Abstract
Recent progress in adjuvant immunotherapy offers hope for improving disease-free survival in high-risk bladder cancer (BC) and renal cell carcinoma (RCC). This review focuses on key trials such as CheckMate 274 and KEYNOTE-564, which show promising results with nivolumab in BC and pembrolizumab in RCC, including a 30% reduction in progression risk. Pembrolizumab also demonstrated overall survival (OS) benefit in RCC. The review also explores the potential of circulating tumor DNA (ctDNA) as a biomarker for better therapy selection and patient stratification. It emphasizes the need for ongoing research to establish survival benefits and suggests integrating biomarkers and risk stratification to optimize adjuvant immunotherapy in BC and RCC.
Introduction
Adjuvant treatment is widely used in various cancers to decrease recurrence and improve survival. However, the role of adjuvant treatment in genitourinary cancers, especially in renal cell carcinoma (RCC) and bladder cancer (BC), is still need to be clarified. For patients with localized disease, radical surgical resection is considered the standard of therapy for both of them. In patients with muscle-invasive bladder cancer (MIBC), cystectomy alone is associated with up to 80% overall cure rate depending disease stage, but the risk of recurrence is up to 40% to 70% and most patients with recurrent disease died of metastatic disease within 3 years of diagnosis.1,2 On the contrary, disease recurrence is observed in almost 30% of patients with RCC, and most relapses arise at distant metastatic sites, resulting in a poor prognosis.3,4 After surgical resection, systemic therapy is required to prevent the recurrence of disease.
The landscape of adjuvant treatment for BC and RCC is marked by complexities and ongoing debate, especially concerning the selection of patients and the type of treatment. Despite numerous trials, consistent results have been elusive, fueling continued discussion in the field. Historically, chemotherapy for BC has been extensively studied, yet recent trials have been halted early due to various challenges. Nivolumab emerges as a viable adjuvant therapy option for BC. For RCC, whereas many studies have been conducted, only sunitinib and pembrolizumab have been shown to effectively delay recurrence and pembrolizumab improved OS. Crucially, the selection of both the patient and the treatment regimen is vital to minimize the risk of overtreatment and its associated adverse effects.
This review concludes its introduction by aiming to shed light on the current state of adjuvant therapies for BC and RCC, with particular emphasis on the role of immunotherapy in this context.
Adjuvant Therapies in Bladder Cancer
Approximately 25% of individuals diagnosed with bladder BC initially present with MIBC. Cisplatin-based neoadjuvant therapy remains the recommended treatment approach over adjuvant therapy due to its survival benefit and the consideration that certain patients may be ineligible for adjuvant treatment due to complications after surgery. 5 Despite the recommendations, neoadjuvant therapy is underused due to factors such as cisplatin ineligibility, frailty, comorbidities, and patient reluctance. 6 The role of adjuvant chemotherapy in patients with MIBC after radical surgery remains unclear. Several older randomized trials yielded controversial results, whereas 3 more recent trials designed to examine this question, those trials had to be terminated prematurely due to the poor accrual.7-9 EORTC 30994 trial was comparing immediate and delayed cisplatin-based combination chemotherapy following radical surgery in patients with pT3 to pT4 or N+ M0 BC. 9 The study included 284 patients, with a median follow-up duration of 7 years. The immediate treatment group had a significantly longer disease-free survival (DFS) than the deferred chemotherapy group, with a median duration of 3.11 years versus 0.99 years (hazard ratio [HR] = 0.54; 95% confidence interval [CI] = 0.40-0.73). The 5-year overall survival (OS) rates were 53.6% versus 47.7%, with a median OS of 6.74 years versus 4.6 years (HR = 0.78; 95% CI = 0.56-1.08). The survival advantage of immediate treatment was more significant in patients without lymph node involvement (HR = 0.37; 95% CI = 0.16-0.83). The meta-analysis conducted by Leow et al 10 demonstrated the progression- free survival (PFS) and OS benefit of cisplatin-based combination adjuvant chemotherapy compared with no adjuvant therapy after radical surgery. Furthermore, a recent meta-analysis of randomized controlled trials (RCTs) involving 1183 patients demonstrated a significant improvement in OS with the use of cisplatin-based combination therapy (HR = 0.82; 95% CI = 0.7-0.96). 11 When HR is adjusted for age, sex, pT stage, and pN stage, the survival benefit of adjuvant chemotherapy becomes clearer (HR = 0.77; 95% CI = 0.65-0.92), with an absolute 9% improvement in survival at 5 years. Although these results demonstrate the advantages of adjuvant chemotherapy in individuals not receiving neoadjuvant cisplatin-based regimens, data about the efficacy of adjuvant chemotherapy in individuals receiving neoadjuvant chemotherapy are relatively limited. In an observational cohort study, the 5-year OS rates for patients who received neoadjuvant chemotherapy were 36.8% for adjuvant chemotherapy and 24.4% for observation. 12 Patients who received adjuvant chemotherapy had a 22% lower risk of mortality (HR = 0.78; 95% CI = 0.61-0.99). However, studies are retrospective and yield inconsistent results.12-16 A meta-analysis of retrospective studies revealed that the use of adjuvant chemotherapy improves OS in patients who have undergone neoadjuvant therapy (HR = 0.83; 95% CI = 0.74-0.94). 17 The demand for alternative therapies has emerged for several reasons. First, some patients in need of adjuvant therapy have already undergone cisplatin-based regimens as neoadjuvant therapy, whereas others are cisplatin ineligible. In addition, there is a pressing need for more robust evidence concerning the effectiveness of adjuvant chemotherapy.
