Abstract
Objective
As monotherapy such as topotecan has reached a plateau of effectiveness, new second-line treatments based on experience have been used in clinical application. This study compared the efficacy and safety of different second-line treatments for advanced small-cell lung cancer (SCLC).
Methods
A total of 380 patients with advanced SCLC were screened selectively in the retrospective study. Adverse events and patient responses were assessed using Common Terminology Criteria for Adverse Events v5.0 and Response Evaluation Criteria for Solid Tumors v1.1. The progression-free survival (PFS) was estimated using the Kaplan-Meier method or Cox survival regression model and compared using the log-rank test.
Results
In the platinum-resistant group, disease control rate (DCR) and median PFS (mPFS) were prolonged in the combination group versus single-agent group (DCR: 49.24% vs 24.39%, P = .004; mPFS: 3.73 vs 1.90 months, P < .001). Grade 3/4 toxicity was similar between the 2 groups (P = .683). The mPFS did not differ among single-agent groups (P = .380). No significant difference was observed in mPFS of different combination therapy groups (P = .170). In terms of platinum-based chemotherapy, the DCR and mPFS were prolonged in irinotecan-platinum group versus taxol-platinum group (DCR: 56.14% vs 9.09%, P = .004; mPFS: 3.87 vs 1.93 months, P = .012). Grade 3/4 toxicity was similar between the 2 groups (P = .614). The mPFS was prolonged in the chemotherapy plus immunotherapy group versus single-agent chemotherapy group (P = .003). In the platinum-sensitive group, the mPFS did not differ between the combination group and single-agent group (P = .200). The mPFS did not differ among different single-agent groups (P = .260) or combination groups (P = .150). There was no difference in mPFS among different platinum-based chemotherapy groups (P = .830).
Conclusions
For patients with platinum-resistant SCLC, combination therapy has shown better efficacy and acceptable toxicity profile than monotherapy. Among combination therapies, irinotecan–platinum has shown better efficacy than taxol–platinum. For patients with platinum-sensitive SCLC, the efficacy of different single-agent or combination therapies was similar.
Introduction
Lung cancer is the second most commonly diagnosed cancer and the leading cause of cancer death, with an estimated 2.2 million new cases and 1.8 million deaths in 2020. 1 Small-cell lung cancer (SCLC) comprises approximately 15% of all lung cancer cases. It is characterized by genetic complexity, high invasiveness, and poor survival. Despite high response to initial treatment, most patients with SCLC will relapse or progress after first-line treatment. More than two-thirds of patients with SCLC present with extensive stage SCLC (ES-SCLC) beyond treatment potential. 2 The 1-year and 2-year overall survival (OS) rates are 58% and 21% for limited-stage SCLC, respectively, and 29.4% and 7% for ES-SCLC, respectively. 3
Unfortunately, few drugs are accessible for patients with SCLC during progression after initial treatment. For SCLC with recurrence ≤6 months, topotecan, lurbinectedin, and clinical trials are reasonable treatment options. For SCLC with recurrence >6 months, the original regimen, topotecan, or lurbinectedin may be considered. Topotecan was the only drug approved for second-line treatment of SCLC in the United States and Europe prior to June 2020, which showed a significant improvement in OS (25.9 weeks vs 13.9 weeks) compared with best supportive care. 4 Furthermore, compared with CAV combination chemotherapy, it had similar activity (24.3% vs 18.3%) and median OS (mOS) (25.0 weeks vs 24.7 weeks). 5 Overall, 7% to 24% of patients reported as achieving a response, and the mOS usually ranges between 6 and 9 months. Undeniably, the clinical efficacy of topotecan is relatively modest, and the hematological toxicity is significant. 6 On the basis of the data reported in a single-arm phase II trial, the objective response rate (ORR), disease control rate (DCR), and mOS of lurbinectedin were 35.2%, 68.6%, and 9.3 months, respectively. 7 Although lurbinectedin was approved by the FDA in 2020, the lack of approval for marketing in some countries, such as China, has limited its access.
