Abstract
Background:
Recurrent or metastatic squamous cell carcinoma of the head and neck (R/MHNSCC) is a challenging malignancy with a poor prognosis and limited treatment options. Nivolumab, an immune checkpoint inhibitor (ICI) targeting the programmed cell death/programmed cell death ligand 1 (PD-1/PD-L1) pathway, has emerged as a promising therapy for these patients. However, identifying biomarkers predictive of response to nivolumab remains critical for optimizing treatment strategies. Previous studies have suggested that PD-L1 expression, as determined by the Combined Positive Score (CPS) and other clinical factors, may influence treatment outcome. This study aims to retrospectively examine whether CPS can be a biomarker by staining PD-L1 with 22 C3 antibody in R/MHNSCC patients treated with nivolumab.
Methods:
This retrospective study reviewed the medical records of R/MHNSCC patients treated with ICIs at Tokai University Hospital from April 2017 to December 2022. We examined the relationship between response rate to ICI therapy, PD-L1 staining, biomarkers, and survival. Statistical analyses included t-test, chi-square test, and Cox regression.
Results:
This study included 92 nivolumab-treated patients. Combined Positive Score was evaluable in 53 of these patients. Patients with a CPS of 15 or higher had better progression-free survival (PFS) (P = .0171), with a median PFS) of 13 months. In the Various Definitions analysis, cisplatin-sensitive patients also had good PFS (P = .0295). The cisplatin-sensitive patient population with a CPS of 15 or higher had the best PFS, with a median of 14 months (P = .006). There was no significant difference in overall survival (OS) by CPS value. Immune-related adverse events did not affect OS or PFS.
Conclusions:
CPS ⩾ 15 and cisplatin sensitivity are promising prognostic markers for nivolumab therapy in R/MHNSCC. Considering these biomarkers in patient selection could maximize the therapeutic benefits of nivolumab. This finding may help to optimize ICI therapy strategies.
Introduction
Immune checkpoint inhibitors (ICIs) have emerged as a highly promising approach in the realm of cancer therapy, notably enhancing patient prognosis and quality of life. One of the key mechanisms by which ICIs exert their therapeutic effects is through the reversal of immune suppression associated with tumor progression, thereby enabling the immune system to effectively target and eliminate cancer cells. 1 These inhibitors target immune checkpoints such as PD-1, PD-L1, and CTLA-4 and have been associated with improved patient outcomes and a better quality of life compared with traditional cancer treatments. 2 Nevertheless, their application presents certain challenges, such as immune-related side effects and resistance to therapy. 3 Further studies are needed to optimize the safety and efficacy of these inhibitors, both as monotherapy and in combination with other therapies.
ICIs are frequently utilized in the treatment of recurrent or metastatic squamous cell carcinoma of the head and neck (R/MHNSCC) because they cause fewer adverse events (AEs) than chemotherapy one and improve patients’ quality of life.4,5 Their outcomes and prognosis are gradually being elucidated.6-8 However, around 60% of R/MHNSCC patients show no response to immunotherapy, and merely 20% to 30% of those treated gain long-term benefits from ICIs.9-11 It is important to identify predictors of ICI effectiveness in terms of healthcare costs and side effects.
In R/MHNSCC, the Keynote048 study 12 demonstrated the efficacy of Combined Positive Score (CPS) as a complementary diagnosis in Pembrolizumab. In contrast, in the CheckMate 141 study, 13 PD-L1 expression (Tumor Proportion Score; TPS) was not a prognostic factor for nivolumab. We hypothesized 2 possible reasons: antibody differences (28-8 vs. 22 C3 antibodies) and differences in PD-L1 measurements (TPS vs. CPS).
The FDA approval of nivolumab and Pembrolizumab for patients with recurrent head and neck squamous cell carcinoma (HNSCC) is a major advance in the treatment options available to these patients, according to a consensus statement from the Cancer Immunotherapy Society. 14 These guidelines provide recommendations on patient selection, therapy sequence, response monitoring, AE management, and biomarker testing. Biomarkers have been explored as prognostic factors in ICI therapy. Previous studies in head and neck cancer by our team reported serum albumin levels, C-reactive protein levels, platelet-to-lymphocyte ratio, neutrophil-to-lymphocyte ratio, lymphocyte-to-monocyte ratio, systemic immune-inflammatory index, and nutritional status score. 15 Studies in patients with metastatic melanoma have also suggested that the dynamics of cytokine levels during treatment can be useful in selecting patients for ICI treatment and predicting treatment response, and have proposed that early reductions in TGF-β and IL-6 may be biomarkers of treatment response. 16 However, there is no established biomarker for predicting the prognosis of ICI therapy.
