Abstract
Background:
Inflammation is the most important deriving force for the development of colitis-associated colorectal cancer (CAC) through the Inflammation-Pretumor dysplasia-CAC sequence. T helper (Th) subsets Th9 and Th17 cells can potentially stimulate inflammation in the ulcerative colitis (UC). Therefore, Th9 and Th17 cells may play a promoting role in the colitis-associated dysplasia (CAD).
Methods:
Using immunohistochemistry (IHC), we evaluated the presentation patterns and densities of T lymphocytes, Th9 and Th17 cells in human UC and CAD tissues.
Results:
A general increasing trend of CD3-positive T lymphocytes, P.U.1-positive Th9 and interleukin (IL)-17A-positive Th17 cells was illustrated throughout the normal-UC-CAD sequence, IHC images showed that these cells were very prominent in the lamina propria, and some cells were also observed in the epithelium in the CAD tissues. Density analysis revealed that numbers of Th9 and Th17 cells were progressively increased in the CAD tissues as compared with the UC and control tissues. In general, densities of Th9 and Th17 cells in the lamina propria were slightly higher in the non-adenoma-like dysplasia (NALD) tissues than that in the adenoma-like dysplasia (ALD) tissues. However, densities of neither Th9 nor Th17 cells in both the ALD and NALD subgroups were associated with the degree of dysplasia in CAD lesions.
Conclusion:
Accumulated Th9 and Th17 cells contribute to the immune cellular composition in the CAD tissues and may represent the early conditional change for the Dysplasia-CAC transition.
Introduction
It is widely accepted that chronic inflammation is thought to be the main driver of colorectal tumorigenesis. There is both convincing clinical and epidemiological evidence to suggest that long-standing and extensive inflammation in the ulcerative colitis (UC) can significantly increase the risk of colitis-associated colorectal cancer (CAC) development,1-5 through an Inflammation-Dysplasia-Cancer pathogenic sequence.3,6,7 Therefore, UC-associated dysplasia (CAD), as one of the most important risk factors for developing CAC, has gained much attention. Clinical and pathological analysis indicated that assessing degree of dysplasia may indicate the risk level for CAC development in patients with UC, 8 and high degree of dysplasia has a increasing potential for CAC development in patients with UC. 9 Clinically, for the early identification of the occurrence and severity of dysplastic lesions in patients with long-standing UC, surveillance colonoscopy with histological examination is strongly recommended. 10 Under the colonoscopy and microscopy, the CAD lesions are histologically classified as two main forms: adenoma-like dysplasia (ALD) and non-adenoma-like dysplasia (NALD). Identification of these two subtypes of CAD is important and necessary, because the recommendations for the clinical management of CAD are based on the types and degree of dysplasia. So far, experts are generally in agreement that an ALD with low- or high-grade dysplasia can be safely removed by colonoscopic polypectomy and accelerated surveillance. In contrast, a high-degree NALD has a high risk of CAC and often requires a colectomy.11-13 Therefore, surveillance colonoscopy with histopathological diagnosis to identify CAD lesions becomes one of the key issues for the prevention and management of CAC in patients with UC.7,11,14-16
To seek the mechanisms for Inflammation-Dysplasia-Cancer transition in patients with long-standing UC, great attempt has been made. Although many genetic/molecular, environmental, and other factors involved in this complex transition process have been identified, 16 the exact mechanisms are still not fully understood. Recently, the promoting effect of T helper (Th) subsets and relevant cytokine products on the development of CAC has been highlighted.17-20 Considerable evidence now suggests that altered immune cellular composition and function in the pretumor microenvironment may reflect the potential and ongoing CAC in the UC,21,22 and analysis of changes in phenotypes and fluctuations of immune cells in the CAD tissues can provide the key insights into the understanding of CAC pathogenesis.17,23
