Abstract
Background:
Monoclonal antibodies that target the PD-1 receptor are emerging as promising therapeutic candidates for the treatment of biliary tract cancers (BTCs). The purpose of the current study was to assess the combination of the camrelizumab with chemotherapy as a first-line treatment for metastatic BTCs.
Methods:
We conducted a prospective single-arm pilot study of PD-1 antibody (camrelizumab 3 mg/kg d1, Q2 W or Q3 W) combined with different chemotherapy regimens as first-line treatment for BTCs. Efficacy endpoints were objective response rate (ORR), disease control rate (DCR), progression free survival (PFS), and overall survival (OS). Treatment-related adverse events (TRAEs) were also evaluated.
Results:
Fourteen patients with histologically confirmed BTCs were evaluated. The ORR was 14.3% (95% CI: 1.8 to 42.8) and the DCR was 64.3% (95%CI: 41.7 to 86.9). The median PFS was 6.5 months (95% CI: 3.8 to 9.2), and the 6- and 12-month PFS rates were 61.6% and 12.3%, respectively. The median OS was 9.9 months (95% CI: 7.6 to 12.2), and the 6-and 12-month OS rates were 74.5% and 26.6%, respectively. All patients displayed at least 1 TRAE., and Grade 3 or 4 TRAEs occurred in 6 (42.86%) patients.
Conclusions:
Camrelizumab combined with chemotherapy as first-line treatment for metastatic BTCs demonstrated acceptable safety and efficacy in our pilot study. These findings warrant prospective controlled clinical trials comparing combinations of camrelizumab and chemotherapy to standard regimens.
Introduction
Biliary tract cancers (BTCs) are being reported with increasing incidence and mortality rates. 1 Only ∼20% of patients are diagnosed with early-stage disease amenable to curative surgery. Furthermore, the efficacy of surgical resection is limited by a high relapse rate of approximately 60%-70%. 2 Cisplatin combined with gemcitabine has been the standard first-line chemotherapeutic regimen for patients with inoperable disease for many years. 3 However, because treatment failures are common, the 5-year survival rates of BTC patients are only 5%-15%. 4 Thus, new therapeutic strategies are essential to improve clinical outcomes.
The development of immune checkpoint inhibitors (ICIs) constitutes a major breakthrough in cancer immunotherapy. ICIs target the immunologic escape mechanisms of malignant cells by reversing T cell inhibition, thereby reducing immune tolerance and improving immune recognition and eradication of tumor cells. 5 Programmed death-1(PD-1) is a protein receptor expressed on the surface of T cells that plays an important role in immunoregulation by downregulating immune responses. PD-1 inhibitors activate innate immune responses against tumor cells by blocking the interaction of PD-1 with its ligand-PD-L1, thereby reversing T cell exhaustion, promoting T cells expansion, and restoring effective antitumor responses. 6 Anti-PD-1 therapy has revolutionized the treatment of many malignancies. The phase III KEYNOTE-189 trial 7 led to the U.S. Food and Drug Administration (FDA) approval of the combination of pembrolizumab (Keytruda®, an anti-PD-1 antibody) and chemotherapy for the treatment of metastatic non-small cell lung cancer without EGFR or ALK alterations. Interim results of the KEYNOTE-158 trial 8 indicated that pembrolizumab exhibited durable antitumor activity and acceptable safety in patients with advanced cervical cancer. On the basis of these results, the FDA granted accelerated approval of pembrolizumab for the treatment of patients with advanced PD-L1-positive cervical cancer with disease progression during or after chemotherapy. In addition, anti-PD-1 treatment has conferred long-term tumor remission and survival benefits in advanced BTCs. In the multicohort KEYNOTE-028 trial, a phase Ib study of pembrolizumab (10mg/kg, Q2 W) in patients with advanced solid tumors, 24 BTC patients with PD-L1 expression experienced an objective response rate (ORR) and disease control rate (DCR) of 17% and 34%, respectively. 9 These results suggest the potential clinical utility of anti-PD-1 antibodies in BTC therapy.
Camrelizumab is a high-affinity, fully humanized, IgG4-κ monoclonal antibody directed against PD-1. Its antitumor activity and safety have been evaluated with favorable results in patients with recurrent or metastatic nasopharyngeal and esophageal carcinoma. 10,11 In our current study, we investigated the safety and efficacy of camrelizumab combined with chemotherapy in the treatment of unresectable BTCs.