This retrospective study aims to determine whether the CPS, obtained by staining PD-L1 with the 22 C3 antibody, can serve as a biomarker in R/MHNSCC patients treated with nivolumab and to examine the association of various factors with overall survival (OS) and progression-free survival (PFS), testing the hypotheses that CPS and various factors are related to OS and PFS in these patients.
Patients and Methods
Study design/sample
This retrospective study included R/MHNSCC patients treated with Nivolumab at Tokai University Hospital in Kanagawa, Japan, from April 2017 to December 2022. Patients were included if they had an Eastern Cooperative Oncology Group (ECOG) performance status of 0 to 2, had adequate organ function, and had received at least one cycle of nivolumab therapy. Exclusion criteria included incomplete medical records, unmeasurable CPS, difficulty tracing patient information, receiving ICI treatment other than nivolumab, and refusal to participate in the study. Nivolumab was administered to patients at 3 mg/kg or 240 mg/body doses every 2 weeks, based on individual patient conditions determined during a multidisciplinary head and neck cancer conference. The treatment continued until either disease progression or unacceptable toxicity occurred, with patients monitored until death or the cutoff date (May 31, 2023). The study received approval from the Institutional Review Board of Tokai University Hospital (22R223) and was conducted in compliance with the principles of the Declaration of Helsinki. Informed consent was waived due to the retrospective nature of this analysis, which used existing administrative and clinical data.
Variables and data collection methods
The primary predictor variable was the CPS. The outcome variables were PFS and OS. Covariates included age, gender, human papillomavirus (HPV) status, prior therapy (chemotherapy, radiation therapy, surgery), and cisplatin sensitivity.
Medical records were reviewed to collect demographic data, treatment details, and outcomes. Clinical response to treatment was assessed every 4 to 12 weeks using computed tomography (CT), and tumor response was evaluated according to the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. Objective response rate (ORR) was defined as the percentage of patients achieving complete response (CR) or partial response (PR) as the best outcome. Disease control rate (DCR) was defined as the proportion of patients achieving CR, PR, or stable disease (SD) as the best outcome; OS was defined as the time from the start of treatment to the date of death or cutoff date, regardless of cause; PFS was defined as the time from the start of treatment to disease progression, death from any reason, or cutoff date, whichever occurred first. Progression-free survival was defined as disease progression, death from any cause, or cutoff date, whichever occurred first. Duration of response was defined as the time from the first response (SD, PR, PD) to disease progression.
Data analyses
All statistical analyses were performed with the statistical software ‘EZR’ (Easy R). 17 Methods included t-test, chi-square test, analysis of variance (ANOVA), and Cox regression analysis. Multiple comparisons were managed using Bonferroni correction. Overall survival and PFS were estimated using the Kaplan-Meier method and evaluated using the log-rank test. The association between ORR and each factor was tested using the chi-square test or Fisher’s exact test for comparisons between 2 groups and the Kruskal-Wallis test for comparisons of 3 or more groups. Cutoff values for CPS were determined by survival Classification and Regression Tree (CART) analysis. A Cox regression model was used to analyse the relationship between inflammatory and nutritional biomarkers and OS or PFS. A multivariate analysis was performed after adjusting for age. Survival CART analysis was used as a machine learning model to identify the CPS most related to OS. Cox regression model analysis, CART analysis, and random survival forest analysis were performed using R software (ver. 4.2.2). Statistical significance was set at P < .05.
PD-L1 staining
Programmed cell death ligand 1 staining with PD-L1 IHC 22 C3 pharmDx ‘Dako’ (Agilent Technologies, California, USA) was used to measure the CPS and TPS. Tumor proportion score is defined as the positivity rate in tumor cells by PD-L1 immunostaining. Combined Positive Score is defined as the number of PD-L1-positive cells (tumor cells, lymphocytes, and macrophages) divided by the total number of tumor cells ×100; a minimum of 100 viable tumor cells must have been present for the specimen to be considered evaluable. 11 A trained pathologist and a head and neck surgeon determined the CPS and TPS values. Microscopic observations were performed at weak (200×) and strong (400×) magnification.
