer. However, their efficacy and safety still need further exploration in patients who diagnosed BTC. This research mainly discusses the efficacy of immunotherapy in the second-line use of cholangiocarcinoma.
Methods:
In total, 126 individuals with BTC diagnosis from 2014 to 2024, who were treated with first-line or neoadjuvant treatment but were evaluated for progression or intolerance, were retrospectively included. All patients received standard chemotherapy, 57 received ICIs in combination with targeted therapy or not, and 69 did not. Patients were divided into simple chemotherapy (SC) and CT. Differences in efficacy, adverse events, progression-free survival (PFS), overall survival (OS), progressive disease (PD), and efficacy of multiple factors and efficacy were analyzed. The primary endpoint is defined as OS. The secondary endpoint is defined as PFS, objective response rate (ORR), disease control rate (DCR), and treatment-related adverse reactions (TRAEs).
Results:
The PFS and OS of 4.68 and 30.26 months for ICIs with or without targeted therapy were proven statistically significant (P = .0012; P < .001). The ORR was 5.26% (3/57) in the CT group and 1.45% (1/69) in the SC group, and the DCR was 54.39% (31/57) compared with 33.33% (23/69). Cox analysis showed that TNM stage, T stage, histology grade, CA199 level, and treatment assessment grade were associated with OS (P < .05). Histologic differentiation (P = .009) and CA199 reduced (P = .003) were proven as independent prognostic factors. The highest grade of 3 to 4 adverse reactions (TRAEs) was a reduction in hemoglobin (29.37%).
Conclusion:
Our work concluded that immunocombined chemotherapy with or without specific treatment showed significant antitumor activity and acceptable safety. Immune checkpoint inhibitors are likely to be a reliable second-line therapy for advanced BTC.
Introduction
Bile duct carcinoma (BTC), in particular BTC, accounts for about 3% of all gastrointestinal (GI) malignancies, and in the hepatocellular carcinoma (HCC), it is the second most common type. However, it has late diagnosis, high malignancy, insensitivity to drugs, and poor prognosis.1,2 Very few patients who diagnosed BTC were able to undergo surgery, and their 5-year survival rate is about 20%.3,4 Limited alternatives are available after diagnosis 5 and got limited improvement in patient. Furthermore, the efficacy of back line is limited further. The standard treatment of the second-line treatment for BTC does not reach a consensus. Several targeted therapies have been tested in large-scale clinical trials, such as HER2 (human epidermal growth factor receptor 2; NCT04722133), 6 FGFR2 (fibroblast growth factor receptor 2; NCT02052778, NCT02924376),7,8 and IDH1 (isocitrate dehydrogenase 1; NCT02989857). 9 Unfortunately, only limited patients are eligible to be applied and benefit from these regimens.
Immune checkpoint inhibitors (ICIs) have been applied to solid tumors, including advanced BTC, 10 especially as a back-line treatment. In Testis and Ovary-specific PAZ domain gene 1 (TOPAZ-1) 11 involving 685 patients with a diagnosis of bile duct cancer, the median time to deterioration of global health status or quality of life was 7·4 months in patients receiving durvalumab in combination with chemotherapy, demonstrating superior efficacy over chemotherapy alone. The KEYNOTE-028 study 12 demonstrated similar results in patients receiving pembrolizumab and mPFS, 5.7 and 1.8 months, respectively. A similar outcome was also demonstrated in the clinical study KEYNOTE-158. 13 Individuals with advanced BTC may be able to benefit from ICIs, but the results need to be further verified.11 -13 Despite to date ICI monotherapy has shown limited efficacy in BTC, the meaningful and durable responses to ICIs in mismatch repair (MMR)-deficient and microsatellite instability (MSI)-high malignancies including biliary cancers suggest that testing patients for MMR, MSI, TMB, and PD-L1 expression is warranted. 14 In addition, there are still differences in a number of clinical trials that get limited progress in the first-line therapy for advanced BTC, and ICIs have shown limited efficacy and the treatment-related adverse reactions (TRAEs).15,16
As said above, although many evidences proved by trials confirmed the reliability of efficacy in other solid tumors, the application of ICIs in second-line treatment of cholangiocarcinoma still remains potential research value. Angela Dalia Ricci’ study found a potential association between BRCAm and response to immune checkpoint inhibitors (ICIs). 17 Therefore, this retrospective study aims to talk about the use of ICIs for BTC in second-line treatment, at the same time, compare the efficacy and safety of immunotherapy combined with chemotherapy whether plus targeted therapy and chemotherapy alone and to provide certain clinical data support for the application of ICIs in the treatment of cholangiocarcinoma in the future.
