Abstract
Background:
Serum Cystatin S (CST4), a secretory protein that inhibits cellular matrix degradation, significantly influences the tumor microenvironment and tumor progression. However, the prognostic value of serum CST4 in gastric cancer (GC) remains unclear. This study aims to explore serum CST4’s utility in GC prognostic assessment.
Methods:
A cohort of 334 patients with GC who underwent radical gastrectomy was assessed. Preoperative serum CST4 levels were measured alongside traditional tumor markers, correlating with clinical data and patient outcomes. The cohort was divided into training and test sets at a ratio of 3:1 for Cox regression analyses, which identified CST4 as an independent risk factor for overall survival (OS) and disease-free survival (DFS). A prognostic model was developed, validated with calibration curves, and its predictive value was evaluated using receiver operating characteristic (ROC) curves. In addition, CST4 expression was correlated with immune cell infiltration using data from The Cancer Genome Atlas (TCGA). Patients were stratified by median CST4 levels, and Kaplan-Meier curves for OS and DFS were plotted.
Results:
Cystatin S was confirmed as an independent risk factor for OS and DFS. Integrating CST4 with traditional markers and TNM pathological staging significantly enhanced the predictive value for prognosis. Cystatin S’s impact on tumor progression is likely mediated through modulation of the immune microenvironment, including immune suppression and evasion.
Conclusion:
Cystatin S is an effective biomarker for GC prognostic assessment, assisting in the evaluation of prognosis and the selection of treatment strategies for patients with GC.
Keywords
Introduction
Gastric cancer (GC) is one of the most prevalent malignant tumors globally, with particularly high incidence and mortality rates in East Asia, including China.1-3 The prognosis of GC is influenced by a multitude of factors, such as tumor staging, patient age, sex, and the biological characteristics of the tumor. 4 In clinical practice, the assessment of prognosis is pivotal for tailoring therapeutic strategies. 5 While the TNM staging system has been instrumental in prognosticating patients with GC, its reliance on anatomical descriptors overlooks critical determinants such as tumor biology and host immune response. 6 Furthermore, conventional tumor markers, including carcinoembryonic antigen (CEA) and carbohydrate antigen 19-9 (CA19-9), have demonstrated limitations in sensitivity and specificity, particularly in early detection and prognosis prediction of GC. 7
In recent years, with the deepening understanding of tumor biology, new biomarkers have been gradually identified and applied in the diagnosis and prognostic assessment of tumors.8,9 Serum cysteine protease inhibitor Cystatin S (CST4), as a secretory protein, has been demonstrated to play a significant role in the development of various tumors.10-12 Cystatin S can influence the degradation of the extracellular matrix (ECM) and the alteration of the tumor microenvironment by inhibiting the activity of cysteine proteases, thereby participating in the regulation of tumor cell adhesion, proliferation, and invasive migration.13,14 Despite recognition of CST4’s potential, current investigations have primarily focused on its diagnostic utility,15-17 leaving its prognostic value, particularly in the context of GC, relatively unexplored.
This study aims to evaluate the predictive value of CST4 for the overall survival (OS) and disease-free survival (DFS) of patients with GC through an in-depth analysis of clinical data and serum CST4 levels, combined with univariate and multivariate Cox regression analysis. Furthermore, by establishing a prognostic model and time-dependent receiver operating characteristic (ROC) analysis, this study will explore the advantages, disadvantages, and potential complementarity of CST4 compared with traditional tumor markers and TNM staging in the prognostic assessment of GC. It is hoped that the findings of this study will provide new perspectives for the prognostic assessment of GC and offer strong reference for clinical treatment decisions.