Definition of factors
Adverse events were recorded using the National Cancer Institute Common Terminology Criteria for Adverse Events version (5.0). Cisplatin-resistant was defined as cancer with documented tumor progression during treatment with cisplatin or relapse within 6 months after cisplatin-based chemoradiation. Cisplatin-sensitive was defined as cancer that recurred at least 6 months after completion of cisplatin-based therapy. Human papillomavirus positive was defined as positive for p16 staining in biopsy specimens. A history of chemotherapy was defined as a history of at least one course of chemotherapy. A history of radiation therapy was defined as curative or prophylactic irradiation. A history of primary resection or neck dissection was defined as a history of surgery. Immune checkpoint inhibitor line was defined as the order in which nivolumab therapy was used in the treatment sequence. Pathological sample was defined as whether the histopathological specimens were obtained from the primary site or metastatic lymph nodes. Type of recurrence was defined as the difference between locoregional recurrence and distant metastasis.
Results
Patient characteristics
Of the 132 R/MHNSCC patients, 92 received nivolumab, and 40 received pembrolizumab; of the 92 who received nivolumab, PD-L1 analysis was available in 53 patients (Figure 1). Of the patients treated with nivolumab, 39 specimens were difficult to stain for PD-L1 due to insufficient specimen volume, biopsy specimens from other hospitals, or specimen deterioration.

Patient flowchart. Of the 132 R/MHNSCC patients, PD-L1 analysis was available for 53 patients.
Their characteristics are summarized in Table 1. There were 47 males and 6 females with a median age of 64 (range: 47-80) years. The primary site was the hypopharynx in 18 patients, the oropharynx in 13 patients, the larynx in 8 patients, the nasopharynx in 6 patients, the maxillary sinus in 4 patients, the oral in 3 patients, and others (unknown primary origin) in 1 patient. The ECOG performance status was 0 in 50 patients and 1 in 3 patients. The disease sites evaluated were locoregional recurrence in 32 patients, distant metastases in 12 patients and locoregional and distant in 9 patients. The line of ICI was one patient for first, 26 patients for second, 20 patients for third, and 5 for fourth. Before receiving nivolumab therapy, 16 patients underwent surgery, 47 received radiotherapy, 51 underwent chemotherapy, and 18 received cetuximab. Chemotherapy-related AEs were present in 13 patients. Pathology specimens were primary in 45 patients and metastatic lymph nodes in 8 patients. Nine patients were positive for HPV. Twenty-nine patients received salvage chemotherapy after nivolumab, including 19 patients on paclitaxel, 4 patients on paclitaxel plus cetuximab, and 6 patients on TS-1 (Tegafur/Gimeracil/Oteracil).
Patient characteristics.

Abbreviations: ECOG, Eastern Cooperative Oncology Group; HPV, human papillomavirus; ICI, immune checkpoint inhibitor.
Measurement of CPS and TPS
The correlation between CPS and TPS is shown in Figure 2. The correlation coefficient was 0.985 (95% CI: 0.972-0.992), indicating a strong correlation between CPS and TPS.

Correlation between CPS and TPS. The correlation coefficient was 0.985 (95% CI: 0.972-0.992), indicating a strong correlation between CPS and TPS.
Figure 3 shows the distribution of patients by CPS. Four patients had a CPS of less than 1, 37 patients had a CPS of 1 or more and less than 20, and 12 patients had a CPS of 20 or more.

Distribution of CPS. 7.5% of patients had a CPS of less than 1, 49.1% had a CPS of 1 to 20, and 43.4% had a CPS of more than 20.
PD-L1 staining
To further illustrate the differences in PD-L1 expression, we present representative immunohistochemical images of both high and low PD-L1 expression, along with corresponding hematoxylin and eosin (HE) stained images (Figure 4). Figure 4A shows a case with high PD-L1 expression, where the tumor cells are strongly stained, indicating significant PD-L1 positivity, accompanied by the corresponding HE image for histopathological context. Figure 4B depicts a case with low PD-L1 expression, with minimal staining observed in the tumor cells, alongside the corresponding HE image. These combined images visually demonstrate the variability in PD-L1 expression and its histological correlation, which may have clinical significance.