Methods
Participant selection
Patients who were diagnosed with advanced BTC during the period 2014 to 2024 at Harbin Medical University Cancer Hospital were included: (1) BTC pathology, including intrahepatic cholangiocarcinoma (ICCA), hilar cholangiocarcinoma (HCCA), distal cholangiocarcinoma (DCCA), vater ampulla carcinoma (VPC), and gallbladder carcinoma (GCA); (2) measurable lesions according to RECIST1.1 18 , 19 ; (3) progression or intolerance after first- or second-line treatment with at least 2 courses and 1 efficacy assessment; and (4) Eastern Cooperative Oncology Group (ECOG) Score 0 to 2. 20 The primary exclusion criteria included: (1) incomplete or missing clinical data; (2) absence of measurable target lesions; and (3) pathological confirmation of other tumors within the past 5 years.
Research methods
Clinical information
Individual data were collected, such as identification, sex, age, height, weight, tobacco and alcohol, pathological diagnosis time, primary tumor site, histological grade, AJCC grade (American Joint Commission for Cancer), T stage, N grade, ECOG score, surgery history, neoadjuvant or adjuvant therapy, combined radiotherapy, ablation therapy, liver artery perfusion chemotherapy, additional ICIs, type of ICIs, additional target treatment, type of target drug, efficacy assessment, progression time, cause of progress, hematologic findings, radiological findings, genetic test results, TRAEs, survival status at final follow-up, time of death, and cause of death.
The research was divided into 2 groups: one was a combination of immunotherapy and chemotherapy whether using target therapy, and the other was a control group (SC group, chemotherapy alone). The 2 groups were treated with standard chemotherapy, including GP (gemcitabine plus cis-platinum), gemcitabine plus S-1 (GS), and gemcitabine plus oxaliplatin (GEMOX). 21 Patients in the combination therapy group were given additional immunotherapy with or without targeted chemotherapy.
Follow-up data included hospital visits and phone calls. The last follow-up visit occurred in May 2024. Overall survival (OS) is defined as the time from the beginning of therapy until death from any cause. Progression-free survival (PFS) is defined as the time from the start of the first cycle of therapy until disease progression or death. The primary end point is defined as OS. At the same time, the secondary end point is defined as PFS, objective response rate (ORR), DCR, and TRAEs.
Response evaluation criteria
Both groups were assessed according to the solid tumor response evaluation criteria (RECIST1.1), which are classified as progressive disease (PD), steady status (SD), partial response (PR), and complete response (CR). 18 Classification TRAEs were used in the Toxicity Criteria version 5.0. The percentage of PDs was defined as the disease control rate (DCR), and the ratio between CR and PR was defined as the total response rate (ORR).
Statistical analysis
Data analysis was performed by R (Version 4.3.2). Continuous variables following a normal distribution are recorded as average values (standard deviation). Continuous variables with non-normal distribution are recorded using standard deviation (first and third quartiles). Two sets of categorical variables were compared using either Fisher exact test or the chi-square test. Single-factor and multiple-factor analysis methods are Cox regression analysis that evaluates the impact of variables on survival rate. Kaplan-Meier survival curves were constructed and compared using the log-rank test. A P value less than .05 was considered statistically significant.