Materials and Methods
Study subjects
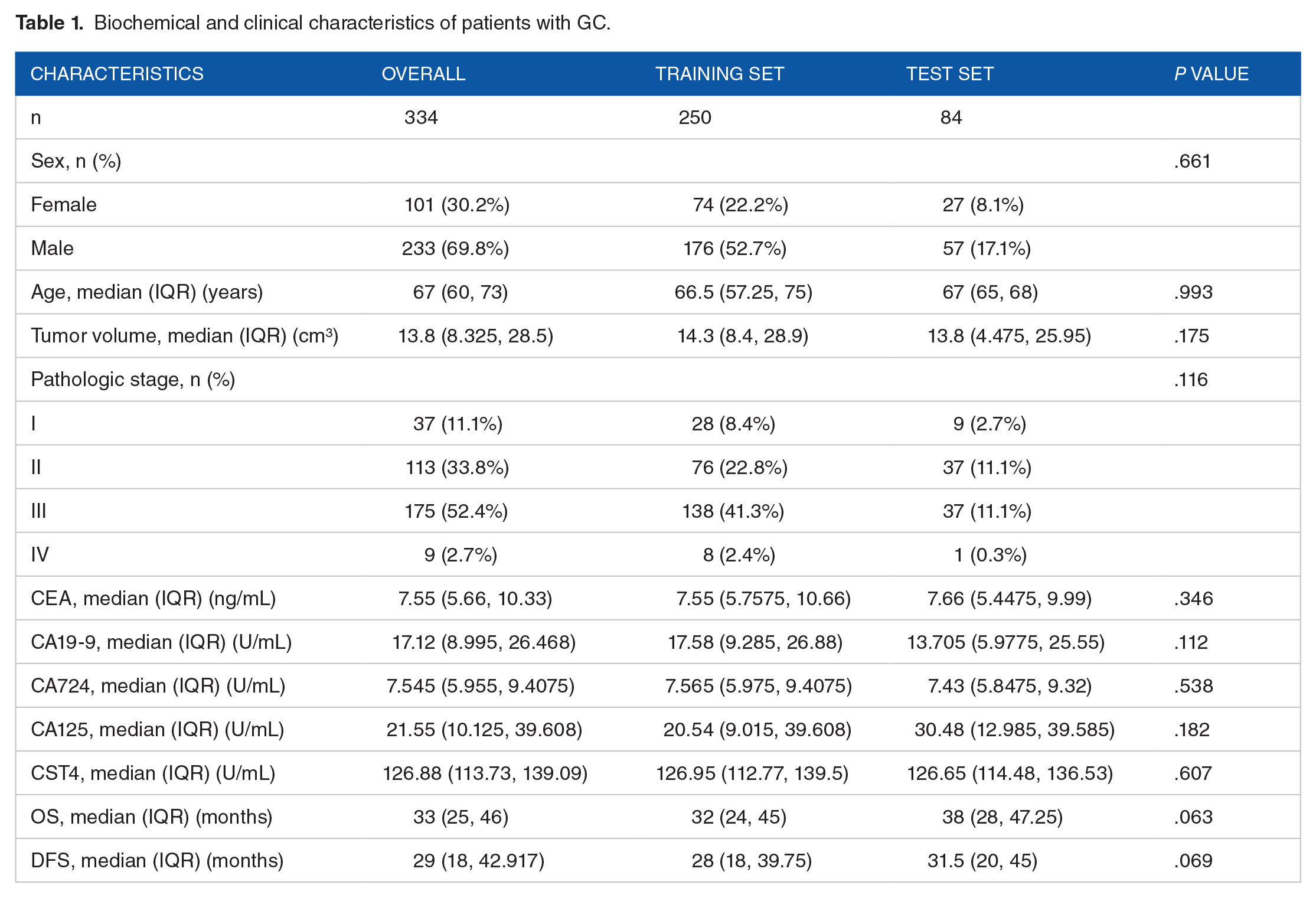
This study included 334 patients with gastric cancer who underwent curative surgery at the Department of Gastrointestinal Surgery, The Affiliated Suzhou Hospital of Nanjing Medical University from June 2019 to May 2024. All patients underwent preoperative serum CST4 and routine serum tumor marker tests. The inclusion criteria for the study subjects were patients diagnosed with gastric cancer, aged 18 years or older, and without other severe complications. The study has been approved by the hospital’s ethics committee and has obtained informed consent from all patients.
Data collection
Clinical data of the patients were collected, including age, sex, tumor pathological staging, tumor size, and so on. The collection of clinical data followed strict privacy protection measures to ensure the confidentiality of patient information.