Hematoxylin-eosin (HE) stained images and representative immunohistochemical images of PD-L1 high and low expression in tumor tissue (400×). In the case with high PD-L1 expression (A), tumor cells and immune cells are strongly stained, indicating that PD-L1 is significantly positive. In the case with low PD-L1 expression (B), tumor cells, and immune cells are not stained.
Treatment response
The treatment response was evaluated according to RECIST version 1.1. Of the 53 patients (Table 2), 9 (17.0%) achieved a CR, 15 (28.3%) achieved a PR, 190 (35.8%) had SD, and 10 (18.9%) had progressive disease (PD). The ORR was 45.3%, and the DCR was 81.1%.
Effectiveness of immune checkpoint inhibitors therapy.

Abbreviations: CPS, combined positive score; DCR, disease control rate; ORR, objective response rate.
Classification and Regression Tree analysis was used to evaluate the cutoff values of CPS in OS and PFS. As shown in Supplementary Figure 1, the cutoff value of CPS in PFS was shown to be 15. The ORR and DCR in patients with CPS less than 15 were 30% and 63.3%, respectively. In patients with CPS 15 or higher, the ORR was 39.1%, and the DCR was 82.6%. There was a favourable trend in the group with CPS 15 or higher.
Survival
Overall survival and PFS were estimated using the Kaplan-Meier method (Figure 5). The median OS was 21 (95% CI, 17-34) months, and the median PFS was 8 (95% CI, 5-12) months. The 1- and 2-year OS rates were 73.2% and 43.9%, respectively. The 1- and 2-year PFS rates were 34.9% and 23.1%, respectively.

Kaplan-Meier curves in the overall population: (A) overall survival and (B) progression-free survival. Survival curves were plotted based on the last survival confirmation date. Two-year survivors are shown as censored at 24 months. The median OS was 21 (95% CI, 17-34) months, and the median PFS was 8 (95% CI, 5-12) months. The 1- and 2-year OS rates were 73.2% and 43.9%, respectively. The 1- and 2-year PFS rates were 34.9% and 23.1%, respectively.
Classification and Regression Tree analysis showed a cutoff value of CPS 2 for OS and CPS 15 for PFS (Supplementary Figure 1). Based on this classification, OS and PFS were estimated using the Kaplan-Meier method (Figure 6). Even with a cutoff value of 2 for CPS, no significant differences were shown in both OS and PFS. On the other hand, a cutoff value of 15 for CPS showed a significant difference in PFS (P = .0171). The median PFS in the CPS less than 15 group was 6 months, and the 1- and 2-year PFS rates were 14.5% and 7.2%, respectively. On the other hand, the median PFS for patients with CPS 15 or higher was 13 months, and the 1- and 2-year PFS rates were 53.7% and 37.2%, respectively, showing a favourable trend.

Kaplan-Meier curves by CPS: (A) overall survival and (B) progression-free survival. Survival curves were plotted based on the last survival confirmation date. Two-year survivors are shown as censored at 24 months. There is no significant difference in OS or PFS with a CPS cutoff 2. A significant difference exists in PFS with a CPS cutoff of 15 (P = .0171). The median PFS for the CPS < 15 group was 6 months, with a 1-year PFS rate of 14.5% and a 2-year PFS rate of 7.2%. The median PFS for patients with CPS 15 or higher was 13 months, with a 1-year PFS rate of 53.7% and a 2-year PFS rate of 37.2%.
Analysis of various parameters
Table 3 shows the OS and PFS regression analysis results in nivolumab-treated patients. Examination of OS showed significant correlations with cisplatin sensitivity, DCR, and ORR. Furthermore, multivariate analysis showed that cisplatin sensitivity was significantly correlated with OS; PFS and factor analysis showed significant correlations with cisplatin sensitivity, DCR, ORR, cetuximab treatment history, and gender, but no significant differences in multivariate analysis.
Cox regression analysis of overall survival and progression-free survival.

Abbreviations: CPS, combined positive score; ECOG, eastern cooperative oncology group; HPV, human papillomavirus; ICI, immune checkpoint inhibitor.
p-values in bold indicate a significant difference.