Results
Clinicopathological features of participants
In total, 126 participants were enrolled with a diagnosis of advanced BTC in the study (Figure 1 and Table 1). Of these, 69 participants were randomly assigned to the simple chemotherapy (SC) group, and remaining 57 were included in the combined therapy (CT) group. As for the CT group, immunotherapy drugs that participants were primarily using PD-1 inhibitors, for example, sintilimab (25), camrelizumab (14), toripalimab (9), and tirelizumab (1). Meanwhile, some individuals in the CT group were treated with targeted therapy drugs such as lenvatinib (14), apatinib (4), bevacizumab (3), donafenib (2), and abrocitinib (1). Both arms were discontinued when individuals cannot tolerant treatment or found disease progression. The baseline table (Table 1) indicated that there were no statistically significant differences in baseline clinical parameters, for instance, the age (P = .214), ECOG score (P = .372), T stage (P = .432), diagnosis (P = .125), N stage (P = .06), liver metastases (P = .245), multiple metastasis status (P = 1), surgical treatment (P = .201), carcinoembryonic antigen [CEA] (P = .662), and CA199 (P = .615). Otherwise, there were several variables that were statistically significant, such as sex (P = .033), whether smoking (P = .001), whether alcohol (P = .008), AJCC grade (P = .012), histological differentiation (P = .031), CEA (P = .01), CA199 (P = .002), chemotherapy regimen (P < .001), type of targeted therapy (P < .001), type of PD-1 ICIs (P < .001), and PD-L1 ICIs (P = .017). Otherwise, all participants received upfront treatment mainly about traditional chemotherapy such as GEMOX, oxaliplatin plus capecitabine (XELOX), and GS. Above all, 42 patients were treated with ICIs, 22 of them were given PD-1 inhibitors such as sintilimab (12), camrelizumab (8), toripalimab (7), pembrolizumab (3), and tislelizumab (2), and the remaining patients were treated with PD-L1 inhibitors, for instance, durvalumab (7) and envafolimab (4). Patients who were identified progression after second-line treatment mainly were treated with single ICI or single chemotherapy such as S-1.

The flow chart of this study.
Comparison of disease characteristics in patients with advanced biliary tract tumors.
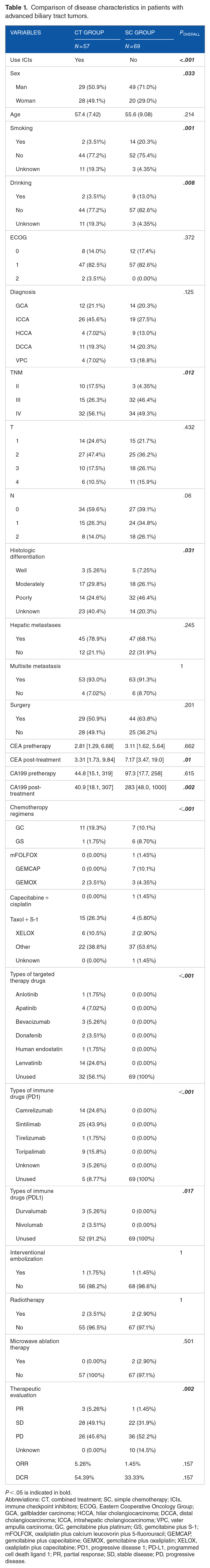
P < .05 is indicated in bold.
Abbreviations: CT, combined treatment; SC, simple chemotherapy; ICIs, immune checkpoint inhibitors; ECOG, Eastern Cooperative Oncology Group; GCA, gallbladder carcinoma; HCCA, hilar cholangiocarcinoma; DCCA, distal cholangiocarcinoma; ICCA, intrahepatic cholangiocarcinoma; VPC, vater ampulla carcinoma; GC, gemcitabine plus platinum; GS, gemcitabine plus S-1; mFOLFOX, oxaliplatin plus calcium leucovorin plus 5-fluorouracil; GEMCAP, gemcitabine plus capecitabine; GEMOX, gemcitabine plus oxaliplatin; XELOX, oxaliplatin plus capecitabine; PD1, progressive disease 1; PD-L1, programmed cell death ligand 1; PR, partial response; SD, stable disease; PD, progressive disease.