The importance of activation of Th subsets Th9 and Th17 cells and their cytokine products interleukin-9 (IL-9) and IL-17A/F in the initiation of inflammation and the development of UC has been previously confirmed.24,25 Studies also reported that Th17 cytokine IL-17A can significantly stimulate the development of CAC26,27 through multiple mechanisms, and blocking IL-17A signal by anti-IL-17A antibody injection significantly inhibit the development of CAC in colitis mice, suggesting a promoting effect of IL-17 on the development of CAC. 28 Regarding the role of Th9/IL-9 in the development of sporadic colorectal cancer (CRC), data from various studies are controversial.29-31 Both antitumor32-34 and protumor effects on the development of sporadic CRC have been reported. However, CAC develops under the background of long-standing inflammation in the UC, which is different to the sporadic CRC. More recent studies have shown an increased expression of Th9 cytokine IL-9 in CAC mouse model30,35 and human CAC tissues. 36 Furthermore, Gerlach et al 37 demonstrated that activated PU.1-positive Th9 cells exhibit a significant promoting effect on the development of CAC through enhanced production of IL-6 in colitis mice. They also showed that the levels of IL-9- and P.U.1-positive Th9 cells were greatly increased in human CAC samples and genetic deficiency of IL-9 in mice or inactivation of PU.1 in T lymphocytes potentially inhibited the growth of CAC in vivo. 37 Therefore, Th9 cells and relevant cytokine IL-9, most likely, hold a promoting effect on the development of CAC under the background of chronic inflammation in the intestine.
Currently, most existing studies have emphasized changes in Th9 and Th17 cells in the established CAC tissues. The contribution of Th9 and Th17 cells to the composition of immune microenvironment in the pretumor dysplastic stage is still incompletely evaluated.
Given the above background, we have, therefore, designed this study to analyze the presentation pattern of Th9 and Th17 cells in the pretumor microenvironment in different histological types of CAD lesions in patients with UC.
Materials and Methods
Patients and biopsies
Colorectal biopsies from 42 patients with CAD taken by colonoscopy from 1998 to 2011 were retrieved from the tissue bank of the Department of Pathology, University Hospital of North Norway according to standardized diagnostic criteria, and inflamed colorectal tissues were taken from 16 UC patients from 2006 to 2008 during the routine surveillance colonoscope examination according to the standard protocol 38 in the Endoscopy Room of Gastroenterology, University Hospital of North Norway. In addition, total 15 morphological normal colorectal biopsies without pathological evidence by the colonoscopic and histological evaluations were used as the controls. No patients or control subjects had a history of sporadic CRC. All colorectal biopsies were immediately immersed in 10% formalin after sampling by colonoscopy and routinely embedded in paraffin within 24 hours for histological examination. Sections (4 μm) were cut and then stained with hematoxylin and eosin (H&E). Histological diagnosis for all the biopsies was determined by experienced pathologists and reviewed and confirmed by a senior gastroenterological pathologist (Dr Sørbye) at the Department of Pathology, University Hospital of North Norway. The study is approved by the Norwegian Regional Ethical Committee (REK) of North and West Norway (Approval IDs 29895 and 419346) respectively, and written concern is obtained from all patients with UC and normal controls. As many CAD patients were dead and the written consent was impossible to collect, waiver of the requirement to obtain written consent from these patients was issued by the REK.
