Abstract
Background:
As a second-line therapy, oxaliplatin/fluorouracil/leucovorin (FOLFOX) remains the standard of care for patients with biliary tract cancer (BTC); however, its efficacy is suboptimal. The aim of this study was to evaluate whether, compared with chemotherapy alone, the immune checkpoint inhibitor (ICI) combination regimen improved the overall survival (OS) in patients with advanced BTC.
Methods:
Patients diagnosed with advanced BTC who received chemotherapy or ICI combination therapy as second-line (L2) treatment between January 1, 2018, and April 1, 2022, were retrospectively identified.
Results:
A total of 98 patients with BTCs were reviewed and recruited: the chemotherapy group (cohort A, n = 40), the chemotherapy plus ICIs group (cohort B, n = 27), and the tyrosine kinase inhibitor (TKIs) plus ICIs group (cohort C, n = 31). The median progression-free survival (PFS) and median OS were 2.6 months (95% confidence interval [CI]: 1.7-4.2) and 7.8 months (95% CI: 5.9-12.0) for cohort A, 4.3 months (95% CI: 2.9-8.4) and 10.9 months (95% CI: 7.67-NA) for cohort B, 5.1 months (95% CI: 4.0-8.3) and 10.1 months (95% CI: 8.23-NA) for cohort C, respectively. The confirmed overall response rates were 7.5% (3/40, cohort A), 22.2% (6/27, cohort B), and 19.4% (6/31, cohort C), whereas the disease control rates were 47.5% (19/40, cohort A), 77.8% (21/27, cohort B), and 77.4% (24/31, cohort C). Grade 3 or higher treatment-related adverse reaction were reported in 20.0% (cohort A), 37.0% (cohort B), and 41.9% (cohort C) of the patients.
Conclusions:
The ICI combination strategy beyond first-line (L1) systemic chemotherapy plays a positive role in advanced BTCs. Both TKIs plus ICIs and chemotherapy plus ICIs could be considered candidates for trials and applied as competitive L2 treatment regimens for advanced BTCs in clinical practice.
Introduction
Biliary system cancer is a heterogeneous group of malignancies with a 5-year survival rate of <10%.1,2 In the advanced disease setting, gemcitabine-based combination regimens as the current standard of care could improve outcomes with a median overall survival (OS) of 11.0 to 13.0 months. However, the objective response rate (ORR) is only 25.0% to 30.0%,3,4 with the second-line (L2) therapeutic options being limited. Thus, the development of novel molecules and identification of novel targets are urgently required in this setting. 5
Based on the pivotal ABC-06 study, oxaliplatin/fluorouracil/leucovorin (FOLFOX) was evaluated as an L2 treatment after progression with gemcitabine/cisplatin (GemCis); however, its efficacy was suboptimal, demonstrating only a modest 1-month OS benefit compared to that with the best supportive care (6.2 vs 5.3 months, P = .031). 6 The NIFTY trial revealed that liposomal irinotecan in combination with 5-fluorouracil (5-FU) and leucovorin (LV) achieved a median OS of 8.6 months and median progression-free survival (PFS) of 7.1 months. 7
Efforts have been made to identify potential targeted strategies for treating biliary tract cancers (BTCs), including multiple pathways such as angiogenesis and other precise targets. The FOENIX-CCA2 study showed that futibatinib achieved an ORR of 42% and median OS of 21.7 months, and the ClarIDHy study reported that ivosidenib achieved a median OS of 10.8 months. 8 However, only a small proportion of patients with specific gene alterations benefit from precision-targeting drugs. In the REACHIN study, the regorafenib group showed an improved PFS compared with that in the placebo group (3.0 vs 1.5 months, P = .004); no patients achieved an objective response, and no survival benefit was found regarding OS. 9 A phase II trial (NCT03873532) involving surufatinib reported a median PFS of 3.7 months and median OS of 6.9 months in 39 patients with BTC. 10
Immunotherapy has revolutionized the treatment of several solid tumors. However, the efficacy of immune checkpoint inhibitor (ICIs) monotherapy in unselected advanced BTC presents polarized results. The ORRs and median OS were 5.0% to 22.0% and 5.7 to 14.2 months in all-comer patients who received programmed cell death 1/ligand 1 (PD-1/L1) inhibitors.11,12 The MOUSEION-03 suggested that the combination of immunotherapy may significantly increase the chance of achieving complete response (CR) compared to that with control treatments, 13 proving a new insight for addressing this dilemma. Thus far, a pooled analysis of 2-phase Ib trials (TQB2450-Ib-05 and TQB2450-Ib-08) against the non-first-line (non-L1) regimen of anlotinib plus TQB2450 for BTC has reported an ORR of 21.2% and a median OS of 15.7 months in 66 patients, 14 which was the optimum finding obtained from the L2 treatment of patients with advanced BTC. Results of other trials demonstrated that, with lenvatinib combined with pembrolizumab, the ORR and median OS were 10.0% to 25.0% and 8.6 to 11.0 months, respectively.14,15 This regimen represents a potential chemotherapy-free option for L2 treatment of BTCs. Emerging evidence supports the potential of chemotherapy plus ICIs as the L1 initial regimen in patients pretreated with BTCs. GemCis plus durvalumab in the TOPAZ-1 study showed substantial clinical activity in patients with BTCs, achieving an ORR of 26.7% and median OS of 12.8 months.16,17 Furthermore, the BilT-03 trial evaluated the utility of adding nivolumab to L2 liposomal irinotecan, 5-FU, and LV in patients with advanced BTCs, with a median OS of 7.5 moths. 18 Although several single-arm trials have preliminarily explored the efficacy of such combinations, the associated survival benefits require further investigation.