Some drugs such as taxol drugs are commonly used in clinical practice in second-line treatment because of their low toxicity profile and somehow comparable clinical activity with topotecan. Nab-paclitaxel is currently approved for advanced non-small cell lung cancer (NSCLC). 8 For SCLC, the first phase II study investigating albumin paclitaxel for relapse SCLC has shown some modest antitumor activity associated with a favorable toxicity profile. 9 The RR and mOS were 8% and 3.6 months in the refractory group, respectively, but 14% and 6.6 months in the sensitive group, respectively. The median progression-free survival (mPFS) was similar in the refractory group (1.8 months) and the sensitive group (1.9 months). However, retrospective studies with large sample sizes necessary to explore the efficacy of taxol drugs comparing with topotecan are limited.
Although the recommended scheme for second-line treatment is a single drug, the second-line treatment of SCLC is not strictly in accordance with the guidelines based on the effect of drug efficacy and toxicity. New second-line treatment options are emerging, such as the trend toward combination therapy. A phase II experiment of camrelizumab plus apatinib showed potential antitumor activity in patients with both chemotherapy-sensitive and chemotherapy-resistant ES-SCLC who had failed platinum-based chemotherapy. 10 In a prospective, single-arm, phase II trial, combining sintilimab with anlotinib as second- or later-line therapy in patients with ES-SCLC showed good clinical efficacy and acceptable toxicity. 11 The above clinical trials confirmed the potential of therapies outside the guidelines as second-line treatment options for patients with ES-SCLC. As a result of the encouraging OS benefit results of IMpower 133 and CASPIAN,12,13 immunotherapy plus platinum-based chemotherapy has become a commonly used standard treatment regimen in first-line settings. Given these results, some studies on immune drugs have been conducted in second-line therapy.
This study aimed to analyze and compare the efficacy and toxicity of different second-line treatments in advanced SCLC, providing a reference for clinical decision-making. Our study retrospectively reviewed 380 patients who received second-line treatments for advanced SCLC.
Materials and Methods
Patients
We retrospectively collected data of patients with advanced SCLC who underwent second-line treatment between January 1, 2017 and July 30, 2022 from the medical records at Jiangsu Cancer Hospital. Patients relapsing or progressing beyond 90 days were defined as platinum-sensitive relapse patients, and those relapsing or progressing before 90 days were defined as platinum-resistant relapse patients. The patients’ basic information (ie, sex and age), therapy regimen and cycle clinical assessment, response, and toxicity were recorded. The inclusion criteria were as follows: (1) patients histologically or cytologically diagnosed with ES-SCLC; (2) patients aged 18 to 80 years; (3) patients who scored 0 to 2 on the Eastern Cooperative Oncology Group performance status; (4) patients who had received second-line treatment after failure of first-line platinum-based therapy; (5) patients included in this study had at least 1 measurable disease. The exclusion criteria were (1) patients aged >80 years; (2) patients with serious heart, liver, kidney, and other important organ diseases; (3) patients with active autoimmune disease or related history; (4) patients for whom follow-up information is not available. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This retrospective study was approved by the Academic Ethics Committee of Jiangsu Cancer Hospital, approval number (NO. [2023]081), and individual consent for this retrospective analysis was waived. We have removed all patient details to ensure the confidentiality of patient information. The reporting of this study conforms to STROBE guidelines. 14
Response and Toxicity
The objective tumor response measures included complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD). CR was defined as the complete disappearance of all target lesions and all non-target lesions, if present. PR was recorded when the sum diameters of target lesions reduced by at least 30%, taking as reference the baseline sum diameters. PD was recorded when the sum diameters of target lesions increased by at least 20%, taking as reference the smallest sum on study, or the appearance of new lesions and/or unequivocal progression of pre-existing non-target lesions. SD was recorded when the sum diameters of target lesions increased to less than PD, or reduced to less than PR. ORR was defined as the percentage of patients with CR and PR. DCR was defined based on the cumulative objective response and stabilization rates (CR + PR + SD). The ORR was evaluated at baseline and every 2-3 cycles of treatment by computed tomography or magnetic resonance imaging, according to the Response Evaluation Criteria in Solid Tumors 1.1. Adverse events (AEs) were assessed using the Common Terminology Criteria for AEs v5.0.