Examination of Th9 and Th17 cells in the UC and CAD tissues by immunohistochemistry
To examine the density changes of total T lymphocytes, T lymphocytic subtypes Th9 and Th17 in the UC and CAD tissues, immunohistochemistries (IHCs) were performed in 4 μm thick paraffin sections cut from the control, UC, and CAD biopsies. In brief, sections were deparaffinized in xylene, rehydrated in a series of graded ethanol until water, and immersed in 0.3% H2O2 solution in methanol for 15 minutes to block endogenous peroxidases. Then, Antigen retrieval was conducted by boiling slides in the ethylenediaminetetraacetic acid (EDTA) buffer solution (pH 8.0) through microwave processing for 3×5 minutes. After cooldown at room temperature and rinsing three times with the solution of phosphate-buffered saline (PBS) with 0.25% Triton-X 100 (PBS-T), sections were incubated the following primary antibodies at 4 ° C overnight: mouse anti-CD3 monoclonal antibody (Clone F7.2.38; purchased from DAKO, Carpinteria, CA, USA) to label T lymphocytes; rabbit anti-human PU. 1 polyclonal antibody (purchased from LifeSpan BioSciences Inc, Seattle, WA, USA) to label Th9 cells and goat anti-human IL-17A polyclonal antibody (Catalog No: AF-317-NA, clone Ile20Ala155, purchased from R&D System; Minneapolis, MN, USA) to label Th17 cells. After which sections were washed with PBS-T buffer solution for 10 minutes and detection was performed with a commercial with Vectastatin Elite ABC Kit (Vector Lab., Burlingame, CA, USA) according to the manufacturer’s instructions and our previous published method 39 ; 3-Amino-9-ethylcarbazole (AEC; Vector Laboratories, Burlingame, CA, USA) was used as chromogen and slides were counterstained with Mayer’s hematoxylin. Negative control slides for each IHC were routinely performed: (1) primary antibodies were substituted with the isotype-matched control antibodies; (2) secondary antibody was substituted with PBS buffer.
Morphometric analysis
Well-oriented IHC stained sections were observed and examined with a light microscope (CX31, Olympus Optical Co., LTD, New York, USA) by two experienced evaluators (Drs Cui and Yuan). As previous studies have revealed that lymphocytes are presented in both the lamina propria and epithelium in the colorectal mucosa,40,41 the densities of T lymphocytes, including Th9 and Th17 cells, were evaluated in the lamina propria and epithelium, individually, according to the method described in our previous publication.40,41 In detail, semiquantitative grading of CD3-positive T lymphocytes, PU.1-positive Th cells and IL-17A-positive Th17 cells in the lamina propria under five optional high-power fields (HPF, magnification ×400) with abundant distribution from each slide were scored as score 0 (nil/HPF), score 1 (1-19 cells/HPF), score 2 (20-49 cells/HPF), and score 3 (over 50 cells/HPF), and the average value of scores from each slide was used for the statistical analysis of positive cell densities.40,41 Cells infiltrated in the colorectal epithelium positive for CD3, PU.1, and IL-17A were absolutely counted under five optional HPF fields with abundant distribution from each slide.40,41 Then, the average value from each slide were used for the statistical analysis of positive cell densities of positive cells in the epithelium.
Statistical analysis
The results were expressed as mean ± SEM unless otherwise stated. Statistical significance was evaluated by the two-tailed Mann-Whitney test and the Kruskal-Wallis test. Values of P
Results
Clinicopathological characteristics of UC patients with CAD lesion
After pathological assessed and reviewed, detailed clinicopathological characteristics for each group are summarized in Table 1. The median age of normal control was 54 years (age range 33-77 years), and the median age of UC patients (n = 16) was 50 years (age range 28-70 years). CAD lesions were histologically classified as ALD (n = 22; age 35-85 years; median age 65 years) and NALD (n = 20; age 25-84 years; median age 58 years) subtypes under the colonoscopy and microscopy. Further analysis showed that CAD degrees in both ALD and NALD biopsies showed that most cases were with low-grading dysplasia (LGD), a few cases with moderate-grading dysplasia (MGD) and only one case with high-grading dysplasia (HGD).
Basic histological information of UC patients and colitis-associated dysplastic patients.

Abbreviations: ALD, adenoma-like dysplasia; HGD, high-grade dysplasia; LGD, lower-grade dysplasia; MGD, moderate-grade dysplasia; NALD, non-adenoma-like dysplasia; UC, ulcerative colitis.