Adjuvant Immunotherapy in Bladder Cancer
The efficacy of immunotherapy has been extensively investigated across various stages of urothelial cancer, including both advanced disease and localized disease with different aims such as adjuvant and neoadjuvant therapies.18-20 The IMvigor 010 trial was the first randomized phase 3 study to evaluate the effectiveness of immuno-oncology (IO) in the adjuvant setting for muscle-invasive urothelial carcinoma (MIUC) (Table 1). The study included patients who were at a high risk of recurrence, pT3, pT4a, or pN+, and either ineligible for or declined adjuvant cisplatin-based combination chemotherapy for patients who had not received neoadjuvant cisplatin-based chemotherapy and ypT2 to ypT4a or ypN+ for patients who received neoadjuvant cisplatin-based regimens. 21 Between October 2015 and July 2018, 809 patients were enrolled to atezolizumab (1200 mg every 3 weeks for 1 year or 16 cycles, whichever first) and observation. The primary efficacy endpoint was DFS, and the study did not meet it after 21.9 months of median follow-up time. Median DFS was 19.4 months for atezolizumab and 16.6 months for observation (HR = 0.89; 95% CI = 0.74-1.08). Programmed cell death ligand 1 (PD-L1) positivity was defined as PD-L1-expressing tumor-infiltrating immune cells covering ⩾5% of the tumor area. Although median DFS was numerically longer in patients with PD-L1 positivity (24.8 months vs 41.4 months), it was not statistically significant (HR = 1.01; 95% CI = 0.76-1.35). Overall survival data were immature at the time of analysis. Eighteen-month OS was 79% for atezolizumab and 73% for the observation arm. It should be noted that a significant proportion of patients, more than one-third, were administered atezolizumab for less than 6 months. In total, 29% of patients discontinued atezolizumab due to recurrence, whereas 15% did so due to adverse effects. Grade 3 or higher TREAs were seen in 16% of the patients treated with atezolizumab.
Phase-III trials about adjuvant use of immune checkpoint inhibitors for adjuvant treatment for bladder cancer.

Abbreviations: CI, confidence interval; DFS, disease-free survival; HR, hazard ratio; ITT, intent to treat; N/R, not reached; OS, overall survival.
PD-L1-expressing tumor-infiltrating immune cells covering ⩾5% of the tumor area; bPD-L1 expression level ⩾1%.
CPS ⩾ 10%.
CheckMate 274 trial was conducted to evaluate the efficacy of adjuvant nivolumab treatment in urothelial carcinoma. 22 Pathological stage criteria for patients enrolled in this study were the same as in the IMvigor 010 study. Seven hundred nine patients who underwent radical surgery were randomly assigned to nivolumab (240 mg every 2 weeks for 1 year) or placebo. The primary endpoints were DFS in the intent to treat (ITT) population and among those with PD-L1 positivity. Disease-free survival was 20.8 and 10.8 months for nivolumab and placebo arms, respectively. Adjuvant nivolumab treatment was associated with a 30% and 45% risk reduction in progression in the ITT and PD-L1-positive populations, respectively. After a median follow-up of 36.1 months, the DFS advantage of nivolumab continues. The median DFS was 22 months vs 10.9 months and 52.6 months vs 8.4 months in the ITT and PD-L1-positive populations, respectively (HR = 0.71; 95% CI = 0.58-0.86 and HR = 0.52; 95% CI = 0.37-0.72). 23 Grade 3 or higher treatment-related adverse events (TRAEs) occurred in 17.9% of patients in the nivolumab arm and 7.2% in the placebo arm. 22 Nivolumab treatment was discontinued in 12.8% of patients due to TRAEs. The initial results of overall OS data were recently reported at the 2024 European Association of Urology (EAU) annual meeting. Adjuvant nivolumab demonstrated a survival benefit in the ITT population, and this benefit was more pronounced in the PD-L1-positive subgroup (HR = 0.76; 95% CI = 0.61-0.96 and HR = 0.56; 95% CI = 0.36-0.86, respectively).