Statistical Analysis
Progression-free survival (PFS) was defined from the time of the first cycle of second-line therapy to disease progression, death from any cause, or last follow-up date. PFS was determined via the Kaplan–Meier method and compared by the log-rank test. Univariate analyses were conducted using Cox proportional hazards regression models. The ORR and DCR were compared by the chi-square test. All statistical analyses were conducted using the SPSS (version 26.0) and R software (version 3.6.3). P < .05 was considered statistically significant.
Results
Patient Characteristics
A total of 380 patients with advanced SCLC received second-line treatment from January 1, 2017 to July 30, 2022 at Jiangsu Cancer Hospital. A total of 142 patients were classified as sensitive relapse and 238 patients were classified as resistant relapse. Baseline clinical and pathological features are summarized in Table 1.
Clinical Characteristics of Patients and Clinical Activity of Second-Line Treatment.
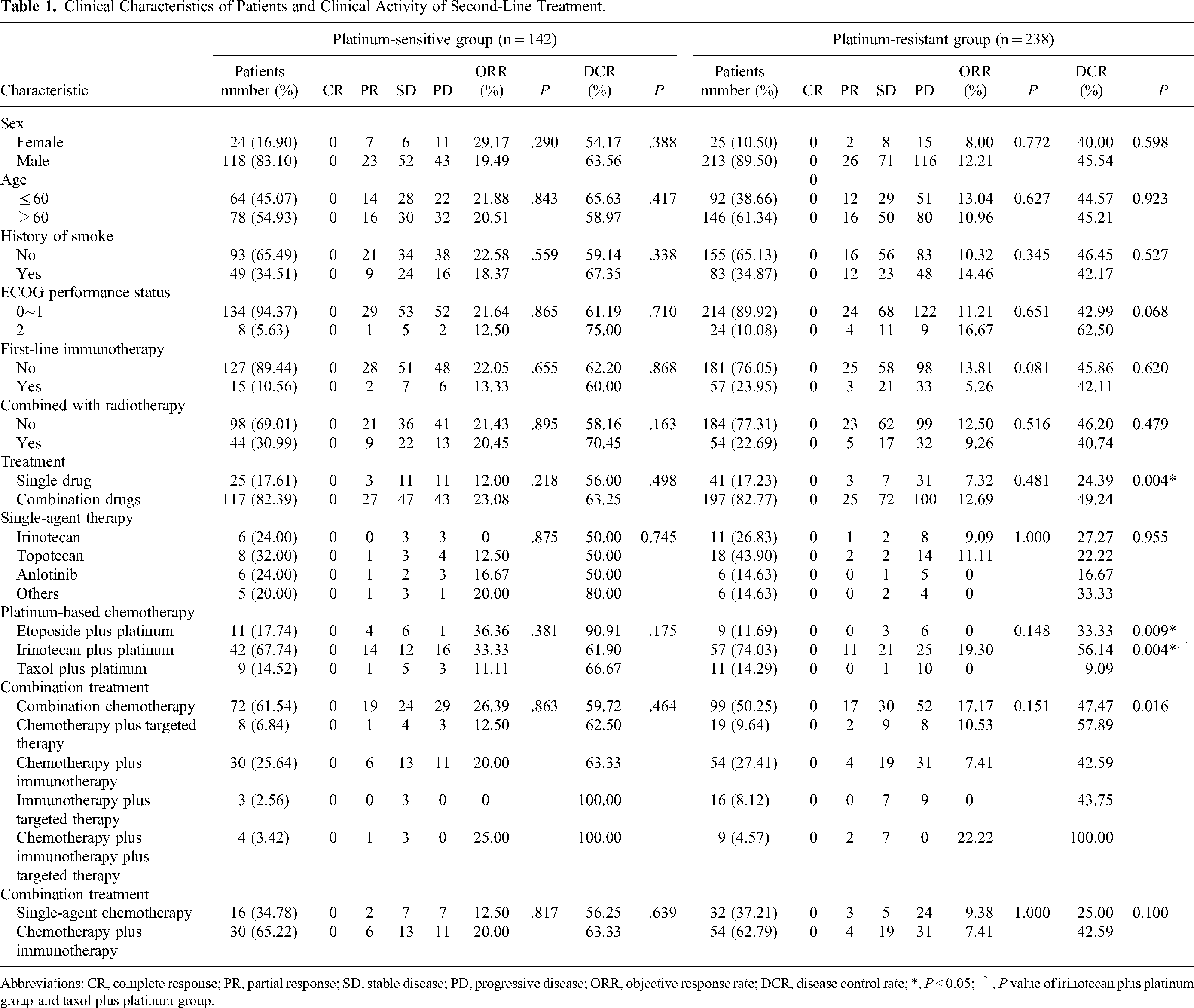
Abbreviations: CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease; ORR, objective response rate; DCR, disease control rate; *, P < 0.05; ^, P value of irinotecan plus platinum group and taxol plus platinum group.
Treatment Characteristics
All 380 patients were assessable for response (Table 1). In the platinum-resistant group, 41 patients received a single drug, which were irinotecan (11/41, 26.83%), topotecan (18/41, 43.90%), anlotinib (6/41, 14.63%). Other monotherapies were grouped together, which were paclitaxel (2/41, 4.88%), apatinib (1/41, 2.44%), etoposide (1/41, 2.44%), toripalimab (1/41, 2.44%), and atezolizumab (1/41, 2.44%). A total of 197 patients were treated with combination drugs, which included combination chemotherapy (99/197), chemotherapy plus targeted therapy (19/197), chemotherapy plus immunotherapy (54/197), immunotherapy plus targeted therapy (16/197), and chemotherapy plus immunotherapy plus targeted therapy (9/197). And 77 patients received platinum-based chemotherapy, including 9 cases of etoposide plus platinum, eleven cases of taxol plus platinum, and 57 cases of irinotecan plus platinum. Fifty-four patients received radiotherapy, and 57 received first-line immunotherapy.
In the platinum-sensitive group, 25 patients were treated with a single drug and 117 received combination drugs. Among the single-agent group, 6 patients received irinotecan, 8 received topotecan, 6 received anlotinib. Other monotherapies included etoposide (2 of 25), toripalimab (2 of 25) and apatinib (1 of 25). Among the combination treatment group, 72 patients received combination chemotherapy, in which 62 received platinum-based chemotherapy such as etoposide plus platinum, taxol plus platinum, and irinotecan plus platinum. Eight patients received chemotherapy plus targeted therapy. Exactly 30 patients received chemotherapy plus immunotherapy. Three patients received immunotherapy plus targeted therapy. Four patients received chemotherapy plus immunotherapy plus targeted therapy. Forty-four patients received radiotherapy, and 15 received first-line immunotherapy.