Figure 6 shows the results of Kaplan-Meier survival curves and log-rank tests by cutoff values for cisplatin sensitivity, HPV, prior cetuximab treatment, and type of recurrence.
An important factor in OS was cisplatin sensitivity, with a significant difference between the 2 groups. Cisplatin sensitivity, prior cetuximab treatment, and type of recurrence were the important factors for PFS, and there were significant differences between the 2 groups in these factors.
Progression-free survival was better in the patient population with a CPS cutoff value of 15 or higher (Figure 6) or cisplatin sensitivity (Figure 7). These results examined Kaplan-Meier curves, as shown in Figure 8, indicating that PFS was better in the cisplatin-sensitive group with a CPS of 15 or higher.

Kaplan-Meier curves: (A) overall survival and (B) progression-free survival according to the cisplatin sensitivity, HPV, prior cetuximab treatment and type of recurrence. Survival curves were plotted based on the last survival confirmation date. Two-year survivors are shown as censored at 24 months. Progression-free survival tended to be better in cisplatin-sensitive patients, patients with no history of cetuximab use, and patients with distant metastases.

Kaplan-Meier curves: progression-free survival according to the cisplatin sensitivity and CPS. The patient population with a CPS of 15 or higher and cisplatin sensitivity showed a trend towards better PFS.
Analysis of immune-related adverse events
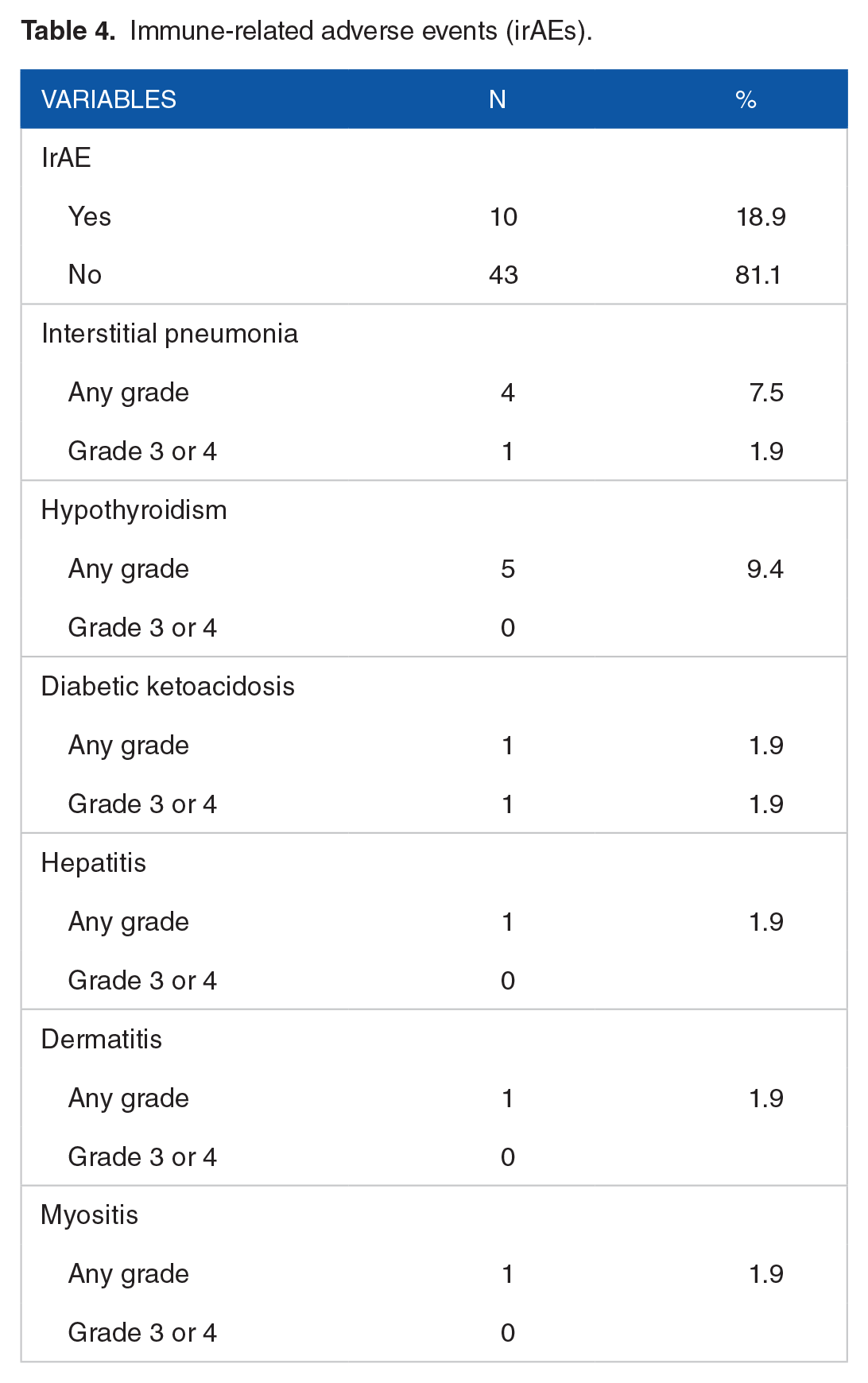
Table 4 shows that immune-related adverse events (irAEs) was observed in 10 patients (18.9%). The most common was hypothyroidism (9.4%), followed by interstitial pneumonia (7.5%), type 1 diabetes (1.9%), hepatitis (1.9%), dermatitis (1.9%), and myositis (1.9%). Grade 3 or higher AEs were observed in one case each in interstitial pneumonia and type 1 diabetes mellitus. As shown in Table 3, there was no significant correlation between the presence or absence of irAEs and OS or PFS.
Immune-related adverse events (irAEs).
Discussion
The aim of this study was to assess the prognostic significance of PD-L1 expression using CPS and TPS in patients with recurrent/metastatic head and neck squamous cell carcinoma (R/MHNSCC) treated with nivolumab. We hypothesized that PD-L1 CPS stained with 22 C3 antibody, would be a valuable biomarker for predicting treatment outcomes. Our specific aims were to determine the correlation between PD-L1 expression and OS and PFS and to compare these findings with existing studies.