Representative clinical trials for second-line treatment of BTC are presented in Supplementary Table 1. Based on these results, we conducted a retrospective study to compare the efficacy and safety of chemotherapy, chemotherapy plus ICIs, and tyrosine kinase inhibitor (TKIs) plus ICIs as L2 therapies for patients with advanced BTC.
Material and Methods
Study design and patients
This study was conducted at 3 centers. All consecutive patients with advanced BTCs who received L2 treatment between January 1, 2018, and April 1, 2022, were enrolled in our cohort, and their data were retrospectively collected (Figure 1). Patients were considered eligible if they had (1) pathologically proven adenocarcinoma and (2) at least one measurable tumor lesion according to the Response Evaluation Criteria in Solid Tumors (RECIST v1. 1), (3) progressed after L1 treatment with a gemcitabine-based combination regimen, and (4) received chemotherapy, chemotherapy plus ICIs, or TKIs plus ICIs for at least 2 cycles of treatment. Patients were excluded if they had (1) been treated with target therapy or immunotherapy in the L1 palliative setting, (2) received radiation therapy or best supportive care alone or other treatment strategy, or (3) been diagnosed with ampullary carcinoma.

The flowchart of the study illustrates the enrollment procedure and the treatment protocol. Center 1 stands for Zhejiang Cancer Hospital; Center 2 stands for Zhejiang Medical & Health Group Hangzhou Hospital; Center 3 stands for Wenzhou Central Hospital.
Data collection
The following clinicopathological parameters were collected: sex, age, primary tumor site, and grade of differentiation, the Eastern Cooperative Oncology Group (ECOG) performance status, surgery, hepatobiliary diseases, body mass index (BMI), systemic therapy (including the number of lines treated, regimen, start date, end date, number of cycles, and adverse reaction), tumor node metastases (TNM) stage, and distant metastatic site. Blood examination data included albumin, carbohydrateantigen 19-9 (CA19-9), carbohydrate antigen 125 (CA125), carcinoembryonic antigen (CEA) levels, and hepatitis B (HBV) infection.
Evaluation criteria
The Response Evaluation Criteria in Solid Tumors (RECIST 1.1) were used to evaluate the tumor response. The images were evaluated separately by 2 investigators. Adverse reactions were recorded according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0 (CTCAE 5.0). The TNM stage was determined according to the American Joint Committee on Cancer standards (AJCC, 8th edition).
The primary outcome was OS, defined as the time from the date of medication initiation until the date of death. Secondary endpoints were PFS (time between medication initiation and radiological disease progression or death of any cause, whichever occurred first). The ORR was defined as the proportion of patients with CR or partial response (PR) of total evaluated. The disease control rate (DCR) was defined as the proportion of patients with CR, PR, and stable disease (SD).