Response and PFS
In the platinum-resistant group, the DCR and mPFS were significantly higher in combination group compared with the single-agent group (DCR: 49.24% vs 24.39%, P = .004; mPFS: 3.73 vs 1.90 months, P < .001, Figure 1A). Among different single-agent groups (including irinotecan, topotecan, anlotinib, and others), no significant differences in mPFS were observed (P = .380, Figure 1B). No significant difference was observed in mPFS of different combination therapy groups (chemotherapy vs chemotherapy plus targeted therapy vs chemotherapy plus immunotherapy vs immunotherapy plus targeted therapy vs chemotherapy plus immunotherapy plus targeted therapy: 3.73 vs 4.73 vs 3.33 vs 3.80 vs 5.33 months, Figure 1C). The DCR and mPFS among the different platinum-based chemotherapy groups (etoposide–platinum, irinotecan–platinum, and taxol–platinum) were significantly different (DCR: P = .009; mPFS: P = .046, Figure 1D). The DCR and mPFS of the irinotecan–platinum group were significantly higher than those of the taxol–platinum group (DCR: 56.14% vs 9.09%, P = .004; mPFS: 3.87 vs 1.93 months, P = .012, Figure 1E). However, there was no differences in mPFS between irinotecan–platinum group and etoposide–platinum (3.87 vs 2.67 months, P = .710). And similar negative result was observed in mPFS between taxol–platinum group and etoposide–platinum (1.93 vs 2.67 months, P = .230). However, compared with the single-agent chemotherapy group, the chemotherapy plus immunotherapy group had a longer mPFS (3.33 vs 1.90 months, P = .003, Figure 1F).

Kaplan–Meier curves in the platinum-resistant group. (A) Progression-free survival (PFS) in the single-agent and combination drug groups. (B) PFS of different single-agent groups. (C) PFS of different combination therapy groups. (D) PFS of different platinum-based chemotherapy groups. (E) PFS in the irinotecan plus platinum group and taxol plus platinum group. (F) PFS in the single-agent chemotherapy group and chemotherapy plus immunotherapy group.
In the platinum-sensitive group, no difference was found in mPFS between the combination group and single-agent group (P = .200, Figure 2A). No significant differences in mPFS were observed among different single-agent groups (P = .260, Figure 2B). There was no difference in mPFS among different combination therapy groups (chemotherapy vs chemotherapy plus targeted therapy vs chemotherapy plus immunotherapy vs immunotherapy plus targeted therapy vs chemotherapy plus immunotherapy plus targeted therapy: 4.53 vs 5.05 vs 4.93 vs 6.90 vs 8.70 months, Figure 2C). The mPFS did not differ among different platinum-based chemotherapy groups (P = .830, Figure 2D). The chemotherapy plus immunotherapy group did not translate into superior mPFS compared with the single-agent chemotherapy group (P = .120, Figure 2E).

Kaplan–Meier curves in the platinum-resistant group. (A) Progression-free survival (PFS) in single-agent and combination drug groups. (B) PFS of different single-agent groups. (C) PFS of different combination therapy groups. (D) PFS of different platinum-based chemotherapy groups. (E) PFS in the single-agent chemotherapy group and chemotherapy plus immunotherapy group.
Combination therapy or monotherapy was a significant factor affecting the PFS in the platinum-resistant group (P < .001). Age, sex, smoke, ECOG score, previously treated with immunotherapy, and combined radiotherapy were not the major factors affecting the PFS in both groups (Table 2).
Univariate Analysis of Factors of Progression-Free Survival.

Safety
As shown in Table 3, 52.6% (200/380) of patients experienced treatment-related AEs, and 19.7% (75/380) patients experienced grade 3 or 4 treatment-related AE. The most common treatment-related AEs were leukopenia (32.6%), followed by nausea/vomiting (8.4%), thrombocytopenia (5.8%), and elevated alanine aminotransferase (ALT) or aspartate transaminase (AST) (3.9%). No grade 5 treatment-related AE was reported. In the platinum-resistant group, the incidences of AEs at any grade and grade 3/4 were similar in the single-agent and combination treatment group (any grade: 39.0% vs 53.3%, P = .096; grade 3/4: 14.6 vs 17.3%, P = .683). The toxicities were similar between the irinotecan plus platinum group and taxol plus platinum group (any grade: 57.9% vs 45.5%, P = .668; grade 3/4: 21.1% vs 9.1%, P = .614). The incidence and severity of grade 3/4 toxicity were similar in the chemotherapy plus immunotherapy group and single-agent chemotherapy group (16.7% vs 18.8%, P = .806).
Treatment-Related Adverse Events for Total Patients and Patients with Platinum-Resistant Relapse.