Increased CD3-positive lymphocytes were observed in the CAD tissues
The CD3-positive lymphocytes were observed in all the cases of control, UC, and CAD (including ALD and NALD) groups (refer to Figure 1). In the control tissues, CD3-positive lymphocytes were mainly present in the lamina propria (arrow-pointed cells in Figure 1A). In addition, some CD3-positive lymphocytes infiltrated in the epithelium (arrowhead-pointed cells in inserted enlarged-image in Figure 1A) were observed.
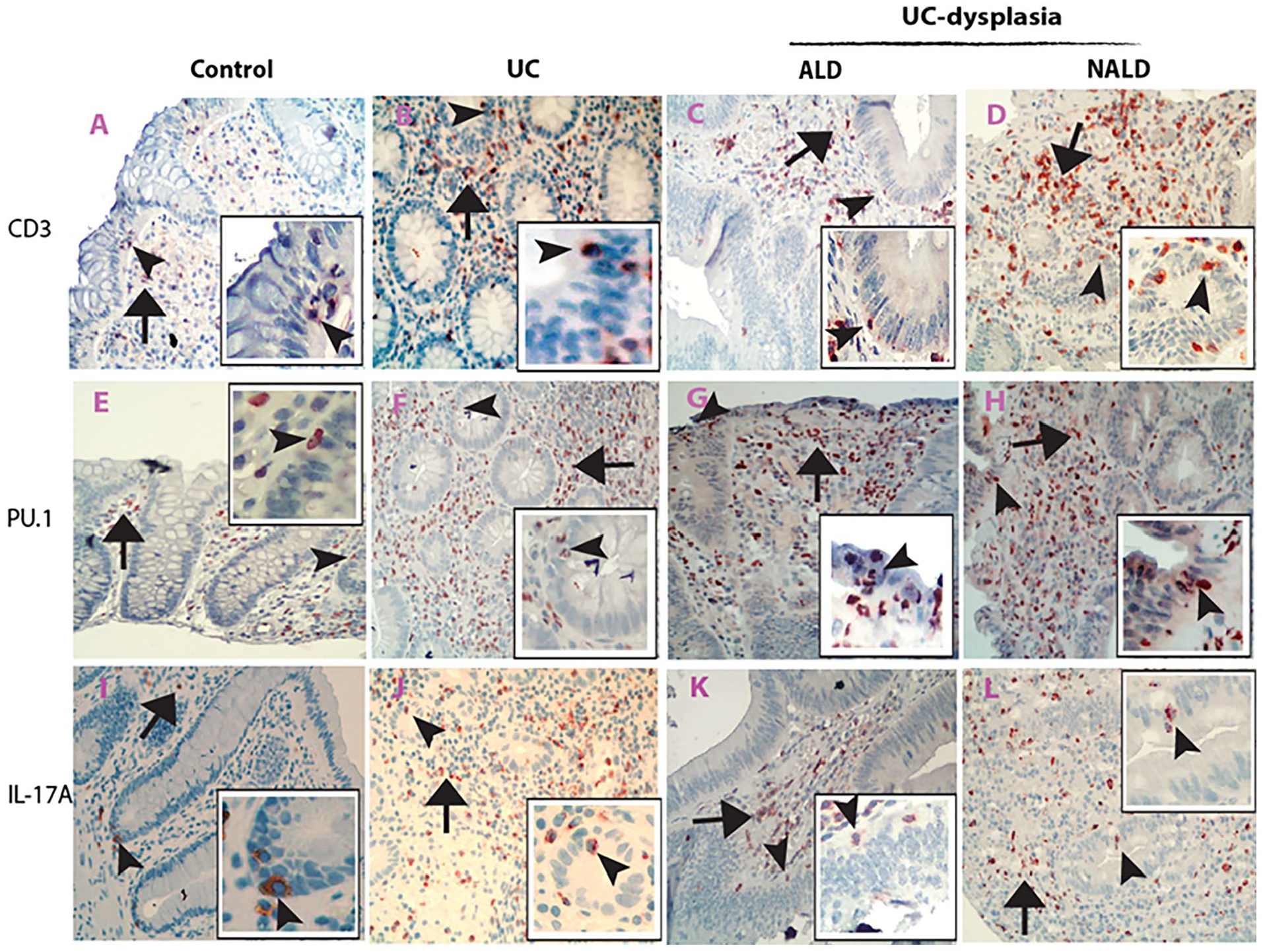
Immunohistochemical (IHC) photographic presentation of CD3-positive T lymphocytes, PU.1-positive Th9, and IL-17A-positive Th17 cells in human UC and colitis-associated dysplastic tissues. Microscopic photos from the control sections showed that CD3-positive T lymphocytes were primarily observed in the lamina propria (black arrow-pointed cells in A), and intraepithelial CD3-positive T lymphocytes were also fund (black arrowhead-pointed cell in inserted enlarged image in A). As expected, increased CD3-positive T lymphocytes in the lamina propria (black arrow-pointed cells) and epithelium (black arrowhead-pointed cells in inserted enlarged images) were shown in both the UC (B) and colitis-associated dysplastic (C for ALD and D for NALD) sections. Microscopic photos of PU.1 IHC sections showed that increased PU.1-positive Th9 cells were observed in the lamina propria (black arrow-pointed cells) and epithelium (black arrowhead-pointed cells in inserted enlarged images) in both UC (F) and colitis-associated dysplastic (G for ALD and H for ALD) sections relative to the control section (E). Similarly, microscopic photos of IL-17A IHC sections showed that IL-17A-positive Th17 cells were increased in both the lamina propria (black arrow-pointed cells) and epithelium (black arrowhead-pointed cells in inserted enlarged images) in the UC (J) and colitis-associated dysplastic (K for ALD and L for NALD) sections as compared with the control section (I) (A to L IHC images, counterstained with hematoxylin, original magnification ×200). ALD indicates adenoma-like dysplasia; NALD, non-adenoma-like dysplasia; UC, ulcerative colitis.
As expected, accumulated CD3-positive lymphocytes were diffusely distributed in the lamina propria (arrow-pointed cells in Figure 1B), and some in the inflamed epithelium (arrowhead-pointed cells in inserted enlarged-image in Figure 1B) in the UC tissues. In the CAD tissues, dense CD3-positive lymphocytes were predominantly accumulated in the lamina propria (arrow-pointed cells in Figure 1C for ALD and Figure 1D for NALD). In the dysplastic epithelium of ALD and NALD where infiltrating CD3-positive lymphocytes were also present, an increase in amount of CD3-positive T lymphocytes in all cases of CAD tissues were observed (arrowhead-pointed cells in inserted enlarged images in Figure 1C for ALD and Figure 1D for NALD).