Results of the AMBASSADOR trial (NCT03244384), which evaluated the use of adjuvant pembrolizumab in patients with MIUC after radical surgery, were recently presented. 24 Patients diagnosed with pT2-4aNx or pTxN+ disease or positive surgical margins after neoadjuvant chemotherapy, or those with pT3-4Nx or pN+ disease or positive surgical margins after surgery without chemotherapy, were eligible for the trial. In total, 702 patients with urothelial carcinoma who underwent radical surgery were randomly assigned to pembrolizumab (200 mg every 3 weeks for 1 year) and observation arms. Dual primary end points were DFS and OS. After 22.3 months of median follow-up, the median DFS was 29 months vs 14 months (HR = 0.69; 95% CI = 0.54-0.87) and adjuvant pembrolizumab demonstrated significant improvement in DFS, regardless of PD-L1 status. However, adjuvant pembrolizumab failed to show OS benefit (median OS was 50.9 months for pembrolizumab and 55.8 months for observation with HR = 0.98; 95% CI = 0.76-1.26). Almost half of the patients (48.4%) experienced grade 3 or higher adverse events (AEs) in pembrolizumab arm.
The TOMBOLA study (NCT04138628) is a phase II clinical trial that aims to evaluate the effectiveness of atezolizumab in patients who have circulating tumor DNA (ctDNA) positivity after neoadjuvant therapy and radical cystectomy. The study’s primary outcome was a complete response, defined as ctDNA depletion, that should go from positive to negative with normal imaging after therapy. IMvigor 011 (NCT04660344) was designed to evaluate the efficacy and safety of atezolizumab (for 1 year) adjuvant treatment compared with placebo in patients with MIBC who have ctDNA positivity after cystectomy. 25 These studies continue to enroll patients.
The role of ctDNA as a biomarker for evaluating the tumor burden is a developing area in solid tumors. Circulating tumor DNA can be differentiated from germline DNA by detecting tumor-specific genetic alterations. Advanced BC is one of the cancers with higher ctDNA levels, along with lung and colorectal cancers. 26 An elevated ctDNA level correlates with an unfavorable outcome in patients with metastatic BC. 27 Furthermore, elevated preoperative ctDNA levels were associated with disease progression and poor survival outcomes following surgery. 28 The presence of ctDNA following cystectomy is strongly associated with the occurrence of metastatic recurrence, with a sensitivity of 100% and a specificity of 98%. 29 These results raise the question of whether ctDNA can guide the adjuvant treatment decision. Circulating tumor DNA analysis of IMvigor 010 showed that 36% of patients were positive for ctDNA at the start of adjuvant therapy. 30 Patients with ctDNA positivity had a greater risk of recurrence in the observation arm than those without (HR = 6.3; 95% CI = 4.45-8.92). There was no difference in DFS between the observation and atezolizumab arms in ctDNA-negative patients; however, DFS improved with atezolizumab treatment in ctDNA-positive patients (HR = 0.58; 95% CI = 0.43-0.79). Furthermore, ctDNA clearance (defined by conversion from ctDNA positive in C1D1 to ctDNA negative in C3D1) is associated with an extended duration of DFS in patients treated with atezolizumab (HR = 0.26; 95% CI = 0.12-0.56). Finally, atezolizumab improves OS in patients with positive ctDNA (HR = 0.59; 95% CI = 0.41-0.86), and ctDNA clearance was associated with a prolonged OS (HR = 0.14; 95% CI = 0.03-0.59). These results suggest that ctDNA may be a marker for adjuvant treatment response. IMvigor011 may provide more precise information on this matter.
Adjuvant Therapies in Renal Cell Carcinoma
Patients with early stage RCC may still experience relapse, underscoring the need for adjuvant treatments that could improve their prognosis. However, indiscriminate treatment can expose these patients to unnecessary risks and adverse effects. To address this, various prognostic scoring systems have been devised for patient selection in RCC adjuvant trials, which are designed to better predict outcomes and guide treatment decisions.
Historical Treatments—Dark Age
Before the 2004 was considered as a dark age of the RCC. Spontaneous tumor regression and the infiltration of immune cells into tumors are thought to play a role in the effectiveness of adaptive immunity in treating RCC. Then, it was proposed that cytokine treatment could activate an immunologic response against cancer cells. Interleukin-2 (IL-2) and interferon alpha (IFN-α) were the only therapeutic options, and response rates to these treatments were 5% to 10% in patients with metastatic RCC. 31 Interleukin-2 and IFN-α monotherapy or combination has also been evaluated in the adjuvant setting after radical surgery.32-36 None of these studies was able to demonstrate a significant improvement in DFS or OS. A combination of IL-2, IFN-α, and 5-fluorouracil also failed to show benefit in adjuvant setting (HR = 0.84; 95% CI = 0.63-1.12 for DFS and HR = 0.87; 95% CI = 0.61-1.23 for OS). 37 In addition, a retrospective study revealed that patients with pT2b-T3cN0 improved from IFN treatment. 38 However, due of the observational study’s selection bias, the proof value is weak.
Tyrosine Kinase Inhibitor Treatments—The Middle Age
Vascular endothelial growth factor (VEGF) is currently recognized as the primary factor involved in the development and the progression of RCC. The activation of the VEGF pathway is accountable for the recruitment, migration, and proliferation of endothelial cells, consequently leading to angiogenesis in the context of RCC. 39 After demonstrating significant improvement in outcomes for patients with metastatic RCC, anti-VEGF agents quickly became the standard of care and the role of anti-VEGF as an adjuvant therapy has been evaluated in several trials (Table 2).