Serum sample collection and processing
All serum samples were collected preoperatively, centrifuged using standard operating procedures, and stored at −80°C until testing. The concentration of serum CST4 was quantitatively detected by enzyme-linked immunosorbent assay (ELISA), using the Human Cystatin 4 (CST4) ELISA Kit from SAB (Sino American Biochemistry Co., Ltd.), catalog number: SEMEK13486.
Detection of tumor markers
Tumor markers in the serum, including CEA, CA19-9, and so on, were detected using an automated immunoassay system.
Follow-up
Long-term follow-up was conducted for all patients, recording their OS and DFS.
Statistical methods and data analysis
Continuous variables with normal distribution are expressed as mean (X) ± standard deviation (SD), and those with skewed distribution are expressed as median/interquartile range. Categorical variables are represented numerically. Statistical analysis was performed using SPSS version 23.0. Normally distributed continuous variables were compared using paired t tests, skewed continuous variables were compared using non-parametric tests, and categorical variables were compared using chi-square tests. The correlation between CST4 and clinical data was statistically analyzed using the Spearman method. The RNAseq data of TCGA-STAD (Stomach Adenocarcinoma) were downloaded and organized from the TCGA database (https://portal.gdc.cancer.gov), and the correlation analysis between CST4 and immune cell infiltration was calculated using the ssGSEA algorithm provided by the GSVA package [1.46.0] with markers of 24 immune cells. The results of the correlation analysis were visualized using the ggplot2 package (v3.3.3). Patients were randomly divided into a training set and a test set in a 3:1 ratio. Univariate and multivariate Cox regression analysis was used in the training set to evaluate the impact of CST4 and other clinical variables on the prognosis of patients with GC. Prognostic models were established using R (v3.6.3), the NomogramEx package (v3.0), and the rms package (v1.3.2), and the accuracy of the models was tested using calibration curves. Time-dependent ROC analysis was used to evaluate the predictive value of various indicators and combined risk factors for prognosis. Survival curves were plotted using the Kaplan-Meier method, and the survival differences between patients with different CST4 levels were compared using the Cox regression test. Survival analysis and visualization were performed using the ggplot2 package (v3.3.3), glmnet package (v4.1.2), survival package, and survminer package. P value of <.05 was considered statistically significant.
The additional methods used in this study are presented in the supplementary materials, and the schematic of this study is shown in Figure 1.

Schematic diagram of the study design.
Results
Association of serum CST4 expression with biochemical and clinical characteristics in patients with GC
The clinical characteristics of the 334 patients with GC are presented in Table 1 and Supplementary Table S1. Correlation analysis revealed a positive correlation between CST4 expression and Pathologic Stage as well as CEA (Figure 2A and B); however, CST4 expression was negatively correlated with OS and DFS (Figure 2C and D) (Table 2).
Biochemical and clinical characteristics of patients with GC.

Association of serum CST4 expression with biochemical and clinical characteristics in patients with GC.
Correlations between CST4 and clinical characteristics in patients with GC.

Bold values indicate statistical significance.
Prognostic value of CST4 expression in postoperative patients with GC
Kaplan-Meier analysis was conducted to evaluate the association between CST4 expression and survival outcomes in patients with GC. The analysis showed that increased CST4 expression was related to reduced OS and DFS in the patients with GC (Figure 3A and B). This finding suggests that CST4 may serve as a prognostic factor for survival in GC. To further validate this, we examined the TCGA database and found that CST4 expression was negatively correlated with OS (Figure 3C), disease-specific survival (DSS) (Figure 3D), and progression-free interval (PFI) (Figure 3E), which supports our Kaplan-Meier analysis results.

Kaplan-Meier analysis of survival outcomes in patients with GC. (A, B) Kaplan-Meier curves of OS (A) and DFS (B) in 334 gastric cancer patients from our cohort, grouped by CST4 expression levels. (C-E) Kaplan-Meier curves of OS (C), DSS (D), and PFI (E) in gastric cancer patients from TCGA database, grouped by CST4 expression levels. OS: overall survival, DFS: disease-free survival. DSS: disease-specific survival, PFI: progression-free interval. P values were determined by log rank test (A-E) (*P < .05, **P < .01, ***P < .001).