Further statistical analyses of cell density grading results revealed that scores of CD3-positive T lymphocyte density in the ALD and NALD subgroups were significantly higher in both the lamina propria (refer to Figure 2A, Control vs ALD vs NALD, P

Graph analysis of density grading scores of CD3-positive T lymphocytes, PU.1-positive Th9, and IL-17A-positive Th17 subset cells in human colitis-associated dysplastic tissues. Graphic statistical analysis of CD3-positive T lymphocytes, PU.1-positive Th9, and IL-17A-positive Th17 cells revealed that densities of CD3-positive T lymphocytes (A for positive cells in the lamina propria, B for positive cells in the epithelium), PU.1-positive Th9 cells (C for positive cells in the lamina propria, D for positive cells in the epithelium) and IL-17A-positive Th17 cells (E for positive cells in the lamina propria) were significantly increased in both the UC (gray bar) and UC-associated ALD (grid bar) and NALD (black bar) as compared with control (white bar, all P
The presentation of PU.1-positive Th9 and IL-17A-positive Th17 cells in the CAD tissues
PU.1-positive Th9 cells in the CAD tissues
The IHC analysis revealed a general tendency of increase in the proportion of PU.1-positive Th9 cells from the control to UC and UC-associated dysplastic tissues. In the control tissues, PU.1-positive Th9 cells were evenly distributed in the lamina propria (arrow-pointed cells in Figure 1E) between intestinal crypts, and intraepithelial positive cells (arrowhead-pointed cells in the inserted enlarged image in Figure 1E) were rarely observed. In the UC tissues, increased PU.1-positive Th9 cells were observed in both the lamina propria (arrow-pointed cells in Figure 1F) and epithelium (arrowhead-pointed cells in the inserted enlarged-image in Figure 1F). In the CAD (both ALD and NALD) tissues, PU.1-positive Th9 cells were significantly increased and predominantly distributed in the whole lamina propria (refer to Figure 1G for ALD and Figure 1H for NALD). Some PU.1-positive Th9 cells were present in the subepithelial region of lamina propria and closely to crypts. In addition, intraepithelial infiltrations of PU.1-positive Th9 cells were also observed in the CAD tissues (refer to inserted enlarged images in Figure 1G for ALD and Figure 1H for NALD).
Analysis of density grading data confirmed that the CAD tissues exhibited increased densities of PU.1-positive Th9 cells in the lamina propria (Figure 2C, Control vs UC vs ALD vs NALD, P
IL-17A-positive Th17 cells in the CAD tissues
The IL-17A-positive Th17 cells were observed in both the lamina propria and epithelium in all groups. In the control tissues, IL-17A-positive cells were at a low density and primarily in the lamina propria (arrow-pointed cells in Figure 1I), only few intraepithelial IL-17A-positive Th17 cells were observed (refer to arrowhead-pointed cells in the inserted enlarged image in Figure 1I). In the UC tissues, increased IL-17A-positive Th17 cells aggravated in both the lamina propria (arrow-pointed cells in Figure 1J) and epithelium (arrowhead-pointed cells in the inserted enlarged-image in Figure 1J). In the CAD tissues, IL-17A-positive Th17 cells were highly accumulated in the whole lamina propria (arrow-pointed cells in Figure 1K for ALD; Figure 1L for NALD) and some cells infiltrated in the dysplastic epithelium (arrowhead-pointed cells in the inserted enlarged-images in Figure 1K for ALD; Figure 1L for NALD).
Density grading results of IL-17A-positive Th17 cells confirmed IHC observations and showed an increasing trend of IL-17A-positive Th17 cell density scores in the lamina propria and epithelium in both the UC and the CAD (ALD and NALD) groups (refer to Figure 2E and F, P
The relationship between densities of PU.1-positive Th9 or IL-17A-positive Th17 cells and the degree of dysplasia in the CAD subgroups (ALD and NALD tissues)
As shown in Table 2 (for ALD) and Table 3 (for NALD), densities of both PU.1-positive Th9 and IL-17A-positive Th17 cells in either the lamina propria or epithelium were not associated with dysplastic degree in both the ALD and NALD subgroups (all P
Densities of T lymphocytes, and subsets Th9 and Th17 cells in different dysplastic degrees of adenoma-like dysplastic (ALD) tissues.

Abbreviations: HGD, high-grade dysplasia; LGD, lower-grade dysplasia; MGD, moderate-grade dysplasia.
P-values were from the Mann-Whitney test.
Densities of T lymphocytes, and subsets Th9 and Th17 cells in different dysplastic degrees of non-adenoma-like dysplastic (NALD) tissues.

Abbreviations: HGD, high-grade dysplasia; LGD, lower-grade dysplasia; MGD, moderate-grade dysplasia.
P-values were from the Mann-Whitney test.