Outcomes and survival analysis
All 126 patients underwent a minimum of 2 regular treatments and had at least 1 efficacy evaluation. In the CT group, 5.26% (3/57) of patients achieved PR, 49.1% (28/57) of patients achieved SD, and 45.6% (26/57) of patients achieved PD. In the SC group, 1.45% (1/69) of patients achieved PR, 31.9% (22/69) of patients achieved SD, and 52.2% (36/69) of patients achieved PD. Both arms showed that no patients achieved CR. The combined treatment group exhibited an ORR rate of 5.26%, compared to 1.45% in the SC group; however, statistically significant difference was not shown (P = .157). The combined arm had a DCR of 54.39%, instead of 33.33% in the SC arm, which did not demonstrate significant difference between the arms (P = .157) (Table 1). By observing the CT group, the most common immune drug was sintilimab (43.9%) and the most common target drug was lenvatinib (24.6%). The survival curve indicated that the CT group showed delightfully significantly higher PFS (4.68 ± 4.93) and OS (30.26 ± 26.54) compared with the SC group, which had OS (17.14 ± 7.19) and PFS (3.50 ± 3.19) (P < .001; P = .0012) (Figure 2). In addition, in the subgroup under the CT group, the OS for the ICIs with target therapy and chemotherapy arm was longer than the ICIs combined with chemotherapy arm, but this difference did not identified statistically significant (64.12 months ± 17.41 vs 43.28 months ± 7.43; P = .85; Figure 3). By analyzing the ICIs combined with chemotherapy group, we found that 1 (3.12%) patient had PR, 15 (46.9%) patients had SD, 16 (50%) patients had PD, the ORR was 3.13%, and the DCR was 50%. In another group of 25 patients who received ICIs combined with targeted therapy and chemotherapy, 2 patients (8%) were evaluated as PR, 13 patients (52%) were identified as SD, and 10 patients (40%) experienced PD, resulting in an ORR of 8% and a DCR of 60%. Although the comparison between the 2 groups showed an improvement in ORR and DCR after adding targeted therapy, unfortunately, this was not statistically significant (P = .157; P = .157) (Table 2).

Kaplan-Meier curves of overall survival (the left) and progression-free survival (the right) between combined treatment and simple chemotherapy groups.

Kaplan-Meier curves of overall survival between the ICIs combined with chemotherapy group and the ICIs with target therapy with chemotherapy group.
Univariate and multivariate Cox analyses of OS prognostic factors.

P < .05 is indicated in bold.
Univariate and multifactorial results of clinicopathological feature associated with OS
It showed that AJCC stage, T stage, histological grade, whether CA199 decreased, and therapeutic evaluation grade of all 126 patients were related to OS after univariate analysis (P < .05, Table 3). It demonstrated that well histologic differentiation (P = .009) and CA199 decreased (P = .003) were independent prognostic factors among the above variables for OS after further multivariate Cox regression analysis (Table 3).
Adverse reactions in 2 groups.

Adverse events
All 126 patients completed a minimum of 2 cycles of the second-line treatment. Table 4 presents the adverse events (AEs) for the enrolled patients. Among the 126 patients, the most frequent nonhematologic AE was stomachache (22.22%), whereas the most prevalent hematologic AE was a decrease in hemoglobin count (74.6%). The most frequent nonhematologic AE in both groups was stomachache (23.19% vs 21.05%), whereas the most common hematologic AE was decreased serum albumin (84.06% vs 61.41%). In the CT group, ulteriorly, decrease in hemoglobin count (31.58%) was the first frequent grade 3 to 4 AE. Conversely, the highest incidence of grades 3 and 4 was a decrease in hemameba count (31.88%). After managed by symptomatic medications, all grade 3 to 4 TRAEs were relieved and did not impact subsequent treatment. Most grade 1 to 2 TRAEs resolved after treatment suspension or symptomatic medication, without impacting subsequent treatment.
Treatment efficacy and survival condition for the combined treatment group.

Abbreviations: ICI, immune checkpoint inhibitors; CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease; ORR, objective response rate; DCR, disease control rate.
Discussion
Our study demonstrated the efficacy and safety of immunotherapy as the second-line treatment for advanced BTC. This outcome further enriches the treatment of cholangiocarcinoma and provides reliable clinical data for follow-up studies.
In our study, the OS showed 30.26 months in the CT arm and 17.14 months in the SC arm, with prolonged 13.12 months (P < .001) that suggests a statistically significant difference. The PFS of the CT group was 4.68 months and the SC group was 3.50 months, showing a statistically significant difference of 1.18 months (P = .0012). By comparing the SC group, the CT arm demonstrated a 3.81% increase in ORR (5.26% vs 1.45%) and a 21.06% increase in DCR (54.39% vs 33.33%). 22 This DCR improvement is 20.39% higher than that reported in the KEYNOTE-028 trial (34%). 22 The use of ICIs may benefit patients who diagnosed BTC in second-line treatment. This result aligns with the findings of the NCT03111732 trial. 23 However, the ORR and DCR did not show statistically significant difference (P = .157; P = .157), which due to the small amount of data. Kim JW’s research (NCT03875235) showed that the ORR for patients receiving durvalumab, tremelimumab, and chemotherapy as second-line treatment was 50% (15/30). 24 This outcome did not show a significant improvement compared with the 70% (33/47) ORR observed in patients using ICIs in the first-line therapy. Compared with ICIs combined with chemotherapy alone, ICIs combined with chemotherapy and target therapy showed a 4.87% increase in ORR (8% vs 3.13%, P = .157) and a 10% increase in DCR (60% vs 50%, P = .157). No statistical differences were observed. We also found that OS was not statistically difference between the ICIs combined with chemotherapy group and the ICIs with target therapy with the chemotherapy group (P = .85).
