Abstract
Background
Bladder cancer in the setting of previous a kidney transplant (KT) is challenging to manage due to complex medical and surgical considerations.
Objective
To provide a comprehensive evaluation of the scope of management of bladder cancer in KT patients, and describe the controversies surrounding these management options.
Methods
A systematic review of studies reporting management of KT patients with bladder cancer and involving ≥3 patients was performed. A narrative review was also performed for various aspects of management such as pathophysiology, surgical considerations, intravesical therapy, immunosuppression and oncological surveillance.
Results
Bladder cancer incidence in KT recipients is 2.8–4.1 times higher than the general population, and there is a notable association with aristolochic acid nephropathy as well as BK virus oncogenesis. Regarding surgical treatment, transurethral resection is preferred for non-muscle invasive tumors, and intravesical BCG for intermediate- and high-risk patients appears to be underutilized despite its safety and associated reduction in recurrence. Radical cystectomy with limited pelvic lymph node dissection, urinary diversion, and consideration of bilateral nephroureterectomy appears to be the safest method of oncological control in muscle-invasive tumors. A switch in immunosuppressive regimens to mTOR inhibitors may be considered in lieu of its antitumor effects. Routine surveillance in KT patients with risk factors for bladder cancer is challenging and may be warranted especially in the Asian population which has a higher rate of urothelial malignancy.
Conclusions
This review provides a thorough summary of management strategies for bladder cancer in the setting of previous KT.
Introduction
The incidence of malignancy following kidney transplantation (KT) is significantly increased compared to the general population.1,2 Correspondingly, malignancy-related deaths after KT account for a significant proportion of total post-transplant mortality, with this figure reported at 18% in one study of over 19,000 patients. 3 Common de novo malignancies occurring after KT include post-transplant lymphoproliferative disorder, skin cancers, and urologic cancers. 1 In particular, bladder cancer is a challenging malignancy to manage even outside the domain of simultaneous KT, with high recurrence rates in non-muscle invasive bladder cancer (NMIBC) 4 and high morbidity in patients with muscle-invasive bladder cancer (MIBC) who undergo radical cystectomy. 5
Several unique challenges are evident in the management of bladder cancer in patients with previous KT. Although the development of malignancy has been associated with common immunosuppressive medications such as calcineurin inhibitors and azathioprine, these medications are essential for allograft maintenance and any changes in regimen may trigger a rejection episode. 6 Moreover, treatment options for bladder cancer such as intravesical bacillus Calmette-Guerin (BCG), mitomycin C (MMC), and systemic chemotherapy have their respective controversies for use in immunosuppressed KT patients; caution must also be exercised in the setting of a precious allograft which will lead to end-stage renal failure if lost through drug-induced kidney injury. Planned surgical treatments for bladder cancer must bear in mind the presence of altered anatomy, previous surgery to the abdomen and bladder, and possibility of simultaneous tumor involvement of the upper urinary tract and/or the allograft itself. In addition, reconstruction options after radical cystectomy (RC) must also take into account the location and amenability to different diversion options of the allograft.
Studies describing the management and outcomes of bladder cancer in KT patients are sparse, with most being case reports or small case series. Hence, we aimed to provide a comprehensive evaluation of the scope of management of bladder cancer in KT patients, and describe the controversies surrounding these management options.
Methods
This was a combined systematic and narrative review of the treatment of KT patients with bladder cancer.
We first performed a systematic review which adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Guidelines. 7 An electronic search strategy was performed by two independent reviewers on PubMed, Embase, and Scopus for relevant articles, without language restrictions. The search was first conducted from database inception to 14 October 2024. The full search strategy is listed in Supplementary Table 1. Retrieved abstracts and full texts were reviewed by two independent investigators; conflicts were resolved via group consensus among all authors in the study.
Studies reporting baseline characteristics and management of KT patients with bladder cancer were included. Studies had to report at least 3 cases to be eligible for inclusion. Single or double-patient case reports, reviews and conference abstracts were excluded. Studies which examined the prevalence of bladder cancer or other urological malignancies in KT patients, but did not provide data on treatment, were also excluded. Where reported, only de novo cases of bladder cancer in KT patients were included for analysis, and patients with pre-existing bladder cancer before transplantation were excluded. Patients with upper tract urinary cancer (UTUC) were also excluded from the study, unless they also had synchronous bladder cancer.
Data from included studies was extracted narratively with the aid of a standardized data collection template with predefined data fields including study characteristics, patient demographics, descriptions of management for bladder cancer, and outcomes. Studies were assessed for risk of bias by two independent investigators using the Joanna-Briggs Institute Critical Appraisal Tools for case series or cohort studies.8,9
Moreover, narrative evidence synthesis was used to discuss various domains of the treatment for bladder cancer, such as pathophysiology, surgical considerations, intravesical therapy, immunosuppression and oncological surveillance. Studies in literature commenting on these aspects, but not necessarily with data that could be extracted as part of the systematic review, were also considered for descriptive analysis to supplement the studies formally included in the systematic review.
Results
Study selection and baseline characteristics
The search strategy retrieved 1745 studies. After 552 duplicates were removed, the remaining 1193 studies were screened by title and abstract. Twenty five studies were identified for full-text review, including 3 which were found via citation searching. Finally, 21 studies10–30 were included for analysis (Figure 1).