Discussion
Previous studies have shown that high proportions of T lymphocytes are found in the UC 42 and CAC microenvironment;43-45 however, the role of diverse Th subsets in the pretumor dysplastic status is not fully understood. In this study, we found that T lymphocytic sub-phenotypes Th9 and Th17 cells were highly aggregated in the CAD tissues. Densities of Th9 and Th17 cells were greatly changed, in which both were increased in the dysplastic lamina propria and epithelium, suggesting a contributory effect of Th9 and Th17 cells to the T lymphocytic phenotypic composition in the inflammatory pretumor microenvironment.
In this study, we first evaluated the presentation of CD3-posiitve lymphocytes in the UC and CAD tissues. The IHC analysis results reveal a great change in the presentation and densities of CD3-positive T lymphocytes in both the UC and UC dysplastic (ALD and NALD) tissues. These results confirmed previous findings and indicated that T lymphocytes are accumulated in the UC and pretumor microenvironment. The Th9 and Th17 cells are T lymphocytic subtypes 46 and can produce numerous cytokines to promote inflammation,24,25,47 and considerable studies have addressed the importance of Th9 and Th17 cells in the development of chronic inflammation in UC.24,37,45,48-51 As chronic inflammation is the most important deriving force for the development of CAC, the dynamic of Th9 and Th17 cell changes in constituting a link between chronic intestinal inflammation and the development of CAC should be considered. Indeed, there are studies to preliminarily confirm the importance of Th17 cells in the formation of CAD in colitis mice.27,28,52 Regarding the role of Th9 cells in the development of CRC, previous studies performed in the sporadic CRC reported controversial findings, both protumor and antitumor effect were shown. 53 However, recent published in vivo and in vitro data have revealed that overexpression of Th9 cytokine IL-9 gene in RKO and Caco-2 cell lines led to an enhanced proliferation by activating c-myc and cyclin D1 signaling pathway, 36 indicating a possible regulatory effect of IL-9 on the growth of CAC. More recently, a study reported that enhanced expansion of Th9 cells and expression of IL-9 (Th9 cytokine) stimulate the CAC development and growth in colitis mice, 37 suggesting a promoting effect of Th9 cells on the development of CAC. In this study, our current findings of abundant Th9 and Th17 cells scattered in the UC tissues provided supportive evidence for the involvement of Th9 and Th17 cells in the pathogenesis of UC. Furthermore, dense PU.1-positive Th9 and IL-17A-positive Th17 cells were identified in the CAD tissues. Density grading analysis also confirmed the IHC observation and revealed that densities of Th9 and Th17 cells in the lamina propria and epithelium were evidently increased in the CAD group compared with the control group, and the density of Th9 cell was even higher than that in the UC group. Therefore, such significant increased numbers of Th9 and Th17 cells in the UC tissues and CAD lesions may provide further supportive evidence to suggest a potential role of Th9 and Th17 cells in the development of CAC. 37
The possible mechanisms of the interaction between Th9 and Th17 in promoting the development of CAC has been hypothesized. For example, there is ample evidence to demonstrate the involvement of mast cells in the maintenance of chronic inflammation in UC and the process of inflammation-associated neoplasia.27,54 Interestingly, previous scientific findings suggested that Th9/IL-9 can greatly enhance the survival and cytokine production of mast cells.55,56 Furthermore, Th17 cells have been found to play a crucial role in the induction and perpetuation of the intestinal inflammation and CAC in UC through releasing different proinflammatory mediators.27,57-60 Whereas data from various studies showed that IL-9 can obviously expanse Th17 differentiation under certain conditions.61,62 Studies also revealed that IL-9 can be produced by Th17 cells and acts as an important mediator to stimulate the inflammation. 63 Therefore, the similar presentation pattern of Th9 and Th17 cells in the CAD tissues may imply that the interaction between Th9 and Th17 cells may have a synesthetic effect to promote the initiation of CAC.