Statistical analysis
The median value (interquartile range) and frequency (percentage) were used to describe continuous and categorical variables, respectively. Quantitative and qualitative variables were compared using analysis of variance (or Kruskal-Wallis H-test, if appropriate) and the chi-square test (or Fisher’s exact test, if appropriate), respectively. Overall survival and PFS were estimated using the Kaplan-Meier method and compared using the log-rank test. Prognostic factors associated with early-progressive disease were evaluated using Cox regression analysis. Statistical analyses were performed using the SPSS (version 25.0) for Windows (SPSS, Chicago, IL, USA) and R software 4.2.2 (R Foundation for Statistical Computing, Vienna, Austria). Statistical significance was set at P value <.05 significant.
Results
Baseline characteristics and treatment
A total of 98 patients with advanced BTCs were eventually eligible: the chemotherapy group (cohort A, n = 40), the chemotherapy plus ICIs group (cohort B, n = 27), and the TKIs plus ICIs group (cohort C, n = 31), with a male/female ratio of 0.8 (18/22), 1.1 (14/13), and 0.6 (12/19), respectively. The median age was 58.5 years (interquartile range [IQR]: 46.5-67) in cohort A, 60 (range: 51-66) years in cohort B, and 58 (range: 53-67) years in cohort C. In the etiology surveillance, 15 patients (37.5%, cohort A), 7 patients (25.9%, cohort B), and 13 patients (41.9%, cohort C) had hepatitis B infection. Most patients showed good physical performance in all cohorts, with an ECOG score of 0 to 1. There were 38 patients (95%) in cohort A, 12 (85.7%) in cohort B, and 25 (92.6%) in cohort C with distant metastases. In cohorts A, B, and C, 21 (52.5%), 15 (55.6%), and 16 (51.6%) patients underwent primary tumor resection, respectively. The patients in the 3 cohorts were well balanced at baseline. Baseline patient demographics and clinical characteristics are summarized in Table 1.
Baseline demographics.

Abbreviations: BMI, body mass index; ECOG, Eastern Cooperative Oncology Group.
Data were presented as n (%) or median with IQR as appropriate.
Treatment
In cohort A, the median duration of chemotherapy injected was 4.0 cycles (95% CI: 3.2-4.8) and 12.1 weeks, and all patients discontinued treatment owing to tumor progression. Various chemotherapy regimens were used and classified into categories: “F- alone” (6, 15%): including S-1 and capecitabine; “F-P” (25, 62.5%): all regimens combining cisplatin/oxaliplatin and S-1/ capecitabine/5-FU; “F-I” (2, 5%): all regimens combining 5-FU and irinotecan; “F-A” (7, 17.5%): all regimens combining capecitabine and albumin paclitaxel.
The median administered cycles of ICIs were 4.6 cycles (95% CI: 3.4-5.7) for cohort B, 5.5 cycles (95% CI: 4.1-6.9) for cohort C, and all patients received at least 2 shots. The usage of ICIs included nivolumab (0, 0%; 1, 3.2%), durvalumab (1, 3.7%; 0, 0%), sintilimab (8, 29.6%; 12, 38.7%), toripalimab (5, 18.5%; 6, 19.4%), carrelizumab (6, 22.2%; 8, 25.8%), pembrolizumab (2, 7.4%; 0, 0%), cadonilimab (0, 0%; 1,3.2%), and tislelizumab (5, 18.5%; 3, 9.7%) in cohort B and cohort C, respectively. The TKI included lenvatinib (10, 33.4%), anlotinib (15, 50.0%), apatinib (4, 13.3%), and regorafenib (1, 3.3%) in cohort C. Twenty-one and 25 patients discontinued treatment owing to tumor progression, respectively. Cohort B was the same as cohort C; one patient discontinued treatment due to intolerable adverse reactions, and 5 patients were continued under medication before the cutoff date.
During the study period, patients received chemotherapy, chemotherapy plus ICIs, or TKIs plus ICIs as L2 treatment, which was chosen based on their condition and chronological background. Dose adjustments for toxicity and patient PS were performed at the discretion of the investigator. A more detailed description of the L2 therapy regimens is provided in Supplementary Table 2.
Efficacy
All patients were available for efficacy assessment and experienced a detectable objective response according to the Response in RECIST 1.1. Figure 2 shows the maximum change in the sum of measurable lesions and the best overall response. The ORR following cohort A, cohort B, and cohort C treatment as the L2 systemic therapy in advanced BTCs was 7.5% (95% CI: 1.6%-20.4%), 22.2% (95% CI: 8.6%-42.3%), and 19.4% (95% CI: 7.5%-37.5%), respectively, with 0 (0%), 1 (3.7%), and 0 (0%) CR, and 3 (7.5%), 5 (18.5%), and 6 (19.2%) PRs. The DCR in cohort A, cohort B, and cohort C was 47.5% (95% CI: 31.5%-63.9%), 77.8% (95% CI: 57.7%-91.4%), and 77.4% (95% CI: 58.9%-90.4%), respectively. Table 2 displays detailed information on the therapeutic responses.