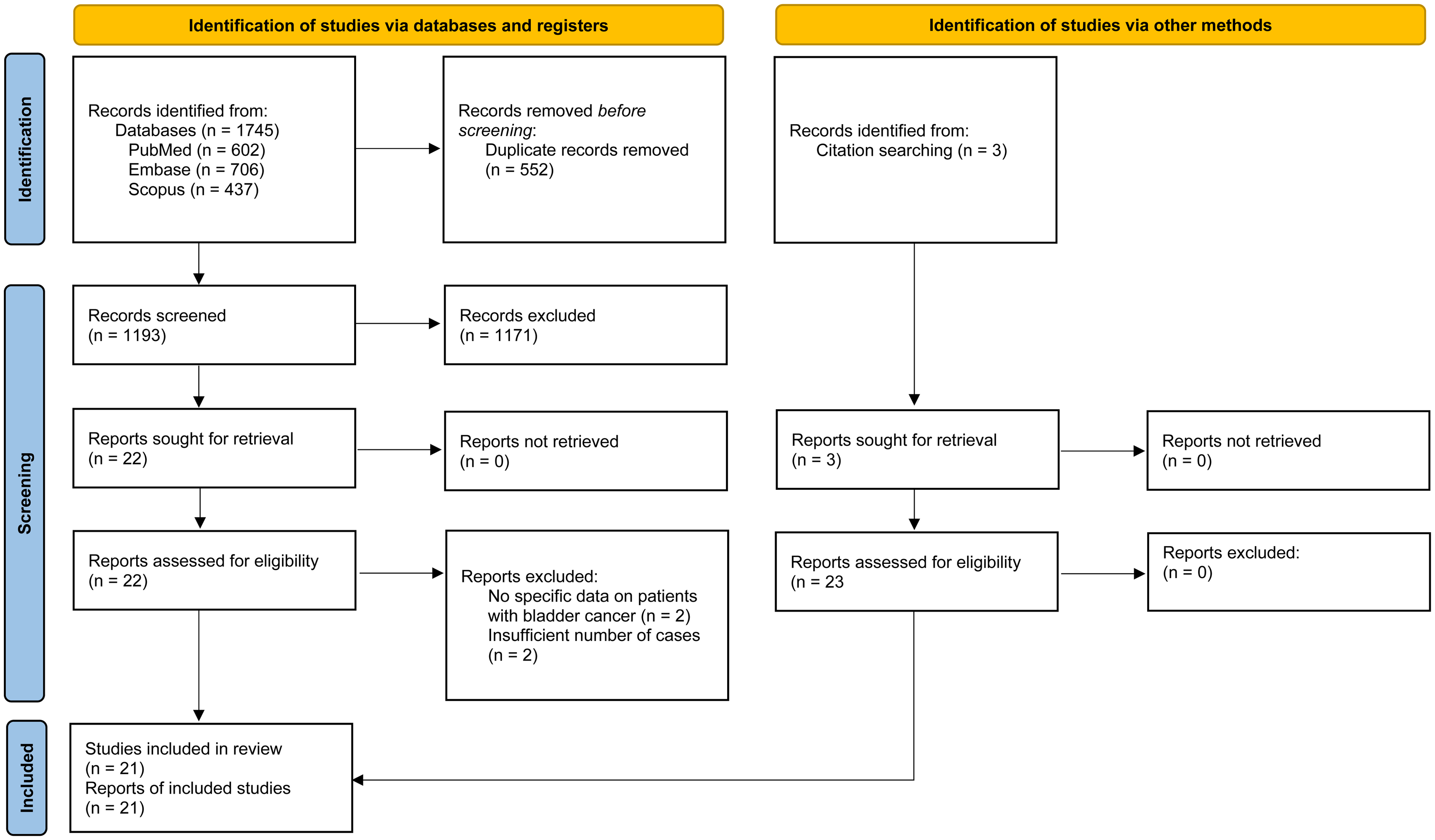
PRISMA flow diagram of included studies.
Included studies were published between 2002 and 2023 (Table 1). The number of included cases per study ranged from 3 to 87. The average age of patients ranged from 42 to 65 years, and there was wide variation in gender distribution. The average time from transplantation to bladder cancer ranged from 2.1 to 10.5 years. Mean follow-up time ranged from 1.0 to 10 years (Table 2). Risk of bias was mostly low to moderate.
Baseline characteristics.
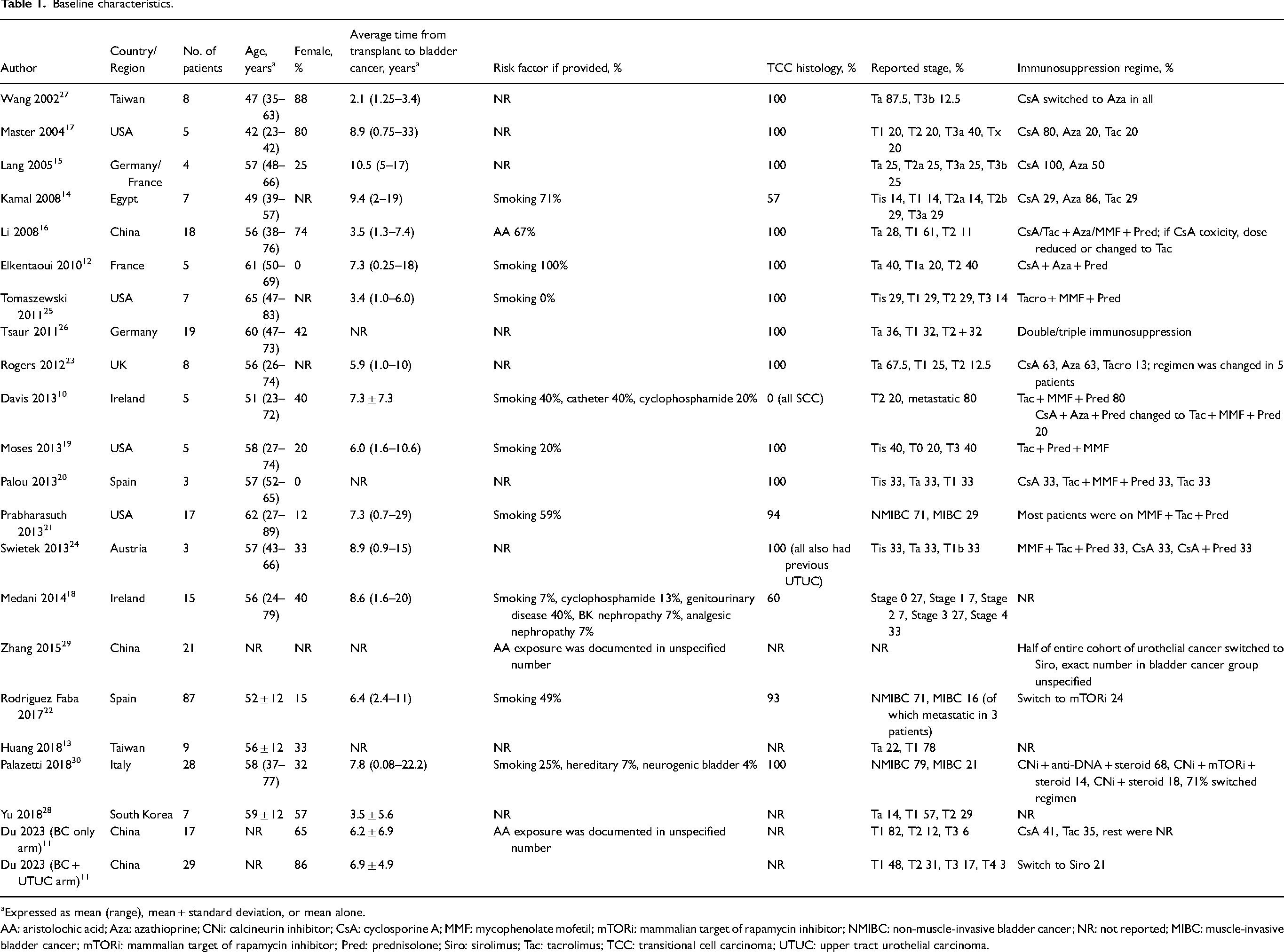
Expressed as mean (range), mean ± standard deviation, or mean alone.