In this study, density grading analysis showed a general trend that scores of Th9 and Th17 cells accumulated in the lamina propria of the NALD subgroup than that in the ALD subgroup. Although the significance of Th9 and Th17 cells distributed in different compartments in dysplastic tissues remains to investigate, this finding may reflect a different spatial distribution pattern between the ALD and NALD subgroups. Therefore, it is necessary to evaluate the exact role of Th9 and Th17 cells in different compartments during the development of CAC in the future.
Limitations of this study must be addressed. In this prior study, only paraffin blocks, but no fresh tissues, from UC and CAD tissues were recruited. Paraffin blocks are suitable for IHC analysis of the presentation pattern, location and density analysis of Th9 and Th17 cells at the cellular level. However, it is impossible for the quantitative analysis of these cell markers by western blot, real-time PCR, and low cytometric techniques. Therefore, the expression of these cells examined by other laboratory techniques must be confirmed in the future. To some extent, several factors may lead to differences in tissue paraffin block quality and affect IHC results. 38 In this study, the sampling of UC and CAD tissues under surveillance colonoscopy were taken by different gastroenterologists and processed to paraffin blocks by different technicians in the Department of Pathology. Studies have also demonstrated that tissue preparations, such as the size, location, orientation, and laboratory process, affect the histological evaluation and diagnosis in inflamed tissues. Previous clinical evidence has also revealed that the quality of bowel preparation prior to colonoscopy examination may also influence the observation and then sampling of inflamed mucosa under colonoscopy in patients with UC. 38 All these factors should be considered when we evaluate IHC results in the future. Furthermore, this study is a preliminary study, and the study sample size is quite small. Findings and conclusion obtained from this small study must be validated in the future project with a big sample size. Finally, due to the limitation of ethic permission for this study, we were unable to include human CAC paraffin blocks to examine Th9 and Th17 cells in the CAC tissues. Therefore, it is important to evaluate the presentation pattern of Th9 and Th17 cells in CAC tissues in the future.
Conclusions
This study evaluated the presentation of Th subsets Th9 and Th17 cells in the CAD lesions and revealed a general tendency of Th9 and Th17 cells to dynamically increase in the whole lamina propria along the Normal-UC-CAD sequence. These findings suggest that Th9 and Th17 cells significantly contribute to the composition of immune phenotypes in the CAD microenvironment and may represent the early conditional change for the Dysplasia-CAC transition under the UC background. Therefore, future studies that highlight the exact effects and mechanisms of Th9 and Th17 cells in the immunopathogenesis of CAC remain to be conducted.
Footnotes
Acknowledgements
We express our sincere gratitude to our colleagues at the Departments of Pathology and Gastroenterology for supplying biopsies.
Author Contributions
GC contributed to the concept or design of this work; or acquisition, interpretation of data and in drafting the article. AY contributed to the conduction of experiments, analysis, and interpretation of data. SWS contributed to pathological analysis, interpretation, and discussion of data, and in reviewing the article. JF contributed to the concept or design of this work, supervision, and review of the article. All authors approved the version to be published.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by grants from Medical Research Program, Northern Norway Regional Health Authority (SFP-44-04) and the Second Affiliated Hospital of Zhengzhou University (202204169).
Availability of Data and Materials
The data that support the findings of this study are available from our hospital, but restrictions apply to the availability of these data, which were used under license for this study, and so are not publicly available. Data are, however, available from the authors upon reasonable request and with permission of our hospital.
Consent for Publication
None consent for publication.
Ethics Approval and Consent to Participate
This study was conducted according to the World Medical Association Declaration of Helsinki and the study protocol has been approved by The North and West Norwegian Regional Ethical Committee of Norway (Approval Numbers: 29895 and 419346). Written consent is obtained from all patients with UC and normal controls. As many CAD patients were dead and the written consent was impossible to collect, waiver of the requirement to obtain written consent from these patients was issued by the Regional Ethical Committee.