Univariate and multivariate Cox regression analyses were used to assess prognostic factors for postoperative OS and DFS in patients with GC. The results showed that CST4, CEA, CA19-9, and Pathologic Stage are independent risk factors for OS (Table 3), while CST4, CEA, and Pathologic Stage are independent risk factors for DFS (Table 4). The risk scores of the independent risk factors derived from the Cox regression analysis for OS and DFS are displayed in Figure 4. The prognostic predictive model was constructed based on the risk factors identified through Cox regression analysis (Figure 5A and B), which effectively forecasts the prognosis of patients with GC at the first, second, and third year post-surgery. The calibration curves demonstrated excellent model fit (Figure 5C and D).
Univariate and multivariate COX regression analyses of prognostic factors related to OS in patients with GC.

Bold values indicate statistical significance.
Univariate and multivariate COX regression analyses of prognostic factors related to DFS in patients with GC.

Bold values indicate statistical significance.

The risk scores derived from the Cox regression analysis for OS (A) and DFS (B). OS: overall survival, DFS: disease-free survival.

Prognostic prediction models of OS (A) and DFS (B) and calibration curve of OS (C) and DFS (D). OS: overall survival, DFS: disease-free survival.
Furthermore, the predictive accuracy of the independent risk factors and combined risk scores model obtained from the Cox regression analysis for OS and DFS at the first, second, and third year post-surgery was measured using time-dependent ROC curves (Figure 6). In the first postoperative year, the area under the curve (AUC) for CST4 in predicting OS (AUC = 0.869) was higher than that for Pathologic Stage (AUC = 0.849) and CA19-9 (AUC = 0.775), but lower than that for CEA (AUC = 0.888). The combined risk score model (AUC = 0.985) showed a significantly higher AUC compared with the individual risk factors (Figure 6A). Similarly, in the first postoperative year, the AUC for CST4 in predicting DFS (AUC = 0.703) was lower than that for Pathologic Stage (AUC = 0.757) and CEA (AUC = 0.744), but the combined risk score model (AUC = 0.847) showed a significantly higher AUC compared with the individual risk factors (Figure 6B). The same pattern was observed in the second and third postoperative years, with the combined risk score model consistently showing a higher AUC than the individual risk factors for both OS and DFS (Figure 6C to F). In summary, these results elucidate the advantages of combined risk scores model for CST4, pathology, and tumor markers in terms of sensitivity and specificity for predictive assessment of gastric cancer.

Time-dependent ROC curves of OS and DFS about the independent risk factors and combined risk scores. (A, B) Time-dependent ROC curves of OS (A) and DFS (B) about the independent risk factors and combined risk scores at the first year post-surgery. (C, D) Time-dependent ROC curves of OS (C) and DFS (D) about the independent risk factors and combined risk scores at the second year post-surgery. (E, F) Time-dependent ROC curves of OS (E) and DFS (F) about the independent risk factors and combined risk scores at the third year post-surgery. OS: overall survival, DFS: disease-free survival.
Notably, during the follow-up period, we conducted surveillance on the postoperative CST4 levels in a subset of patients. We were delighted to discern a marked reduction in CST4 levels subsequent to surgery. The CST4 levels demonstrated a significant correlation with the tumor progression of patients, including complete response (CR), partial response (PR), and progressive disease (PD). These findings, which we have detailed in Figure 7, suggest that CST4 not only serves as a prognostic marker but also as a potential indicator of treatment response and tumor progression.