AA: aristolochic acid; Aza: azathioprine; CNi: calcineurin inhibitor; CsA: cyclosporine A; MMF: mycophenolate mofetil; mTORi: mammalian target of rapamycin inhibitor; NMIBC: non-muscle-invasive bladder cancer; NR: not reported; MIBC: muscle-invasive bladder cancer; mTORi: mammalian target of rapamycin inhibitor; Pred: prednisolone; Siro: sirolimus; Tac: tacrolimus; TCC: transitional cell carcinoma; UTUC: upper tract urothelial carcinoma.
Treatment and outcomes.
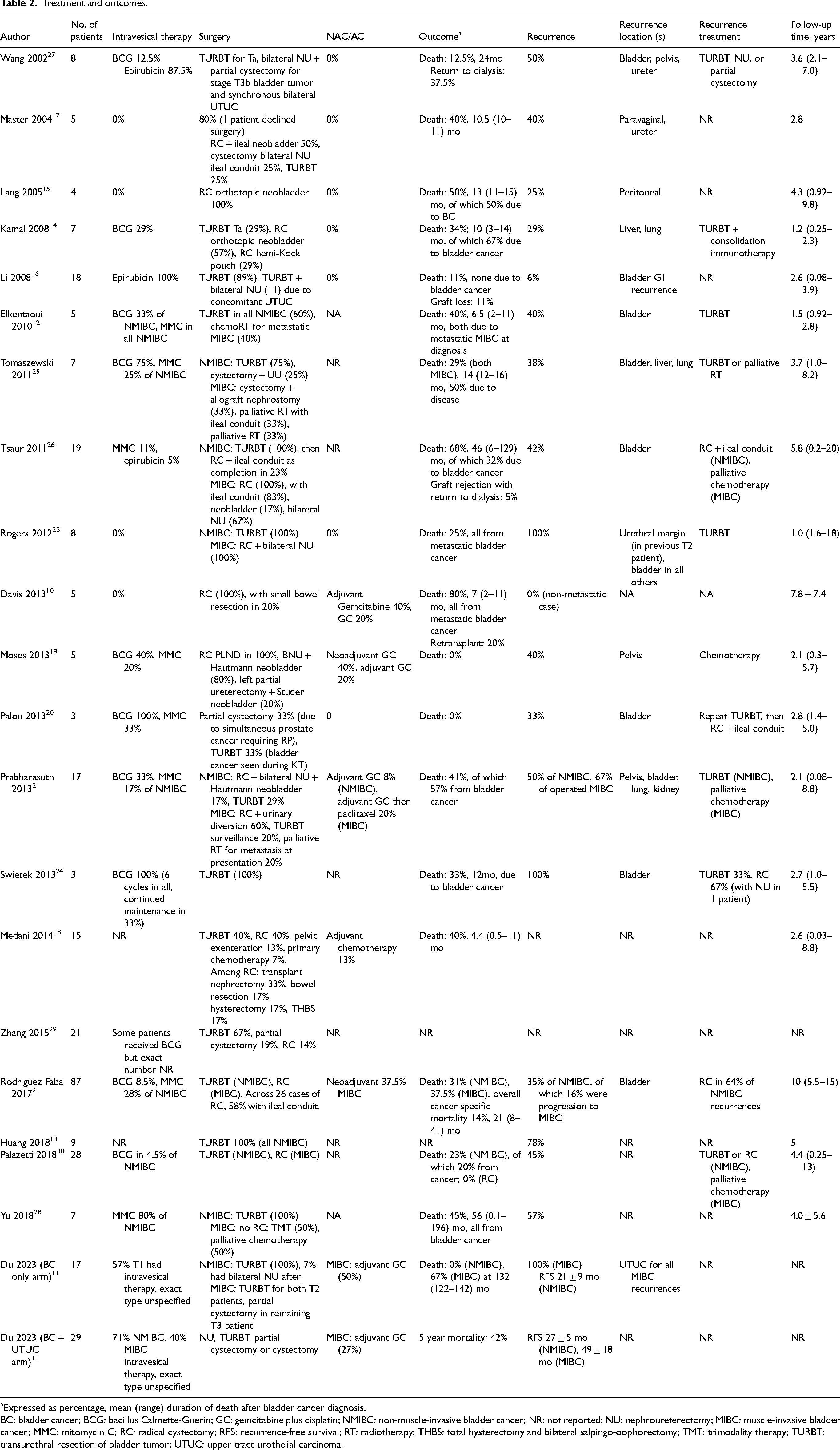
Expressed as percentage, mean (range) duration of death after bladder cancer diagnosis.