The main finding was that PD-L1 CPS was significantly associated with PFS but not OS, suggesting that CPS may be a potential biomarker to predict the efficacy of nivolumab in R/MHNSCC.
Of the 132 R/MHNSCC patients, 92 received nivolumab treatment (Figure 1). This is because nivolumab is used in Japan for R/MHNSCC patients with prior radiation chemotherapy or chemotherapy due to the Japanese insurance system. Of these 92 patients, PD-L1 analysis was available in 53 patients.
In R/MHNSCC, PD-L1 expression was a useful prognostic factor for pembrolizumab therapy 12 but not for nivolumab therapy. 13 Two possible reasons for this are differences in antibodies and differences in PD-L1 assays; Maule et al 18 reported that the 22 C3 antibody is the most sensitive immunohistochemical companion diagnostic assay for PD-L1 expression in tumor cells and can identify PD-L1 positivity more frequently than the 28-8 antibody. In addition, PD-L1 expression by TPS and CPS has been reported to be similar in non-small-cell lung cancer in a study using 22 C3 antibody.19,20 In light of this, in this study, we performed PD-L1 staining using 22 C3 antibody with respect to nivolumab therapy. As shown in Figure 2, there were no differences in CPS and TPS assay methods, and a strong correlation was observed. Therefore, we focused on CPS in this study because the effect of antibody differences was considered significant.
Of the 53 patients, 23 (43%) had PD-L1 CPS of 20 or more and 49 (92%) had PD-L1 CPS of 1 or more (Figure 3). This was similar to the Keynote048 study 12 (43% had a CPS of 20 or more, and 85% had a CPS of 1 or more).
This study’s median OS was 21 months, and the 12 month OS rate was 73.2%. The median PFS was 8 months, and the 12 month PFS rate was 34.9%. These results showed a trend towards better OS trend than the Checkmate 141 study 13 and Japanese clinical use reports,21,22 but comparable PFS.
The CART analysis in this study derived a CPS cutoff value of 2 or 15. Based on this classification, OS and PFS were estimated using the Kaplan-Meier method. When the CPS cutoff value of 2 was considered, there were no significant differences in both OS and PFS. On the contrary, when comparing the CPS less than 15 and 15 or more groups, PFS was significantly better in the CPS 15 or more patient population in both median (6 vs 13 months), 1-year PFS rate (14.5% vs 53.7%) and 2-year PFS rate (7.2% vs 37.2%; P = .0171). Objective response rate and DCR in the patient population with CPS 15 or higher tended to be better than in the population with CPS less than 15 but insignificant.