Postoperative serum CST4 levels and their association with patient prognosis. (A) In patient 1, postoperative CST4 levels indicated a favorable response to surgery and chemotherapy, with the patient achieving a complete response (CR), maintaining low serum CST4 levels that gradually decreased. (B) Patient 2 experienced tumor recurrence 8 months after surgery and chemotherapy, with poor response to subsequent chemotherapy, radiotherapy, and further radiotherapy, remaining in progressive disease (PD), with sustained elevation of serum CST4 levels. (C) Patient 3 had a good response to surgery and chemotherapy, achieving a complete response (CR) upon postoperative review, with a significant reduction in serum CST4 levels. Two years postoperatively, the patient’s condition progressed with hilar lung metastasis and entered progressive disease (PD). After chemotherapy, radiotherapy, immunotherapy, and targeted therapy, the patient achieved a partial response (PR), with tumor reduction. Preop, preoperative; RT, radiotherapy; mFOLFOX6, Oxaliplatin + Leucovorin + 5-Fluorouracil; SOX, Oxaliplatin + S-1; TCF, Docetaxel + Cisplatin + 5-Fluorouracil.
Mechanisms of CST4 affecting gastric cancer prognosis
In our comprehensive analysis of the role of CST4 in gastric cancer prognosis, we used data from the TCGA-STAD database to uncover several key mechanisms. GSEA enrichment analysis of CST4-correlated genes revealed a significant positive correlation with pathways involved in ECM formation (Figure 8A and B). Through the STRING database, we identified a notable interaction network between CST4 and the CTS family genes (CTSA, CTSB, CTSL, CTSW) (Figure 8C), further pinpointing proteins that may collaborate with CST4 to modulate gastric cancer prognosis. Correlation analysis within the TCGA database exposed significant links between CST4 and ECM proteins (Figure 8D), suggesting CST4’s role in the dynamics of the tumor microenvironment. The impact of CST4 expression on ECM-related genes, including COL1A1, LAMA1, FN1, and ELN, at both the protein and mRNA levels was further confirmed by knocking down CST4 in gastric cancer cells (Figure 8E and F). Using transcriptomic data from the TCGA database and employing the ssGSEA algorithm based on the principle of linear support vector regression for deconvolution, we inferred immune cell infiltration across different tissues and found that CST4 expression in gastric cancer was negatively correlated with the infiltration of CD8+ T cells, T helper cells, and Tcm cells (Figure 8G to J), while it was positively correlated with the infiltration of TReg cells (Figure 8K). This elucidates a potential mechanism by which CST4, through interactions with the CTS enzyme family, inhibits ECM degradation, thereby affecting immune cell infiltration. In addition, a positive correlation was identified between CST4 expression and TGF-β2 (Figure 8L), which may underlie the observed correlations between CST4 expression and immune cell infiltration in patients with GC.

Mechanisms of CST4 affecting gastric cancer prognosis. (A) Heatmap of top 50 CST4-positively correlated genes in TCGA-STAD database. (B) Mountain plot of GSEA enrichment analysis for CST4-positively correlated genes in TCGA-STAD database. (C) Exploration of CST4-interacting proteins via STRING database (https://version11.string-db.org/). (D) Correlation analysis of CST4 with extracellular matrix proteins in TCGA database. (E) Western blot showed the protein expression of extracellular matrix-related genes COL1A1, LAMA1, FN1, and ELN when CST4 was knocked down in gastric cancer cells (MKN-45, HGC27). (F) qRT-PCR detected the mRNA expression of extracellular matrix-related genes COL1A1, LAMA1, FN1, and ELN when CST4 was knocked down in gastric cancer cells (MKN-45, HGC-27). (G) Analysis of immune cell infiltration in gastric cancer patients by CST4 expression in the TCGA database. (H-K) The correlation scatter plot demonstrated the correlations between CST4 expression and infiltration of CD8+ T cells (H), T helper cells (I), Tcm cells (J), and TReg cells (K). (L) Correlation between CST4 expression and TGFB2 expression. The data are representative of 3 independent experiments. Quantitative data are shown as mean ± SD (E, G), and P values were determined by 2-tailed unpaired Student’s t test (H-L) (*P < .05, **P < .01, ***P < .001).