BC: bladder cancer; BCG: bacillus Calmette-Guerin; GC: gemcitabine plus cisplatin; NMIBC: non-muscle-invasive bladder cancer; NR: not reported; NU: nephroureterectomy; MIBC: muscle-invasive bladder cancer; MMC: mitomycin C; RC: radical cystectomy; RFS: recurrence-free survival; RT: radiotherapy; THBS: total hysterectomy and bilateral salpingo-oophorectomy; TMT: trimodality therapy; TURBT: transurethral resection of bladder tumor; UTUC: upper tract urothelial carcinoma.
Tumor characteristics
Most studies comprised predominantly transitional cell carcinoma (TCC; urothelial) histology except one study in which all cases were MIBC of squamous cell carcinoma (SCC) histology. 10 There was considerable variation in the percentage of MIBC across included studies. Cases of metastatic bladder cancer at presentation were reported in two studies.10,22 Twelve studies identified risk factors for the development of bladder cancer other than immunosuppression in their cohort; these included smoking and aristolochic acid-containing drugs (especially in the Asian population, due to herbal medicine consumption). In the study of patients with bladder SCC, all patients had risk factors such as long-term urinary catheterization, smoking, or cyclophosphamide use. 10 Oddly, a predominance of females was seen in some of studies, although not in the largest study of 87 patients, 22 suggesting that this may be more due to chance from low sample size rather than approximation of true prevalence.
Immunosuppression
Immunosuppressive regimens were reported in 18 studies. Regimens mainly consisted of combinations of cyclosporine A or tacrolimus, azathioprine or mycophenolate mofetil, and prednisolone. A change in regimen was performed in some patients, which typically involved switching cyclosporine A to tacrolimus or sirolimus, or a reduction in cyclosporine A dose.
Treatments
As with bladder cancer in the general population, TURBT was performed for most NMIBC cases and RC for operable MIBC cases. In some patients, partial cystectomy was performed to avoid compromise of the ureter of the transplanted kidney. Notably, eight studies reported on patients who underwent bilateral nephroureterectomy of the native kidneys, either as treatment for concomitant UTUC, or as a prophylactic measure to reduce recurrence in the upper urinary tract. Ileal conduit and ileal neobladder were the two commonest approaches for urinary diversion. Patients who had metastatic disease at presentation were typically commenced on palliative radiotherapy and/or chemotherapy. Transplant nephrectomy alongside RC for two cases was reported in one study. 18 In another study, a percutaneous allograft nephrostomy tube was placed due to scar tissue and adhesions limiting small bowel mobilization and creation of a urinary diversion. 25
The use of intravesical therapy was reported in 19 studies and varied considerably. BCG utilization ranged from 0–100%. Other intravesical therapies included epirubicin and MMC. Gemcitabine plus cisplatin was the typical adjuvant chemotherapy of choice. Only one study reported preoperative renal function alongside perioperative chemotherapy, and gemcitabine plus cisplatin was given in three patients: neoadjuvant in a patient with T2 disease and estimated creatinine clearance (eCrCl) of 34, partial dose neoadjuvant in a patient with T2 disease and eCrCl of 47 and declining renal function, and adjuvant in a patient with T3 disease and eCrCl of 73. Neoadjuvant chemotherapy was only reported in one study, 22 which was received by 37.5% of MIBC patients.
Outcomes
Graft loss was not seen in all but two studies. In one study, only a single patient out of 19 developed chronic graft rejection requiring return to dialysis 62 months after transplantation. In the other study, 1-year and 5-year graft survival was 92.5% and 68.4%. Five patients had acute rejection after immunosuppressant dose reduction (CsA or Tac) during TCC treatment, of which 2 experienced graft loss.
Mortality was reported in all studies and varied considerably, reflecting differences in the proportion of NMIBC, MIBC and metastatic tumors at diagnosis. For instance, 80% of MIBC patients in one included study 10 died from bladder cancer with metastatic recurrence; all-cause mortality was only seen in 31% of NMIBC patients in another study; 22 and no deaths were seen in the NMIBC cohort of the bladder cancer arm of another study. 12 Similarly, the prevalence of cancer recurrence (whether local or metastatic) exhibited marked variation across studies, ranging from 6% to 100%. Recurrence was typically treated with TURBT for NMIBC recurrences; RC or partial cystectomy for MIBC recurrences, and palliative chemotherapy for metastatic recurrences.
Discussion
This review provides an overview of the current literature on treatment of bladder cancer in patients with previous KT. Several unique aspects of bladder cancer in KT are present in this group, which may affect treatment decisions. Our use of PRISMA-compliant systematic review technique also provides a rigorous method of literature analysis.
Included studies in this systematic review were mostly of low to moderate quality, with most being case series with low numbers of patients, and the largest included cohort having 87 patients in total. Nonetheless, given that patients with both bladder cancer and previous KT are very rarely encountered in daily practice, this represents the best attainable evidence to date. Several themes were seen across included studies, and are further detailed below:
- Bladder cancer in KT recipients was almost always TCC, and notable risk factors besides immunosuppression included smoking and aristolochic acid. - For NMIBC, TURBT was the treatment modality of choice, but intravesical BCG for intermediate- and high-risk NMIBC was uncommon. - For MIBC, RC with limited PLND and urinary diversion was the treatment modality of choice. Bilateral nephroureterectomy was considered for concomitant UTUC, or as a prophylactic risk-reducing measure. - If a switch in immunosuppressive regimens was performed, it was typically switched from a cyclosporine-containing regimen to a tacrolimus or sirolimus-containing regimen. Dose reduction of cyclosporine was also reported. - Mortality and cancer recurrence varied considerably among studies, and was likely confounded by the low number of patients per study.