The efficacy of each group: (A) the maximum of change of the sum of the target lesions and (B) best overall response per RECIST1.1 according to the treatment regimen.
Treatment summary and therapeutic responds.

Abbreviations: DCR, disease control rate; ORR, objective response rate.
Compared with cohort A, patients in cohort B and C with the ICIs recombination regimen presented the prolonged PFS (2.6 months, 95% CI: 1.7-4.2 vs 4.3 months, 95% CI: 2.9-8.4 vs 5.1 months, 95% CI: 4.0-8.5; P = .0056) (Figure 3A), but not OS (7.8 months, 95% CI: 6.3-12 vs 10.9 months, 95% CI: 9.7-12.1 vs 10.1 months, 95% CI: 1.9-18.4; P = .11) (Figure 3B). The PFS rates at 12 and 24 weeks were 45% and 17.5% in cohort A, 59.3% and 25.9% in cohort B, and 74.2% and 32.3% in cohort C. The OS rates at 1 year were 25%, 25.9%, and 29.0%, respectively.

Survival curves between groups: (A) the Kaplan-Meier method estimated the PFS length in all cohorts and found a marginal superiority for PFS in the immunotherapy combination group. The PFS rate on 12 weeks were 45%, 59.3%, and 74.2%, and 24 weeks were 17.5%, 25.9%, and 32.3%, respectively, with a median PFS of 2.6, 4.3, and 5.1 months (P = .0056); (B) the Kaplan-Meier method estimated the OS length in all cohorts and found no statistical differences in the OS. The OS rate on 1 year were 25%, 25.9%, and 29.0%, respectively, with a median OS of 7.8, 10.9, and 10.1 months (P = .111).
Multivariate analysis included sex, age, ECOG status, tumor site, osseous metastasis, surgery, and second-line treatment. The results demonstrated that second-line treatment (P = .008) was an independent prognostic factor for PFS (Figure 4).
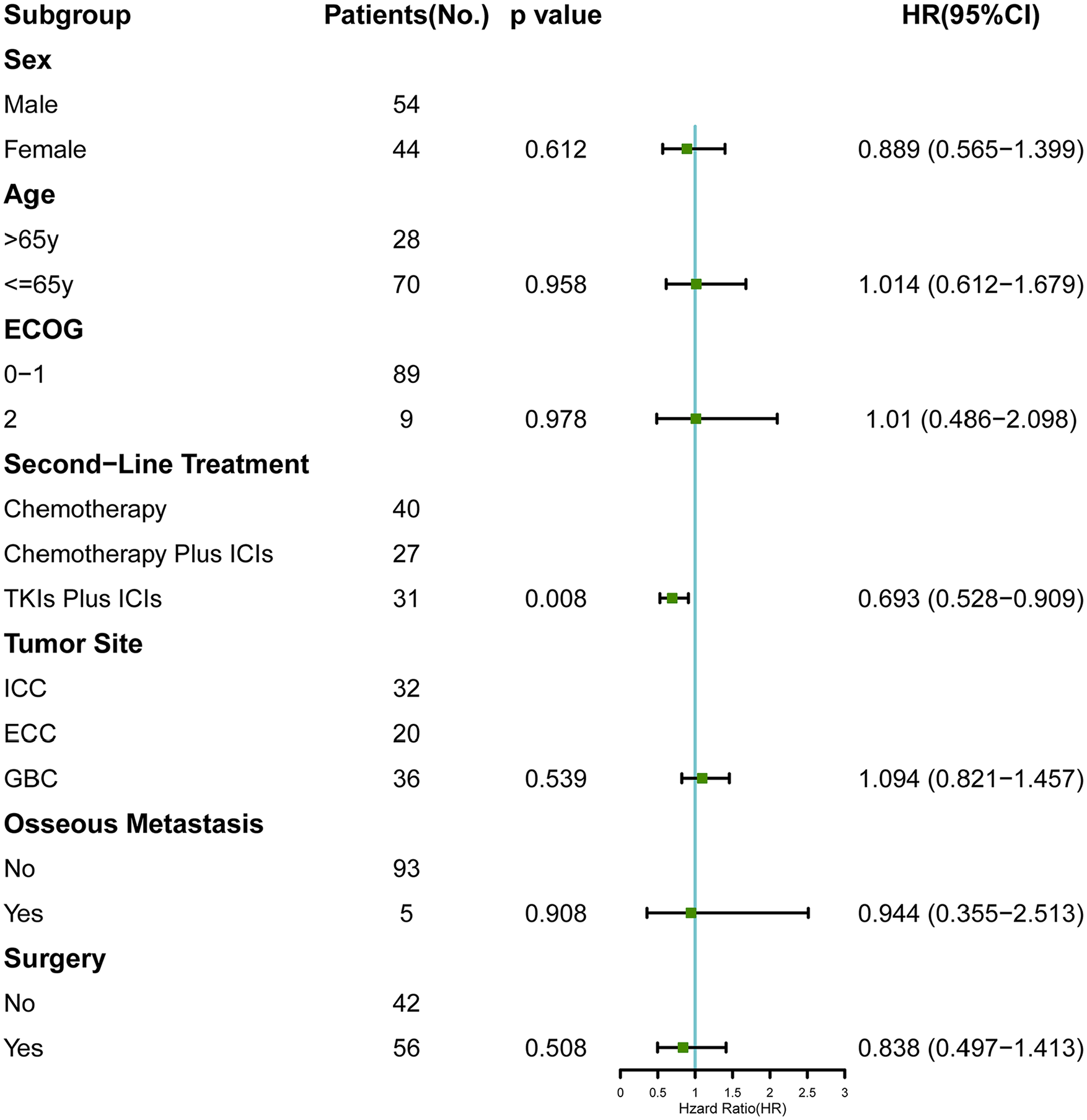
Multivariate analysis of PFS. Analyses showed that PFS benefit was observed according to second-line treatment.
Tolerability and safety