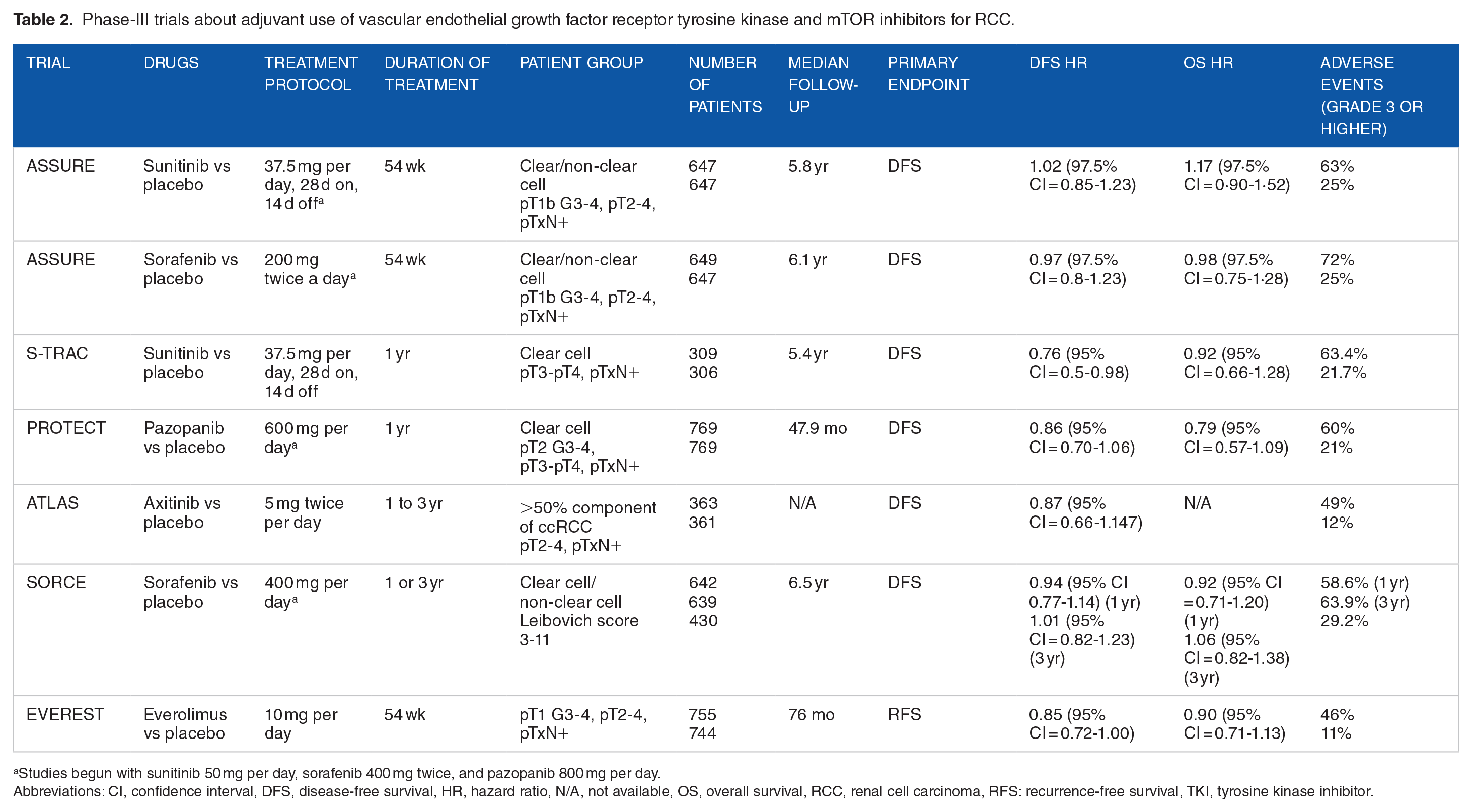
Phase-III trials about adjuvant use of vascular endothelial growth factor receptor tyrosine kinase and mTOR inhibitors for RCC.
Studies begun with sunitinib 50 mg per day, sorafenib 400 mg twice, and pazopanib 800 mg per day.
Abbreviations: CI, confidence interval, DFS, disease-free survival, HR, hazard ratio, N/A, not available, OS, overall survival, RCC, renal cell carcinoma, RFS: recurrence-free survival, TKI, tyrosine kinase inhibitor.