Patients and Methods
Patients
We conducted a prospective single-arm pilot study from July 2018 to December 2019. Inclusion criteria were: (1) histologic or cytologic diagnosis of a BTC (intrahepatic cholangiocarcinoma [iCCA], extrahepatic cholangiocarcinoma [eCCA], or gallbladder carcinoma [GBC]; (2) no prior immunotherapy or systemic treatment; (3) at least 1 measurable lesion as defined by the Response Evaluation Criteria in Solid Tumor (RECIST version 1.1) criteria; and (4) an Eastern Tumor Collaborative Group Physical Status score of 0-1. Written informed consent for immunotherapy as participation on the trial was obtained from all patients before starting the combination therapy. Patient data were collected from medical records. We had access to patient identifying information during and after data collection. This study was approved by the ethical committee of our hospital (2018 Medical Review No .050).
Treatment Procedures
Patients were given camrelizumab in combination with different chemotherapeutic regimens until the onset disease progression (PD), intolerable toxicity, or withdrawal of consent. Camrelizumab was given intravenously 3mg/kg every 2 weeks or every 3 weeks on day 1 of chemotherapy regimens. Regimens included FOLFOX-4 (oxaliplatin 85 mg/m2 IV on day 1; levoleucovorin calcium 200 mg/m2 over 2 h on day 1; fluorourea 400 mg/m2 IV on day 1 followed by 600 mg/m2 civ over 22 h for 2 days, every 2 weeks), GEMOX (Gemcitabine 800 mg/m2 IV on day 1; oxaliplatin 85 mg/m2 IV on day 1, every 2 weeks), TS (paclitaxel liposome 175 mg/m2 IV on day 1, S-1 60 mg twice daily PO for 14 days, every 3 weeks) or S-1 monotherapy (60 mg twice daily PO for 14 days, every 3 weeks). Individualized treatment regimens were designed by a multidisciplinary team.
Efficacy Assessments and Safety Monitoring
We assessed tumors using computed tomography (CT) or magnetic resonance imaging (MRI) at baseline and every 8 weeks thereafter. Tumor response was determined through radiological review and judged according to RECIST guidelines. The primary efficacy endpoints were objective response rate (ORR) (the percentage of patients with a confirmed complete/partial response (CR/PR) as per RECIST version 1.1); disease control rate (DCR) (the proportion of patients who have had a complete or partial response or stable disease (SD) per RECIST version 1.1); progression free survival (PFS) (time from the initial treatment to first documentation of disease progression or death); and overall survival (OS) (time from initial treatment to death).
Safety was determined by the frequency and severity of adverse events (AEs). Assessments included monitoring of vital signs, symptoms, and hematological parameters. AEs and complications during treatment were closely observed and recorded. All toxicities or adverse effects were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (v4.0).
Statistical Analysis
Eligible patients who received at least 1 dose of camrelizumab combined with chemotherapy were included in the efficacy and safety analyses. Baseline characteristics and efficacy data were presented as median (range) or number of patients (percentage). The Kaplan-Meier method was used to estimate median OS and PFS. Two-sided 95% confidence intervals (CIs) were calculated by the Brookmeyer-Crowley method. Safety results were displayed in the form of a number and a percentage of patients with AEs. P < 0.05 was considered statistically significant. All statistical analyses were conducted using SPSS 22.0 software (IBM, SPSS, Chicago, IL, USA).
Results
Patients Characteristics
Between July 5, 2018, and November 30, 2019, 24 patients were screened, and 14 patients were considered eligible. Clinicopathological characteristics of these 14 patients are listed in Table 1. The median age was 50.5 years (range, 36-70). There were 10 male and 4 female patients. iCCA was the most common malignancy (n = 9, 64.28%), followed by eCCA (n = 3, 21.43%) and GBC (n = 2, 14.28%). Nine patients had single metastases, and 5 had multiple metastases. The liver was the most common metastatic site (n = 7, 50%). Five patients received palliative resection before receiving camrelizumab and chemotherapy combination treatment.
Clinicpathological Characteristics.

GEMOX was the most common chemotherapy regimen (9 of 14 patients). Two patients received FOLFOX-4 or TS, respectively, and one patient received S-1. The median treatment cycle was 5.5 (range, 1-11). At data cutoff on November 30, 2019, median follow-up was 1.9 months (range, 0.03 to 14.5 months). Three iCCA patients and 2 eCCA patients were still undergoing treatment (Figure 1).

Flow diagram of the study.
Efficacy
The ORR and DCR were 14.3% (95% CI: 1.8 to 42.8) and 64.3% (95% CI: 41.7 to 86.9), respectively. No patient achieved CR, 2 (14.29%) exhibited PR, 7 (50%) had SD, and 5 (35.71%) experienced PD (Table 2). The waterfall plot for the maximal change of the target lesions in these 14 patients is presented in Figure 2.