In total, 8 patients (20%), 10 patients (37.0%), and 13 patients (41.9%) reported and experienced ⩾grade 3 adverse reactions in cohort A, cohort B, and cohort C. Same as cohort A, cohort B had a relatively high proportion of decreased appetite (10%, 11.1%) and bone marrow suppression (10%, 7.4%). In addition, patients in cohort B experienced nausea (7.4%), fatigue (7.4%), and elevated aspartate aminotransferase levels (7.4%). The most common ⩾grade 3 adverse reactions were decreased appetite (12.9%), thrombocytopenia (9.7%), and immune-related thyroiditis (9.7%) for cohort C. Detailed information of adverse reactions is shown in Table 3. Most patients were advised to continue taking the medication at a reduced dosage or to receive medical support. Two patients withdrew from treatment due to intolerable adverse reactions, including one with grade 4 palmar plantar erythrodysesthesia syndrome in cohort B and one with grade 4 increased thrombocytopenia in cohort C. Patients with immune-related adverse reactions were treated with low-dose corticosteroids and recovered from the condition.
Adverse events ranking.

Discussion
Although limited, the ABC-06 trial was the first randomized phase III clinical trial to demonstrate the benefits of FOLFOX in the L2 systemic treatment regimen for BTCs. However, there is no robust evidence for an optimal therapy for L2. To our knowledge, this is the first retrospective study to evaluate the additional role of ICIs with chemotherapy or TKIs compared with chemotherapy alone in patients with advanced BTC who have been previously treated with chemotherapy. Although some clinicians have already used ICIs with chemotherapy or TKIs in this setting, they do so without knowing the magnitude of their benefits.
Owing to its retrospective nature, the inclusion criteria were not strictly defined in this study. Currently, the gemcitabine-platinum combination is considered the standard frontline treatment. In our study, approximately 70% of patients received GemCis or GS as L1 treatment, whereas the remainder received other gemcitabine-based combinations. Our results reflected routine practices and highlighted the preferential use of GemCis and GS in China. Otherwise, those patients with ECOG 2 or less who could tolerate oral intake were also eligible, which was different from that followed in other clinical trials.