Several limitations of the systematic review are acknowledged. Meta-analysis of different management strategies was not possible in light of the small number of patients. Similarly, the effect of baseline characteristics and various treatments on survival metrics such as overall survival, recurrence-free survival and cancer-specific survival could not be analyzed due to low numbers of patients in most included studies. Certain factors previously reported to increase death from post-KT malignancy include increased age, pretransplant history of malignancy and deceased-donor kidney transplantation,
3
but these results were from an analysis of all malignancies and not bladder cancer specifically. Factors such as deceased-donor versus living-donor grafts, comorbidity or frailty indices, and tumor genetics would be important factors to consider in the treatment of bladder cancer patients, but were sparsely reported in included studies and literature.
Pathophysiology and natural history of bladder cancer in KT recipients
Compared to the general population, cancer incidence in KT recipients is 2.8–4.1 times higher; specifically for urothelial cancers, it is around 3.1–3.5 times higher, and tends to present at a younger age. 31 The exact risk appears to be higher in Asian compared to Western populations. Tsaur et al. 26 found that the overall incidence of urothelial cancer was 0.4% in a European population, with the majority having NMIBC. In contrast, urothelial malignancy is the most common post-KT malignancy in Chinese patients 29 with up to 4.55% incidence in one study. 32 Tumors in this population have a higher prevalence of aggressive disease, multifocal tumors, and upper tract involvement. These findings appear to be due to exposure to aristolochic acid in Asian populations, which is a component of some herbal medicines and has known associations with nephropathy, urologic malignancy, and reduced graft survival.33,34 Aside from these risk factors, most common immunosuppressants used in the setting of KT are associated with neoplasia, with the notable exception of mammalian target of rapamycin inhibitors (mTORi). This class of immunosuppressants blocks of the downstream effector of the PI3 K/Akt/mTOR pathway, which is implicated in urothelial carcinogenesis, hence explaining the lower risk of oncogenesis in patients on sirolimus and everolimus. 35
The role of virus-induced carcinogenesis in the development of post-KT bladder cancer remains controversial. BK virus has strong oncogenic potential stemming from the large T antigen, which inactivates Rb and p53 tumor suppressor gene products. 36 Bladder cancer incidence in KT patients with BK virus nephropathy is reported to be 2.2 times that of other KT patients; 37 it has also been associated with an 8.21-fold higher risk of urothelial cancer in the transplanted urinary tract 38 and urologic malignancies of atypical histology. 39 Strong evidence for BK virus as a causal factor for bladder cancer has also been demonstrated using immunohistochemistry. 40 Interestingly, this correlation appears to exist only in KT patients and is absent in non-KT patients with bladder cancer. 41 Viral reactivation has been associated with bladder cancer according to urine DNA load studies, 42 and polyomavirus replication rates are an independent risk factor for bladder cancer post-transplantation. 43 These findings raise suspicion for a greater role of BK virus in bladder carcinogenesis in KT patients, as opposed to the general population wherein immunocompetence may restrain BK virus-associated carcinogenesis. Human polyomavirus has also been associated with cases of post-KT UTUC in Taiwan, 44 and human papilloma virus with several cases in the United States. 45 However, whether treatment of these viral infections leads to regression of bladder cancer remains to be elucidated.
NMIBC
Transurethral resection of bladder tumor
For NMIBC, the gold standard treatment of TURBT differs little in KT patients as compared to the general population. However, it should be noted that tumor recurrence for NMIBC is higher in KT patients compared to non-KT patients with end-stage renal disease, 13 which may be due to several factors such as higher tumor aggressiveness and lower utilization of intravesical BCG therapy.
Intravesical therapy – BCG
The use of intravesical therapies, especially BCG immunotherapy, in KT patients is controversial. Although intravesical BCG is a guideline-recommended adjuvant therapy after TURBT for intermediate- and high-risk NMIBC patients, the evidence for immunosuppressed KT patients is low due to lack of a sizable population for randomized trials. There exist reported cases of disseminated mycobacterial infection following intravesical BCG, which can be potentially fatal; this risk is thought to be higher in immunosuppressed patients such as those with a KT. 46 There are concerns that chronic steroid therapy may lower the host's immune response to BCG immunotherapy and hence decrease its efficacy. Conversely, there are also theoretical concerns about the Th1 inflammatory response from BCG contributing to a heightened immune response to the allograft, resulting in rejection. 47 The immune response to BCG has also been implicated in cases of acute kidney injury, which may prove disastrous for the allograft kidney. 48
Nonetheless, several studies have shown a favorable safety profile for intravesical BCG in this population. Palou et al. 20 found no change in renal function across 3 patients, and did not report any cases of tuberculous infection. A systematic review of 7 patients in the setting of KT 49 found no cases of disseminated infection although there was one failure of BCG therapy. Another review of 238 patients with all types of solid organ transplants including KT 47 found a low complication rate of 12%, all of which were Clavien-Dindo Grade 1. Moreover, in this study, disease-free survival in patients who received intravesical BCG after TURBT was numerically higher than those who underwent TURBT only (47% versus 35%). Huang et al. 13 compared NMIBC patients with and without KT, finding a higher rate of cancer recurrence in the KT group (78% versus 38%, p = 0.032). The largest included cohort study of 87 KT patients with bladder cancer 22 had 71 NMIBC patients, of which only 29 received adjuvant intravesical therapy – BCG induction only in 2 patients, BCG induction and maintenance in 4 patients, and MMC in 20 patients. Univariate analysis showed a significant reduction in bladder recurrence in patients who received BCG compared to those who did not (p = 0.043). For the MMC-treated patients, only one complication of urinary tract infection was reported.