Efficacy Evaluation in the Research According to RECIST v 1.1.

Abbreviations: RECIST, Response Evaluation Criteria in Solid Tumors; CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease; ORR, objective response rate; DCR, disease control rate; CI, confidence interval.

Waterfall plots of the changes in the size of target lesions.
At data cutoff, 5 (35.71%) PFS events and 9 (42.86%) deaths had occurred. The median PFS was 6.5 months (95% CI: 3.8-9.2), and the median OS was 9.9 months (95% CI: 7.6 to 12.2). The 6- and 12-month PFS rates were 61.6% and 12.3%, respectively, whereas the 6- and 12-months OS rates were 74.5% and 26.6%, respectively (Figure 3A and B). Nine patients who received GEMOX experienced a median PFS of 8.3 months (95% CI: 4.3-12.2), and median OS was similar to the entire group. The 6- and 12-month PFS and OS rates of the GEMOX group were relatively higher than entire group (Figure 3C and D). The ORR and DCR in GEMOX group were 22.2% and 52.2%, respectively.
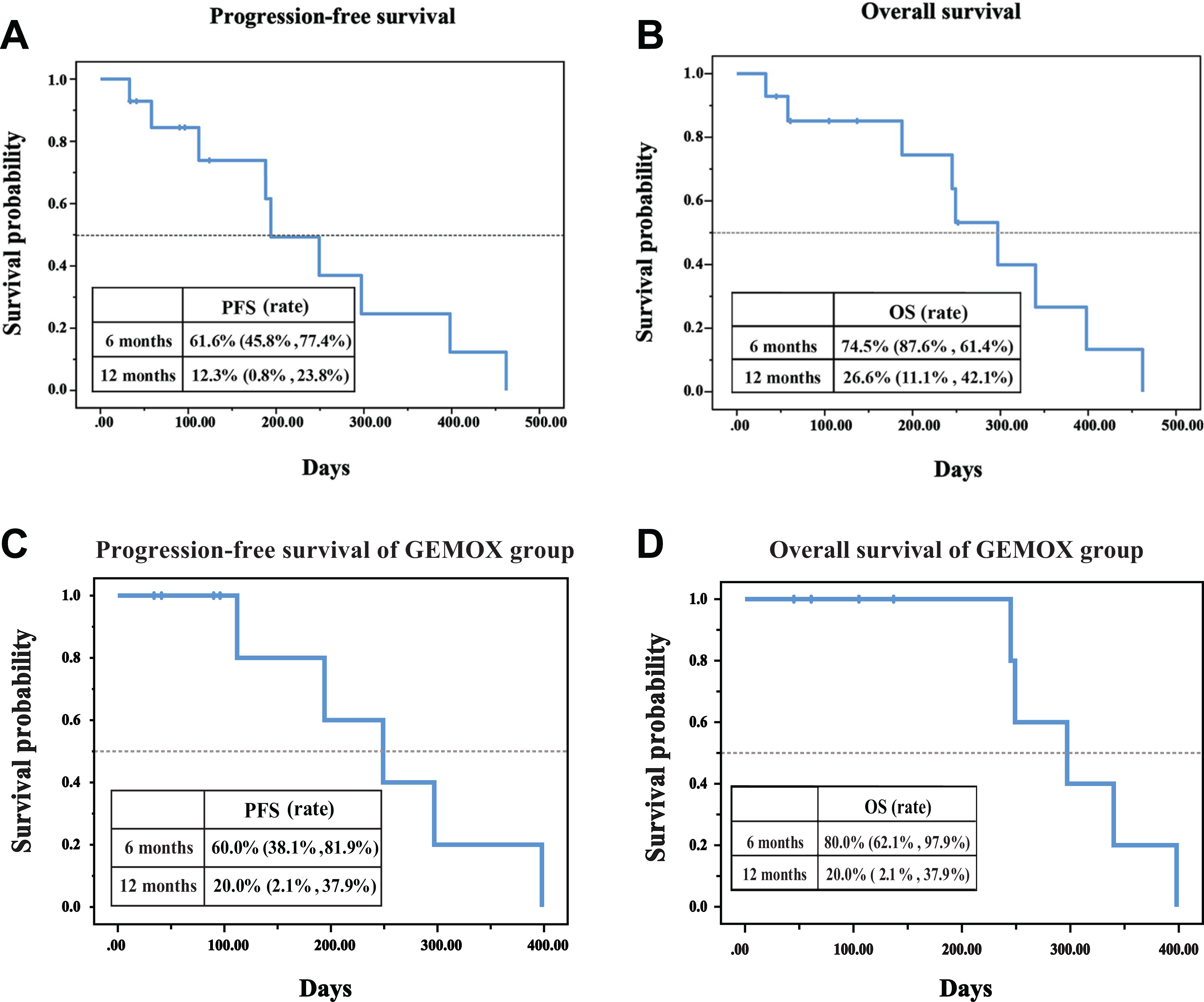
A, Kaplan-Meier estimates of progression free survival. B, Kaplan-Meier estimates of overall survival. C, Progression free survival analysis of GEMOX group. D, Overall survival analysis of GEMOX group.