The efficacy of chemotherapy as an L2 treatment for patients with BTCs in this study was similar to that reported in previous studies, with an ORR of 7.5%, median PFS of 2.6 months and median OS of 7.8 months. Two systematic reviews of 761 and 1391 patients showed that the response rate for the use of second-line therapy was only 7.7%, with a median PFS of 2.6-3.2 months and median OS of 6.5-7.2 months. 19 Of note, fluorouracil alone is not certain to have been inferior to doublet chemotherapy as an L2 therapy (mOS:6.5 month 95%CI 5.43—NA vs 95%CI 5.97—12.7, P = 0.46), and previously several lines of evidence also indicating this point.20,21
Our study results showed that chemotherapy plus ICIs and TKIs plus an ICIs regimen as L2 treatments demonstrated active and promising trends in advanced BTC. Compared with chemotherapy, this combination regimen prolonged the survival duration of L2 systemic therapies and significantly improved the median PFS. Currently, several ongoing clinical trials on the TKIs plus ICIs combination regimen as L2 therapy in advanced BTCs have remained in a single-arm, phase II, small-sample stage. Supplementary Table 2 summarizes ongoing trials registered with ClinicalTrials.gov. Emerging evidence supports the potential of chemotherapy plus ICIs as the L1 initial regimen in patients pretreated with BTCs. GemCis plus durvalumab in the TOPAZ-1 study showed substantial clinical activity in patients with BTCs, achieving an ORR of 26.7% and a median OS of 12.8 months. In immunotherapy exploration, there are large differences in the ORR and OS in unselected populations. Our study shows an “trailing effect.” In cohort B, one patient discontinued pembrolizumab after 12 cycles of maintenance therapy, whereas the anticancer treatment effect persisted, with a PFS of 29.4 months and an OS of 39.8 months. In addition, our study showed that the differences in efficacy between TKIs plus ICIs and chemotherapy plus ICIs were subtle, without statistical significance, in terms of tumor response and survival. Although the efficacy of TKIs plus ICIs was associated with modestly prolonged PFS, it failed to translate into a survival benefit.
In this study, the incidences of grade 3 or higher adverse reactions were 20%, 37%, and 41.9%, respectively. In addition, one patient (3.7%; 3.2%) each in cohorts B and C discontinued treatment owing to adverse reactions. A systematic review of 32 studies, which included 2,324 participants, evaluated the safety of ICIs plus antiangiogenic agents in malignancies and reported an overall adverse reaction of approximately 60%. 22 However, the combination of TKIs and ICIs in this study exhibited a favorable safety profile, consistent with the findings of a previous study on lenvatinib and pembrolizumab. 23 For anlotinib plus a PD-1/L1 inhibitor and chemotherapy plus durvalumab, the incidences of grade 3 or higher was 25.8% and 62.7%, respectively.14,16 The set up of the “Three None Wards (No pain, No vomiting, No thrombus)” in the research centers were contributed to reducing the incidence of adverse reactions, which provide better care and optimal support for patients. Although it seems chemotherapy plus ICIs was associated with better safety, the grade 3 or higher adverse reactions in each of the group were manageable. Therefore, if ongoing studies continue to identify promising biomarkers, immunotherapy regimens may become a new standard of care for L2 therapy in BTCs. In particular, senile patients with inferior performance status can obtain new opportunities.
Knowledge gaps make it difficult to formulate individualized immunotherapies in BTCs patients with BTC. At present, in the absence of clear and reliable biomarkers to predict the efficacy of ICI in the population, it is necessary to conduct phase III clinical trials based on biomarkers to screen patients most likely to benefit from immunotherapy. Remarkably, although immunotherapy benefits patients with BTC, it faces 2 challenges: treatment resistance and hyperprogression, both of which affect the efficacy of immunotherapy and accelerate tumor progression. The high heterogeneity and frequency of gene mutations in BTC, combined with its complex and diverse tumor microenvironment, are the main internal and external reasons for immunotherapy resistance and hyperprogression. The identification of tumor gene characteristics and immune microenvironment subtypes is helpful in judging the efficacy of immunotherapy. Combination immunotherapy is a feasible method to overcome the challenges associated with immunotherapy resistance and hyperprogression. In addition, exploring peripheral blood markers and in vitro models can predict immunotherapy resistance and hyperprogression and adjust the treatment regimen accordingly. Answers to these questions may benefit patients with metastatic and locally advanced disease, leading to greater possibilities of curing this challenging disease.