In view of these findings, the benefit of BCG in reducing recurrence in intermediate- to high-risk NMIBC appears to supersede the potential risks of disseminated infection and graft rejection, which appear to be extremely rare side effects. In fact, the underutilization of BCG treatment (or substitution of intravesical MMC/epirubicin) may be a contributing factor to the poorer recurrence-free survival seen in KT patients as compared to the general population.13,28,50
Intravesical therapy – others
Use of intravesical MMC, epirubicin and gemcitabine was reported in some included studies. Epirubicin was used in all 18 patients in the study by Li et al., 16 and no deaths due to bladder cancer were reported during follow-up. Prabharasuth et al. 21 offered intravesical options of BCG, MMC and thiotepa, and treatment was determined by patient and physician discretion. There was no mention of treatment by NMIBC risk category as is recommended by current guidelines. Novel gene therapies such as nadofaragene firadenovec were not used, but these may represent a feasible treatment option as they do not come with a risk of disseminated disease as in BCG. Indeed, this gene therapy has shown promise in long-term follow-up of non-KT patients with BCG-unresponsive bladder cancer, with good safety outcomes. 51
MIBC
Radical cystectomy and pelvic lymph node dissection
RC for bladder cancer in the setting of previous KT is a challenging operation due to altered anatomy, adhesions from previous surgery, and the importance of preserving allograft urinary drainage to avoid compromise to renal function.
For KT patients, pelvic lymph node dissection (PLND) is typically performed radically on the contralateral side of the graft and to a limited extent on the ipsilateral side. Moses et al. 19 reported that pelvic recurrence occurred in 40%, but it is unclear with such a small sample of patients whether this was associated with the suboptimal PLND. However, from studies in patients without KT, crossover lymphatic drainage is a common phenomenon and unilateral PLND may still miss lymph nodes even in strictly unilateral tumors. 52 This common practice reflects the trade-off between complete oncological clearance and preservation of a precious allograft.
For reconstruction after RC, surgeons must consider the length of the graft ureter and avoid over-tensioning during anastomosis to prevent disruption of blood supply and causing ureteric stenosis. In the largest case series of Rodriguez Faba et al., ileal conduit was the most frequent method of urinary diversion employed in 58% of patients undergoing RC, but functional outcomes and survival outcomes specific to the MIBC group were not reported. For these patients with complex and altered anatomy, an ileal conduit is the most accommodating method of urinary reconstruction and can also allow for postoperative radiation of the pelvis in unresectable disease. 25 Orthotopic diversion with neobladder is a feasible alternative as outlined by Moses et al.; all patients were continent in the daytime postoperatively, although 60% had mild nighttime stress urinary incontinence. 19 The authors identified the transplant ureter via indigo carmine in all patients, and also by frozen section in 60% (3 of 5) patients. Bilateral native nephroureterectomy was performed in 80%. Kamal et al. 14 also found excellent outcomes for orthotopic neobladders, with all 5 patients having adequate renal function, acceptable serum creatinine and electrolyte values, and ability to void spontaneously. This technique however requires additional considerations for preservation of the length of the graft ureter.
Overall, RC with urinary diversion and limited ipsilateral PLND appears to be a feasible and safe option for patients with previous KT.
Concomitant UTUC
Several studies included patients with both bladder cancer and UTUC. Most urothelial carcinoma typically originates in the recipient's urinary system rather than the donor's, due to a combination of the longer period of exposure to triggers such as smoking and aristolochic acid in the recipient and immunomodulation of the graft which may have some antitumor effects. 11 In contrast to patients without KT, nephroureterectomy of native kidneys does not affect renal function. Bilateral nephroureterectomy can serve as a risk-reducing operation even if concomitant UTUC is not present. In one study, concomitant T3b bladder cancer and bilateral UTUC was treated not with radical cystectomy but with partial cystectomy and bilateral nephroureterectomy, ostensibly to avoid disruption of the anastomosis between the bladder and the allograft ureter. 27 Several cases of partial cystectomy in simultaneous bladder and UTUC were also reported by Du et al. 11 Interestingly, in this study, recurrences in all 3 patients who underwent RC for MIBC without concomitant UTUC were in the upper urinary tract. Ideally, the bladder tumor would have to be small, localized to the contralateral side of the transplant or the bladder dome, and far from the transplanted ureter anastomosis, for partial cystectomy to achieve adequate oncological margins without loss of the anastomosis to the transplant ureter; 53 nonetheless RC is usually preferred in view of the risk of recurrence.
In a study of 35 cases of urothelial cancers in KT recipients, 13 bilateral nephroureterectomies were performed, of which 2 were for known bilateral UTUC, and of which 4 of the remaining 11 had previously undetected UTUC. Two of 15 patients who initially had unilateral nephroureterectomy eventually needed contralateral nephroureterectomy for a subsequent tumor. Hence, simultaneous bilateral nephroureterectomy with RC should be conducted even in the setting of isolated MIBC, in view of the high recurrence rates in the native urinary tract, low risk of the procedure as native kidneys contribute little to existing urinary function, and difficulty with upper tract surveillance.
Trimodality therapy
Trimodality therapy (TMT) consists of maximal TURBT followed by concurrent chemoradiation, and is a feasible alternative for patients who strongly desire bladder preservation. TURBT has been discussed prior and chemotherapeutic considerations are discussed in the next section. The distinctive feature of TMT from the aforementioned treatments for bladder cancer is its use of pelvic radiotherapy, which has theoretical effects on the nearby graft. Treatment regimens typically involve 64–66 Gy of maximum radiation dose to the tumor. 54 However, reports of pelvic radiation in the setting of KT are promising. In a series of 10 patients with urothelial cancer (including 4 cases of bladder cancer) receiving 3D conformal radiotherapy or intensity-modulated radiation therapy, no cases of graft failure directly related to radiation occurred. 55 Studies of patients receiving pelvic radiotherapy for other pelvic malignancies such as prostate and anal cancer also reported no significant effect on graft function.56,57 There exists only one case report of TMT for bladder cancer in KT, wherein the patient successfully completed the treatment course without graft loss or recurrence at 55 months after diagnosis. 58 Overall, the limited evidence in literature suggests that TMT may still be a feasible alternative to RC for KT patients, although there is no data on whether it is oncologically equivalent to RC in this setting.