Median PFS and OS did not differ according to primary tumor (iCCA vs eCCA vs GBC), sex (male vs female), and chemotherapy regimen (FOLFOX-4 vs GEMOX vs TS vs S-1) (P > 0.05 for all). Of note, we found that patients with single metastases and those who received >4 cycles of combination treatment tended to have longer median PFS (P = 0.026 and P = 0.006, respectively) and OS (P = 0.007 and P = 0.002, respectively) (Figure 4 and Figure 5).

Subgroup analysis of mPFS. A, mPFS by gender. B, mPFS by primary tumor location. C, mPFS by chemotherapy regimen. D, mPFS by treatment cycles. E, mPFS of patients by number of metastatic sites.

Subgroup analysis of mOS. A, mOS by gender. B, mOS by primary tumor location. C, mOS by chemotherapy regimen. D, mOS by treatment cycles. E, mOS by number of metastatic sites.
Safety
Treatment-related adverse events (TRAEs) occurred in all patients. The most frequent TRAE was vomiting (n = 7, 50%), followed by fever (n = 6, 42.86%), nausea (n = 5, 35.71%), fatigue (n = 4, 28.57%), and anorexia (n = 3, 21.43%). Grade 3/4 TRAEs occurred in 6 (42.86%) patients. These included 4 vomiting episodes, 3 neutropenic episodes, and single occurrences of fever, anorexia, drug allergy, hepatitis, leukopenia, and elevated aspartic aminotransferase. Camrelizumab-related AEs of any grade occurred in 5 (35.71%) patients, including 2 hemangiomas and 2 episodes of colitis, and single episodes of hypothyroidism and hepatitis. All symptoms responded to supportive care. No drug-related deaths occurred (Table 3).
Treatment Related Adverse Events (TRAEs).

Discussion
Biliary tract cancers (BTCs) are a heterogeneous group of hepatobiliary malignancies, comprised primarily of iCCA, eCCA, and GBC). Although BTCs account for only 0.7% of malignant tumors and 3% of gastrointestinal malignancies in adults, their incidence and mortality are steadily increasing. 12 -14 In addition to the combination of gemcitabine and cisplatin as the standard first line chemotherapy schedule for patients with inoperable BTCs, fluoropyrimdine-based or gemcitabine-based chemotherapeutic regimens are also commonly used. 15 However, the efficacy of these treatments is still quite limited. 4 To improve clinical outcomes of BTC, novel therapeutic approaches that include the development of new agents 16 ; liver-directed therapies 17 -19 ; and the targeting of fibroblast growth factor receptor fusion rearrangements and isocitrate dehydrogenase-1 and -2 mutations 20 are being evaluated. However, none of these efforts have yet altered the current standard of care.
Immunotherapy that targets the PD-1/ PD-L1signaling pathway has emerged as promising treatment for many malignancies. Pembrolizumab (Keytruda ®), nivolumab (Opdivo ®) and atezolizumab (Tecentriq ®) antagonize PD1 and PD-L1 binding, and have shown encouraging results in melanoma, non-small-cell lung, prostate, renal-cell, and colorectal cancers. 21,22 In the KEYNOTE 189 trial, pembrolizumab combined with chemotherapy dramatically prolonged OS and PFS in advanced non-small-cell lung cancer. 7 Results from a phase Ib/II study revealed that the combination of pembrolizumab with gemcitabine and nab-paclitaxel led to TelCentris a 100% DCR and tolerable toxicity in pancreatic cancer patients. 23 Several clinical trials, such as the KEYNOTE 028 and KEYNOTE 158 trials and a phase II trial of nivolumab have indicated the potential clinical utility of anti-PD-1/PD-L1 immunotherapy in the treatment of BTCs. 9,24,25
We evaluated the safety and efficacy of camrelizumab combined with chemotherapy in metastatic BTCs. Our findings (median OS and PFS of 9.9 months [95% CI: 7.6 to 12.2)]and 6.5 months [95% CI: 3.8-9.2] respectively, and DCR of 64.3%) contrast to the results of a Phase III trial of cisplatin–gemcitabine therapy for BTCs that resulted in a median PFS of 8.0 months (95% CI: 6.6-8.6) and a tumor control rate of 81.4%. 3 However, the patient characteristics of the 2 studies were substantially different. The Phase III trial enrolled patients with locally advanced disease (23.8%) in addition to patients with metastases, whereas all of our patients had metastatic disease. Because BTC is an extremely heterogeneous disease, we conclude that varying baseline patient characteristics contributed to the discordant results of these 2 studies.