Number of patients with an event (percent). Abbreviations: ALT, alanine aminotransferase; AST, aspartate transaminase.
Discussion
Previous clinical trials have suggested the potential of combination therapy as a second-line treatment for SCLC. In our study, for patients with platinum-resistant relapse, both short-term and long-term efficacy were better in the combination treatment group than in the monotherapy group. Considering that patients with poor PS tend to prefer monotherapy due to their inability to tolerate treatment-related toxicity, we screened both groups for confounding factors. The results showed that the baseline characteristics of the 2 groups were basically balanced and comparable. Multivariate analysis showed that combination therapy or monotherapy was indeed a significant factor affecting the PFS in the platinum-resistant group. Given that different drugs act on various targets or cells, synergy or combination therapy may achieve greater therapeutic effects at the cost of similar side effects. Further safety comparisons were made between the 2 groups. Although the incidence of any grade and grade 3/4 AE was numerically higher in the combination group than in the monotherapy group, the difference was not statistically significant. Therefore, the toxicity of combination therapy was considered acceptable.
Among the single-agent groups, single-agent irinotecan or single-agent anlotinib failed to show better mPFS than topotecan in both the platinum-resistant group and platinum-sensitive group. Only a few studies have compared the efficiency of topotecan and irinotecan monotherapy for SCLC second-line therapy. A recent phase 3 part of the RESILIENT trial indicated that liposomal irinotecan did not translate into superior OS compared with topotecan, which had just been reported in a press release. 15
Among the platinum-based chemotherapy groups in the platinum-resistant group, the irinotecan–platinum group had higher DCR and mPFS than the taxol–platinum group. The toxicities between the 2 groups were similar. To date, there is no consensus that irinotecan–platinum is superior to other platinum–chemotherapy regimens. A previous study demonstrated that irinotecan–platinum treatment shows extended PFS compared with taxol–platinum treatment (P < .05), 16 which was consistent with our results. Moreover, several studies have shown no difference in efficacy between irinotecan–platinum treatment and taxol–platinum treatment.17,18 The above treatment regimens warrant further comparisons and investigations involving a large sample size or prospective studies.
Unfortunately, different platinum-based chemotherapy regimens had similar basic progression control times in patients with platinum-sensitive SCLC. And no significant difference was noted in mPFS between monotherapy and combination treatment. Overall, our study failed to show any better regimen than topotecan for platinum-sensitive SCLC second-line therapy. Two phase 3 trials of platinum-based chemotherapy in the second-line treatment of sensitive relapsed SCLC have reported positive results.19,20 The first trial was a comparison study of triplet chemotherapy with cisplatin, etoposide, and irinotecan versus topotecan as monotherapy. Although triplet chemotherapy achieved better outcomes than topotecan (mOS: 18.2 vs 12.5 months, P = .0079; mPFS: 5.7 vs 3.6 months, P < .0001; ORR: 84% vs 27%, P < .0001), the triplet regimen was not adopted as standard care worldwide mainly because of excessive toxicity. 20 The second trial, a randomized phase 3 French trial in 2020, reported that rechallenge chemotherapy consisting of the same regimen (carboplatin–etoposide) can lead to a better ORR (49% vs 25%, P = .004) and mPFS (4.7 vs 2.7 months, P = .004) compared with single-agent topotecan. 19 Our analysis failed to further compare etoposide–platinum with topotecan due to the limited sample size, so retrospective studies with large sample sizes are expected in the future.