The ASSURE trial is the first randomized phase-III study investigate the role of for tyrosine kinase inhibitor (TKI) treatment in adjuvant settings. ASSURE study compared sunitinib and sorafenib versus placebo, and the primary endpoint was DFS. The patients were assigned at random to 1 of 3 arms: sunitinib 50 mg twice daily orally for the first 4 weeks of each 6-week cycle, sorafenib 400 mg twice daily orally for the first 4 weeks of each cycle, or placebo. This study failed to meet the primary endpoint. There was no improvement in DFS and OS with sunitinib or sorafenib compared with placebo. 40 Furthermore, in the active treatment groups, there was a substantial occurrence of grade 3 or greater side effects.41,42
S-TRAC is the second pivotal TKI trial which investigated the efficacy of sunitinib in RCC. 43 Unlike the ASSURE study, only patients with clear cell histology and high-risk have been included. The primary endpoint was DFS, which revealed a significant difference between the sunitinib and placebo groups, with a median DFS of 6.8 years versus 5.6 years, respectively (HR = 0.76; 95% CI = 0.59-0.98). Nevertheless, a considerable proportion of patients (44%) who were undergoing treatment with sunitinib experienced severe adverse effects, which compelled them to discontinue the treatment before completing the 1-year. In 2017, the Food and Drug Administration (FDA) approved sunitinib as an adjuvant therapy for high-risk, locoregional clear cell RCC (ccRCC) patients. Subsequently, a secondary analysis of the study did not demonstrate an improvement in OS (HR = 0.92; 95% CI = 0.66-1.28). 44 However, sunitinib was associated with a 26% reduced risk of relapse in high-risk patients. Treatment discontinuation due to AEs was 28.1% and grade 3 or higher TRAEs occurred in 63.4% of patients in the sunitinib arm. The current National Comprehensive Cancer Network (NCCN) recommendations classify adjuvant sunitinib for stage III RCC as a category III recommendation. The appropriateness of DFS as a metric, the presence of variation among patient populations, and the suitability of individual doses remain issues of ongoing debate.
The PROTECT study investigated the efficacy of pazopanib as adjuvant therapy for patients with locally advanced RCC who were at high risk of relapse. 45 Patients with non-ccRCC were excluded, as with S-TRAC. Starting dose of pazopanib was reduced to 600 mg once daily due to AEs during the study. Unfortunately, no DFS benefit was observed for pazopanib 600 mg once daily compared with placebo (HR = 0.86; 95% CI = 0.70-1.06). By contrast, the secondary analysis of DFS in the 800 mg pazopanib subgroup of the ITT cohort (n = 403) yielded a DFS benefit (HR = 0.69; 95% CI = 0.51-0.94), but in this group of patients, there was a higher rate of treatment discontinuation because of adverse effects.
Subsequently, the SORCE study evaluated the efficacy of sorafenib in the adjuvant treatment of RCC. 46 Patients with intermediate- or high-risk ccRCC or non-ccRCC were randomly assigned to sorafenib (1 or 3 years of treatment) or placebo. The SORCE study showed that there was no DFS (HR = 1.01; 95% CI = 0.82-1.23 for 3 years of sorafenib vs placebo and HR = 0.94; 95% CI = 0.77-1.14 for 1 year of sorafenib vs placebo) or OS (HR = 1.06; 95% CI = 0.82-1.38 for 3 years of sorafenib vs placebo and HR = 0.92; 95% CI = 0.71-1.20 for 1 year of sorafenib vs placebo) benefit of adjuvant sorafenib treatment.
The ATLAS study, which compared axitinib and placebo, was ended early at a predefined intermediate analysis because it failed to reveal a DFS benefit (HR = 0.87; 95% CI = 0.66-1.147). 47 The study also categorized patients into 2 groups based on their likelihood of tumor recurrence. There was no benefit shown in DFS in either the highest-risk (pT3 with FG 3 or pT4 and/or N+, any T, any FG) or the lower-risk group (pT2 or pT3 with ⩽G2) (HR = 1.0; 95% CI = 0.62-1.66 and HR = 0.73; 95% CI = 0.52-1.02, respectively)
EVEREST study examines the efficacy of the mammalian target of rapamycin inhibitor everolimus as an adjuvant treatment in a randomized double-blinded RCT. In that trial, individuals with intermediate-high risk (pT1G3-4N0 to pT3aG1-2N0) or very high risk (pT3aG3-4 to pT4G-any or N positive) for kidney cancer recurrence were given everolimus 10 mg once a day for 54 weeks. 48 Recurrence-free survival (RFS) was prolonged with everolimus than with placebo in the very-high-risk group (HR = 0.79; 95% CI = 0.65-0.97) but not in the intermediate-high-risk group (HR = 0.99; 95% CI = 0.73-1.35). 48
Immunotherapy—Golden Age
Immunotherapy is a swiftly advancing therapeutic approach in the field of oncology. Monoclonal antibodies targeting immune checkpoints, programmed death-1/-ligand 1 (PD-1/L1), and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) have demonstrated remarkable effectiveness in the treatment of metastatic RCC. Immunotherapy combinations became the standard of care for the first-line treatment of advanced RCC.49,50 The effectiveness of immune checkpoint inhibitors in the adjuvant treatment of the RCC after nephrectomy began to be evaluated (Table 3).