The JCOG1113 study 26 compared the efficacy and AEs of gemcitabine plus S-1 (GS) and gemcitabine plus cisplatin (GC) in recurrent or unresectable BTCs. Patient baseline characteristics were more similar to those in our study. The median OS of GC recipients was 13.4 months (95% CI, 12.4-15.5), whereas that of GS recipients was 15.1 months (95% CI, 12.2-16.4); the median PFS was 5.8 months (95% CI, 5.5-7.0) with GC and 6.8 months (95% CI, 5.4-8.0) with GS; the response rates were 32.4% (48/148) in the GC arm and 29.8% (42/141) in the GS arm. In a phase II 27 single-arm study of camrelizumab plus FOLFOX4 or GEMOX for advanced HCC or BTC, the ORR and DCR of 43 evaluable BTC patients were 7% and 67.4%, respectively, in contrast to 22.2% and 52.2%, respectively, in our GEMOX recipients. The differences in response rates may have no clinical significance because of the diversity of patient populations and characteristics in the aforementioned studies. Over-all, these data suggest that the addition of anti-PD-1 receptor immunotherapy in our study resulted in acceptable efficacy based on previous experience with conventional first-line chemotherapy.
The camrelizumab/chemotherapy combination in our study did not cause any previously unreported toxicity, and did not generate a safety signal compared to previous experience with standard chemotherapy. Adverse events were tolerable and typically responded to symptomatic treatment.
Our multidisciplinary team selected chemotherapeutic regimens that were individualized for each patient. Several studies have demonstrated a promising survival benefit and a mild toxicity profile of FOLFOX4 in BTCs. 28,29 Gemcitabine-cisplatin and GEMOX are the most commonly used regimens in BTCs. 30 A recent match-pair analysis 31 found no clinically relevant or statistically significant differences in response and clinical benefit rates of the two regimens. However, GEMOX caused a lower incidence of hematologic toxicities and was also easier to administer to patients with borderline renal and cardiac functions.Another standard treatment of GS also showed considerable survival benefit in several study. 26 While taking the patients willingness as well as the fearness of poor oral medication management into consideration, we choose GEMOX regimen as combined chemoterapy for 9 patients. Kim et al 32 reported sufficient antitumor activity of paclitaxel liposome combined with gemcitabine. However, in consideration of hematologic toxicity and our patients’ relatively low performance status, we selected S-1 as either monotherapy or in combination with paclitaxel liposome for 3 patients, because S-1 has shown promising efficacy and mild toxicity. 33,34 The chemotherapy regimens that we applied not only correspond with standard guidelines, but were also selected according to each patient’s physical condition.
Our study has several limitations. First, this is a prospective, single-arm pilot study, which might limit the interpretation of the results. Second, the small sample size and heterogeneous patient population may contribute to selection and recall biases. Third, we did not determine PD-L1 expression and tumor mutational burden (TMB) statuses. These 2 biomarkers may inform patient selection for cancer immunotherapy. 35 -37 The absence of PD-L1 expression and TMB status data precluded the identification of patients most likely to benefit from this regimen. Although these limitations somewhat weaken the reliability and validity of our conclusions, the ‘real-world’ data are still beneficial for the design of future studies.
In conclusion, we report an early pilot study to investigate the safety and efficacy of the combination of camrelizumab and first-line chemotherapy in BTCs. The addition of camrelizumab yielded an acceptable tumor control rate and survival benefit in patients with inoperable BTCs. In addition, there were no serious TRAEs. Our results suggest that the novel therapeutic strategy of combing an anti-PD-1 antibody with traditional chemotherapy may have clinical utility to improve outcomes of BTCs. The acceptable safety and efficacy suggested by our pilot study support the feasibility of conducting controlled clinical trials comparing combined immunotherapy and chemotherapy to standard chemotherapeutic regimens.
Footnotes
Authors’ Note
Yi Yu, MD, Shanshan Huang, MD PhD, Jun Chen, MD, contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by grants from the National Natural Science Foundation of China (#81860427, #81760432, #81660402, and #81660405), Department of Health of Jiangxi Province Projects (#2019A058), Department of Science and Technology of Jiangxi Province Projects (#20202BAB216028).