Our analysis identified TNM-III (P = .003), TNM-IV (P = .006), T3 (P = .024), T4 (P = .025), well and moderately differentiated histology (P = .001, P = .007), decreased CA199 (P = .006), SD (P = .025), and PD (P = .014) as independent prognostic factors related to OS (Table 2). These findings demonstrate the effectiveness of combining immunotherapy with chemotherapy, whether plus targeted therapy, for second-line therapy of advanced BTC.
Immunotherapy has shown significant therapeutic effects in various carcinomas and is approved for first-line treatment of advanced biliary duct carcinoma. Treatment-related adverse reactions were observed in both groups. Consistent with prior clinical studies for immunotherapy, common TRAEs include leukopenia, neutropenia, thrombocytopenia, hyperbilirubinemia, hypoalbuminemia, and stomachache. Most patients can be managed with dose adjustments or symptomatic treatment, avoiding the need for discontinuation due to TRAEs.
Our findings indicate that combining immunotherapy with chemotherapy, whether plus target therapy, is safe and effective management as a second-line therapy for advanced cholangiocarcinoma. Nevertheless, our study has some limitations. First, it prevents drawing meaningful conclusions on the efficacy whether pulsing target therapy for treating advanced BTC patients, due to the limitation of sample size. We think that promoting actively regular checkups can help address this issue. Second, there is inherent selection and bias because individuals are mainly concentrated in a single center. Therefore, combined with multicentered, cross-regional research collaborations are necessary. Third, assessing PD-1/PD-L1 expression levels were not available in ICI-treated individuals, which limiting our ability to further explore the impact of gene expression on immunotherapy. Encouraging genetic testing may solve the problem. Fourth, this study also did not record gene detection information so that it was impossible to explore the correlation between related genes and efficacy. Fifth, some baseline variables remain differences. With the deepening of the study of tumor microenvironment, the efficacy of immunotherapy may be further improved. Next 5 years, with the maturity of CAR-T therapy, the treatment of cholangiocarcinoma can be taken to a higher stage. The survival benefit of combining immunotherapy with or without targeted therapy, with chemotherapy, for second-line treatment of advanced BTC patients remains uncertain. Further prospective, multicenter cohort studies with large samples and extended follow-up periods are necessary.
Conclusions
In summary, combining immunotherapy with chemotherapy, with or without targeted therapy, significantly improves ORR, OS, and PFS in second-line advanced BTC patients while maintaining acceptable tolerability compared with chemotherapy alone. Multicenter studies are necessary to evaluate the long-term prognosis improvements associated with this treatment.
Footnotes
Acknowledgements
The authors acknowledge all individuals who were included that allowed us to collect and analyze their clinical characteristics. They thank all the doctors at Harbin Medical University Cancer Hospital for providing us with assistance within our capabilities.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Conception/design: CJ. Provision of study material or patients: CJ, LY, and LK. Collection and/or assembly of data: CJ, LY, LK, TF, LC, LH, QM, YC, and WW. Data analysis and interpretation: CJ, LY, LK, TF, ZM, and Q.L. Conceptualization, methodology, and validation: ZM Supervision, conceptualization, resources, and writing: QL and JT. Article writing and final approval of article: All authors.
Availability of Data and Materials
The data sets used and/or analyzed during this study are available from the corresponding author on reasonable request.
Ethics Approval and Consent to Participate
The authors confirm that all methods were conducted according to the principles of the Declaration of Helsinki and were approved by The Institutional Review Committee and the Medical Ethics Committee of the Third Hospital of Harbin Medical University (Approval No: KY2023-22, Date: February 15, 2023). Written informed consents have been obtained from all patients.
Consent for Publication
All participants have signed an informed consent form indicating their consent to publish clinical data relevant to them in the article.