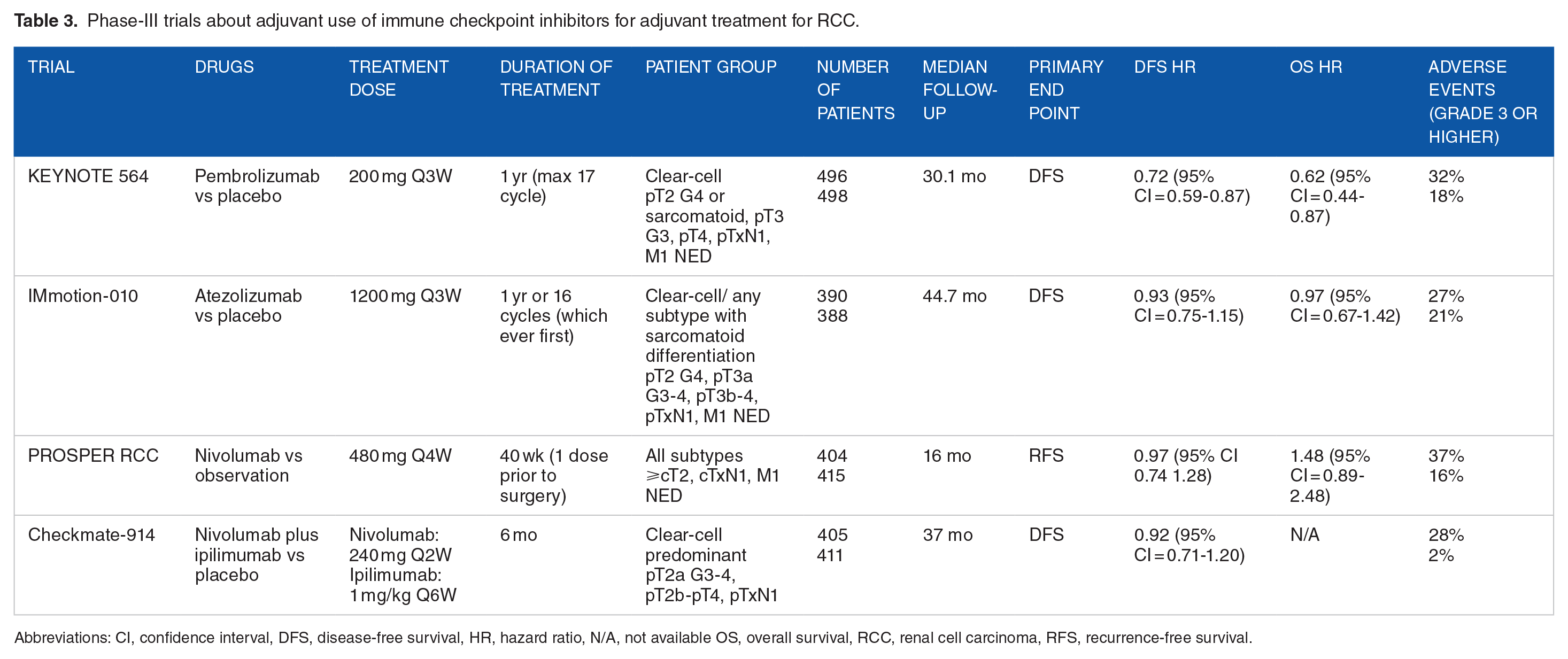
Phase-III trials about adjuvant use of immune checkpoint inhibitors for adjuvant treatment for RCC.
Abbreviations: CI, confidence interval, DFS, disease-free survival, HR, hazard ratio, N/A, not available OS, overall survival, RCC, renal cell carcinoma, RFS, recurrence-free survival.
KEYNOTE-564 is a randomized phase III study that compared pembrolizumab and placebo for 1 year in patients with intermediate-to-high risk or high risk or M1. 51 Protocol-defined intermediate-high-risk criteria included pT2 with nuclear grade 4 or sarcomatoid differentiation, pT3 and high-risk criteria included pT4 or regional lymph-node metastasis or stage M1 tumors with no evidence of disease (NED). Pembrolizumab 200 mg or placebo was administered intravenously once every 3 weeks for 1 year or until disease recurrence or unacceptable toxicity. The primary endpoint was DFS according to the investigator. Disease-free survival was significantly prolonged in patients treated with pembrolizumab (HR = 0.68; 95% CI = 0.53-0.87) at the prespecified period of interim analysis, when the median time from randomization to the data cutoff date was 24.1 months. In subgroup analyses, pembrolizumab showed a DFS benefit in patients with M0 tumors (HR = 0.74; 95% CI = 0.57-0.96) and M1 tumors with NED (HR = 0.29; 95% CI = 0.12-0.69). Patients with a PDL1 combined positive score (CPS) of ⩾1 had a greater DFS benefit (HR = 0.67; 95% CI = 0.51-0.88) than those with a PDL1 CPS of <1.24-month OS rates were of 96.2% for pembrolizumab and 93.8% for placebo. Following these findings, the FDA approved the adjuvant pembrolizumab on November 17, 2021, for adjuvant treatment of RCC. With a median follow-up of 57.2 months, an updated analysis of KEYNOTE-564 showed a 28% lower risk of recurrence or death in the pembrolizumab arm compared with the placebo arm. Furthermore, adjuvant pembrolizumab treatment was associated with survival benefit (HR = 0.62; 95% CI = 0.44-0.87). 52
IMmotion010 is a phase III, randomized, placebo-controlled trial evaluating the efficacy of the PDL1 inhibitor atezolizumab as an adjuvant therapy option for patients with RCC with a clear cell or sarcomatoid component and a high risk of recurrence. 53 As with KEYNOTE-564, the high-risk definition included pT2 with nuclear grade 4 or sarcomatoid differentiation, pT3 and high-risk criteria included pT4 or regional lymph-node metastasis or stage M1 (NED). Patients randomized atezolizumab or placebo once every 3 weeks for 16 cycles or 1 year. Disease-free survival was similar between patients in the 2 groups after a median follow-up time of 44.7 months. Disease-free survival was 57.2 months for the atezolizumab group and 49.5 months for placebo group (HR = 0.93; 95% CI = 0.75-1.15). Two-year DFS rates were 67% versus 65% in atezolizumab and placebo arms, respectively.