Metastatic disease
Current guidelines recommend treatment of metastatic bladder cancer with systemic chemotherapy or immunotherapy. 59 In patients with a kidney transplant, the nephrotoxic side effects of common agents such as cisplatin may warrant dose adjustment to avoid graft loss. Immunotherapy agents such as PD-1 inhibitors may theoretically lead to graft loss due to production of T cells against donor alloantigens. In a systematic review of case reports of KT patients treated with PD-1 inhibitors for various cancers, the rate of graft loss was shown to be 82%, with most cases of rejection occurring in days to weeks after initiation. 60 Yet, in another study of 17 KT patients treated with nivolumab for solid tumors, no cases of graft loss occurred over a median follow-up of 28 months. Results of the latter study suggest that keeping to the baseline immunosuppressive regime at initiation would not affect expected antitumor efficacy and may reduce the risk of allograft rejection caused by initiation of immunotherapy. 61 There is no data in literature regarding the use of the novel antibody-drug conjugate enfortumab vedotin, or fibroblast growth factor receptor inhibitors such as erdafitinib, for bladder cancer in KT recipients, and is an important area for future research as these therapies become increasingly employed.
Immunosuppression regime
Despite the possible contributions of immunosuppression to de novo cancer, continued immunosuppression is usually required to prevent graft rejection and return to dialysis. In the setting of treatment for bladder cancer, dose modification may be required to avoid toxicity. 14 Several studies also performed a switch of immunosuppressive medication, the most common being from cyclosporine A to sirolimus or tacrolimus.10,11,23,29 With regard to tacrolimus, the superior graft survival seen compared to cyclosporine 62 may have driven the switch in some studies, especially given that some patients were already on a cyclosporine regimen before the proven efficacy of tacrolimus. mTORi such as sirolimus have demonstrated antiproliferative effects in other settings; it is also associated with lower cancer risk post-transplantation, especially non-melanotic skin cancer, 63 and this effect is most pronounced in patients who convert from a pre-established immunosuppression. However, the higher risk of mortality seen on meta-analysis for KT patients on sirolimus 64 makes this unsuitable option at the outset of transplantation. Everolimus is currently in wider use than sirolimus, especially in combination with tacrolimus. Palazetti et al. 30 performed a regimen switch introducing mTORi alongside dose reduction or cessation of calcineurin inhibitors, and did not report any instances of graft loss. On equipoise, a switch to an mTORi such as everolimus should be considered in patients who present with post-transplant cancer, including bladder cancer, to take advantage of its antiproliferative effects; this was also demonstrated in the large study by Rodriguez Faba et al. 22
There has been discussion in literature regarding the role of reduction of immunosuppression during treatment of advanced or muscle-invasive tumors. Despite the risk of increased rejection rates, the immunosuppressive effects of chemotherapeutic cytotoxic agents used in adjuvant treatment of MIBC or as part of palliative chemotherapy for metastatic disease may compensate for the reduction in primary immunosuppressive therapy. 17 In such patients, the renal toxicity of cytotoxic agents such as cisplatin must also be considered. Immune checkpoint inhibitors used for cancer treatment in the presence of a solid organ transplant have been reported to cause graft rejection in 41% of patients, and 71% of those went on to experience permanent graft failure. 65 BK virus nephropathy may be treated with reduction of immunosuppression,66,67 and there is also some evidence to suggest that mTORi immunosuppression reduces the rates of virus-related events compared to calcineurin inhibitors. 68 However, there is no data on whether elimination of BK virus causes regression of associated bladder tumors. Hence, in patients whose graft function is already suboptimal at the time of bladder cancer diagnosis, there may be a consideration to withdraw immunosuppression and focus on cancer treatment, while discussing a return to renal replacement therapy with the patient.
Surveillance
Due to the heightened malignancy risk in KT patients, the role of surveillance remains controversial, with surveillance bias contributing to the higher on-paper detection rates of malignancy compared to patients on dialysis. Rogers et al. suggested urine cytology in cases of microscopic hematuria and detailed anatomical studies in those with risk factors or visible hematuria. 23 Most cases of bladder cancer in included studies were detected due to gross hematuria and not via routine surveillance. Post-TURBT, routine cystoscopic surveillance is necessary similar to NMIBC management in patients without KT.
The epidemiology of post-KT bladder cancer also varies across populations, which may have an impact on surveillance. In view of the markedly higher incidence of post-KT urologic malignancy, routine urinalysis and ultrasonography evaluation were recommended in the Asian population by Zhang et al. 29 The addition of cystoscopy to evaluation of patients with microscopic hematuria was also advocated by Davis et al. 10 Urine cytology screening for patients with BK virus reactivation post-transplantation was advocated by Roberts et al. in view of the possible causal relationship of BK virus with bladder carcinogenesis. 69 In a study by Roumeguere et al., 70 patients with a KT due to aristolochic acid nephropathy underwent routine cystoscopy with trigonal zone biopsy and urine cytology even if cystoscopy was normal, due to the high risk of urothelial cancers in this population.