In this study, CPS was not related to OS but tended to be associated with PFS. It has been reported that there was no significant correlation between PD-L1 expression and OS in HNSCC. 23 Although the carcinomas differ, several studies of nivolumab therapy for advanced gastric cancer have reported that CPS is the only clinical factor associated with PFS 24 and that CPS is associated with PFS but not OS. 25 Response to salvage chemotherapy after nivolumab therapy has also been reported,26-28 and the response to salvage chemotherapy also influences OS. Therefore, it is possible that this study did not find a difference in OS according to CPS values.
A study 29 on PD-L1 expression in HNSCC reported higher expression of PD-L1 in lymph node metastases than in primary tumors and heterogeneity within tumor specimens in 52.2% of cases. However, in this study, CPS was measured and examined in 45 primary lesions and 8 cervical lymph nodes, and there was no significant difference between the different collection sites. In addition, although there are few reports on temporal heterogeneity of PD-L1 expression, it has been pointed out that it may change from the initial diagnosis during disease progression in other carcinomas.30,31 Although there are many unknowns regarding changes in PD-L1 expression in HNSCC, it has been reported that the concordance rate of PD-L1 expression between initial and recurrent lesions is around 64-67%, even at the same site. 30 Therefore, spatial and temporal heterogeneity of PD-L1 expression may have affected the results.
In recurrent ovarian cancer, patients in the platinum-sensitive group are more likely to respond to subsequent chemotherapy than those in the platinum-refractory group and have a better prognosis.31,32 This concept has recently been used for R/MHNSCC. In this study, 66% of patients were platinum-sensitive, a population not included in the CheckMate 141 study. 13 CheckMate-141 enrolled only platinum-refractory R/MHNSCC patients and did not study platinum sensitivity. The KEYNOTE 048 study 12 showed that Pembrolizumab was effective in platinum-sensitive R/MHNSCC. Nivolumab may also be effective in platinum-sensitive R/MHNSCC. Okamoto et al 33 reported a study of nivolumab therapy in a platinum-sensitive patient group (median PFS: 9.6 months). Hori et al 34 said that in R/M-HNSCC patients treated with nivolumab therapy, PFS was significantly better in the platinum-sensitive group than in the platinum-refractory group (median PFS: 13 weeks vs 38 weeks, P = .006). Similarly, in this study, OS and PFS were significantly better in the platinum-sensitive patient population.
From the above, as shown in Figure 8, we divided the patients into platinum-sensitive/refractory and CPS 15 or higher/less than 15. It is suggested that PFS is best in the group of patients who are platinum-sensitive and have a CPS of 15 or higher.
Most irAEs with Nivolumab treatment are reported to be of low grade.22,35 In this study, 18.9% of patients experienced irAEs of all grades, but only 2 patients (3.8%) had grade 3 or higher. Most were low-grade and allowed patients to continue nivolumab therapy while dealing with the AEs. These results suggest that nivolumab has a manageable safety profile in real-world clinical practice. In addition, patients who experienced irAEs of any grade have been reported to have a better prognosis than those who did not. 21 ,35-37 However, in the univariate analysis of this study, the presence or absence of irAEs was not related to OS or PFS.
Biomarkers related to ICIs were also examined. A recent meta-analysis of anti-PD-L1 in head and neck cancer reported that anti-PD-L1 was more effective in female patients, patients with local recurrence, and HPV-positive patients. 9 Still, these differences were insignificant in this study. It has also been reported that an overall performance status (PS) of 0 to 1 is significantly associated with OS,22,36 and it is thought that Nivolumab administered while maintaining PS reflects the host’s immune status, resulting in better OS and PFS. In this study, all patients had PS 0 to 1, making it difficult to examine the differences in PS. This is because our department generally administers ICI therapy to patients in good general condition, ie, those with good PS. Furthermore, decreased efficacy of nivolumab in patients previously treated with cetuximab has been reported. 38 Cetuximab is thought to suppress interferon gamma-induced PD-L1 expression in HNSCC cell lines. 39 Indeed, PFS was significantly worse in the patient population previously treated with cetuximab in this study (P = .0186).