PROSPER is a phase III, randomized, open-label trial that investigates nivolumab in perioperative setting for stage T2 or greater or lymph-node-positive M0 RCC of any histology. 54 Trial design is 1 dose nivolumab before surgery followed by 9 adjuvant doses after surgery. Interim analysis after a median follow-up of 16 months showed no difference in RFS between groups (HR = 0.97; 95% CI = 0.7-1.28). Overall survival data were immature, and the study was terminated because of futility.
CheckMate 914 is a randomized, phase III trial that compared the efficacy of ipilimumab plus nivolumab versus placebo (part A) or nivolumab monotherapy versus ipilimumab plus nivolumab versus placebo (part B) as adjuvant therapy in patients with ccRCC at high risk of recurrence. 55 High risk was defined using the same criteria as the KEYNOTE-564 and IMmotion010 trials, with the addition of patients with tumor stage 2 and histological grade 3 disease. Checkmate-914 did not meet the primary efficacy endpoint of DFS for nivolumab with ipilimumab versus placebo (HR = 0.92; 95% CI = 0.71-1.19) after 37 months of median follow-up. Disease-free survival at 24 months was 76.4% and 74% for ipilimumab plus nivolumab and placebo groups, respectively.
The ongoing RAMPART study (NCT03288532), a phase III trial, is investigating the effectiveness of adjuvant durvalumab and tremelimumab in patients with ccRCC or non-ccRCC. The study has 3 groups: no treatment, durvalumab alone (monthly for a year), or a combination of durvalumab and tremelimumab. The primary endpoints are DFS and OS, whereas secondary endpoints include metastasis-free survival and RCC-specific survival time, with completion expected by July 1, 2024. LITESPARK 022 (NCT05239728) aimed to compare the efficacy and tolerability of hypoxia-inducible factor- 2 (HIF-2) inhibitor belzutifan with pembrolizumab against a placebo plus pembrolizumab as an adjuvant treatment for intermediate-high or high-risk ccRCC, including M1 NED. The primary endpoint was DFS, and the secondary endpoint was OS.
Discussion
Adjuvant therapy has established its efficacy across a range of malignancies, becoming a cornerstone of treatment in oncology. However, the intricacies of determining who should receive such therapy and which drugs to use present a considerable challenge. In making these decisions, it is crucial to balance the potential benefits of treatment against its possible adverse effects and the overall impact on the patient’s quality of life. These multifaceted considerations are vital in the strategic planning of adjuvant therapy protocols.
Muscle-invasive bladder cancer treatment consists of a combination of cystectomy, radiotherapy, and chemotherapy. However, as the disease stage increases, OS rates dramatically decrease. The 5-year survival rate for locally advanced BC is approximately 38%, whereas it plunges to 6% for metastatic BC. 56 These survival rates highlight the need for developing new treatment options to decrease recurrences and improve survival. Immunotherapy emerges as a promising adjuvant treatment alternative, especially for patients who cannot undergo cisplatin-based therapy or have previously received cisplatin treatment in the neoadjuvant phase. Immunotherapy is currently the only adjuvant treatment option besides chemotherapy. Although IMvigor 010, CheckMate 274, and AMBASSADOR trials have similar study designs, their results are inconsistent. Atezolizumab did not demonstrate any significant DFS or OS benefit, whereas pembrolizumab showed a DFS benefit and nivolumab showed a DFS and OS advantage. The experimental arms of these 2 studies exhibited similar DFS: 19.4 months for IMvigor010 and 22 months for CheckMate 274.21,23 However, there is a difference of nearly 6 months in DFS between the 2 control arms (10.9 months for CheckMate 274 and 16.6 months for IMvigor 010). The IMvigor 010 and AMBASSADOR trials were designed as an observational-control study, whereas the Checkmate 274 trial was a placebo-control trial. In addition, the patients participating in the studies have different characteristics. CheckMate 274 and AMBASSADOR had more patients with upper urinary tract disease (21% for CheckMate 274, 21.9% for AMBASSADOR, and 6.7% for IMvigor 010), whereas IMvigor 010 had more patients with positive lymph nodes (52% for IMvigor 010, 49.8% for AMBASSADOR, and 47.3% for CheckMate 274). In contrast to the CheckMate 274 and IMvigor 010 trials, patients with positive surgical margins were eligible for the AMBASSADOR trial. Therefore, it is not appropriate to directly compare these studies.