Discussion
CST4 has been demonstrated to play a significant role in the development of various malignancies.10-12 Studies by Zhang et al 10 have indicated that the overexpression of CST4 promotes the invasiveness of gastric cancer by activating the ELFN2 signaling pathway. Guo et al 11 discovered that CST4 facilitates lymph node metastasis of esophageal cancer cells through the VEGF-MAPK/ERK-MMP9/2 pathway. Yang et al 12 investigated how CircRNA circ_0023984 promotes the progression of esophageal squamous cell carcinoma by regulating the miR-134-5p/Cystatin S axis. Although CST4’s role in tumorigenesis has been extensively studied, most research has focused on its mechanisms within tumor tissues. However, as a secretory protein, CST4’s detectability in serum suggests its potential as a tumor marker. Current research on serum CST4 has primarily concentrated on its diagnostic utility for cancer.15-17 For instance, research by Cai et al 15 has shown that the combined detection of serum CST4 and DR-70 aids in the early diagnosis of colorectal cancer. Studies by Huang et al 16 have also highlighted the significance of serum CST4 in the diagnosis of colorectal cancer. Zhou et al 17 further confirmed the clinical application value of serum CST4 in combination with tumor markers for the diagnosis of digestive system malignancies. In Supplementary Figure S1, the differential expression of the CST4 protein in gastric cancer (GC) tissues and adjacent non-neoplastic tissues was validated using immunohistochemistry and data from The Cancer Genome Atlas (TCGA). However, in this study, diverging from previous research on the diagnostic value of CST4, we have elucidated the role of serum CST4 in the prognostic assessment of gastric cancer. We found that the expression level of serum CST4 is an independent prognostic factor for OS and DFS in patients with GC. Kaplan-Meier analysis further substantiated this observation, showing that increased CST4 expression was associated with reduced OS and DFS, suggesting that CST4 may have a role in tumor progression and patient survival.
The correlation between postoperative CST4 levels and tumor progression, as described in Figure 7, presents an interesting observation. These results hint at CST4’s potential as a prognostic marker and a bioindicator of treatment response and tumor progression. However, we remain cautious about drawing a definitive causal link between CST4 and metastasis or recurrence based on our current dataset. Further research with expanded patient cohorts and in-depth molecular analyses would be beneficial to better understand the mechanisms through which CST4 may influence tumor dynamics.
Through time-dependent ROC curve analysis, we further validated the effectiveness and superiority of the risk score model combining CST4 with pathology and conventional tumor markers in the prognostic assessment of gastric cancer. This finding provides a new biomarker and prognostic model for gastric cancer and offers a new dimension for the application of CST4 in oncology. While the tumor-node-metastasis (TNM) staging system is a proven method for assessing patient prognosis, our findings suggest that integrating CST4 levels might offer additional value by providing a more nuanced understanding of tumor behavior and tumor microenvironment. 18 By incorporating CST4 levels into existing prognostic frameworks, clinicians could better stratify patients based on their risk profiles, potentially guiding more personalized treatment strategies. This could be particularly beneficial in determining candidates for aggressive therapies or closer monitoring.
Our analysis of public databases revealed a positive correlation between CST4 expression and the infiltration of regulatory T cells (TReg cells), while a negative correlation was observed with the infiltration levels of central memory T cells (Tcm), helper T cells, and CD8+ T cells. This suggests that CST4 may exert a complex regulatory role within the tumor immune microenvironment. This could be attributed to the properties of CST4 as a protein that inhibits the activity of cysteine proteases, where CST4 suppresses the degradation of the ECM by inhibiting the activity of cysteine proteases (CTS family),14,15 mediating the infiltration of immune cells with tumoricidal effects such as Tcm, helper T cells, and CD8+ T cells. 19 In addition, the positive correlation between CST4 and TGF-β2 expression may influence the activation of TGF-β receptors and the activity of its downstream signaling molecules, thereby suppressing the proliferation of activated T cells and effectively inhibiting the differentiation and effector functions of T cells. 20 Furthermore, the activation of the TGF-β pathway can promote the differentiation and function of TReg cells, indirectly promoting the infiltration of TReg cells.21,22 We also found that CST4 expression in patients with GC is positively correlated with CEA and Pathologic Stage. Carcinoembryonic antigen is a tumor-associated antigen that is typically overexpressed when tumor cells proliferate rapidly.23,24 The TNM pathological staging is the most commonly used indicator for assessing the progression of digestive tract tumors. 6 By elucidating the potential role of CST4 in the tumor immune microenvironment, we have also well understood the relationship between CST4 expression and tumor cell proliferation and invasion, which is well corroborated by the correlation between CST4 expression and CEA and Pathologic Stage.