With the wide application of immunotherapy in first-line settings, some studies on immune drugs have been conducted in second-line or later-line therapy. In single-arm phases I and II trials, about 11% to 19% of patients with relapsed SCLC showed a consistent clinical response in nivolumab or pembrolizumab monotherapy, with 21% having a 2-year survival rate.21–23 Unfortunately, in a subsequent phase III clinical trial (CheckMate 331), nivolumab monotherapy showed no promising data in OS than chemotherapy (7.5 vs 8.4 months, P = .11). 24 Similarly, the IFCT-1603 trial did not show a significant efficacy signal for atezolizumab monotherapy compared with chemotherapy (mOS: 9.5 vs 8.7 months, P = .60). 25 These results suggested that anti-programmed cell death protein-1 (PD-1)/programmed death ligand-1 (PD-L1) monotherapy may have limited efficacy in relapse SCLC. In the 2022 World Conference on Lung Cancer, a phase 2 study of temozolomide combined with nivolumab in the treatment of relapse SCLC was conducted. Seven of the 25 evaluable patients achieved response with an ORR of 28%, which was better than the historical data of immune monotherapy for relapse SCLC. 26 Another phase 2 study of pembrolizumab in combination with paclitaxel was shown to be effective with an acceptable tolerability profile in patients with refractory ED-SCLC. 27 In our study, this trend of increased benefit from immunotherapy combined with chemotherapy was observed in the platinum-resistant group, in which the immunotherapy group had a longer mPFS than the single-agent chemotherapy group. However, the encouraging PFS benefit results by using immunotherapy were not observed in the combination chemotherapy group. In the platinum-sensitive group, the immunotherapy group did not translate into superior mPFS compared with single-agent chemotherapy or combination chemotherapy. Thus, the immunotherapy plus single-agent chemotherapy regimen in platinum-resistant SCLC may be an important direction to explore.
Some patients cannot tolerate chemotherapy toxicity, another treatment such as immunotherapy combined with targeted therapy thus be explored for this population. A phase II study (Passion trial) demonstrated that camrelizumab plus apatinib exhibited potential antitumor activity in patients who had failed platinum-based chemotherapy with an acceptable toxicity profile. 10 At the same time, the ANCHER-04 phase II clinical study showed that penpulimab combined with anlotinib had significant efficacy in patients who had failed platinum-based chemotherapy. The ORR was 42.86%, the DCR was 71.43%, and the mPFS was 4.62 months, respectively. 28 However, in our study, immunotherapy combined with targeted therapy did not show better mPFS compared with other treatment regimens.
Previous clinical trials on NSCLC suggest that first-line immunotherapy has a positive effect on the efficacy of posterior therapy. PFS2 is defined as the time from randomization to a second disease progression or death. When the immunotherapy group was cross-treated with other treatments after progression, it showed significantly longer PFS2 than the control group (Keynote 407 trial: 13.8 vs 9.1 months, P < .001; Keynote 024 trial: 18.3 vs 8.4 months, P < .001).29,30 However, in our study, the mPFS did not significantly differ between the 2 groups in the platinum-resistant group, as well as in the platinum-sensitive group. Our results suggested that first-line immunotherapy may not exert a consistent positive promoting effect in SCLC. Given the inherent limitations of retrospective studies, further research is still necessary.
There are several limitations in this study. The current analysis was based on a small sample from a single institution and limited by missing data on certain baseline characteristics of interest, such as comorbidities. And our study time is not long enough. Therefore, the OS data was lacking. Our results suggest the possibility of clinical treatment, rather than reaching definitive conclusions. Besides, given this was a retrospective study, the calculation and justification of the sample size selection were lacking in this study. Thus, larger prospective studies are needed to confirm our findings.
Conclusions
For patients with advanced platinum-resistant SCLC in second-line treatment, combination therapy has shown better efficacy and acceptable toxicity profile than monotherapy. Among different combination therapies, irinotecan–platinum has shown better efficacy than taxol–platinum. For patients with advanced platinum-sensitive SCLC in second-line treatment, the efficacy of different single-agent or different combination therapies was similar.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The study was approved by the Academic Ethics Committee of Jiangsu Cancer Hospital, approval number (NO. [2023]081).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, 82172872; Social Development Project of Jiangsu Province, BE2021746; “Six Talent Peaks” of Jiangsu Province, WSN-039; Guangzhou Life Oasis Public Service Center, Health Research Exchange Project 2-38; and Suqian Sci & Tech Program, S202208.