Although guidelines recommend neoadjuvant therapy for MIBC in cisplatin-eligible patients, there is a disconnect between guidelines and daily practice.5,57 Indeed, adjuvant therapy is more commonly used in clinical practice. 58 So, RCTs are needed to investigate the efficacy of adjuvant therapy, particularly in patients received neoadjuvant treatment. Although there are risk stratification scores and nanograms available to predict oncological outcomes after cystectomy, none of them are currently used to guide adjuvant treatment decisions.59-61 The presence of markers such as ctDNA and interventions to increase immunotherapy efficacy can improve the decision-making process for selecting patients for adjuvant therapy while protecting patients from unnecessary therapies and related adverse effects. 62
Approximately 40% of patients developing progression after nephrectomy justifies the need for adjuvant therapies in RCC patients with a high risk of relapse. 63 The adjuvant treatment of RCC immunotherapy is promising, but careful patient selection and risk-benefit analysis are required to avoid overuse. In recent years, vascular endothelial growth factor receptor (VEGFR)-TKIs and immune-checkpoint inhibitors (ICIs) have notably improved survival outcomes for patients with metastatic RCC. However, in the adjuvant setting for RCC, outcomes from randomized phase III trials using VEGFR-TKIs have been inconsistent. The S-TRAC trial alone showed a benefit in DFS, which was not reflected in OS. Sunitinib is a category 3 recommendation for adjuvant therapy by the NCCN. Its widespread use in daily practice is limited due to the lack of demonstrated OS benefit and high toxicity rates. Immunotherapy was certainly a milestone in the history of RCC treatment. Based on the results of the KEYNOTE-564 trial, pembrolizumab monotherapy is currently approved for use in the adjuvant setting for RCC. However, only KEYNOTE-564 has described a significant DFS and OS advantage for immunotherapy as an adjuvant therapy. The potential of immunotherapy as a treatment in the adjuvant context shows promise; however, there is a significant risk of relapse. In the KEYNOTE-564 study, it was shown that 22.7% of patients in the intervention group experienced relapse. Moreover, research into adjuvant immunotherapies is necessary, but efforts must be focused on alternating and enhancing adjuvant options, taking into account the emergence of new therapeutic classes in the RCC therapeutic scene.
In the discussion of our review on adjuvant therapy for renal BC and RCC, several limitations should be acknowledged. First, our review primarily focused on phase 3 RCTs, which may overlook insights from phase 1 to 2 trials or real-world data that could offer crucial context on treatment efficacy, tolerability, and safety. Second, inherent heterogeneity in the inclusion criteria among the studies we reviewed, particularly concerning disease stages for RCC, poses challenges for direct comparisons and may affect the generalizability of our conclusions. In addition, our study primarily focused on analyzing aggregated data rather than examining data at the individual patient level, which could provide further valuable information. Finally, despite our comprehensive coverage of various aspects of adjuvant therapy in BC and RCC, the evolving landscape of oncology research means that newer findings and advancements could emerge post-publication, necessitating ongoing updates to our understanding.
Conclusions
In conclusion, surgical intervention continues to be the cornerstone of managing localized RCC and BC, yet the substantial risk of postoperative recurrence underscores the necessity for adjunctive therapies. Pembrolizumab for RCC and nivolumab for BC have demonstrated a marked reduction in recurrence risks. Nevertheless, the absence of an OS advantage, coupled with a current lack of biomarkers for patient-treatment alignment in everyday clinical settings, as well as issues related to toxicity, restricts their broader application. In addition, the rate of recurrence despite treatment with pembrolizumab or nivolumab indicates the potential requirement for alternative therapeutic options beyond immune checkpoint inhibitors. Therefore, the choice to employ adjuvant ICI treatments for RCC and BC should be a collaborative, informed decision-making process with the patient, carefully weighing the anticipated benefits against the possible toxicities and the reality of no confirmed OS improvement.
Footnotes
Author Contributions
SB, HB, and YÜ involved in conceptualization. SB and HB involved in investigation. YÜ involved in methodology. YÜ involved in supervision. SB and HB involved in visualization. SB and HB contributed to writing—original draft. YÜ contributed to writing—review and editing.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interest:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Hatice Bolek and Sevinc Balli declared no conflicts of interest to report. Yüksel Ürün declared research funding (Institutional and personal) from Turkish Oncology Group. Yüksel Ürün has served on the advisory board for Abdi-İbrahim, Astellas, AstraZeneca, Bristol Myers-Squibb, Eczacıbası, Gilead, Janssen, Merck, Novartis, Pfizer, and Roche. Yüksel Ürün received honoraria or has served as a consultant for Abdi-İbrahim, Astellas, Bristol Myers-Squibb, Eczacıbasi, Janssen, Merck, Novartis, Pfizer, and Roche.