Despite providing new insights into CST4’s role in the prognostic assessment of gastric cancer, this study has some limitations. First, the sample size of this study is relatively small, which may affect the generalizability of the results. Second, this study only analyzed the expression level of serum CST4 and did not consider other molecular markers that may affect prognosis. In addition, although we have preliminarily elucidated the mechanism by which CST4 inhibits the degradation of the ECM by inhibiting the activity of cysteine proteases (CTS family), thereby mediating the infiltration of immune cells, further mechanism validation and in-depth exploration of the specific molecular mechanisms of CST4’s role in the tumor immune microenvironment are still necessary. To address these limitations, we will further verify the prognostic value of CST4 by expanding the sample size, combining other molecular markers, and adopting a multicenter study design. Moreover, in-depth research on the specific mechanisms of CST4’s role in the tumor immune microenvironment will help develop new cancer immunotherapy strategies. Furthermore, we will explore the expression patterns of CST4 in different tumor types to further investigate its prognostic value in other malignancies and study the combined application of CST4 with other immunotherapy targets to enhance the efficacy of cancer treatment.
Conclusions
This study elucidates the pivotal role of serum Cystatin S (CST4) in the prognostic assessment of gastric cancer, thereby introducing a novel biomarker for clinical management. Distinguished from prior research that has concentrated on the diagnostic utility of CST4, our study highlights its prognostic significance, offering a novel perspective for the personalized treatment and surveillance of patients with GC. We anticipate that our findings not only contribute a new dimension to the application of CST4 in oncology but also illuminate new directions for future research and clinical practice.
Supplemental Material
sj-docx-1-onc-10.1177_11795549241311404 – Supplemental material for Serum Cystatin S (CST4): A Novel Prognostic Marker for Gastric Cancer
Supplemental material, sj-docx-1-onc-10.1177_11795549241311404 for Serum Cystatin S (CST4): A Novel Prognostic Marker for Gastric Cancer by Chao Gu, Shan Chen, Lining Huang, Chenliang Cao, Renshun Yuan, Zhongyang Kou, Weiwei Chen, Haihua Shi and Xiaodong Gu in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
The authors wish to thank the Core Facility of the Affiliated Suzhou Hospital of Nanjing Medical University for its help in the detection of experimental samples.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Gusu Health Talent Programme Research Project (No. GSWS2023058).
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
CG, SC, and LH were responsible for the conception and design of the study. HS and XG provided administrative support. The provision of study materials or patients was managed by CG, HS, and XG. Data collection and assembly were performed by CG, SC, LH, CC, RY, ZK, WC, XG, and HS. Data analysis and interpretation were carried out by CG, SC, LH, CC, RY, ZK, WC, XG, and HS. The manuscript was written by CG. All authors gave their final approval of the manuscript before submission.
Availability of Data and Materials
The data supporting the results of this study are available from the corresponding author upon reasonable request.
Consent for Publication
We have obtained written informed consent from all participants, or their legal representatives, including for deceased subjects, for the publication of this manuscript. This consent encompasses the use of any potentially identifiable data or images related to their case. The Ethics Committee of The Affiliated Suzhou Hospital of Nanjing Medical University has reviewed and approved the consent process, ensuring that the publication adheres to ethical standards and maintains the privacy and confidentiality of all individuals involved in the study.
Ethics Approval and Consent to Participate
This study was approved by the Ethics Committee of The Affiliated Suzhou Hospital of Nanjing Medical University (Approval Number: K-2019-166-K01, Date of Approval: 2019-08-06), in compliance with the Declaration of Helsinki. All participants provided written informed consent before enrollment in the study. The study was conducted in full accordance with ethical standards and all applicable legal requirements.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