Surveillance for UTUC is challenging in post-KT patients. As native kidneys in KT patients are often non-secretory and anuric, the lack of an excretory phase on contrast-enhanced CT scans reduces sensitivity of picking up small tumors. The discovery of native kidney hydronephrosis on imaging has been linked to detection of UTUC after KT, with an odds ratio of 35 compared to KT recipients who did not have hydronephrosis, but this finding has poor positive predictive value. 71 Adding to these challenges is the precipitous renal function in KT patients and the risk of contrast-induced nephropathy of the precious allograft. These factors also contribute to the practice of risk-reducing bilateral nephroureterectomy in the same setting as cystectomy for bladder cancer, as mentioned earlier. Finally, several unique technical challenges of UTUC management and pre/post-operative surveillance exist in KT patients, mainly related to difficulty with endoscopic access to transplant ureters. The success of transplant ureteric orifice access via ureteroscopy ranges from 72% to 100% among studies in literature, and comes with a higher risk of ureteric injury during ureteroscope manipulation.72–74
Conclusions
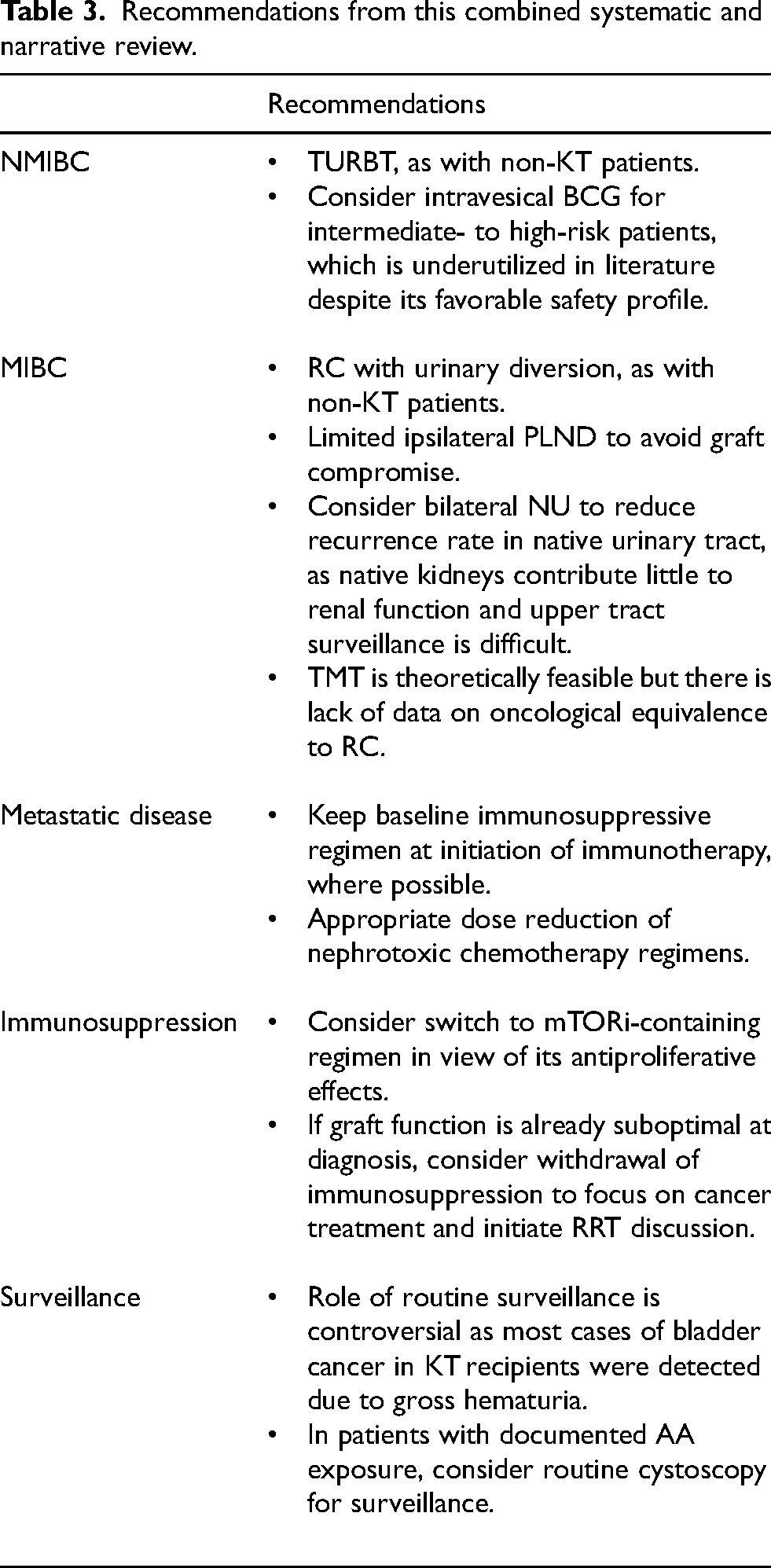
This review summarizes management strategies for bladder cancer in the setting of previous KT (Table 3). RC with limited PLND and urinary diversion and consideration of bilateral nephroureterectomy appears to be the safest method of oncological control in MIBC, while TURBT with intravesical BCG for intermediate- and high-risk NMIBC appears to be underutilized in this group despite its safety and associated reduction in recurrence. A switch in immunosuppressive regimens to mTORi may be considered in lieu of its antitumor effects. Routine surveillance in KT patients with risk factors for bladder cancer is challenging but may be warranted especially in the Asian population which has a higher rate of urothelial malignancy. Further research and longer follow-up of large transplant databases are required to elucidate optimal strategies for bladder cancer treatment in the KT setting.
Recommendations from this combined systematic and narrative review.
Supplemental Material
sj-docx-1-blc-10.1177_23523735251321986 - Supplemental material for Management of bladder cancer in kidney transplant recipients: A narrative review
Supplemental material, sj-docx-1-blc-10.1177_23523735251321986 for Management of bladder cancer in kidney transplant recipients: A narrative review by Khi Yung Fong, Ee Jean Lim, Wei Zheng So, Edwin Jonathan Aslim, Ho Yee Tiong and Valerie Huei Li Gan in Bladder Cancer
Footnotes
ORCID iDs
Author contributions
Khi Yung Fong: Methodology, Software, Formal Analysis, Investigation, Data Curation, Writing – Original Draft, Visualization
Ee Jean Lim: Methodology, Investigation, Writing – Original Draft, Investigation
Wei Zheng So: Methodology, Investigation, Writing – Review & Editing
Edwin Jonathan Aslim: Investigation, Writing – Review & Editing
Ho Yee Tiong: Conceptualization, Investigation, Writing – Review & Editing, Supervision
Valerie Huei Li Gan: Conceptualization, Methodology, Writing – Review & Editing, Supervision, Project Administration
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data sharing statement
This manuscript makes use of publicly-available data from published studies, therefore no additional data are available for sharing.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