However, immunological and biochemical markers could not be obtained from all patients. This study showed that a reasonable combination of immunotherapies in an unselected population could lead to a survival benefit. Therefore, further discussions should be conducted on the application of combined immunotherapy in clinical practice. In clinical studies, we observed a bias between the overall treatment response of real-world patients and data from clinical studies, which may be related to the patient status, suggesting that hierarchical management of patients is important; For patients with tolerable liver function and good general condition, the “Immune combination” regimen should be recommended, while conversely, the efficacy and safety should be balanced, and a milder routine chemotherapy regimen should be recommended. In addition, subgroup analyses based on other studies have shown that patients with gallbladder cancer (GBC) appear to benefit more from targeted combination immunotherapy than those with intrahepatic cholangiocarcinoma (ICC) or extrahepatic cholangiocarcinoma (ECC). We need to recognize that, while immune-based regimens can improve patient outcomes, if patients experience adverse reactions during treatment, it is a future direction to explore how to decrease the dosage and adjust the scheme to ensure a curative effect.
This study had several limitations. First, it was retrospectively designed with a relatively small sample size, which might have contributed to sample bias. Second, owing to the lack of biomarker information in this study, it was not possible to explore the dominant immunotherapeutic population. Finally, this study included patients with heterogeneous BTC with various disease statuses and therapeutic regimens. However, this heterogeneity represents the clinical practice of BTC treatment and may be extrapolated to other cohorts. Although the data were collected retrospectively, our study population was well annotated with a low rate of missing data, thereby highlighting their robustness.
Conclusions
This multicenter retrospective study demonstrates that an ICI combination strategy beyond L1 systemic chemotherapy plays a positive role in advanced BTCs. Taking into consideration the manageable toxicity profile and numerically higher ORR and better PFS, ICI combination therapy could be considered a candidate for L2 trials and turn into a competitive L2 treatment for advanced BTCs in clinical practice.
Supplemental Material
sj-docx-1-onc-10.1177_11795549241272469 – Supplemental material for An Active Trend of Immunotherapy Combination Regimen as Second-Line Therapy Towards Advanced Biliary Tract Cancer
Supplemental material, sj-docx-1-onc-10.1177_11795549241272469 for An Active Trend of Immunotherapy Combination Regimen as Second-Line Therapy Towards Advanced Biliary Tract Cancer by Haimin Weng, Pengfei Zeng, Yuemiao Chen, Qi Xu and Jieer Ying in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-2-onc-10.1177_11795549241272469 – Supplemental material for An Active Trend of Immunotherapy Combination Regimen as Second-Line Therapy Towards Advanced Biliary Tract Cancer
Supplemental material, sj-docx-2-onc-10.1177_11795549241272469 for An Active Trend of Immunotherapy Combination Regimen as Second-Line Therapy Towards Advanced Biliary Tract Cancer by Haimin Weng, Pengfei Zeng, Yuemiao Chen, Qi Xu and Jieer Ying in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-3-onc-10.1177_11795549241272469 – Supplemental material for An Active Trend of Immunotherapy Combination Regimen as Second-Line Therapy Towards Advanced Biliary Tract Cancer
Supplemental material, sj-docx-3-onc-10.1177_11795549241272469 for An Active Trend of Immunotherapy Combination Regimen as Second-Line Therapy Towards Advanced Biliary Tract Cancer by Haimin Weng, Pengfei Zeng, Yuemiao Chen, Qi Xu and Jieer Ying in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
The authors thank all patients and their families for their participation.
Author Contributions
Substantial contributions to the study design: JY and QX. Study data acquisition: HW, PZ, and YC. Analysis of the study data: HW and PZ. Interpretation of study data: HW, PZ, JY, and QX. HW, PZ, and YC wrote or contributed to writing the manuscript. Revised content: JY and QX. This article has been read and approved by all authors. All the authors have met the authorship requirements.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Natural Science Fund of Zhejiang Province (grant no. LTGY23H160010), Medical and Health Science and Technology Program of Zhejiang Province (grant no. 2021KY087), and Key Laboratory of Prevention, Diagnosis, and Therapy of Upper Gastrointestinal Cancer of Zhejiang Province (grant no. 2022E10021).
Availability of Data and Materials
The data collected and analyzed in this study can be accessed by the corresponding author upon reasonable request.
Consent for Publication
Not applicable
Ethical Consideration and Consent
Approval of the Research Protocol by an Institutional Review Board: Studies involving human participants were reviewed and approved by the Institutional Ethics Committees of Zhejiang Cancer Hospital, Zhejiang Medical & Health Group Hangzhou Hospital, and Wenzhou Central Hospital (IRB-2023-491). The requirement for informed consent was waived by the IRB because the study was retrospective.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