Our study has several strengths and limitations. The strengths include using a well-defined patient cohort and applying the 22 C3 antibody for PD-L1 staining, which has been shown to be highly sensitive. The limitations are as follows. First, this is a retrospective study conducted at a single institution, and the relatively small sample size may limit the generalizability of the results. In addition, patient chart variation and potential measurement errors must be considered when interpreting the findings. Second, the determination of PD-L1 positivity involves some degree of subjectivity, and sample quality and spatial and temporal heterogeneity of PD-L1 expression may affect the results. Therefore, it is recommended that future studies use more standardized assessment criteria in combination with molecular biological methods. Third, the assessment of PD-L1 expression is based solely on immunohistochemical methods and not on molecular biological techniques such as PCR or flow cytometry. Therefore, differences in PD-L1 expression in individual cell subpopulations could not be assessed. Future studies should incorporate a broader range of analytical methods to thoroughly examine the spatial and temporal heterogeneity of PD-L1 expression.
Our study suggests that PD-L1 CPS could be a valuable biomarker for predicting PFS in R/MHNSCC patients treated with Nivolumab. The manageable safety profile of Nivolumab and its potential efficacy in platinum-sensitive patients highlights its clinical significance. Future research should focus on larger, multi-centre studies to validate PD-L1 CPS as a prognostic biomarker and explore the underlying mechanisms affecting PD-L1 expression and treatment response.
Conclusions
The CPS of nivolumab was significantly associated with PFS and is considered a promising biomarker. Furthermore, patients with CPS ⩾ 15 and positive cisplatin sensitivity had better PFS on nivolumab treatment, suggesting that these factors determine prognosis.
Supplemental Material
sj-tif-1-onc-10.1177_11795549241290030 – Supplemental material for Combined Positive Score and Cisplatin Sensitivity Are Prognostic Factors for Response to Nivolumab Therapy for Recurrent Metastatic Squamous Cell Carcinoma of the Head and Neck
Supplemental material, sj-tif-1-onc-10.1177_11795549241290030 for Combined Positive Score and Cisplatin Sensitivity Are Prognostic Factors for Response to Nivolumab Therapy for Recurrent Metastatic Squamous Cell Carcinoma of the Head and Neck by Hiroaki Iijima, Akihiro Sakai, Koji Ebisumoto, Go Ogura, Mayu Yamauchi, Takanobu Teramura, Aritomo Yamazaki, Takane Watanabe, Toshihide Inagi, Ryoko Yanagiya, Ai Yamamoto, Hiroshi Ashida, Yoshiyuki Ota, Yurina Sato, Naoya Kobayashi, Daisuke Maki, Naoya Nakamura and Kenji Okami in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
We would like to thank Dr Mebae Ichikawa for her help with the pathology specimens.
Author Contributions
Hiroaki Iijima, MD: Conception, data management, formal analysis, investigation, and writing (draft).
Akihiro Sakai, MD, PhD: Methodology, supervision, formal analysis, project management, writing-review and editing.
Koji Ebisumoto, MD, PhD: Data curation, formal analysis, writing-review and editing.
Go Ogura, MD, PhD: Pathology, histological evaluation.
Mayu Yamauchi, MD: methodology, software, research, resources.
Takanobu Teramura, MD: Data curation, formal analysis.
Aritomo Yamazaki, MD: research, materials, resources.
Takane Watanabe, MD: surveys, materials, resources.
Toshihide Inagi, MD: Research, Resources.
Ryoko Yanagiya, MD: Research, Resources.
Ai Yamamoto, MD: data curation, formal analysis.
Hiroshi Ashida, MD: research, materials, resources.
Yoshiyuki Ota, MD: research, materials.
Yurina Sato, MD: surveys, materials, resources.
Naoya Kobayashi, MD: research, materials.
Daisuke Maki, MD, PhD: writing, reviewing, editing, and resources.
Naoya Nakamura, MD, PhD: Pathology, histological evaluation.
Kenji Okami, MD, PhD: Supervise, manage projects, write reviews, and edit.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
All relevant data have been presented in this manuscript.
Ethical Approval
The study received approval from the Institutional Review Board of Tokai University Hospital (22R223)
Patient Consent and Consent for Publication
The Institutional Review Board waived the requirement to obtain informed consent because this study was retrospective.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
